Abstract
Heavy metal wastes generated from mining activities are a major concern in developing countries such as Iran. Increasing concentrations of these metals in the soil make up a severe health hazard due to their non-degradability and toxicity. In this study, batch washing experiments were conducted in order to investigate the removal efficiency of zinc by biodegradable chelates, tartaric acid. For this purpose, soil samples were collected from the zinc contaminated soil in the region of the Angouran, Zanjan, Iran. Hence, optimization of batch washing conditions followed using a three-level central composite design approach based on the response surface methodology. The results demonstrated that the effects of pH, tartaric acid concentration, and interaction between selective factors on the zinc removal efficiency were all positive and significant (P < 0.05). An optimum zinc removal efficiency of 89.35 ±2.12% was achieved at tartaric acid concentration of 200 mM l−1, pH of 4.46, and incubation time of 120 min as the optimal conditions. Accordingly, response surface methodology is appropriately capable to determine and optimize chemical soil washing process to remediate heavy metal polluted soil.
Keywords
Introduction
Toxic heavy metal contamination of soil is a major environmental problem throughout the world because of their long residual time, strong concealment, toxicity, and other characteristics (Wei et al., 2019). Due to the anthropogenic activities such as mining and industrialization, most soils of rural and urban environments may accumulate the heavy metals high enough to cause risks to human health and ecosystems (Wuana et al., 2010). Zinc (Zn) has been classified as a priority hazardous contaminant by United States Environmental Protection Agency (Moon et al., 2012). Water-soluble Zn not only can contaminate groundwater that lead to potentially adverse impact on human health but also can interrupt the activity in soils as it unsafely influences the activity of microorganisms and earthworms.
A variety of in situ and ex situ remediation techniques have been developed to contain, clean up, or restore heavy metal-contaminated soils, such as soil flushing, surface capping, solidification, electrokinetic extraction, phytoremediation, and vitrification. These techniques can be classified into five categories: physical, chemical, electrical, thermal, and biological remediation or three divisions: transformation-based (e.g. stabilization/immobilization), containment-based (e.g. capping/encapsulation), and transport-based (e.g. extraction/removal) methods (Liu et al., 2018).
Generally, these soil remediation methods utilize different working mechanisms and indicate specific application advantages and limitations. These techniques differ markedly in effectiveness and cost in field practices (Khalid et al., 2017). Among the techniques of remediation, soil washing can be a practical option because of its simplicity, low cost, short duration, and high efficiency. Soil washing is a combined physical and chemical process for heavy metals elimination from contaminated soil by washing the soil ex situ with specially formulated solutions. Soil washing depends upon washing solutions to mobilize heavy metals by changing soil acidity, solution ionic strength, redox potential, or complexation. An ideal washing solution should dramatically improve the solubility and mobility of heavy metal contaminants yet interact weakly with soil constituents and should be nontoxic and biodegradable (Liu et al., 2018). In this regard, the effectiveness of washing is thoroughly related to the ability of chelating agents to dissolve Zn contaminant in soils. Naturally, the selection of the extracting solution does not depend on extraction effectiveness alone, but involves economic and environmental issues as well. An array of chemicals have been tested to formulate effective washing solutions: tartaric acid (TA), hydrochloric acid, polyglutamic acid, fluorosilicic acid, formic acid, acetic acid, sulphuric acid, nitric acid, phosphoric acid, oxalic acid, citric acid, DTPA, EDTA, NTA, EDDS, carbonate/bicarbonate, ammonium acetate, dithionite, isopropyl alcohol, subcritical water, etc. (Alghanmi et al., 2015; Bilgin and Tulun, 2016; Liu et al., 2018; Yang et al., 2012; Zhu et al., 2015). The optimal washing conditions including the effective concentration of the washing solution and agitation time are case-specific and have to be determined by small-scale trials.
Particularly, EDTA is identified as the most effective chelating agent to remove Zn from soils because of its strong chelating ability for cationic heavy metals and its applicability for broad range of soil types (Wirosoedarmo et al., 2018; Xia et al., 2019). However, permeability has been significantly reduced when EDTA is contacted with certain types of soil, showing that batch washing with EDTA may necessitate extended clean-up time. In addition, the high cost of EDTA and its low biodegradability degree has precluded its use in remediation of metal contaminated sites. Accordingly, it has been replaced by natural biodegradable chelates, TA being the most prominent (Dermont et al., 2008; Wuana et al., 2010; Yu et al., 2019), coupling with only minor impact on the physical and chemical properties of the soil (Römkens et al., 2002).
To develop an efficient and cost effective batch washing by TA, particularly for scale up applications, the operating conditions must be optimized. The one-variable-at-a-time approach as a classical optimization method not only requires a lot of experiments, but also frequently and easily ignores the alternative effects between components. To overcome these problems, organized and efficient process will be achieved when mathematical optimization techniques in terms of response surface methodology (RSM) were applied (Ibrahim and Elkhidir, 2011; Kakelar and Ebrahimi, 2016; Motamedimehr and Gitipour, 2019; Yakout et al., 2019).
RSM model could be used to predict the best performance conditions by using a proper experimental design. In addition, the modelling of system using RSM assists interpreting of multivariate phenomena and provides improvements to enhance the possibility of process scaling (Alkhatib et al., 2015).
Gharibzadeh et al. (2018) applied RSM to evaluate the effect of the influencing parameters on phenanthrene removal efficiency in soil washing process by using the nonionic surfactant and found optimal operational conditions to achieve the highest removal efficiency (Gharibzadeh et al., 2018). Fereidonian Dashti et al. (2018) used RSM to calculate the outcomes of process variables and their role in reaching ideal conditions. Their results showed that the actual data fitted well with the predicted data (Fereidonian Dashti et al., 2018). Goleij and Fakhraee (2017) employed RSM with CCD to evaluate the effects of solution pH, contact time, temperature, initial heavy metal concentration, and adsorbent dosage on the removal efficiency of the heavy metals (Goleij and Fakhraee, 2017). The results of all researches showed that RSM is appropriately capable to determine and optimize conditions to achieve the highest removal efficiency and also good fitted between the actual data with the predicted data.
Concerning advantages in using RSM, it has been performed effectively in the optimization of soil washing condition on Zn removal efficiency. Accordingly, the primary objective of this research was to investigate the removal of Zn from a severely contaminated soil by batch washing method with TA as an eco-friendly biodegradable chelant. For this purpose, we developed a quadratic polynomial model, using a central composite design (CCD) based on RSM.
Material and methods
Soil sampling
The test soil was obtained from the top soil layer (0–20 cm) around the Zn mine called Angouran mine located in Zanjan Province, Iran (Lat. 47° 15′ 17″, Long. 35° 44′ 37″). The Zn contaminated soil was air-dried and passed through a 2 mm nylon mesh sieve in order to remove large particles and obtain a homogeneous soil size. After drying, the soil sample was stored in plastic cans at room temperature for subsequent use in experiments. Contaminated soil characterization included the analysis of soil texture, pH, electrical conductivity (EC), cation exchange capacity (CEC), soil organic matter (SOM), calcium carbonate equivalent (CCE), and total Zn content. Soil texture was classified by using hydrometer method as described by Gee and Bauder (1986). By dichromate oxidation, SOM content was measured, and soil pH and EC were measured in a soil–solution ratio of 1:5 (w/v) using pH and EC meter (Rowell, 1994). Determination of CEC and CCE was conducted by saturation of soil with 1 M NH4OAc at pH 7 and the acid neutralization method, respectively (Rowell, 1994). The total Zn content of soil was obtained by using a flame atomic absorption spectrophotometer (Shimadzu AA-7000) after digesting soil sample with HNO3–HCl–HClO4 mixture at a 1:2:2 ratio (v/v/v). The selected physicochemical properties of the polluted soil sample are summarized in Table 1.
Physicochemical features of contaminated soil before washing.

CCE: calcium carbonate equivalent; CEC: cation exchange capacity; EC: electrical conductivity; SOM: soil organic matter.
The contaminated soil was calcareous (CCE = 21%) and slightly alkaline (pH = 7.77). Soil texture was classified as clay loam. The total Zn concentration (5657 mg kg−1) in the soil was much more than the standard level for soil used primarily for food crop production.
Single factor experiments
Single factor experiments were conducted to find out the effective variables on the Zn removal efficiency and their appropriate range by using the one-variable-at-a-time approach in this study. This phase was included in chemical batch washing experiments in which three parameters that may affect the removal efficiency were TA concentration, pH, and washing time. Batch washing experiments were performed using a soil–solution ratio of 1:10 (w/v) in 100 ml acid-rinsed polycarbonate plastic bottles. For each run, 5 g of the contaminated sample was added to 50 ml of the washing reagent. For investigation of the effect of TA concentration on Zn removal, different concentrations ranging from 5 to 400 mM were chosen. The pH effect experiments used 200 mM TA at different pH values (2.0–10.0), adjusted with a diluted HNO3 and/or NaOH solution. Moreover, 200 mM TA solution was prepared for determining the effect of the washing time on removal efficiency, which was performed at different times (15–300 min). A mechanic shaker stroke was used to prepare suspensions at a speed of 150 r min−1 for a predetermined time and then centrifuged at this speed of 4000 r min−1 for 10 min. The solution was filtered with filter paper (0.45 µm) and the Zn level measurement was done at all stages by an atomic absorption spectrophotometer. It should be noted that duplicate experiments were conducted for all samples.
RSM
RSM-based CCD was used to optimize the removal efficiency of Zn from contaminated soil by TA. This approach was used for the determination of combined effects of TA concentration (X1), pH (X2), and washing time (X3) on the Zn removal yield from soil as a dependent variable (Y). In this experiment, variables specifically TA concentration (0–300 mM), pH (2–10), and washing time (5–240 min) were chosen as three independent variables and the domains were determined using a single factor test (Table 2). In this study, CCD including eight cubic points, six axial points (α = ±1.68), and six replicates at the centre point (α = 0) were chosen as the experimental points.
Experimental range and level of independent variables.

Experimental response was assessed to get the optimum values of the variables. The factors were coded at five levels from −1.68, −1, 0, +1, and +1.68 as defined by equation (1)

Results and discussion
Preliminary batch washing experiment
In order to determine which variable and how it affects the response variable, effect plots of independent variables on Zn removal are depicted in Figure 1(a) to (c). It is clear that all of selected variables significantly affected removal yield. As given in Figure 1(a), the lowest value of Zn removal by distilled water is without TA, with a Zn removal yield of less than 0.01%. Consequently, Zn has been insoluble in water. The removal of Zn becomes more by increasing the concentration of TA from 10 to 200 mM, and therefore, this range is chosen as the range of TA concentration for CCD design based on RSM. Above 200 mM, the removal efficiency is constant and does not vary considerably. This can be expressed by the existence of other heavy metals in soil such as lead, cadmium, and arsenic, that they compete with Zn to wash with TA (Wang et al., 2015).

Single factor washing experiments for investigating limit of Zn removal as a function of TA concentration (a), pH (b), and washing time (c).
For Zn removal, it is obvious from Figure 1(b) that pH is important factor which negatively affects removal efficiency. In general, pH has major impact on soil’s ability to retain heavy metals, in which metal desorption from soil constituents increases as pH decreases. Decreasing solution pH leads to a rapid reduction in net negative surface charge and consequently declines soil’s affinity for metal ions (Wu et al., 2003).
Results showed that when pH was decreased from 9 to 2, removal yield increased remarkably from 8.43 ± 2.08% to 60.63 ± 3.85%, while remaining almost constant for lower pH values. Removal with increasing solution pH was characteristic of ‘S’ pattern (Figure 1(b)). Desorption trend can be divided into three phases based on removal efficiency: (1) the greatest removal yield at pH of 2.0, with more than 60.0% of efficiency at which Zn removal decreased gradually; (2) the removal decreasing phase at pH 2.0–4.0, at which Zn desorption decreased significantly, with efficiency decreasing from 50 to 8.5%; and (3) the minimal desorption phase at pH > 7, at which the extraction yield levelled off. Consequently, the efficiency of Zn removal from polluted soil by the acidic condition is better than alkaline conditions. Thus, the pH range for RSM optimization was selected from 4 to 8.
Generally, it has become evident that increasing of washing time causes higher removal efficiency from contaminated soil (Fabbricino et al., 2013). The results of the washing time influence on Zn removal are presented in Figure 1(c). This factor showed a positive effect, which means that the extraction efficiency increases when the incubation time was at the high level. Increasing the washing time from 15 to 120 min enhanced extraction from 7.16 ± 3.7% to 57.52 ± 3.17% demonstrating that removal efficiency depends on the duration of washing time. There was no change in the removal rate above this time and therefore, the effective time range of Zn removal with TA is from 15 to 120 min. In the following, the optimization results for Zn removal are described using the RSM from the CCD.
Optimization of the batch washing process
Optimization was performed in three levels based on three factors by the CCD. The design matrix and the results of the RSM tests are presented in Table 3. This design was evaluated in accordance with the second order polynomial model using multiple regression based on equation (2)


Central composite design coded variables of the observed responses and predicted values for Zn removal.

TA: tartaric acid.
In this equation, X1, X2, and X3 are representatives of the independent variables of pH, TA concentration, and washing time, respectively. The calculated values of Zn removal efficiency have been determined by quadratic polynomial model (equation (3)) and are presented in Table 3. The results show that there is a good agreement between CCD predicted values of unease activity and experimental data.
Zn removal variation from 3.87 to 70.57% represents the significance of optimization for heavy metal extraction from soil. The results indicate that a good agreement between CCD forecast values and observed experimental data according to the correlation coefficient (R2= 0.945) and root mean square error (RMSE = 4.6%) as shown in Figure 2. This implies that the CCD model can explain 94.5% variation in the response.

Comparison between the experimental results and predicted values for Zn removal by the suggested CCD model.
Analysis of variance (ANOVA)
The results of ANOVA of the quadratic response surface are given in Table 4. Based on Table 4, efficiency of Zn removal is significantly influenced by both linear and interaction parameters (P-value < 0.05). This implies that the RSM model could describe the effect of independent variables on the extraction yield of this study very well.
ANOVA results for Zn removal.

ANOVA: analysis of variance; TA: Tartaric acid; DF: Degree of freedom.
R-sq = 0.94, R-sq(adj) = 0.89.
In ANOVA table, the larger the F-value, the more probable that the variance contributed by the RSM model is significantly larger than random error. As the obtained F-value in ANOVA table, 19.34, is noticeably higher than the F critical value tabulated (3.02 at an α level of 0.05), it can be concluded that the model is predicting the experimental data well. This result confirms the prediction power of the fitted model.
The estimated regression coefficients, t-value, and p-value for linear, quadratic, and interaction effects of the variables are summarized in Table 5, at 95% significance level. To evaluate the significance of each term of model, P-value was used. According to Table 5, all three independent variables and interaction effect between TA concentration and time are statistically significant (P-value < 0.05) explaining Zn removal efficiency.
Estimated regression coefficients, t-values, and p-values.

To further elucidate the experimental design results, Pareto analysis (equation (4)) was used for calculation of percentage influence of each factor on Zn removal

Based on Pareto analysis, it was found that the pH (X1, 67.25%) was the most effective parameter in this process. Among the variables, TA concentration (X2, 15.94%) and interaction between TA concentration and time (X2X3, 7.38%) created the main effects on the removal efficiency (Figure 3). These three terms totally include 90.57% of total influences on Zn removal.

Pareto graphic analysis to determine the effectiveness of each variable in the response.
In order to get the optimum CCD model for Zn removal, the final quadratic polynomial model is presented by removing the insignificant variables (p-value > 0.05) as follows (equation (5))

The residual plots are established for the model adequacy checking in Figure 4(a) to (d). As depicted in Figure 4(a), the response model for Zn removal was fixed to the normal distribution. This implies that neither response transformation was needed nor there was any apparent problem with normality (Özer et al., 2009). As it can be observed in Figure 4(b), there is no systematic pattern in the plot indicating that the variance of original observations is constant for all values of the response. Similarly, the histogram of residuals shows a pattern that the residuals are normally distributed for all observations (Figure 4(c)). In summary, all of the plots in Figure 4 revealed that the model is adequate to describe the Zn removal.

(a) Normal probability plot of the residuals, (b) residuals of the RSM model versus the fitted values, (c) histogram of the residuals distribution, and (d) residuals of the RSM model versus the observed values order.
Effect of independent variables by contour plots
To visualize the effects of all factors and the interactions clearly, contour plots – which are developed based on quadratic polynomial model – are shown in Figure 5(a) to (c). To prepare the contour plots, two variables were changed in the experimental ranges and other variable was considered to be constant.
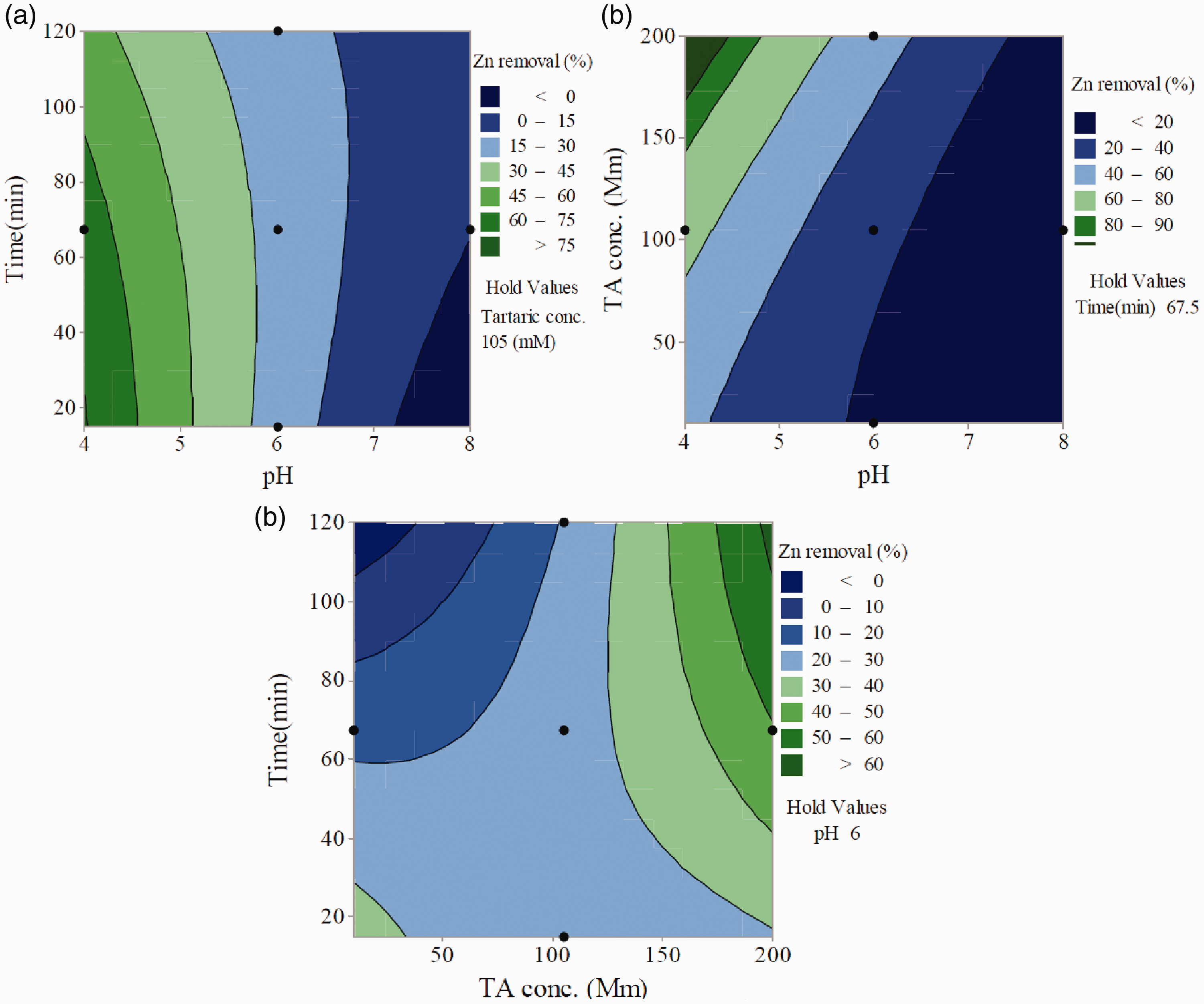
Response surface plots showing the interactions between the variables affecting Zn removal efficiency by TA: (a) interaction between the time and pH, (b) interaction between the TA concentration and pH, and (c) interaction between TA concentration and washing time. TA: tartaric acid.
In Figure 5(a), the combined effect of pH and time is shown at a TA concentration of 105 mM. The change in pH from 8 to 4 resulted in a notable increase of the extraction efficiency from 15 to 75%; in other words, at low pH, provocation processes may lead to improvement in metal removal efficiency. As the pH of the solution decreases, the adsorption capacity of the cationic heavy metals onto soil decreases. This happens because during adsorption at lower pH values the hydrogen ions compete with the metal ions and subsequently, the dissolution of metal compounds increases. Accordingly, this is expected as the extractability of Zn as a cationic heavy metal enhances at high acidity. Another explanation for the decrease in removal efficiency at higher pH could be attributed to the dissociation of functional groups on the active sites of the chelating agent as well as the solution ion chemistry (Begum et al., 2013; Bianchi et al., 2008; Dermont et al., 2008; Jelusic et al., 2013; Wang et al., 2014).
The simultaneous effect of TA concentration and pH on extraction efficiency is shown in Figure 5(b). Experiments were conducted at 67.5 min reaction time with variation of pH from 4 to 8 and TA concentration from 10 to 200 mM. As shown in Figure 5(b), increasing TA concentration at higher pH, the Zn desirability gradually enhanced. The effect of TA concentration was more remarkable at high acidity, and thus maximum Zn removal was observed at higher pH as mentioned above, with increased TA concentration.
These results supported the previous findings in another aspect that increasing pH generally caused an increase in the metal adsorption (Elwakeel et al., 2015; Yakout et al., 2019). It is reasonable to conclude that the magnitude of desorption varies with pH. There is no general agreement on a single mechanism responsible for this behaviour, but strong adsorption of metal hydroxo complexes, hydrolysis of heavy metal on exchange sites, competition of protons for adsorbing sites, and acid catalysed dissolution of reactive oxide sites may be involved (Al-Wakeel et al., 2015; Elwakeel and Yousif, 2010; Sun et al., 2001).
The interaction effect of TA concentration and time is illustrated in Figure 5(c). The CCD model was applied with TA concentration changing from 10 to 200 mM and washing time from 30 to 150 min at constant pH of 6. Figure 5(c) illustrates that removal efficiency is enhanced by increasing the TA concentration, but the effect of time appears to be more complicated. According to the results of the CCD model (Table 4), the effect of washing time was not statistically significant on removal efficiency (P-value > 0.05). In other words, metal desorption was not directly influenced by washing time. However, the influence of washing time was only significant in terms of interaction with TA concentration (P-value < 0.05). At high concentration of chelating agent as reaction time increased, the removal efficiency of metal contaminant was also enhanced for TA washing, while at low concentration by increasing reaction time extraction yield declined probably due to readsorption of heavy metal by contaminated soil (Figure 5). Similar to our results, Tejowulan and Hendershot (1998) demonstrated that the heavy metal desorption rate did not depend on washing time, whereas Zn removal was dependent on chelating agent concentration (Tejowulan and Hedershot, 1998).
It has been demonstrated that low molecular weight organic acids such as TA were able to influence the release of heavy metals from different soils and increase their solubility in bulk soil through the formation of soluble metal organic acid complexes. Hence, addition of organic acids, which chelate with Zn2+ and Zn–OH+, may result in smaller amounts of Zn2+ adsorbed onto soil surface. The high concentration of the TA (chelator), the less Zn2+ is adsorbed, and correspondingly, more Zn2+ is desorbed (Yang et al., 2006).
Optimize washing conditions
The optimum values of TA concentration, pH, and reaction time were determined by a response optimizer in order to attain the highest removal efficiency (Table 6). To verify the predicted conditions, the confirmation experiments were performed with three replications at the predicted optimum conditions of RSM. It was found out that the maximum removal efficiency (89.35 ± 2.12%) was attained when the values of each parameter were set at the optimum values. The obtained maximal removal was in good agreement with the value predicted from the CCD model (95%) suggesting that the statistical optimization strategy used in this study was successful to attain the maximal heavy metal desorption.
Optimal values of the independent variables, experimental, and RSM predicted Zn removal.

SD: standard deviation of the four replicates; TA: tartaric acid.
Conclusion
The remediation of Zn-contaminated soil by TA, a biodegradable chelate, was investigated through soil washing process. The desorption conditions of zinc(II) ions in batch washing system were optimized by RSM-based CCD. The relationship between the Zn removal from contaminated soil by TA and the independent variables was developed via the quadratic polynomial function with high determination coefficients (R2= 0.94) and RMSE 4.6%. The obtained optimal Zn removal efficiency (89.35 ± 2.12) under optimized conditions, which was determined as pH 4.46, TA concentration 100 mM, and reaction time 120 min, was in agreement with the value predicted from the CCD model (95%). Furthermore, the results showed that the pH (X1, 67.25%) was the most effective parameter in desorption process. Among the variables, TA concentration (X2, 15.94%) and interaction between TA concentration and time (X2X3, 7.38%) created the main effects on the removal efficiency. Based on the obtained results, RSM was appropriate for optimizing the Zn desorption process by batch washing from contaminated soil.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
