Abstract
Ag-Y, Ce-Y, and Cu-Y zeolites were successfully prepared via the liquid phase ion exchange method as adsorbents for removing dibenzyl disulfide and dodecyl mercaptan in transformer insulating oil. The physicochemical properties of the adsorbents were investigated by X-ray diffraction, X-ray fluorescence, and N2 adsorption–desorption. The removal efficiency increased rapidly with increased dosage of Ag-Y (0.5–1.0 wt%) and Ce-Y, Cu-Y (1.0–3.0 wt%), and became slower with its increased dosage. Kinetic studies showed the adsorption on these adsorbents can be described by a pseudo-second-order model, and the rate constants increased with increasing temperature, indicating the endothermic nature of the adsorption. The activation energies (Ea) calculated for dibenzyl disulfide and dodecyl mercaptan were over 40 kJ mol−1 which clearly indicated that the adsorption process on the three adsorbents was dominated by chemical adsorption.
Introduction
Mineral insulating oils play a crucial role in power transformers as electrical insulation, and this application requires long-term thermal stability and stable insulation properties (Jian Li et al., 2011; Scatiggio et al., 2008). In recent years, the proportion of transformer failures caused by corrosive sulfur has increased gradually and has garnered increased attention (Flora et al., 2014; Scatiggio et al., 2009). Corrosive sulfur attacks copper windings to form a conductive substance (Cu2S) that leads to increased electrical conductivity and dielectric loss of the insulating paper. This results in the wire insulation strength being gradually weakened and eventually leads to transformer insulation breakdown (Khan and Rajan, 2015; Toyama et al., 2009).
Many researchers have focused on solving the problem of transformer insulation failure caused by corrosive sulfur in oil to promote the stable operation of power systems (Kato et al., 2013; Lukić et al., 2012; Maina et al., 2009; Matejkova et al., 2017). Using adsorbents to remove dibenzyl disulfide (DBDS) and other corrosive sulfur in insulating oil is a promising alternative, because this method can remove impurities under environmental conditions and improve the physical and chemical properties of insulating oil. Na-Y zeolite is widely studied as an adsorbent because of its open pore structure and large number of cation exchange sites (El-Nabarawy and Samra, 1994; Fungaro and Graciano, 2007; Murthy et al., 2013; Zhang et al., 2008). Song et al. (2015, 2016) reported that Ag-Y, Cu-Y, AgCe-Y, and CuCe-Y were effective for adsorption of thiophene or benzothiophene from commercial fuels via π-complexation and direct S–M interaction. Lv et al. (2014) reported that Ag-Y had good adsorption capacity for disulfide in model liquefied petroleum gas.
The modified Na-Y zeolites have a high sulfur adsorption capacity. Moreover, to the best of our knowledge, there is little research focusing on adsorption kinetics of corrosive sulfur in transformer oil by the modified Na-Y zeolites. To solve the insulation failures caused by corrosive sulfur in transformer oil, in this work we prepared Ag-Y, Ce-Y, and Cu-Y by the liquid phase ion exchange method and studied the factors that control the adsorption process. The textural properties, elemental composition, and crystal structure of the prepared adsorbents were characterized. The pseudo-first-order and the pseudo-second-order kinetic models were selected to correlate experimental data to gain additional understanding of the adsorption behavior of those adsorbents.
Experimental
Materials
Na-Y zeolite was purchased from Sino Petroleum Corporation. DBDS (99.99%) and dodecyl mercaptan (NDM, 99.99%) were purchased from Sigma-Aldrich Corporation. Noncorrosive refined base oil was purchased from China National Petroleum Corporation. AgNO3, Ce(NO3)3·6H2O, Cu(NO3)2·3H2O, and petroleum ether (all of A.R.) were purchased from Sinopharm Chemical Reagent Corporation. ICAP 6300 inductively coupled plasma optical emission spectrometer (ICP-OES)” was purchased from Thermo Fisher Scientific Co. Ltd. The main operating parameters of ICP-OES: cooling gas flow 12.0 l min−1, auxiliary gas flow 1.0 l min−1, atomizing gas flow 0.50 l min−1; RF power 1350 W; automatic integration; retesting three times; integration time 5 s; analysis line 182.036 nm.
Preparation of adsorbents
The Ag-Y, Ce-Y, and Cu-Y zeolites were prepared according to the liquid phase ion exchange method. Na-Y zeolite as a starting material was contacted with three different solutions made of 150 ml distilled water containing 2.40 g AgNO3, 2.28 g Ce(NO3)3, and 2.06 g Cu(NO3)2, respectively, at 80°C for 24 h. Each solution was stirred, filtered, and the filtrate was washed, dried, and then calcined at 450°C for 24 h. The ion exchange process was repeated, re-immersion of the solids in the nitrate solutions at 80°C for an additional 24 h. After filtering, washing, and drying at 120°C for 24 h, the three adsorbents were obtained.
Material characterization
The characterization of the textural properties of the Ag-Y, Ce-Y, and Cu-Y was performed by using U.S. Quantachrome Quadrasorb SI-3MP autosorb IQ3 Gas Sorption Analyzer. The surface area, pore volume, and pore diameter were measured based on the multipoint Brunauer–Emmett–Teller (BET) method. The crystal structures of adsorbent were identified by powder X-ray diffraction (Rigaku D-MAX2500VB X-ray diffractometer) and the patterns were collected from 5 to 80°C with 1.2°/min. The elemental compositions of the three adsorbents were analyzed using X-ray fluorescence spectrum (PANalytical B.V. AxiosmAX-minerals).
Preparation of model solution
DBDS and NDM, which cause transformer insulation faults, were used as representative sulfur compounds in transformer oil. Two model solutions containing 100 mg kg−1 sulfur in base oil were prepared.
Adsorption experiment
The 100 g model solution was used to evaluate the desulfurization capacity of each of Ag-Y, Ce-Y, and Cu-Y, and the adsorption kinetics experiments were carried out by controlled experiments in a batch system. In a typical adsorption experiment, a certain amount of adsorbent (name based on the mass ratio of adsorbent to 100 g model solution) was placed in a beaker along with a model solution and stirred continuously at different adsorption temperatures. The sulfur content was then analyzed at fixed intervals.
The adsorption capacity was calculated according to equation (1)
The sulfur removal rate (%) was calculated using the following equation (2)
Results and discussion
Characterization of adsorbents
Figure 1 shows that the main diffraction peak and cell parameters of the obtained adsorbent are similar to Na-Y, which indicates the structure of Na-Y remains. The crystallinity of Ag-Y and Ce-Y decreased obviously after ion exchange, which might be attributed to dealumination in the process of ion exchange and calcination. The XRD characteristic peaks of Ag, Ce, and Cu and their oxides did not appear, indicating that the Ag, Ce, and Cu ions had become highly decentralized into the lattice location of zeolites and occupied the cationic position.

XRD spectroscopy of Na-Y, Cu-Y, Ag-Y, and Ce-Y.
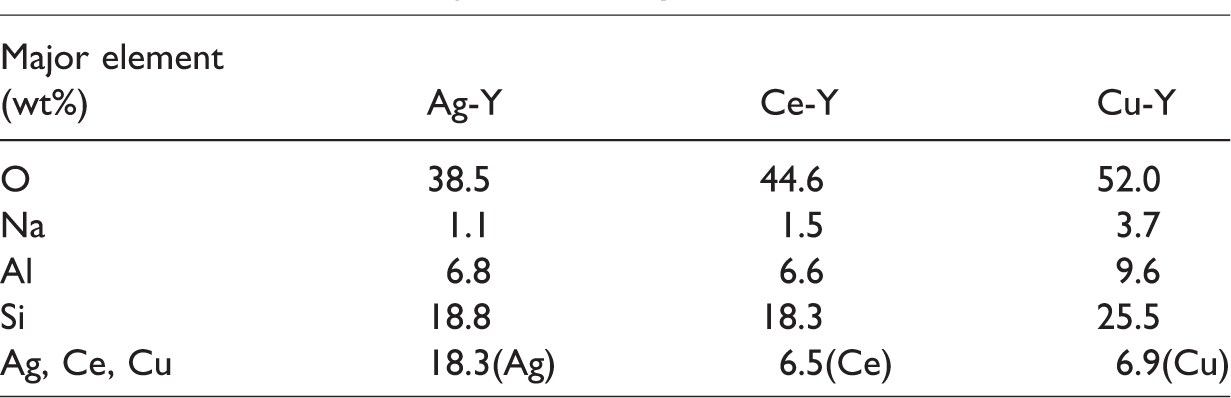
The elemental compositions of the adsorbents are shown in Table 1. The content of metal ions in each of the adsorbents prepared by ion exchange differs from the others with the mass fraction of Ag at 18.3% being the highest. The calculated exchange capacity of Ag, Ce, and Cu ions is 1.7, 0.5, and 1.1 mmol g−1, respectively. This is due to the fact that ion exchange capacity is related to the valence state of cations and the transition metals with valence states similar to Na are beneficial to the ion exchange process.
The elemental composition of Ag-Y, Ce-Y, and Cu-Y.
The surface area, pore volume, and pore diameter of adsorbents are important factors affecting the adsorption performance. Table 2 shows the specific surface area of Na-Y is 632.1 m2 g−1 and the pore volume is 0.2589 ml g−1. Lattice collapse, calcination, sintering, and pore plugging may result in the decrease of specific surface area and pore volume of Ag-Y, Ce-Y, and Cu-Y adsorbents. However, the BET surface area of Ag-Y, Ce-Y, and Cu-Y zeolites did not decrease significantly during modification, indicating the micropore structure of zeolite Y remained. Because the transition metal atom is much larger than the Na atom, the introduction of transition metal ion makes the pore size of the adsorbent increase, which is beneficial for having the sulfide enter into the modified zeolite pore and improving the desulfurization performance of the adsorbent.
The BET analysis of Cu-Y, Ag-Y, and Ce-Y.

Effect of contact time
The data from adsorption experiments of corrosive sulfur on Ag-Y, Ce-Y, and Cu-Y were plotted as a function of time at 313.15 K. As shown in Figure 2, the removal percentage of DBDS and NDM on Ag-Y increased rapidly in the first 15 min but then increased slowly. Beyond the adsorption of 2 h, removal remained almost unchanged. It is also observed that initially DBDS and NDM were adsorbed rapidly on Ce-Y and Cu-Y within 30 min, but subsequently, adsorption rates decreased and the removal curve became stable after 2 h of contact time. We can also see the desulfurization performance is Ag-Y > Cu-Y > Ce-Y at the dosage of 1.0 wt% adsorbent. It can be explained in terms of the hard–soft acids and bases (HSAB) principle, which states that soft acids prefer to associate with soft bases, and hard acids prefer hard bases (Lv et al., 2014). According to the classification of hard and soft acids in HSAB theory, Ag (I) is a soft acid, Cu (II) is an intermediate acid, and Ce (IV) is a hard acid, so Ag-Y showed the strongest adsorption ability for soft bases, such as DBDS and NDM. In addition, the mass fraction of Ag in Ag-Y is 18.3%, which is greater than the mass fraction of Cu in Cu-Y (6.9%) and Ce in Ce-Y (6.5%), so the Cu-Y and Ce-Y showed a poor adsorption performance compared to Ag-Y. Moreover, the pore diameter of Ag-Y is 1.784 nm, while the pore diameter of Ce-Y and Cu-Y is 1.727 and 1.754 nm, respectively, giving a slight advantage to Ag-Y since large pore diameter is better for the adsorption of sulfide. Additionally, from the kinetic results below, chemical adsorption must be considered. Molecular orbital calculations had confirmed that π bond formed between sulfur atoms, Cu+ and Ag+ (Yang et al., 2003). The different adsorption capacities can be attributed to the π complexation of Ag-Y, Ce-Y, and Cu-Y.
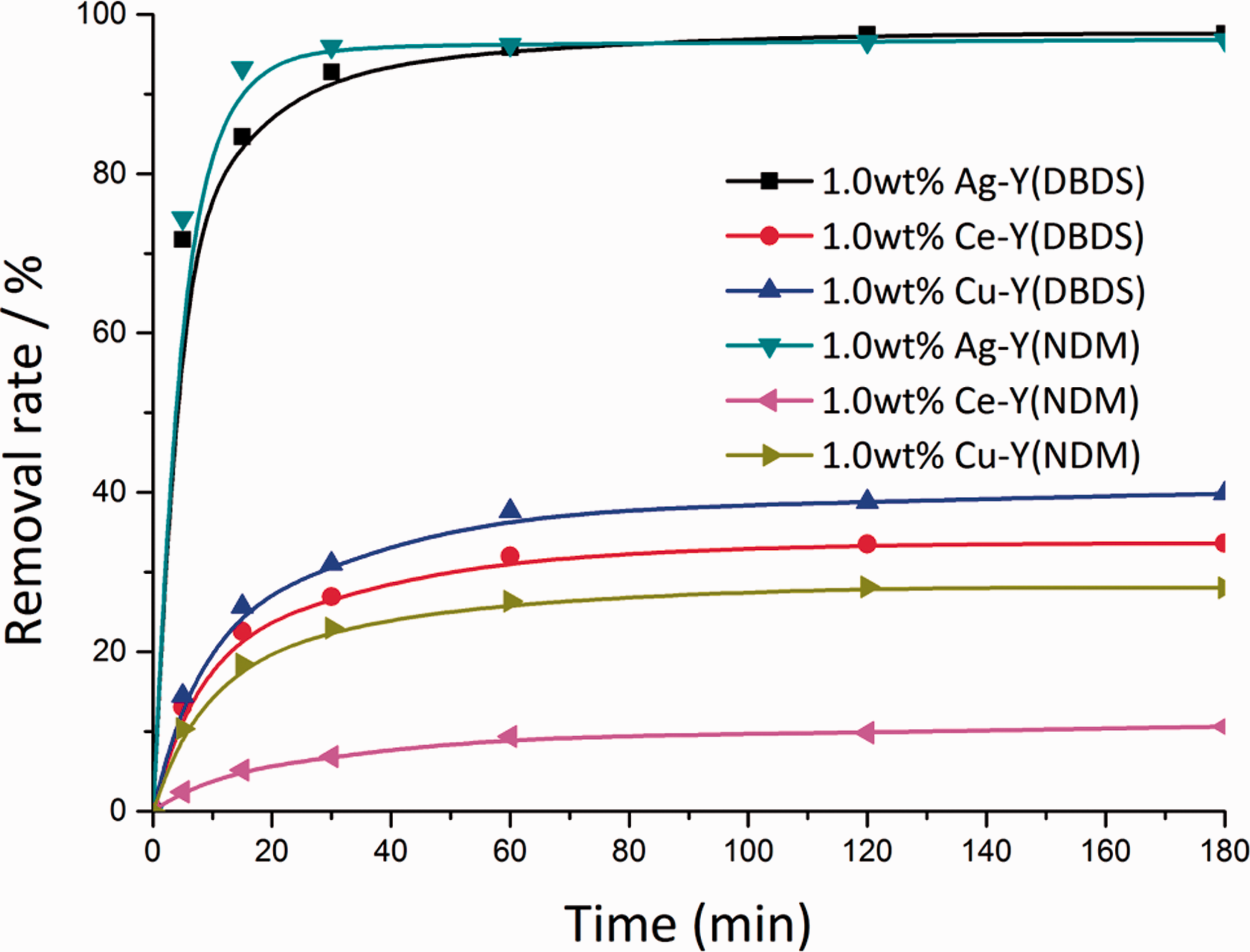
Effect of adsorption time of removal of DBDS and NDM on Ag-Y, Ce-Y, and Cu-Y at 313.15 K. DBDS: dibenzyl disulfide; NDM: dodecyl mercaptan.
The results found that as-prepared Ce-Y and Cu-Y possessed a better adsorption performance for DBDS, possibly due to the electron density of the sulfur atoms of DBDS being greater than NDM, a condition that would enhance direct interaction between DBDS molecules and metal ions (S–M). However, it is worth noting that as-prepared Ag-Y had larger adsorption capacity for NDM than for DBDS. Considering that Ag+ has a high polarity valence electron, the small-sized NDM molecules more easily entered the pores and was adsorbed on Ag-Y.
Effect of dosage
In order to increase the adsorption capacity and utilization ratio of adsorbent to the maximum extent, the influence of adsorbent dosage on adsorption of DBDS and NDM was investigated. As shown in Figure 3, when the adsorbent dosage of Ag-Y was increased from 0.5 to 1.0 wt%, the removal percentage of DBDS and NDM was increased. When the dosage of Ag-Y exceeded 1.0 wt%, there was no further remarkable increase in the percentage of desulfurization removal. Figures 4 and 5 show that when the dosage range of Ce-Y and Cu-Y was 1.0–3.0 wt%, the desulfurization performance improved. Upon further increase of the dosage of Ce-Y and Cu-Y to 5.0 wt%, the desulfurization performance no longer increased significantly. With the increase of adsorbent dose, the desulfurization performance was not obviously improved.

Effect of dosage on removal of DBDS and NDM over Ag-Y at certain time. DBDS: dibenzyl disulfide; NDM: dodecyl mercaptan.

Effect of dosage on removal of DBDS and NDM over Ce-Y at certain time. DBDS: dibenzyl disulfide; NDM: dodecyl mercaptan.

Effect of dosage on removal of DBDS and NDM over Cu-Y at certain time. DBDS: dibenzyl disulfide; NDM: dodecyl mercaptan.
Adsorption kinetics modeling
The analysis of adsorption kinetics is important in designing adsorption system and can provide a better understanding of the adsorption process. Thus, the kinetics of DBDS and NDM adsorption process on Ag-Y, Ce-Y, and Cu-Y were analyzed using pseudo-first-order and pseudo-second-order models. Correlation coefficient (R2) was selected to assess the conformity between the model predictions and experimental data.
The pseudo-first-order kinetic model is expressed as equation (3)

The pseudo-second-order kinetic model can be expressed as equation (4)

Figure 6 gives the nonlinear fitting curves of adsorption of DBDS and NDM on 1.0 wt% Ag-Y, 3.0 wt% Ce-Y, and 3.0 wt% Cu-Y, and the results of fitting parameters are listed in Table 3. As can be seen in Figure 6, the two adsorption kinetic models can fit to the experimental data well. In Table 3, the experimental data were better fitted to pseudo-second-order kinetic model in terms of higher values of R2 > 0.992, indicating that the pseudo-second-order models were slightly more capable of describing the adsorption kinetics process.

The nonlinear fitting curves for DBDS and NDM adsorption on Ag-Y, Ce-Y, and Cu-Y at different temperatures (■: experimental; ——: pseudo-first-order; - - - - -: pseudo-second-order). DBDS: dibenzyl disulfide; NDM: dodecyl mercaptan.
Pseudo first-order and pseudo second-order model constants and correlation coefficients for (DBDS and NDM) adsorption on Ag-Y, Ce-Y, and Cu-Y.
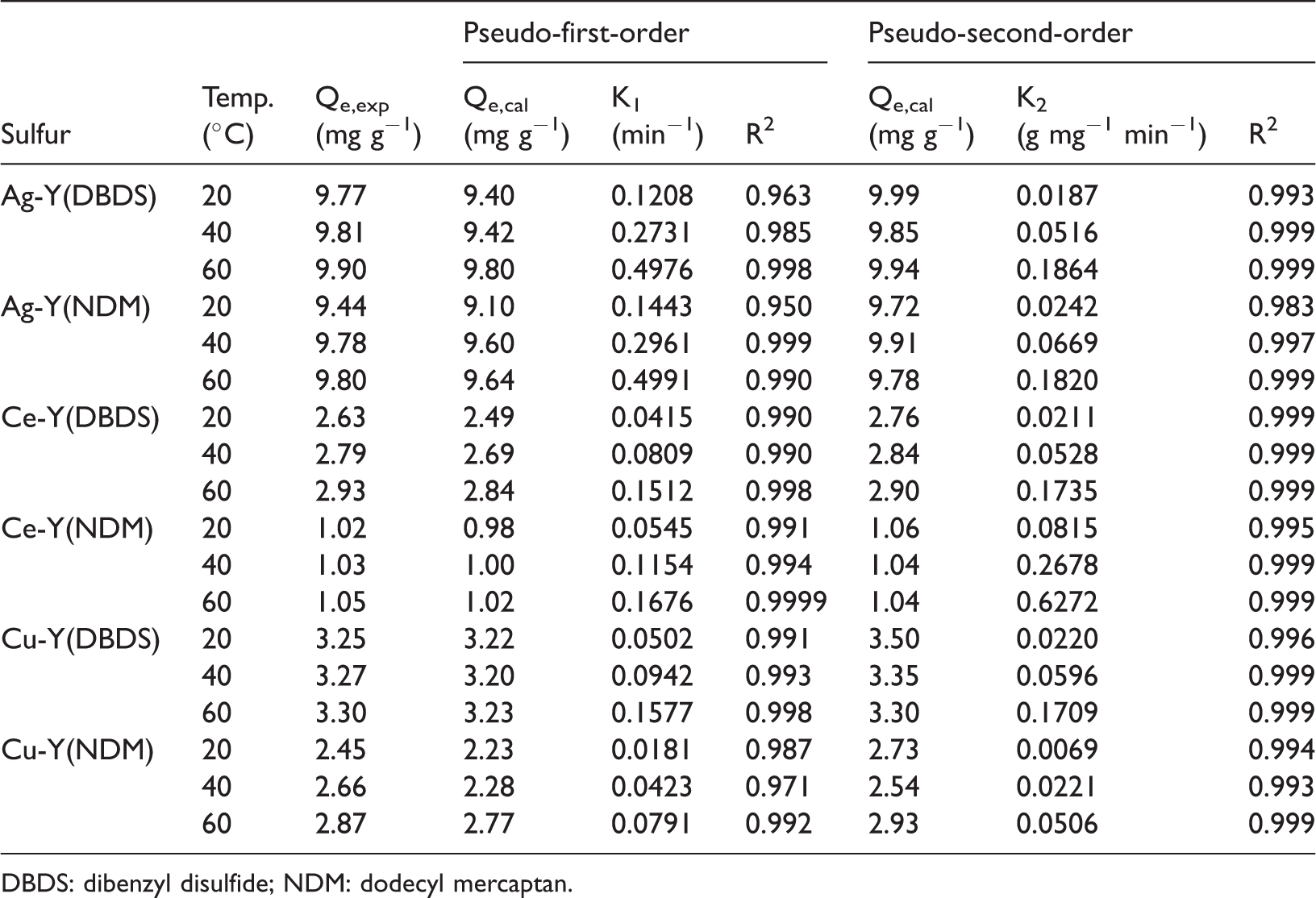
DBDS: dibenzyl disulfide; NDM: dodecyl mercaptan.
Activation parameters of the adsorption process.

DBDS: dibenzyl disulfide; NDM: dodecyl mercaptan.
Effect of temperature on adsorption kinetics
Increasing the system temperature reduces the viscosity of transformer oil, resulting in an increase in diffusion rate and a new equilibrium adsorption capacity. As shown in Figure 6, the adsorption process of DBDS and NDM on Ag-Y was faster to reach equilibrium at higher temperature. The rate constants (k1 and k2) and the adsorption capacity of DBDS and NDM on Ag-Y, Ce-Y, and Cu-Y were found to increase with temperature, showing that higher temperatures are beneficial to the process of adsorption, and indicating the endothermic nature of these adsorption processes. The effect of adsorption temperature on the adsorption rate can be expressed with the Arrhenius equation (5)


Arrhenius plot for the adsorption of DBDS and NDM on zeolites. DBDS: dibenzyl disulfide; NDM: dodecyl mercaptan.
Generally, a value of Ea above 40 kJ mol−1 indicates chemical adsorption and stronger bonds (Anirudhan and Radhakrishnan, 2008; Volchek et al., 2011). The results show that the calculated activation energy (Ea) for the adsorption of DBDS and NDM on Ag-Y, Ce-Y, and Cu-Y was all over 40 kJ mol−1, suggesting the adsorption was chemical adsorption which was consistent with the kinetic fitting results.
Conclusions
In conclusion, the prepared adsorbents are suitable for removing corrosive sulfur in transformer oil, and adsorption efficacy was in the order of Ag-Y > Cu-Y > Ce-Y. When the adsorbent dosage of Ag-Y was increased from 0.5 to 1.0 wt%, the removal rate of DBDS and NDM was rapidly increased and 97.5% DBDS and 96.6% NDM removal by Ag-Y was achieved at the dosage of 1.0 wt% at 313.15 K within 2 h which was better than Ce-Y and Cu-Y at dosage of 3.0 wt%. When the dosage of Ce-Y and Cu-Y range in 1.0–3.0 wt%, the desulfurization performance of DBDS and NDM improved rapidly. As further increase of the dosage of Ce-Y and Cu-Y to 5.0 wt%, the desulfurization performance no longer increased significantly.
Kinetic analysis indicates that the adsorption processes on Ag-Y, Ce-Y, and Cu-Y can be well described by pseudo-second-order kinetic models. The activation energies for the adsorption of DBDS and NDM on Ag-Y, Ce-Y, and Cu-Y were greater than 40 kJ mol−1, suggesting the adsorptions of DBDS and NDM were dominated by chemical adsorption.
Footnotes
Acknowledgements
The author(s) are also thankful to Yining Tang and Wei Qu for analyzing the results of characterization.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


