Abstract
In this study, a novel and simple approach to fabricate zirconium dioxide (ZrO2) nanofibers by critical bubble electrospinning and subsequent thermal treatment was reported. The influences of different matrixes, weight percentages, heating rates, post-processing techniques, and with/without carbonization were discussed. The fiber size grew linearly with the increasing amounts of particles added. In addition, ZrOCl2/PAN composite fibers were converted to ZrO2 nanofibers after high-temperature calcination, whose crystal structure was cubic phase of ZrO2 after 1000°C, and the obtained ZrO2 nanofibers shows excellent high-temperature-resistant adsorption and separation properties.
Introduction
Economic development is now in an important transition phase, but heavy pollution caused by traditional high energy consumption has been paid more and more attention, while ceramic fiber synthesis has attracted a great deal of research interest at home and abroad. Compared to common high-temperature-resistant materials, nanofibers that possess well-defined size and large surface area-to-volume ratio are quite popular with a wide range of applications (He et al., 2012; Zhang et al., 2008). Numerous methods such as, drawing (Torquato et al., 2002), template-assisted synthesis (Lin et al., 2002; Liu et al., 2013), self-assembly (Hartgerink et al., 2001), electrospinning (Greiner and Wendorff, 2007; Li et al., 2016), centrifugal spinning (Zhang and Lu, 2014), and so on have been applied to prepare high-temperature-resistant nanofibers for energy materials.
Electrospinning is recognized as a simple and efficient technique for the production of polymer nanofibers (Liu and He, 2018; Yang et al., 2011), but its yield cannot meet the urgent needs of commercial applications and, therefore, bubble-spinning is proposed and some research evolvements have been achieved so far (Chen et al., 2014; Liu and He, 2017; Zhao et al., 2017). Because this method of spinning is to use polymers’ or melts’ broken bubbles or membranes for fabrication of nanomaterials, it can be a good candidate for the mass production of nanofibers (He, 2012). If the bubble wall remains unbroken under critical condition, there will be more polymer jets produced for the one same bubble. Therefore, the parameters are controlled to form a novel and efficient way for achieving abundant nanofibers by continuous unbroken bubbles, which is called critical bubble electrospinning.
Zirconium dioxide (ZrO2) fibers, a kind of one-dimensional (1D) inorganic metal oxides, are arguably one of the most fascinating materials owing to the unique mechanical and high-temperature resistance, which are widely applied in insulation (Lewis, 2013), filtration (Bass et al., 2012; Biswas et al., 2012; Khan et al., 2013; Maximous et al., 2010), enhancement component for ceramics (Kanka, 2011), solar cells (Koo et al., 2011), batteries (Kusachi et al., 2012), and catalysts (Cheung and Gates, 1998). Due to a combination of unique properties such as superior refractoriness, good mechanical strength, high ionic conductivity, low thermal conductivity, high thermal expansion coefficient and excellent corrosion resistance (Ali and Raj, 2010), ZrO2 thin films are in greater demand (Li et al., 2015).
In this paper, ZrO2 nanofibers were successfully fabricated from immiscible organic/inorganic blends by critical bubble electrospinning followed by thermal treatment, during which unwanted constituents were removed and the desired ceramic phase was obtained. The comparison among different matrix as-spun fibers was made to reveal the corresponding characteristics. Moreover, different weight percentages of ZrO2 particles, heating rates, post-processing parameters, and with/without carbonization were considered to observe the distinctions and to optimize parameters. Meanwhile, critical bubble electrospinning can maintain the bubble to be unbroken over a period of time, which provides the best candidate for mass production of nanofibers with good quality. In order to meet the ever-growing requirements of ZrO2 nanofibers, mass production of ZrO2 nanofibers for high-temperature-resistant adsorption and separation could be obtained in a facile and effective way.
Experimental
Materials
Polyvinyl alcohol 1788 (PVA), polyvinyl pyrrolidone k90 (PVP), polyacrylonitrile (PAN) with an average molecular weight of 150,000 g/mol purchased from Shanghai Macklin Biochemical Co., Ltd., zirconia nanoparticles (99.99%) purchased from Beijing HWRK Chem Co., Ltd., and zirconyl chloride octahydrate (ZrOCl2·8H2O, 98% or higher, Acros Organics, USA) were used in the experiments. Ethanol alcohol (C2H5OH, 99%), polyethylene glycol 20000, and N,N-dimethylformamide (DMF, 73.10) were from National Medicine Group Chemical Reagent Co., Ltd. All materials used were without further purification.
Preparation of composite gels
PVP k90 was dissolved in ethyl alcohol (PVA 1788/ethyl alcohol, or PAN/DMF), and ZrO2 particles and PEG 20000 were added to get a homogeneous mixed solution, which was then mixed with 50% ZrOCl2·8H2O/DMF and stirred to get the ultimate suspension for experiments. The percentages of the components are listed in Table 1.
Composites for the prepared samples.

Electrospinning set-up
Schematic illustration of the critical bubble electrospinning set-up is shown in Figure 1, which includes a high voltage generator, a gas pump, a solution reservoir, and a collector. Here, 30 kV was chosen, the solution surface-to-collector distance in the experiment was also altered into a comfortable length of 20 cm. Polymer solution was added into the solution reservoir, which was connected to a gas pump through a hollow metal electrode and positive voltage was applied to the electrode with a high voltage power generator (CPS-40K03VIT, Chungpa EMT Co., Korea). The relative humidity was controlled at 40% and the ambient temperature was about 25°C. During the process, the bubbles produced by air standing on the solution surface can generate multiple jets to produce nanofibers.

Device for critical bubble electrospinning.
After treatment
The polymer–matrix composites obtained were placed in a vacuum oven for 24 h at room temperature to remove the solvent residuals. The as-spun nanofibers were stabilized at 280°C for 2 h in air with a heating rate of 5°C/min, afterward they were carbonized at 800°C for 2 h in argon with a heating rate of 5°C/min. Finally, the ZrO2 and carbon composite nanofibers were converted to ZrO2 nanofibers in air at 1170°C (considering that the tetragonal phase of ZrO2 can form from the monoclinic phase at 1170°C at a maximum temperature limit of 1200°C) for 4 h with a heating rate of 0.5°C/min. They were compared to confirm the optimal matrix nanofibers; besides, different weight percentages of ZrO2 particles, pre-oxidation, and with/without carbonization, different heating rates, and different post-processing methods were handled in comparison to determine the optimal parameters.
Characterization
For scanning electron microscopy (SEM) investigation, a Hitachi-4800S was used. The diameters of nanofibers were calculated by measuring at least 100 fibers randomly using Image J program. X-ray diffraction (XRD) patterns were obtained using a Siemens D5005 Diffractometer with Cu Ka radiation (λ = 0.154 nm) in a 2θ range from 10° to 70°. IR spectra were obtained on Nicolet 5700 FT-IR spectrometer with a resolution of 0.25 cm−1. Simultaneous TGA/DSC Q600 (10°C/min) was used to test the heat stability in air furnace. Brunauer–Emmett–Teller (BET) surface area and pore size were measured by a volumetric sorption analyzer (Gemini VII 2390 Series physiosorption analyzer from Micromeritics coupled with SmartPrep 065 degassing unit).
Results and discussion
The morphology of different as-spun fibers
The SEM images of different polymer–matrix composites, ZrO2 ultra-fine fibers, and diameter distributions of the ZrOCl2/PVP, ZrOCl2/PVA, and ZrOCl2/PAN fibers are shown in Figure 2. The average diameters of the obtained matrix as-spun fibers ranged from 100 nm to 1100 nm. ZrO2 fibers obtained were relatively smaller than their as-spun counterparts because of the shrinkage caused by the removal of polymer. It can be seen that the morphologies and diameters of the composite nanofibers had significant differences among different polymers. As shown in Figure 2(a1), the ZrOCl2/PVP fibers fabricated had smooth surface; however, some burrs were stuck to the edges of the fibers, which may be influenced by improved conductivity. Figure 2(b1) shows that the ZrOCl2/PVA nanofibers packed up and down and were smooth and compact. The morphologies of the ZrOCl2/PAN fibers are presented in Figure 2(c1), and a huge difference can be seen with the appearance of some growth patterns. Comparing the diameter distributions of the three composite nanofibers (Figure 2(a3), (b3) and (c3)), ZrOCl2/PVA nanofibers were the thinnest between 100 and 600 nm. However, certain conglutinations occurred between fiber and fiber in Figure 2(a2) owing to strong hygroscopicity of PVP, and fibers derived from ZrOCl2/PVA nanofibers in Figure 2(b2) were found to be uneven where some were as large as 3000 nm, whereas others were as thin as about 400 nm. In Figure 2(c2), ZrO2 nanofibers achieved from ZrOCl2/PAN nanofibers were relatively homogenous, which can be explained by the fact that the composite fibers containing PAN had good thermal stability (Kim and Yang, 2003; Zussman et al., 2005). Therefore, we chose PAN as the matrix of as-spun fiber to prepare ZrO2 nanofiber.
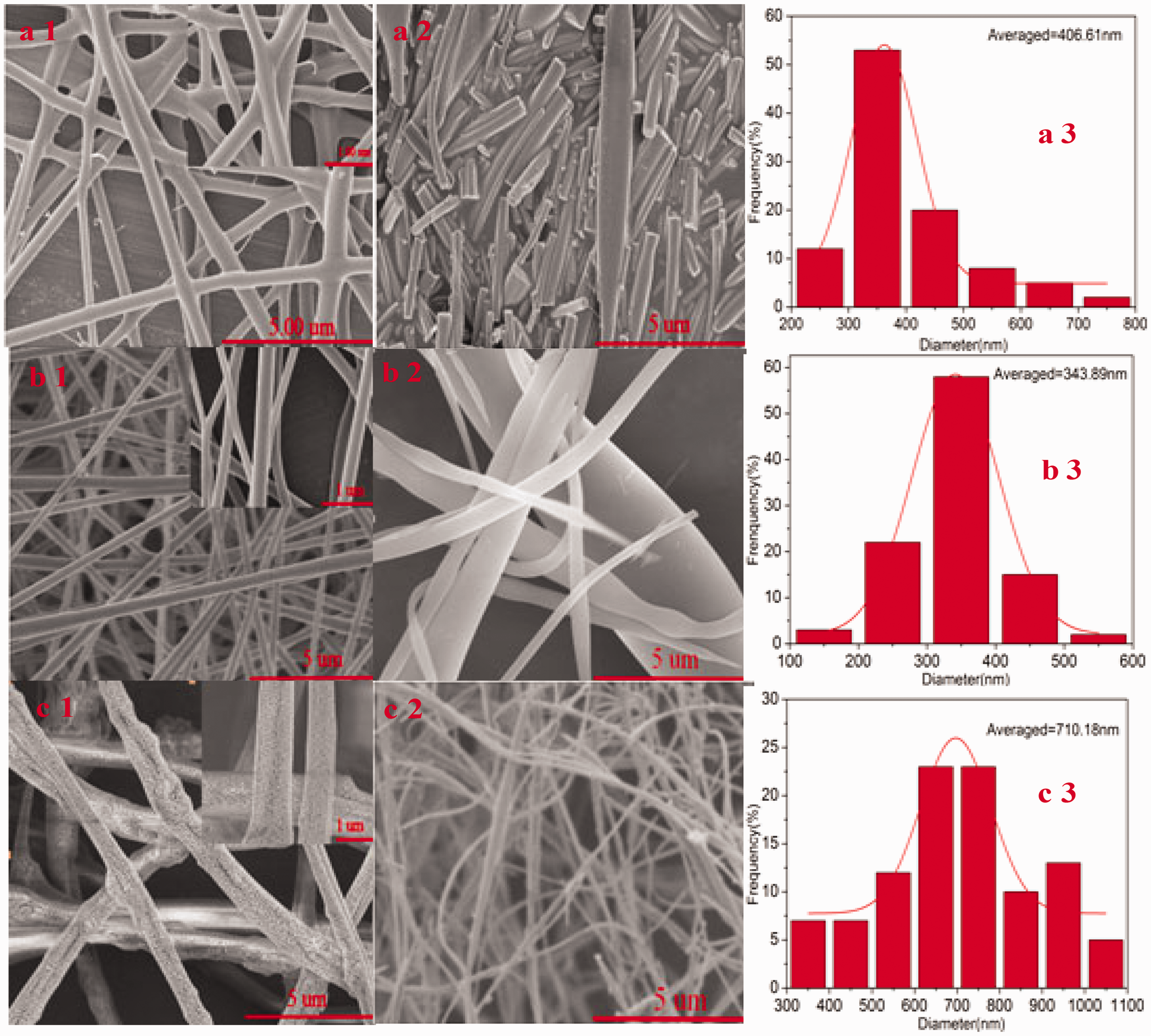
FESEM micrographs of matrix as-spun nanofibers by critical bubble electrospinning: (a1) ZrCl2/PVP; (b1) ZrCl2/PVA; (c1) ZrCl2/PAN and their corresponding ZrO2 nanofibers calcined (b1, b2, b3) and the relevant diameter distributions of composite nanofibers (c1, c2, c3).

Scanning electron micrographs of zirconia fibers after heating at: (a) 800°C; (b) 1000°C; and (c) 1200°C with a heating rate of 5°C/min.
The influence of different parameters on ZrO2 fiber morphology
Figure 3(a) to (c) illustrates that the temperature significantly influenced ZrO2 inorganic nanofibers. The as-spun nanofibers were dealt with in air after stabilization without carbonization, and the morphologies changed as the temperature rose from 800°C to 1000°C and 1200°C with a heating rate of 5°C/min. The fibers became more smooth and continuous; however, if it exceeded 1000°C, fibers began to rupture, which may be because crystalline grains grew with the increasing temperature.
The influence of heating rate on obtaining inorganic nanofibers at 1000°C was shown in Figure 4(a) to (c). The products with the heating rate of 20°C/min and 10°C/min were just membranes with bits of fibers. In contrast, when the rate was 5°C/min, the fibrous shape was very evident. As a result, if the heating rate was lower, the better inorganic ZrO2 fibers after calcination could be obtained.

Scanning electron micrographs of zirconia fibers after heating at 1000°C with the rate of: (a) 20°C/min; (b) 10°C/min; and (c) 5°C/min.
Different post-processing techniques are shown in Figure 5. Figure 5(a) shows that the surface appeared to get uneven without ZrO2 addition, pre-oxidation, or carbonization. After 0.2 g of ZrO2 particles were added into the mixture as stabilizer to avoid the occurrence of phase transition, ZrO2 nanofibers became smooth as shown in Figure 5(b) though some particles adhered to the boundaries of fibers. In contrast, the fibers after stabilization and carbonization as seen in Figure 5(c) but without ZrO2 particle addition were continuous and a little rough. Flat ZrO2 nanofibers were formed as seen in Figure 5(d), while 0.2 g ZrO2 particles were added with stabilization and carbonization. Therefore, the post-processing techniques were important for the fabrication of inorganic nanofibers.

Scanning electron micrographs of ZrO2 fibers at 1000°C with different post-processing techniques: (a) no ZrO2 addition, no stabilization, no carbonization; (b) 0.2 g ZrO2 particles, no stabilization, no carbonization; (c) no ZrO2 addition, stabilization, carbonization (the heating rate was 5°C/min up to 900°C); (d) 0.2 g ZrO2 particles, stabilization and carbonization (the heating rate was 5°C/min up to 900°C).
Referring to mass content of ZrO2 nanoparticles, e.g. 0.05 g, 0.1 g, 0.2 g, and 0.4 g, respectively, the size distributions of ZrO2 nanofibers originating from those conditions are shown in Figure 6. The sizes of nanofibers increased linearly, which might be affected by the mass of particle additions in the suspension because the size of incorporation influenced the fibers directly (Bezemer et al., 2006). Of course, the content of the nanoparticles should not be too big, otherwise the spinning property of the suspension would be destroyed.

ZrO2 nanofiber diameter as a function of ZrO2 particle content in the suspension.
Characterization of ZrO2 ultra-fine fibers
The thermal performance of ZrOCl2/PAN fibers was illustrated by TGA–DTG–DSC in air as shown in Figure 7. It contained three major weight loss regions in the TGA graph. The weight loss region below 240°C could be attributed to the evaporation of the residual solvents and moisture, and the weight loss in the range of 240–380°C was due to the burnout of chlorine, decomposition, and carbonization of polymer, respectively (Sun et al., 2015). Moreover, the weight loss between 380°C and 900°C corresponded to the expulsion of water molecules formed through dehydroxylation of the materials along with the removal of residual carbon via oxidation, and some changes among different crystalline phases might happen at the same time (Sun et al., 2015). There was no significant weight loss above 900°C, and the remaining weight was about 60%, indicating that chlorine was eliminated and polymer was decomposed completely at this temperature. Then the remaining substance had great high-temperature resistance properties.
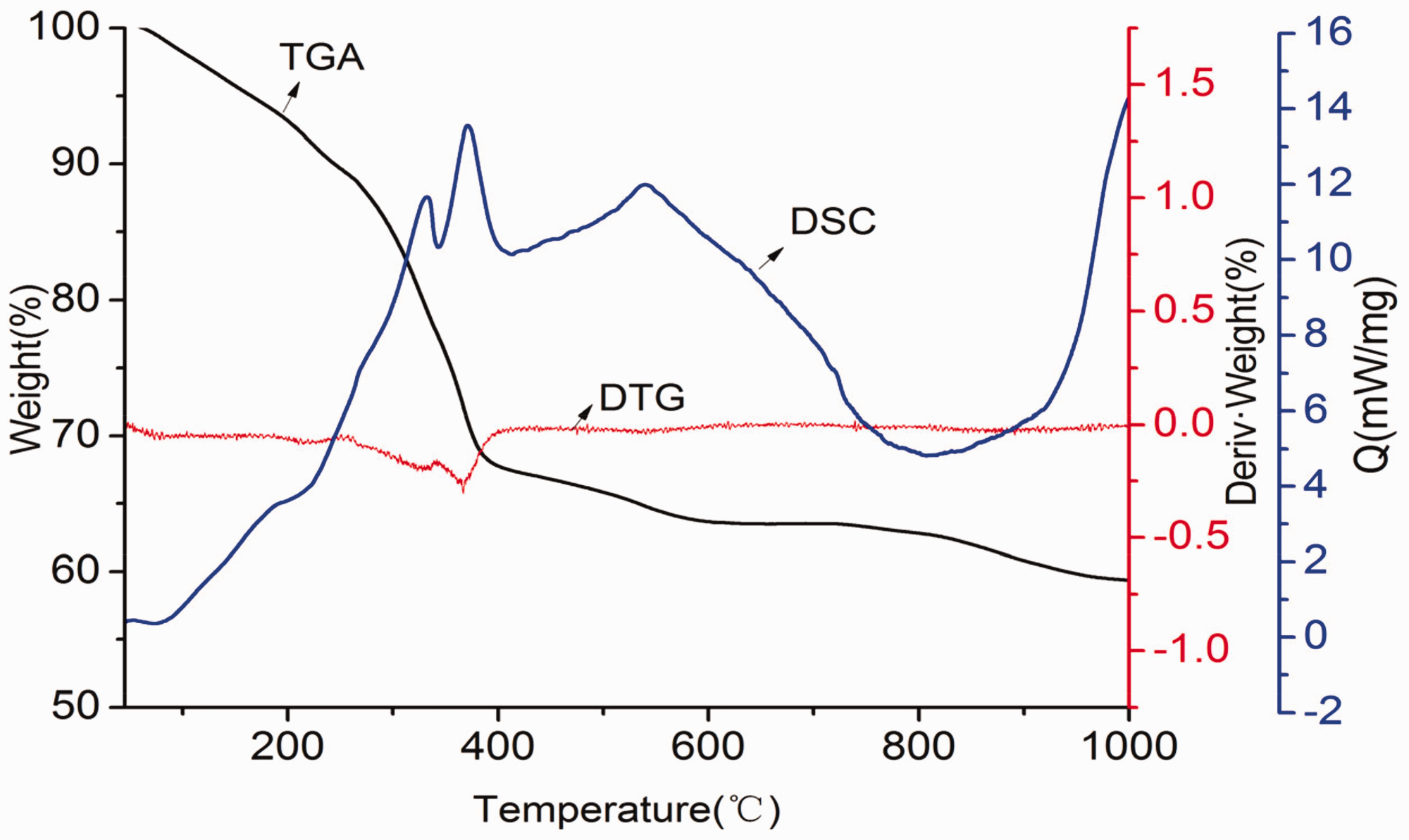
TGA–DTG–DSC curves of ZrOCl2/PAN fibers fabricated by critical bubble electrospinning.
XRD was performed to investigate the crystal structure of the ZrOCl2/PAN fibers at several different calcination temperatures to detect the remaining substance, and the result is shown in Figure 8 to make a simultaneous contrast. It indicated that the diffraction peaks observed for the initial ZrO2 additives were monoclinic phase substance. The patterns had obvious changes after calcination at 1000°C. The crystallization of ZrO2 could be seen after being heated below 1000°C, where the monoclinic phase and the tetragonal phase of ZrO2 mainly existed (Liu et al., 2009; Yogo, 1990; Zussman et al., 2005). When the temperature reached 1000°C, whose diffraction peaks were located at 2θ (°) = 29.9, 34.7, 50.0, 59.7, and 62.5, and 64.0 were corresponding to the planes of (111), (200), (220), (311), and (222) respectively, which agreed with the cubic phase of ZrO2 (Hartridge, 2001; Pullar et al., 1998; Ritzert et al., 1998).

XRD curves of PAN fibers (1) before and (2) after pre-oxidation at 280°C, (3) ZrOCl2/PAN fibers, ZrOCl2/PAN fibers with 0.2 g ZrO2 particles (4) before and (5) after calcination at 400°C, 600°C (6), 800°C (7), 1000°C (8).
In order to examine the influence on the formation of fibers from temperature, IR spectra of the ZrOCl2/PAN nanofibers and what calcined after 400°C, 600°C, 800°C, 1000°C. The results were displayed in Figure 9. The peak of about 3400 cm−1 was assigned to the –OH groups from H2O. Due to the decomposition of PAN and zirconium oxychloride, the peaks at about 2930, 1551, 1422, 1322, 1093 cm−1 corresponded to the stretching of C–H, C–C, C–O, C–N, O–H, respectively. The peak appearing at 654 cm−1, 923 cm−1, and 1448 cm−1 could be assigned to Zr–O–Zr vibrations; meanwhile, the peak of 564 cm−1 Zr–O and the peak of 1630 cm−1 Cl–O became weaker and weaker. In conclusion, the ZrOCl2/PAN fibers changed into ZrO2 nanofibers completely after high-temperature calcination.

FTIR spectra of PAN/ZrOCl2 fibers and fibers treated at different temperatures of 400°C, 600°C, 800°C, 1000°C, respectively.
Surface area and pore size distribution are two important factors that influence the separation performance of nanofibers. Figure 10 illustrates the nitrogen adsorption–desorption isotherms and the pore size distribution of the ZrO2 fibers treated after 1000°C, respectively. In the adsorption–desorption isotherms, the relative lower pressure region (< 0.2P/Po) was corresponded to micropores. In contrast, the additional adsorption at high-relative-pressure range (>0.8P/Po) showing a hysteresis loop was associated with mesopore adsorption (Kim et al., 2007). The isotherm of the ZrO2 fibers can be classified as IV-type, which is a characteristic feature of micropores and mesopores mostly ranging from 1 to 50 nm as seen from Figure 10(b). The table embedded reported the surface parameters, including specific BET surface area and total pore volume, showing the largest specific surface area of 268.423 cm2/g with a total pore volume of 0.146 cm3/g. Therefore, combining with the high-temperature-resistant property, this porous structure with high specific surface area can play an important role as a strong high-temperature-resistant separation.
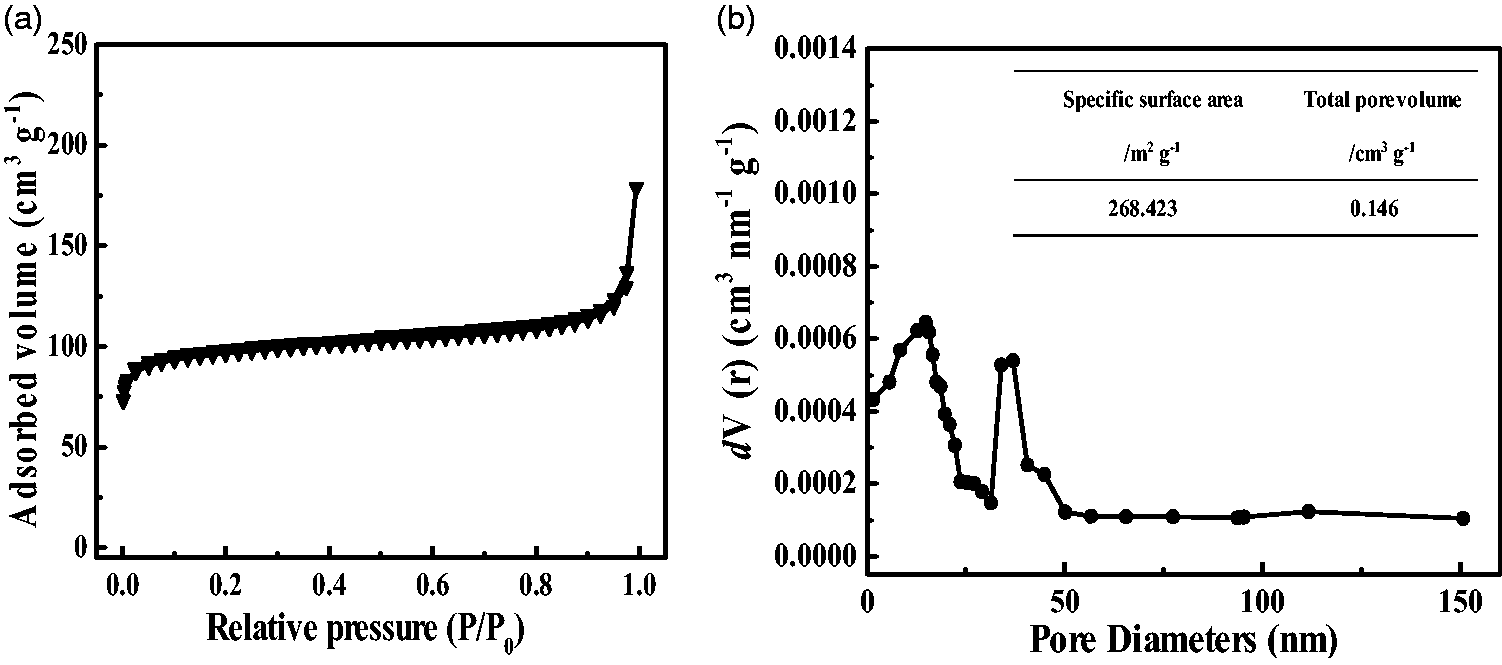
(a) Nitrogen sorption isotherms and (b) pore-size distributions of the ZrO2 fibers treated after 1000°C calculated using the BJH method. The insets are the related specific surface area and total pore volume.
Conclusions
In this paper, we successfully fabricated ZrO2 nanofibers by the critical bubble electrospinning method and heat treatment. Their diameters could be easily controlled by various parameters, and fiber’s morphology could be adjusted from smooth surfaces to unsmooth ones as well, then they can be applied to different fields according to the demands. In summary, PAN is confirmed to have the advantages of thermostability to make inorganic nanofibers during the process. At 1000°C, the smaller heating rate, some ZrO2 additions, stabilization, and carbonization conducted at the same time can avoid phase transition to help the obtained ZrO2 nanofibers to be more smooth and continuous. The increasing ZrO2 concentration makes the nanofiber diameter to grow linearly. ZrOCl2/PAN fibers change into ZrO2 nanofibers after high-temperature calcination and the crystal structure of the composite fibers after the calcination temperature of 1000°C is the cubic phase of ZrO2. Besides, ZrO2 nanofibers are characteristic with micropores and mesopores mostly ranging from 1 to 50 nm and they have the largest specific surface area of 268.423 cm2/g with a total pore volume of 0.146 cm3/g. The results above prove that the critical bubble electrospinning method is simple to fabricate intact ZrO2 ultrafine fibers, which is found to be promising for high-temperature-resistant adsorption and separation. The critical bubble electrospinning can also be used to control macromolecule orientation in nanofibers (Tian, He & He, 2018; Tian & He, 2018; Tian, Li & He, 2018).
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Excellent Ph.D. Program of Zhejiang Sci-Tech University (Grant No. 11110231281803), Research and Innovation Project for College Graduates of Jiangsu Province (Grant No. KYLX16_0135), and it has been sponsored by China Scholarship Council (Grant CSC No. 201606920066).
