Abstract
This paper describes a silk fibroin-based nanofibrous membranes loaded with antitumor drugs curcumin and 5-fluorouracil using electrospinning technique. The concentration of curcumin/5-fluorouracil in the silk fibroin solution for electrospinning was optimized to be 0.15/0.25, 0.3/0.5, and 0.45/0.75 wt%. The morphology, hydrophilic property, pore size, secondary structure, and antitumor drugs release of nanofibrous membranes were measured. The diameter of nanofibrous membranes ranged about 100–200 nm. The results indicated that electrospinning process did not influence the secondary structure and the drug content of antitumor drugs, and dual drugs encapsulated in the silk fibroin nanofibrous membranes were released in a steady and consistent process. In conclusion, the silk fibroin-based drug-loaded membranes can be useful as biomaterials with antitumor function.
Introduction
Colorectal cancer (CRC) is one of the most common malignant tumors in the word, because of its higher incidence and mortality rate. According to the reports from World Health Organization, the number of newly diagnosed cases of CRC each year nearly equals the number of annual mortality (Jordan et al., 2003; Angelis PMD et al., 2006). In recent decades, combination of stent placement and drug delivery has been reported to treat the advanced CRC. In particular, 5-fluorouracil (5-FU) has been reported as incorporated into a polydioxanone stent for the treatment of patients with CRC (Jordan et al., 2003; M P et al., 2006). But the shortcoming of 5-FU are short half-life (1020 min), low utilization rate (10–30%), and toxic effects on normal cells (Akbulut et al., 2004; He et al., 2014). Curcumin (CUR) is a significant activator inhibitor of NF-kappaB (NF-κB), and it was reported to inhibit proliferation of tumor cell and induce apoptosis of tumor after coordinating with 5-FU (Patel et al., 2008; Sharma et al., 2006; Shishodia et al., 2003). The transcription factor NF-κB is up-regulated in many cancer cells where it contributes to development of the pro-survival, antiapoptotic state (Shishodia et al., 2004; Weber et al., 2006). In fact, recent studies indicated the significant additive antitumor effects of 5-FU combined with CUR in vitro (Weber et al., 2005). Consequently, the purpose of this study was to develop a novel antitumor drugs release system into which 5-FU and CUR were incorporated and function simultaneously. Our hypothesis is that the combinational use of the two drugs may not only improve the therapeutic, targeting, and antitumor effect of the device, but also decrease toxicity of 5-FU. Hence, it is significant to incorporate antitumor drugs into a biomaterial through surgically implant in order to obtain the local sustained release properties of antitumor drugs.
Silk fibroin (SF) has good biocompatibility, biodegradability, and the unique mechanical properties, as biomaterials have been widely used in drug delivery and biomedical field (Jin et al., 2004; Wenk et al., 2011). Silk-based biomaterials display unique physicochemical and biological properties, while the spinning process changes the microstructure and affects its water solubility and mechanical performance. The SF nanofibers have high specific surface area and porosity, which is conducive to cell adhesion and drug loading (Keten et al., 2010; Liu et al., 2018; Numata et al., 2012; Tao et al., 2017; Wu et al., 2018; Liu et al., 2018).
Tissue engineering material is the current research hotspot, especially SF nanofibrous membranes made by electrospinning technology are expected to have wide applications in drug delivery systems (DDSs). SF nanofibers produced by electrospinning technology can be directly processed into membranes (Xie et al., 2018), biomedical tubular scaffolds, and layered coating on the surface of materials (Hu et al., 2015; Li et al., 2013). After implantation of SF nanofibrous membranes, it can achieve antitumor drugs release function locally. Furthermore, the diameter of the fiber and the percentage of drug can be easily adjusted by adjusting the process parameters, in order to meet the requirements of DDSs.
In the present work, CUR/5-FU was loaded into the SF nanofibrous membranes using electrospinning technique. The morphology, hydrophilic test, pore size distribution, secondary structure, and drug release properties of nanofibrous membranes were examined.
Materials and methodologies
Materials
Mulberry silk was purchased from Shengzhou Xiehe silk Co., Ltd, China. Anticancer active ingredients—5-FU and CUR—were obtained from Sigma USA. Poly(ethylene glycol) (PEG) was purchased from Dow Chemical Company, USA. Other reagents and solvents were from Thermo Fisher Scientific.
Preparation of electrospun SF/drugs nanofibrous membranes
The SF solution was prepared following in accordance with degumming of silk, dissolved silk by lithium bromide, dialysis to remove impurity ions, centrifugation to remove solid precipitation, and determination of SF solution concentration (Wei et al., 2011; Zhou et al., 2013). The SF solution obtained (∼7 wt%) was concentrated to 20, 23, and 26 wt% placing in a dialysis bag (M.W. cutoff: 3500 Da) immersed in a bath containing 15 wt% PEG solution (molecular weight: 20,000 Da). Three different drug membranes were prepared comprising CUR at 0.15, 0.3, and 0.45 wt% and 5-FU at 0.25, 0.5, and 0.75 wt% to SF by weight, respectively. The above dual drugs were dissolved into 50, 60, and 70 wt% PEG-400 using ultrasonic cell disruptor (JY92-IIDN, China) at a certain rate (40 wt%) for 30 min for accelerating dissolution. Then, spinning solution consisting of drugs and SF was added into the plastic syringe with a stainless steel needle (inner diameter is 0.5 mm), and the plastic syringe was placed into the high-speed rotating electrospinning machine (NEC-10, Kato Tech Co., Ltd, Japan). The spinning voltages were 20, 22, and 24 kV; the distances between the tip of needle and the tip–target distance were 16, 18, and 20 cm; the injection speeds of spinning solution were designed as 0.2, 0.4, and 0.6 ml h−1, respectively. All the samples were vacuum dried at 60°C for 12 h in order to remove solvents before testing.
Characterization of electrospun SF/drugs nanofibrous membranes
Microscopic observations
The surface morphology of the membranes was examined using a scanning electron microscopy (SEM) (Hitachi S-4800, Tokyo, Japan). All membranes were sprayed with gold prior to imaging and then observed at a voltage of 3 kV. The fibers in membranes were imaged by using an inverted microscope (Olympus phase contrast microscope TH4 200, Osaka, Japan).
Hydrophilic test
In order to investigate the hydrophilicity of membranes, the apparent contact angle was examined by Krüss DSA 100 (Krüss Company, Germany) using sessile drop (5 µl drop of pure water) methods. The apparent static contact angle values were produced by ImageJ software (National Institute of Mental Health, USA).
Pore size distribution
Pore size distribution of membranes was examined by a capillary flow porometry (Porometer 3G, Quanta chrome Instruments, USA). All tested membranes were made into circular shape with 15 µm thickness and 25 mm of diameter. During this process, the Porometer 3G was applied by the approach of squeezing out the wetting liquid (Porofil) through the pores in samples. Air pressure was automatically placed on the other side of membranes, during which air flow through the pores was accurately recorded. Independent devices ensure the highest quality accuracy of data.
Structural characterization
The secondary structure of drug-loaded membranes was examined by using Fourier Transform Infrared Spectroscopy (FT-IR) with a MIRacle™ attenuated total reflection Ge crystal cell in the reflection mode (Nicolet 5700, Thermo Electron Corp, Waltham, MA). FT-IR wave numbers were set from 4000 to 400 cm−1 during 32 scans, with 2 cm−1 resolution. The internal crystalline structure of drug-loaded membranes was examined via an X-ray diffractometer (X’Pert-Pro MPD, PANalytical BV, Almelo, Netherlands) using a Cu Kα radiation source. Two dimensional X-ray diffraction patterns were obtained at the following irradiation settings: Cu, Kα, wavelength 0.154 nm, range 5–60°, 40 kV, and 35 mA at a scanning rate of 5° min−1.
Antitumor drugs release
The antitumor drug-loaded membranes were prepared by steam treatment for 6 h and then transferred into sealed centrifuge tubes containing 2 ml phosphate-buffered saline (PBS), pH = 7.4 ± 0.2, at room temperature. The drug release medium solution was rocked by shaking water bath at 100 cycles min−1. One milliliter released solution of samples was collected from soaking solution, meanwhile, 1 ml new PBS added into the original solution. Microplate Reader (Bio-TeK synergy H1, USA) measured the drug release property of CUR/5-FU, then CUR was sensitively tested by instrument at a wavelength of 425 nm, while 5-FU was measured at a wavelength of 265 nm.
Statistical analysis
Data are presented as mean ± SEM (n = 5–10), and all data sets were expressed in terms of their mean and standard deviation. Statistical analysis was performed using a one-way analysis of variance. P-values < 0.05 were considered significant.
Results and discussion
Morphology of electrospun SF/drugs nanofibrous membranes
The SEM of SF/drugs nanofibrous membranes with different concentrations is shown in Figure 1. The experimental results indicated that the nanofiber diameters of membranes increased with the concentrations of SF. For example, the concentrations of SF increased from 20 to 26 wt% when the concentration of PEG-400 was 70 wt%, the diameter of fibers with uniform distribution increased from 476 ± 49 to 736 ± 63 nm. Meanwhile, the results show that reducing the concentrations of SF to below 20 wt% can cause the unevenness of drafting in the process of electrospinning, as shown in Figure 1(g) and (h). The addition of PEG-400 is propitious to eliminate the string of beads on SF nanofibers electrospun from low-viscosity solution. With the concentrations of PEG-400 increased from 50 to 60 wt% when the concentration of SF was 23 wt%, the diameter of fibers reduced from 216 ± 17 to 172 ± 15 nm, as shown in Figure 1(d) and (e). But as the concentrations of PEG-400 increased continuously beyond 70%, the diameter of nanofibers turned to be thicker and appeared as beaded structure, as shown in Figure 1(c), (f), and (i). In summary, nanofibers with the concentrations of SF/PEG (23/60 wt%) were uniform in size and thickness, and the surface of nanofibrous membranes was dense and smooth.

SEM images of SF/drugs nanofibrous membranes with different concentrations: (a)–(i) represents 26/50, 26/60, 26/70, 23/50, 23/60, 23/70, 20/50, 20/60, and 20/70 wt%, respectively.
In this study, spinning voltage, spinning speed, and spinning distance have a major impact on nanofibers, and the spinning solution consisting of SF/PEG (23/60 wt%) was tested (as given in Table 1). SEM images of nanofibrous membranes with different processing parameters are shown in Figure 2. The experimental results indicated that the diameters of the nanofibers decreased first with the spinning voltage and distance and then increased rapidly with spinning speed. The relationship between diameter and processing parameters of SF/PEG nanofibers is given in Table 1. In summary, the optimum process parameters were obtained by orthogonal experimental design. For example, spinning voltage was 24 kV, spinning distance was 16 cm, and spinning speed was 0.2 ml h−1. The technique parameters can be applied in the process of preparing SF/PEG nanofibrous membranes loaded with different concentrations of CUR/5-FU next.

SEM images of nanofibrous membranes with different processing parameters as given in Table 1.
The electrospun SF/PEG nanofibrous membranes with different processing parameters.
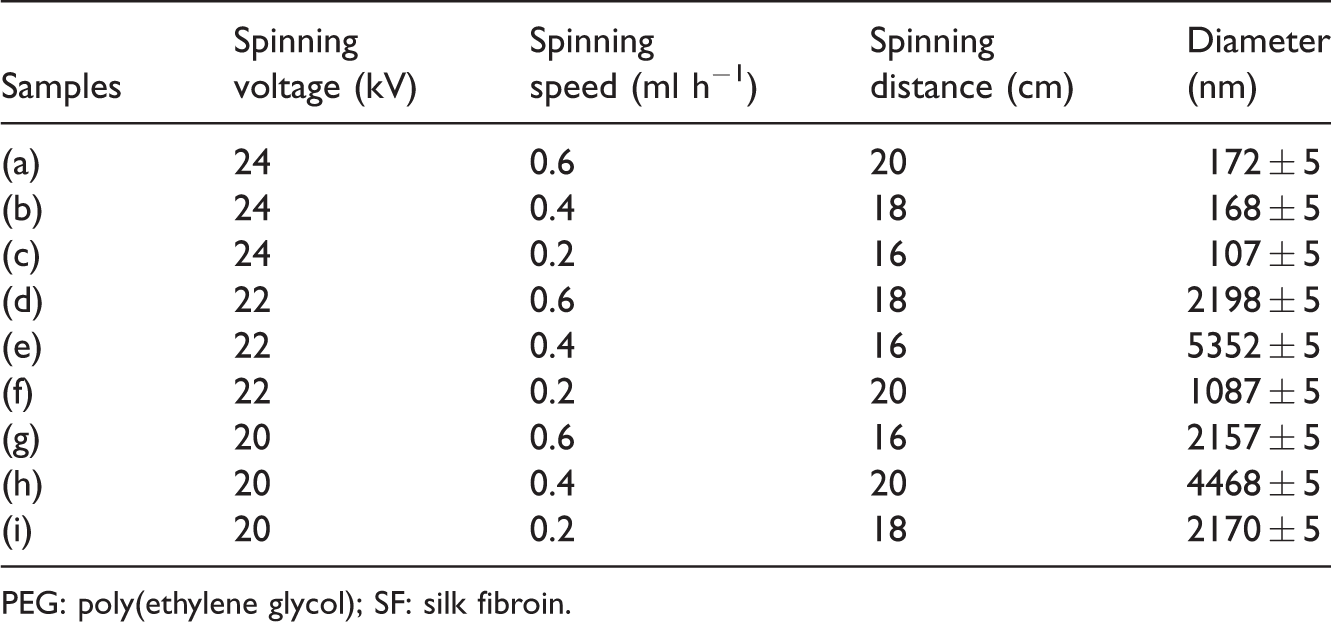
PEG: poly(ethylene glycol); SF: silk fibroin.
The surface morphology of electrospun SF/PEG nanofibrous membranes loaded with different concentrations of CUR/5-FU was observed by optical microscopy and SEM (Figure 3). The diameter of SF/PEG nanofibers was 113 ± 15 nm, and few beads and fiber bonds were found. Moreover, the electrospinning process is smooth and stable. This is because the adding of PEG-400 increased the viscosity of solutions which is beneficial for the formation of uniform structure of nanofibers in the membranes. With the addition of CUR/5-FU (0.15/0.25 wt%), the diameter of nanofibrous membranes increased to 137 ± 15 nm. As the concentrations of CUR/5-FU increased from 0.3/0.5 to 0.45/0.75 wt%, the diameter of fibers with uniform distribution increased from 146 ± 15 to 214 ± 15 nm. As the concentrations of CUR/5-FU increased, the diameter of nanofibers became thicker and appeared as beaded structure. The reason is that the high loading drugs did not disperse fully within the electrospinning solutions; the insoluble drugs decreased the surface tension.

SEM images of electrospun SF/PEG nanofibrous membranes loaded with different concentrations of CUR/5-FU: (a)–(d) represents membranes of plain SF/PEG, CUR/5-FU at 0.15/0.25, 0.3/0.5, and 0.45/0.75 wt%, respectively.
Hydrophilic test
The hydrophilicity of drug membranes is important because the DDS often come into contact with various biological fluids during the treatment process. As shown in Figure 4, SF/PEG nanofibrous membranes showed a hydrophobic surface with a contact angle of 63.9 ± 0.2° at the beginning. As the concentrations of CUR/5-FU increased continuously, the contact angle is gradually decreasing from 137.8 ± 0.9° to 147.9 ± 0.7°. SF/PEG nanofibrous membranes loaded with CUR/5-FU showed a hydrophobic surface, which is significantly higher than SF/PEG nanofibrous membranes, indicating the adding of CUR/5-FU influenced the hydrophilicity of drug membranes. This is also consistent with the findings in previous studies.

Images of water contact angle of electrospun SF/PEG nanofibrous membranes loaded with different concentrations of CUR/5-FU: (a)–(d) represents membranes of plain SF/PEG, CUR/5-FU at 0.15/0.25, 0.3/0.5, and 0.45/0.75 wt%, respectively.
Pore size distribution
Pore size distributions of the electrospun drug-loaded nanofibrous membranes were examined by a capillary flow porometry, as shown in Figure 5. The experimental results indicated that the pore size of SF/PEG nanofibrous membranes was smallest, and the pore distribution was more uniform. With the addition of CUR/5-FU (0.15/0.25 wt%), the pore size of the membranes increased from 23.1 ± 0.5 to 24.2 ± 0.5 µm. When the drug loadings increased to 0.45/0.75 wt%, the pore size of the membranes increased to 42.7 ± 0.5 µm, and the uneven fiber structure, bonds, and beads were found in the membranes. Hence, there is a significant relationship between fiber diameter and pore size distribution, which is consistent with the findings of previous studies.
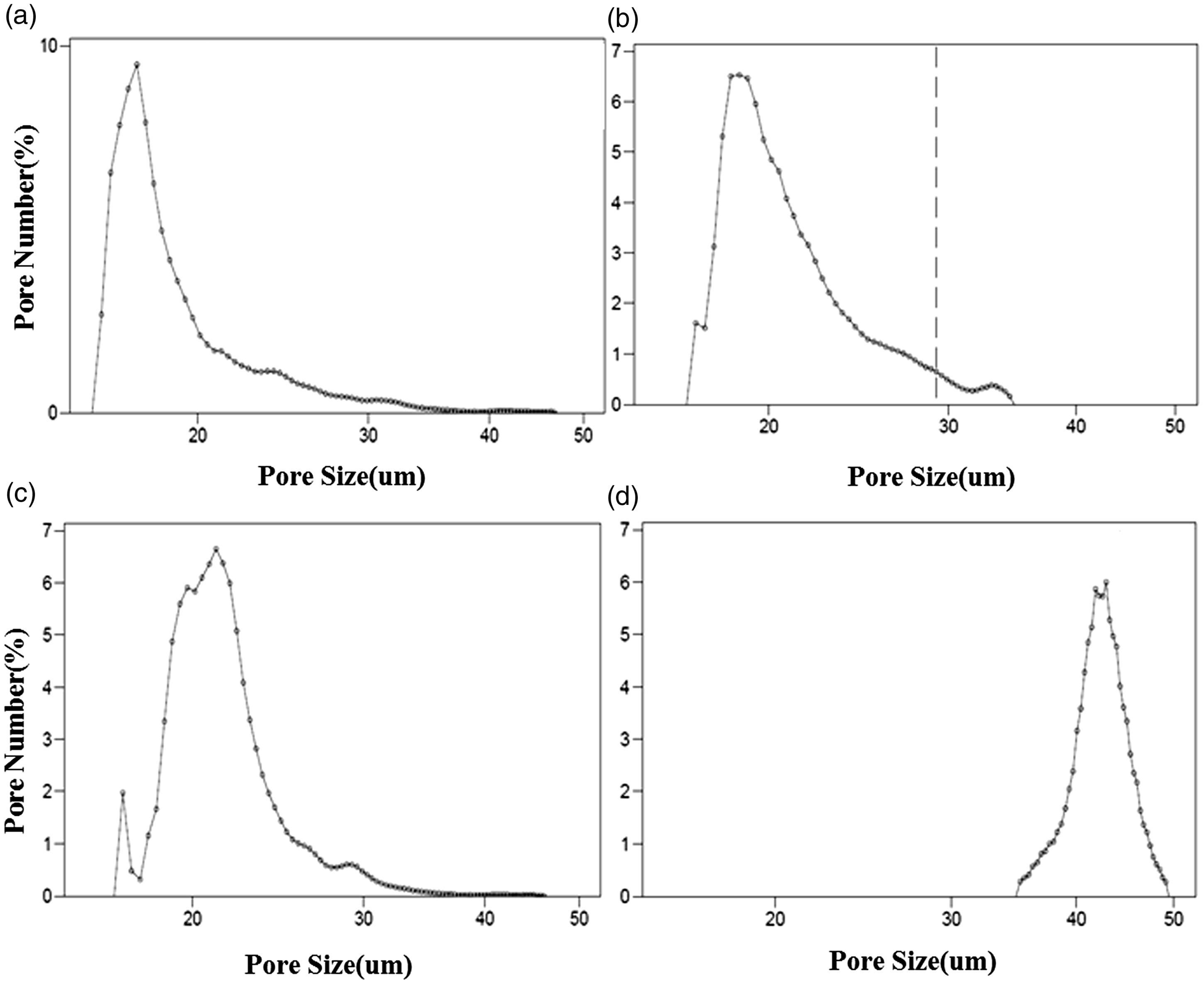
Pore size distribution of electrospun SF/PEG nanofibrous membranes loaded with different concentrations of CUR/5-FU: (a)–(d) represents membranes of plain SF/PEG, CUR/5-FU at 0.15/0.25, 0.3/0.5, and 0.45/0.75 wt%, respectively.
Secondary structures of electrospun SF/PEG nanofibrous membranes
According to the reports, the characteristic peaks for silk I structure were found around 1652 cm−1 (Amide I), 1543 cm−1 (Amide II), 1242 cm−1 (Amide III), and 669 cm−1 (Amide V), and silk II peaks around 1626 cm−1 (Amide I), 1532 cm−1 (Amide II), 1236 cm−1 (Amide III), and 696 cm−1 (Amide V) (Tsukada et al., 2010; Um et al., 2001). As shown in Figure 6(a), a major peak at 1626 cm−1 in the Amide I region accompanied with β-sheet structure formation was found in silk/PEG nanofibrous membranes (a). PEG did not appear as characteristic peaks in the Amide I and II band region. By contrast of the curves (b–d) of 0.15/0.25, 0.3/0.5, and 0.45/0.75 wt% of CUR/5-FU-loaded membranes, a major peak at 1652 cm−1 in the Amide I region accompanied with random coil structure was found. It may be because that the adding of CUR/5-FU can create an effect on the structure and vibration of the original characteristic peaks. It is evident that the dual drugs were loaded into the membranes and their chemical structure of drugs was integrated.

FT-IR spectra (a) and WAXD pattern (b) of electrospun SF/PEG nanofibrous membranes loaded with different concentrations of CUR/5-FU: (a)–(d) represents membranes of plain SF/PEG, CUR/5-FU at 0.15/0.25, 0.3/0.5, and 0.45/0.75 wt%, respectively.
Based on previous literature, the characteristic peaks for α-helix structure were located at 12.1°, 19.8°, 24.1°, 28.5°, and 33.3°, and β-sheet structure located at 9.1°, 18.8°, and 20.5° (Wang et al., 2012). As shown in Figure 6(b), few additional peaks were observed among all nanofibrous membranes—only the intensities were changed. In summary, dual drugs have no obvious influence on crystallization and the secondary structure of SF materials.
Dual drugs release properties
The antitumor drugs release percentage of 0.3/0.5 wt% of CUR/5-FU-loaded membranes is shown in Figure 7. The drug-loaded membranes showed a burst release behavior within 72 h and then remained stable during 210–400 h. The dual drugs of CUR/5-FU-loaded membranes could fully release up to 400 h. The residual dual drugs kept inside of the fibers in the membranes would be sustainably released during the degradation processes. In summary, antitumor drugs encapsulated in the SF/drugs nanofibrous membranes can release in a steady and sustained manner.

In vitro drug release percentage of 0.3/0.5 wt% of CUR/5-FU-loaded membranes; data presented are the mean (±) standard deviation of at least four measurements (n = 5).
Conclusions
In this work, SF/PEG nanofibrous membranes loaded with CUR/5-FU and attached to the surface of CRC stents were developed using electrospinning technique. As a result, CUR and 5-FU were loaded in SF/PEG nanofibrous membranes with a set of concentrations. The morphology, hydrophilic test, pore size distribution, and secondary structure of nanofibrous membranes were examined. In summary, nanofibers with the concentrations of SF/PEG (23/60 wt%) produced by the optimum technique parameters were uniform in size and thickness, and the surface of nanofibrous membranes was dense and smooth. Meanwhile, dual drugs have no obvious influence on crystallization and the secondary structure of SF materials. The adding of CUR/5-FU influenced the hydrophilicity and the pore size distribution of drug membranes, and the released amount of double drugs raised when the drug loading percentage increased. The experimental result of drug release properties in vitro performed a steady and consistent process and sustained for 400 h, thus prospective application for the treatment of CRC in the future.
Footnotes
Acknowledgments
The authors would like to thank the support of China Postdoctoral Science Foundation, University Science Research Project of Jiangsu Province (16KJB540003) and Science and Technology Project of Suzhou (SYG201638 and SYG201849), and projects with code 2018194 and 2018054.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (51603140 and 81603457), Natural Science Foundation of Jiangsu Province (BK20150372), and “National Key R&D Program of China (No. 2017YFC1308800).”
