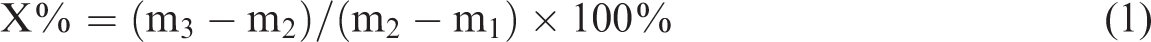Abstract
In this study, 4A zeolite was prepared from opal waste rock by hydrothermal method and applied in ammonium ion adsorption. To optimize synthesis conditions, the effect of crystallization time (1–8 h), crystallization temperature (65–115°C), Na2O/SiO2 (0.6–2.0), H2O/Na2O (20–70), and SiO2/Al2O3 (1.0–3.5) was investigated. X-ray diffraction, scanning electron microscope imaging, cation exchange capacity, static water adsorption, Fourier transform infrared spectroscopy, and N2 adsorption–desorption isotherm were used for assessing properties of 4A zeolite. Adsorption experiments were performed by 1.0 g l−1 4A zeolite with NH4+ solution (5–300 mg l−1) for 4 h at room temperature. The experiment results revealed with a crystallization time of 3 h, a crystallization temperature of 85°C, Na2O/SiO2=1.0, H2O/Na2O = 40, and SiO2/Al2O3=2.0, the 4A zeolite synthesized had excellent performance with cation exchange capacity of 2.93 mmol (g dry zeolite)−1 and static water adsorption of 22.3%. The adsorption process was described by Freundlich model (R2>0.99) and the maximum adsorption capacity could reach to 53.11 mg g−1. The experimental results provided a novel approach for the utilization of opal waste rock, which is produced during the mining of opal-rich palygorskite, and for the synthesis of 4A zeolite and the removal of ammonium ion.
Introduction
The increase of ammonium ion pollution in waters has drawn extensive concern owing to its harmful effects, such as the eutrophication of lakes and rivers, low dissolved oxygen, harm for aquatic life, and the corrosion acceleration of soil materials (Cheng et al., 2017; Huang et al., 2015). Ammonium ion in wastewater is mainly released from municipal, agricultural, or industrial effluents, as well as the utilization of fertilizers, including NOx, NH4+, NO3−, and nitrogen containing organics (Liu et al., 2013a; Malekian et al., 2011). Therefore, the dispose of ammonium ion pollution in water counts a great deal. Conventional removal methods for ammonium ion from wastewater comprise steam stripping, adsorption, ion exchange, and biological nitrification–denitrification (Sun et al., 2017a; Zadinelo et al., 2015). Adsorption has achieved wide-ranging attention for its simplicity and high efficiency for elimination of ammonium ion (He et al., 2016). Amongst adsorption materials for NH4+ ions of aqueous solutions and wastewater, zeolite is especially promising because of the considerable ions exchange capacity and intense affinity for NH4+ compared with other adsorbents (Mazloomi and Jalali, 2016).
4A zeolite, whose ideal chemical structure formula is Na12[(AlO2)12(SiO2)12]·27H2O, is a type of crystalline aluminium-containing silicate with homogeneous microchannels (García et al., 2015; Shen et al., 2017). Negative charges are formed via the replacement of Si with Al in the tetrahedral framework and are compensated by Na+ ions (Ojuva et al., 2015a). As is well known, 4A zeolite has extensive use in fields of chemical industry, environmental conservation, metallurgy, medicine, and so on because of its excellent performance as ion exchangers, adsorbents, and catalysts (Ayele et al., 2015; Müller et al., 2015). The synthesis of 4A zeolite conforming to industry standard of China based on inexpensive and widely available alumina-silica materials under mild condition is of great importance for industrial applications.
Many materials have been explored to synthesize 4A zeolite, including kaolin, coal gangue, fly ash, and palygorskite. (Aldahri et al., 2016; Ayele et al., 2016; Cardoso et al., 2015; Jiang et al., 2014; Maia et al., 2015; Qian and Li, 2015; Tang et al., 2017; Wu et al., 2017; Zhou et al., 2013). A report by Aldahri et al. (2016) put forward that fly ash could be utilized to synthesize Na–P zeolite by using microwave-assisted hydrothermal treatment. Additionally, Zhou et al. (2013) synthesized ZSM-5 zeolite by palygorskite and found that the product has excellent catalytic performance. Sun et al. (2017b) also synthesized 4A zeolite from palygorskite at crystallization temperature of 90°C for 12 h. And in this paper, 4A zeolite was synthesized at 85°C for 3 h with opal as the raw material. A lens body and strip of opal waste rock (OWR) commonly existed in palygorskite mines, with approximately 100,000 t of OWR produced during the palygorskite mine exploitation only in Xuyi County every year (Chen et al., 2003). A previous study showed that the main component of OWR in a palygorskite mine is opal-CT, which can react with alkaline easily (Wilson, 2014). Moreover, a small quantity of palygorskite and dolomite along with scarcely any quartz or illite existed in the OWR. Based on the advantages above, high quality 4A zeolite was expected to be synthesized from OWR.
The aim of this work was to synthesize 4A zeolite with OWR as raw material and to determine the optimum synthesis parameters. The resulting 4A zeolite was then applied to investigate its capacity to remove ammonium ion. As it is known, a study about the synthesis of 4A zeolite from OWR has not been reported. This work provides a new material for synthesis of 4A zeolite and a new method for the removal of ammonium ion. Importantly, a novel method to utilize OWR producing during palygorskite mine exploitation was achieved.
Experimental
Material preparation
4A zeolite was synthesized based on hydrothermal method by using OWR of palygorskite mine from Xuyi County, Jiangsu Province, China. Natural OWR underwent extrusion, crushing, and screening to obtain OWR particles of 200 mesh (0.075 mm). OWR powder was added to 20% hydrochloric acid solution under 70°C with medium-speed stirring for 4 h to eliminate impurities, such as palygorskite and dolomite. The mixed solution then was centrifuged and dried (110°C), resulting in purified OWR samples for further investigation. Based on given molar ratio of SiO2/Al2O3, Na2O/SiO2, and H2O/Na2O, OWR sample (3 g) after acid leaching, NaAlO2, NaOH, and deionized water were blended. The admixture was transferred into a flip mixer and heated to the desired temperature. After the reaction finished, the admixture was washed, centrifuged, and dried overnight to get final sample for further tests.
Material characterization
The chemical composition of OWR sample before and after acid leaching was analysed on Shimadzu XRF-1800 instrument by Rh radiation. The mineralogy of the material and products was examined via X-ray diffraction (XRD) patterns by Dandonghaoyuan 2700 diffractometer with scan range of 3–70° and scan speed of 4°/min. Scanning electron microscope (SEM) imaging was performed on a JSM-6490LV instrument. Fourier transform infrared spectroscopy (FTIR) spectra of the product were obtained by using a spectrophotometer (VERTEX70, Japan) based on transmission technique under normal temperature and pressure with digital detector. And the measuring range was from 400 to 4000 cm−1 with KBr and samples powder mixed at a mass ratio of 100:1. The N2 adsorption–desorption isotherm and pore size distribution analyses of 4A zeolite were carried by Novawin 3000e Surface Area and Pore size Analyzer.
Material testing
Static water adsorption (SWA)
SWA was measured according to the procedure in the National Standard of GB/6287-86 of China; 1.5 g of synthetic product after annealing at 550°C for 1 h was used to adsorb water at 35 ± 1°C for 24 h. All weights were surveyed by electronic balance with the precision of 0.1 mg. SWA can be calculated using the following formula
Cation exchange capacity (CEC)
CEC was judged according to the procedure in the National Light Industry Standard of QB 1768-2003 of China. By adding little bit of HCl or NaOH, CaCl2 solutions with original concentration of 5 mmol l−1 regulated pH to 10. The aforementioned solution with 1.0 g l−1 synthetic product was shaken under vigorous stirring conditions for 20 min at 35 ± 1°C. After the adsorption process and after centrifugation, the supernatant was assessed by an atomic absorption spectrometer (Wayee WYS 2200, China). Next, the CEC was calculated from the residual Ca2+ concentration compared with original Ca2+ concentration. The CEC was tabbed with mmol (g dry zeolite)−1.
Adsorption experiments
Series of experiments were proceeded with three materials and OWR of 1.0 g l−1 with NH4+ solution of specified concentrations (5, 10, 20, 50, 100, 150, 200, and 300 mg l−1). The NH4+ solution pH was adjusted to 7–8 by tiny amounts of NaOH and HCl of different concentration and then mixture was placed on flip mixer for specific time at room temperature. After sorption experiments, the mixture was centrifuged for 5 min by centrifuge with the speed of 3500 r min−1. The concentration of NH4+ in liquid supernatant was determined by spectrophotometry. The removal degree of NH4+ was figured out from the gap between original concentration and remnant concentration of NH4+. The removal capacity (Qe, mg g−1) could be calculated using following formula
Results and discussion
Characterization of OWR
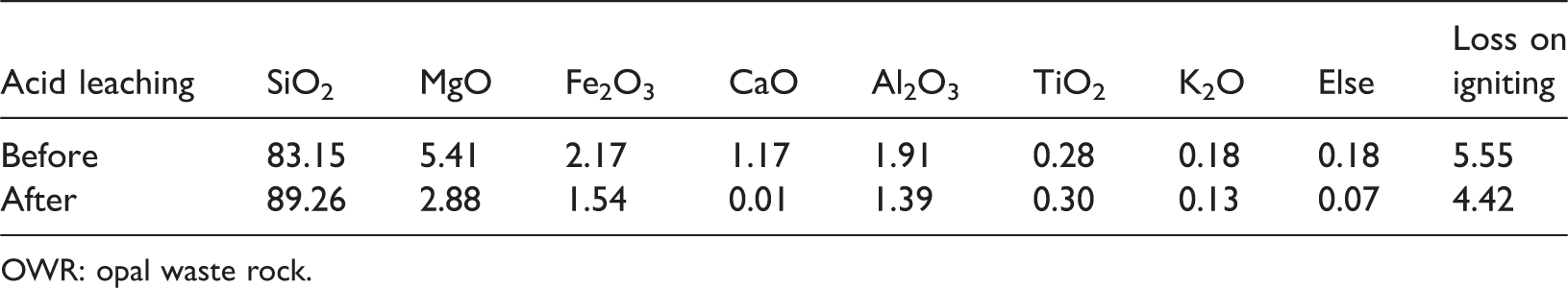
Table 1 shows the chemical composition of OWR before and after acid leaching. It is observed that, compared with natural OWR, the SiO2 mass of OWR after acid leaching increases from 83.15 to 89.26% and impurities, such as MgO, Fe2O3, CaO, also clearly decrease. In other words, the impact of impurities for synthesis of 4A zeolite is diminished. Figure 1 presents XRD patterns of raw OWR and purified OWR. These reflections at 2θ = 20.9° and 35.7° are found and identified as tridymite, the reflection of 2θ = 21.6° is identified as cristobalite, the reflection at 2θ = 8.3° is identified as palygorskite, and the reflection at 2θ = 30.9° is identified as dolomite according to the comparison with standard pattern. The pattern indicates that the OWR is mainly composed of tridymite and cristobalite with lower crystallinity. Thus, the material in this study can be reacted with alkaline more easily. Moreover, from XRD pattern of purified OWR, the reflection of palygorskite becomes weaker, and the reflection of dolomite vanishes when compared with raw OWR, i.e. the structure of palygorskite is destroyed and the dolomite is removed. More importantly, SEM images of OWR (Figure 2(a) and (b)) show the surface pores of raw OWR are mostly blocked by clay minerals, and obvious porous structure can be observed on the surface of purified OWR, indicating that impurities of OWR were removed after acid leaching, thereby enhancing reactivity.
Chemical composition of OWR before and after acid leaching (%).
OWR: opal waste rock.
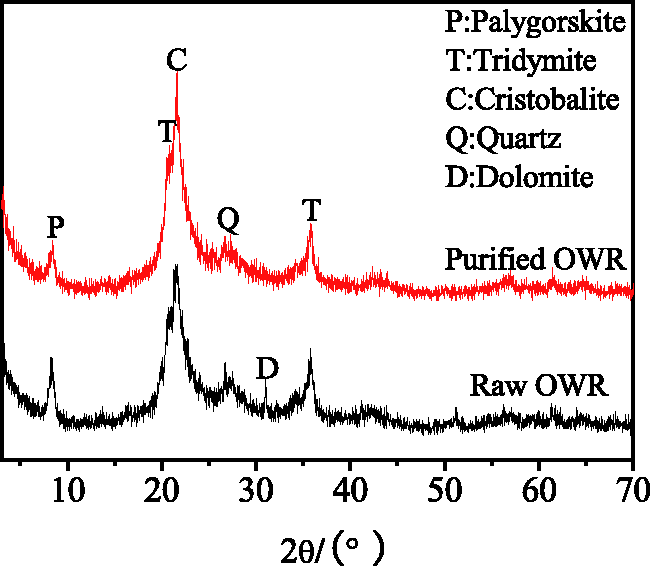
XRD patterns of OWR before and after acid leaching. OWR: opal waste rock.

SEM images of raw OWR (a) and purified OWR (b), 4A zeolite crystallized for 1 h (c), 3 h (d, e, f), 5 h (g), 6 h (h), and EDS analysis (i) of 4A zeolite crystal (dotted square in (e)).
XRD analysis
In this paper, the influences of crystallization time, crystallization temperature, Na2O/SiO2, H2O/Na2O, SiO2/Al2O3 on the synthesis products were investigated. The conditions for synthesis 4A zeolite based on OWR are shown in Table 2. The XRD patterns of synthetic products synthesized at different times are presented in Figure 3(a). Regardless of the different crystallization times, the peaks of 4A zeolite can be obviously seen in all products, and the crystallinity of products increase and then decrease with extending crystallization time. The crystallinity is most improved when crystallization time is just 3 h. The crystallization process is different from the results of the report by Hu et al. (2014), where merely some faint peaks of 4A zeolite appear when crystallization time is 3 h.
Experimental parameters for the synthesis of 4A zeolite from OWR.
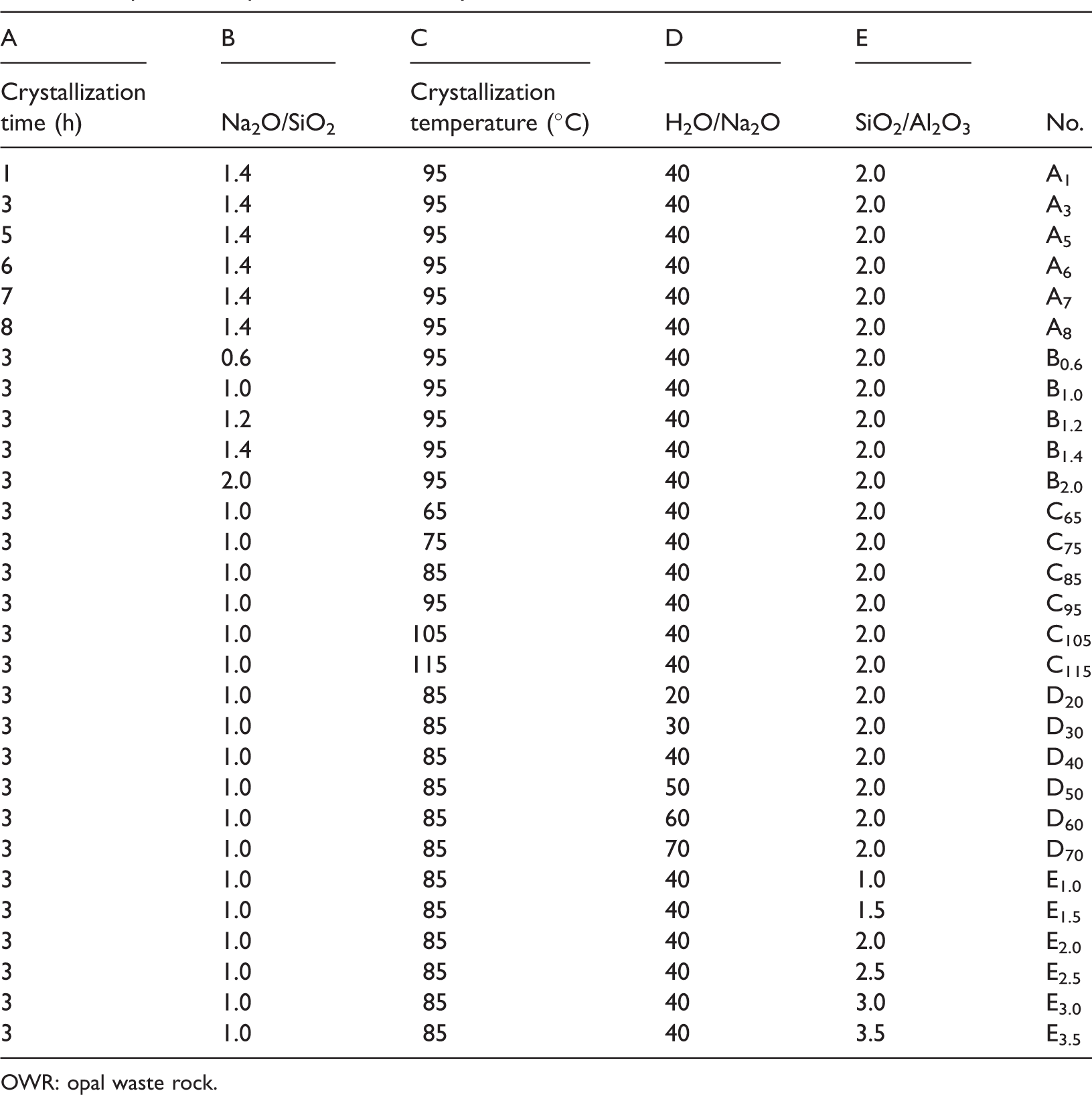
OWR: opal waste rock.

XRD patterns of synthetic products synthesized at different conditions.
The XRD patterns of the 4A zeolite with different Na2O/SiO2 (0.6, 1.0, 1.2, 1.4, 2.0) are displayed in Figure 3(b). When 4A zeolite is synthesized with Na2O/SiO2 of 0.6, small peaks of 4A zeolite appear suggesting the generation of a small quantity of 4A zeolite. The crystallinity of product achieves a rapid growth when Na2O/SiO2 increases from 0.6 to 1.0. Scarcely any changes are observed in XRD patterns when Na2O/SiO2 is more than 1.0. These results indicate that the value of Na2O/SiO2 of 1.0 is adequate for synthesis of 4A zeolite crystals with high crystallinity, in accordance with the results of a published report (Ismail et al., 2010). Regarding 4A zeolite samples at different crystallization temperatures, the XRD patterns are presented in Figure 3(c). When synthesis temperature is 65°C, the master peak is weak cristobalite, showing that OWR cannot form 4A zeolite at 65°C. When crystallization temperature is increased to 75°C, the peak of cristobalite is replaced by faint peaks of 4A zeolite indicating the opening formation of 4A zeolite. With temperature increased to 85°C, the clear enhancement of 4A zeolite peaks can be observed. The results should be assigned to that the increase of temperature will accelerate both nucleation and crystal growth (Liu and Wang, 2017). Compared with that of 85°C, the peaks of 4A zeolite of 95, 105, and 115°C do not exhibit much change.
As shown in Figure 3(d), the crystalline degree of products increases with the decrease of H2O/Na2O. However, when H2O/Na2O decreases to 20, the reflection of sodalite is observed, conforming to the results of published reports (Ma et al., 2010), in which the decrease of H2O/Na2O, that is to say, the raise of alkali dosage, will lead to the generation of sodalite. By changing the dosage of NaAlO2, the effect of SiO2/Al2O3 on property of 4A zeolite is also studied. XRD patterns of 4A zeolite synthesized at different SiO2/Al2O3 for 3 h are exhibited in Figure 3(e). Just when SiO2/Al2O3 molar ratios vary from 1.0 to 3.0, the peaks of 4A zeolite emerge revealing that 4A zeolite will be produced at a narrow value zone of SiO2/Al2O3. Similar results are also observed in the study made by Liu et al. (2013b). When SiO2/Al2O3 molar ratio is 1.0, the peaks of gibbsite are observed in the XRD pattern, implying that OWR just translates into aluminium hydroxides when existing in the solution of excess aluminium and alkaline. The amorphous peaks when the SiO2/Al2O3 value increases to 3.5 display the absence of 4A zeolite.
SEM analysis
Spherical particles coupled with some incomplete unformed crystals are observed in Figure 2(c) when crystallized for 1 h, indicating that 0–1 h is the nucleation stage, and 4A zeolite has not begun to crystallize. The sample crystallized for 3 h in Figure 2(d) shows cubic shape, suggesting that 4A zeolite has been formed; these results agree with former studies (Zayed et al., 2017). The crystals of 4A zeolite synthesized for 3 h show homogeneous cubic morphology as presented in Figure 2(e) and (f) revealing SEM image under lower magnification. As displayed in Figure 2(g) and (h), when crystallization time is 5 and 6 h, the crystal size of 4A zeolite do not have great change compared with 4A zeolite of 3 h. Figure 2(i) presents the energy dispersive spectroscopy (EDS) patterns of 4A zeolite crystal, i.e. the region delimited by the rectangle in Figure 2(e), and the O, Na, Si, and Al are consistent with 4A zeolite (Wang et al., 2015).
CEC and SWA analysis
The CEC and SWA results of all 4A zeolite are shown in Figure 4. As exhibited in Figure 4(a), CEC and SWA get a substantial decrease with crystallization time less than 3 h. When the crystallization time is over 3 h, the SWA of products do not have much change, whereas CEC decreases, except at the point of 5 h. The reason is that with the extension of crystallization time, 4A zeolite would translate into sodalite which had inferior exchange capacity leading to CEC decrease. In addition, in Figure 4(b) and (c), when Na2O/SiO2 and temperature are lower than 1.0 and 85°C, CEC and SWA undergo a significant decrease. That decrease hints at the fact that Na2O/SiO2 and the temperature will influence the performance of synthetic 4A zeolite, consistent with XRD results displayed in Figure 3(b) and (c). As presented in Figure 4(d) and (e), with the raise of H2O/Na2O and SiO2/Al2O3, CEC and SWA first rise and then drop. CEC and SWA achieve the maximum value of 2.92 mmol (g dry zeolite)−1 and 20.3% when H2O/Na2O is 40 (Figure 4(d)) and SiO2/Al2O3 is 2.0 (Figure 4(e)), respectively. 4A zeolite based on OWR without acid leaching was also synthesized at Na2O/SiO2 of 1.0, H2O/Na2O of 40, SiO2/Al2O3 of 2.0, crystallization temperature of 95°C for 6 h; its CEC and SWA results are 2.34 mmol (g dry zeolite)−1 and 19.7%, respectively.

CEC and SWA of synthesis product synthesized at different conditions ((a) Na2O/SiO2=1.4, T = 95°C, H2O/Na2O = 40, SiO2/Al2O3=2.0; (b) t = 3 h, T = 95°C, H2O/Na2O = 40, SiO2/Al2O3=2.0; (c) t = 3 h, Na2O/SiO2=1.0, H2O/Na2O = 40, SiO2/Al2O3=2.0; (d) t = 3 h, Na2O/SiO2=1.0, T = 85°C, SiO2/Al2O3=2.0; (e) t = 3 h, Na2O/SiO2=1.0, T = 85°C, H2O/Na2O = 40).
FTIR analysis
FTIR spectra of 4A zeolite crystallized at 95°C for 0, 1, 3, 5 and 6 h are shown in Figure 5. Three bands at 475, 785, and 1090 cm−1, ascribed to OWR (Eckert et al., 2015), can be obviously seen when crystallization time is 0 h. The bands at 785 and 1090 cm−1 are, respectively, ascribed to symmetrical and asymmetrical stretching vibration of the Si–O–Si, corresponding to amorphous silica (Jiang et al., 2012). All the 4A zeolite exhibit absorption bands at approximately 3435 and 1651 cm−1 both ascribed to O–H vibrations of the absorbed water (Su et al., 2016). When the mixture crystallized for 1 h, the bands at approximately 991, 700, and 447 cm−1 appear in the product and correspond to sodium aluminosilicate gels (Jiang et al., 2012). In addition, when crystallization time is 1 h, the weak band at 554 cm−1 emerges which is assigned to D4R vibration, the second major frame of 4A zeolite (Wang et al., 2014), indicating the partial formation of 4A zeolite. Four bands at 462, 559, 670, and 1001 cm−1, assigned to 4A zeolite (Sapawe et al., 2013), appear distinctly with crystallization time increasing to 3 h. The band at 667 cm−1 is attributed to the symmetrical stretching vibration of T–O bond (T represents Si or Al) and 1001 cm−1 attributed to asymmetrical stretching vibration of internal tetrahedron (Ni et al., 2014). All 4A vibrational bands decrease slightly with crystallization time extending to 6 h; this result is in accord with the XRD result as shown in Figure 3(a).

FTIR spectra of the products crystallized under different times.
N2 adsorption–desorption isotherm analysis
The N2 adsorption–desorption isotherm and pore size distribution of 4A zeolite were presented in Figure 6. That N2 adsorption–desorption isotherm displayed the typical IV-type isotherms according to the IUPAC standard (Thommes et al., 2015). And from the pore size distribution curves, the pore size of 4A zeolite showed a distribution of multipeaks but mainly mesopores. After calculation, the N2-BET, the total pore volume, and the average pore diameter of 4A zeolite were 26.9 m2 g−1, 7.64 × 10−2 cc g−1, and 11.3 nm.
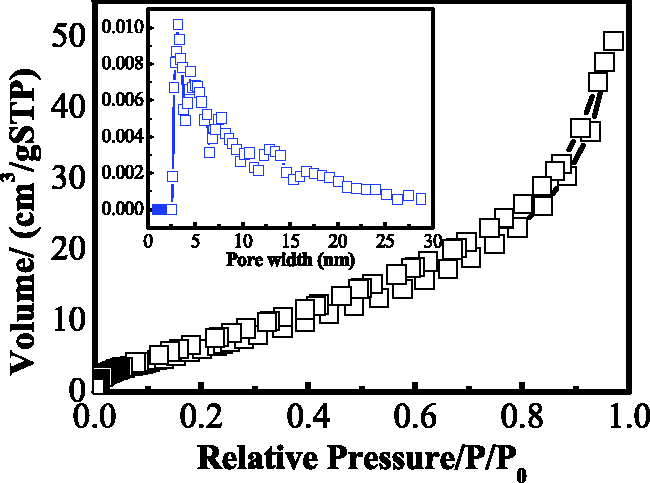
N2 adsorption–desorption isotherm and pore size distribution of 4A zeolite (t = 3 h, Na2O/SiO2=1.0, T = 85°C, H2O/Na2O = 40, SiO2/Al2O3=2.0).
Ammonium ion removal experiments
The effect of time on ammonium ion sorption onto 4A zeolite was performed in Table 3. The removal amounts of ammonium ion raised with the prolongation of the adsorption time and the equilibrium was obtained very rapidly within 90 min. A slight increase was observed when the adsorption time increased from 90 to 180 min. We set the adsorption time to 4 h in the adsorption experiment of this work to guarantee the adsorption equilibrium.
Effect of adsorption time on removal of ammonium ion.

Experimental conditions: C0 = 50 mg l−1, m/V = 1.0 g l−1, I = 1 mmol l−1 NaCl, T = 298 K, pH near to neutral; 4A zeolite: t = 3 h, Na2O/SiO2=1.0, T = 85°C, H2O/Na2O = 40, SiO2/Al2O3=2.0.
Figure 7(a) displays the adsorption isotherms of ammonium ion on materials a, b, c and OWR. a, b, and c represent 4A zeolite samples under t = 3 h, Na2O/SiO2=1.0, T = 85°C, H2O/Na2O = 40, SiO2/Al2O3=2.0; t = 3 h, Na2O/SiO2=1.4, T = 95°C, H2O/Na2O = 40, SiO2/Al2O3=2.0; t = 3 h, Na2O/SiO2=1.0, T = 85°C, H2O/Na2O = 40, SiO2/Al2O3=2.5. From Figure 7(a), the removal capacities of three materials for ammonium ion increase in the sequence of a>b>c, in agreement with the trend of CEC, as presented in Figure 4. Moreover, the sorption capacity of OWR is far worse than materials a, b, c (4A zeolite).

(a) Adsorption isotherms of ammonium ion on three materials and OWR, (b) linear plot of Langmuir isotherm, and (c) linear plot of Freundlich isotherm (Qe (mg g−1): the removal capacity for NH4+; Ce (mg l−1): the equilibrium concentration of NH4+).
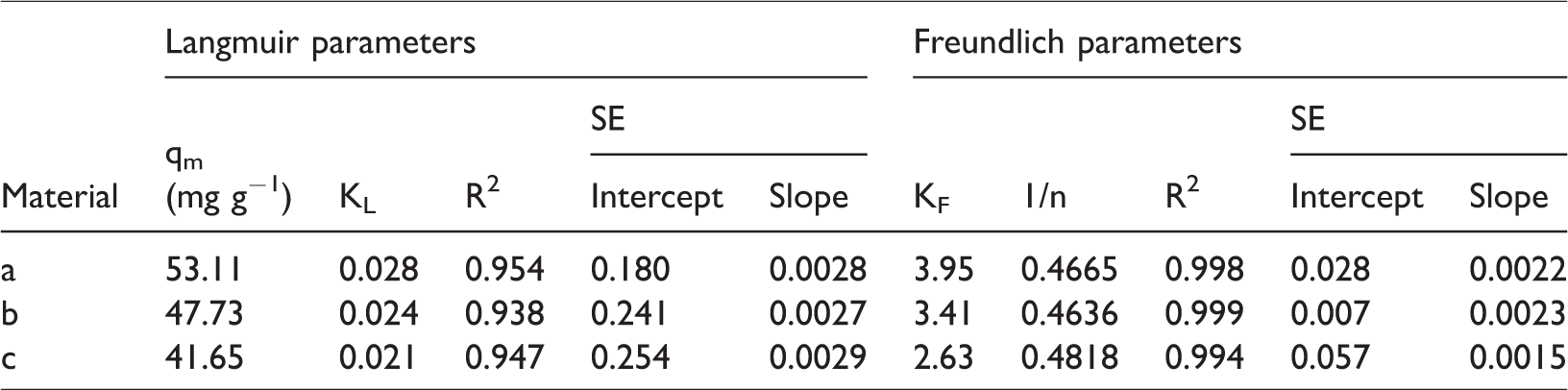
Figure 7(b) and (c) shows linear plots of Langmuir and Freundlich models; relative parameters of two models are calculated out and listed in Table 4. Although both Langmuir and Freundlich models fit the experimental adsorption data, Freundlich model has higher R2 (0.998, 0.999, 0.994) than Langmuir model (0.954, 0.938, 0.947) and lower SE according to Table 4. Therefore, Freundlich model is more suitable to fit experiment data. Moreover, the maximum adsorption capacity of ammonium ion on material a is 53.11 mg g−1. The maximum adsorption capacity of some commonly used adsorbents is listed in Table 5.
Relative parameters based on the Langmuir and Freundlich models.
A summary of the maximum adsorption capacity of some commonly used adsorbents for ammonium ion removal.

As we all know, 4A zeolite had superior ability of ion exchange and in the uptake process of vast majority cation, ion exchange was the main mechanism (Ojuva et al., 2015b; Zavareh et al., 2018). The exchange process for NH4+ removal by 4A zeolite could be depicted as following

Conclusions
In this study, 4A zeolite was successfully synthesized from OWR via acid pretreatment, followed by hydrothermal treating under temperate condition. Based on results of XRD, SEM, CEC, SWA, and FTIR, 4A zeolite has great performance when synthesized under Na2O/SiO2 = 1.0, H2O/Na2O = 40, SiO2/Al2O3 = 2.0, crystallization time of 3 h, and crystallization temperature of 85°C. Under those conditions, 4A zeolite was synthesized with CEC of 2.93 mmol (g dry zeolite)−1, SWA of 22.3%, and NH4+ ion adsorption capacity up to 53.11 mg g−1. The special pattern and pore structure of OWR of the palygorskite mine and the open channels via acid pretreatment all contribute to the fast translation of OWR into 4A zeolite. This research provides not only a new material for synthesis of 4A zeolite but also a new application of OWR. The adsorption properties of 4A zeolite as heavy metal adsorbent will be further studied in the following work.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank for the financial support from the National Natural Science Foundation of China (NSFC) (41572028), The Xuyi Palygorskite Application Technology development and Industrialization Center of Chinese Academy of Sciences, China (201503), Engineering Research Center of non-metallic minerals of Zhejiang Province and Key Laboratory of Clay Minerals, Ministry of Land and Resources for support this study.