Abstract
The adsorption of CO2 and CH4 on carbon molecular sieves was studied based on the adsorption potential theory, which is widely used in gas adsorption on microporous adsorbents. The methods to obtain the adsorption density in the adsorbed phase, including Ozawa’s method and the empirical method, and the methods to calculate the virtual saturation vapor pressure, including Dubinin’s method and Amankwah’s method, were discussed. A functional exponential form proposed in this study could describe the adsorption characteristic curve better than the cubic polynomial and logarithmic function used in the previous literature. A new model, which expresses the correlation of adsorbed amount, temperature, and pressure, was proposed and verified experimentally. The model provides a reliable way to predict the adsorption isotherms of CO2 and CH4 on carbon molecular sieves at different temperatures according to the data measured at a certain temperature and to select a proper kind of carbon molecular sieve in CO2/CH4 separation.
Introduction
The adsorption potential theory, first proposed by Polanyi in 1914 to describe physical adsorption of gases on solids (Dubinin, 1960), has been commonly applied in adsorption behavior of H2, CH4, N2 (Amankwah and Schwarz, 1995; Dubinin, 1960; Ozawa et al., 1976; Wang et al., 2010), and other gases on microporous homogeneous carbonaceous materials such as activated carbon. It also has gradually attached importance in adsorption process between gases and heterogeneous carbonaceous materials such as coal (Hill, 1952; Huan et al., 2015; Rudzinski and Everett, 1992). The concept was further developed by Dubinin (1960) into the theory of volume filling of micropores. The range of application of the potential theory includes adsorption not only close to critical points (Rangarajan et al., 1995) and dew points (Aranovich and Donohue, 1996) but also at high, moderate, and low pressures (Grant and Manes, 1966; Lewis et al., 1950). The adsorption potential theory is the theory of thermodynamics, which belongs to the macroscopic performance of adsorption equilibrium rather than involving microcosmic mechanism (Zhao, 2005). The advantageous feature of the theory for describing gas adsorption is that a single adsorption characteristic curve, which is independent of temperature, can be obtained. If the characteristic curve is known, then the adsorption uptake of the adsorbate on the adsorbent at different temperatures can be predicted (Hao et al., 2014).
Research on the application of adsorption potential theory has been reported frequently and the area of its application has also been enlarged recently. Su et al. (2006) found that the adsorption in coal and gas system can be explained by adsorption potential theory. Su et al. (2008) proposed a method to predict the adsorption data of coal bed gas on coal by adsorption potential theory at 303 and 333 K and verified by experiments of measuring the adsorption process of gas on three different coals from Hedong mine, Qingshui Basin, and Fuxin Basin. Wang et al. (2010) studied adsorption equilibrium of xenon on activated charcoal and found that the predicted data at 303 and 323 K, which were obtained from adsorption characteristic curve according to the adsorption data at 293 K based on adsorption potential theory, fitted well with the experimental data at the corresponding temperature. Yang et al. (2010) utilized the characteristic curve of N2 on Yangquan coal, which was drawn by adsorption data at 77 K, to predict the adsorption process at 303 K and found that the values were consistent with the experimental data. Zhang et al. (2008) studied the properties of the adsorption characteristic curve of CH4 on coal with this theory and formulated a new temperature–pressure comprehensive adsorption model for adsorption of CH4 on coal. Du and Wu (2007) obtained the affinity coefficients of the H2-zeolites adsorption system and derived a formula for the generalized adsorption function to describe supercritical adsorption of H2 on zeolites according to the adsorption potential theory. Liu et al. (2015) deduced a calculation model of partition coefficient of volatile organic compounds and studied the mechanism of its emission between air and building materials based on this theory. Askalany and Saha (2017) proposed a new thermodynamic formula on adsorbed phase volume, which was based on the potential theory, to calculate the isosteric heat of adsorption. Shapiro and Stenby (1998) formulated a multicomponent adsorption theory according to the theory, which could be applied in larger ranges of pressures and temperatures.
CO2 and CH4 are the main components in biogas and landfill gas and the separation of their mixture has attracted more attention in recent years (Li et al., 2017; Llano-Restrepo, 2010; Sizova et al., 2015). Carbon molecular sieves (CMSs), a kind of solid adsorbents which are commonly used to separate the two gases, have many advantages compared with active carbons and zeolites, such as narrow PSDs, strong hydrophobicity, great resistance to acids and alkalis, and excellent thermal stability (Bai et al., 2013; Grande and Rodrigues, 2007; Liu et al., 2016; Yu et al., 2012). Several indexes like separation factor and selectivity are usually adopted to evaluate the separation effect of CMS, which are obviously influenced by temperature (Álvarez-Gutiérrez et al., 2016; Duan et al., 2016; Tamnanloo et al., 2014; Wu et al., 2015). In order to select suitable adsorbents to separate CO2 and CH4 from their mixture, it is necessary to measure the adsorption uptake of a single pure component at different temperatures, which consumes large amounts of work and time (Krishna, 2012). Meanwhile, the adsorption of CO2 and CH4 on CMS is physical (Bai et al., 2013) so that the adsorption behavior could be explained by Polanyi adsorption potential theory. Therefore, based on the theory, adsorption data at one temperature are needed and then the adsorption behaviors of the gas on CMS at the other temperatures can be derived. It can be more convenient than measuring adsorption isotherms at every condition.
Additionally, literature on the application of the adsorption potential theory in adsorption process of CO2 and CH4 on CMS is relatively less compared with that on activated carbons and coals. In this research, the adsorption of CO2 and CH4 on CMS is studied through adsorption potential theory and the effects of the parameters in this theory on the adsorption characteristic curves are discussed. The feasibility of the theory, which is utilized to predict the adsorption uptake of CO2 and CH4 on CMS at different temperatures, is verified by contrasting predicted data with experimental ones. Besides, a relational expression of adsorption amount, temperature, and pressure is formulated and demonstrated, which is expected to be used in prediction of the adsorption of CO2 and CH4 on CMS and in the selection of a reliable CMS to separate the two gases.
Material and methods
Materials
The adsorbent materials used in this study are common commercial CMSs made by phenolic resin. They are numbered as CMS-A, CMS-B, and CMS-C, and their characteristics are listed in Table 1.
The physical parameters of the CMS samples.

CMS: carbon molecular sieve.
Pure component adsorption isotherms
The adsorption isotherms of pure CO2 and CH4 on CMS were measured through an Intelligent Gravimetric analysis (IGA-100B, UK) at the pressure of up to 1 MPa and at the temperatures of 298, 308, and 318 K, respectively. To avoid the influence of water and other gases adsorbed on the adsorbents, the samples were pretreated in a vacuum at 473 K for 4 h before the measurements. The purity specifications of CO2 and CH4 were both 99.99%.
Methods
Polanyi adsorption potential theory
The adsorption potential theory assumes that the adsorbents have a gravitational field in certain space away from the surface (Meng et al., 2016). Thus, the gas molecules around the solid surface are immediately adsorbed once moving into this field, which is called adsorption space. The theory suggests that the interaction force in the adsorption system belongs to dispersion force, which is irrelevant with temperature. That means in the adsorption space, the relationship between the adsorption potential and the adsorbed phase volume (also can be called adsorption volume) changes little as the temperature varies. The adsorption characteristic curve, which is described by the adsorption potential and the adsorption volume, is almost constant in spite of the variation of temperature for the same adsorption system. Consequently, it is realizable that the adsorption isotherms at different temperatures can be obtained and the relationship among adsorbed amount, temperature, and pressure can be established according to the adsorption characteristic curve which is drawn from the adsorption data at one temperature. This is the unique feature of the adsorption potential theory, which is not possessed by other adsorption theories.
The concept of adsorption potential reflects the work required to attract per unit mass of gas from the infinite boundary to a certain position around the adsorbents, which can be described as equation (1) (Bai et al., 2014; Meng et al., 2016)

Methods to describe the adsorption characteristic curve
The saturation vapor pressure in equation (1) is used for adsorbates when the temperature is below the critical temperature of the adsorbate. However, when above the critical temperature, there is no concept of the vapor pressure; hence the use of virtual saturation vapor pressure was proposed (Amankwah and Schwarz, 1995; Hao et al., 2014). In this study, two approaches that existed in the literature to calculate the virtual saturation vapor pressure are utilized, and the expression of which is given as follows.
Dubinin’s method is represented as equation (2) (Do, 1998; Dubinin, 1960; Srinivasan et al., 2012)

Amankwah’s method is represented as equation (3) (Do, 1998; Dubinin, 1960; Hao et al., 2014)

The computational formula of the adsorption volume per unit mass of adsorbents is given as equation (4) (Meng et al., 2016)

There are several methods to calculate the adsorbate density ρad in the adsorbed phase. Here, the Ozawa’s method and the empirical formula are considered.
Ozawa et al.’s (1976) method to compute ρad is given as equation (5)

The empirical method is presented as equation (6) (Meng et al., 2016; Ozawa et al., 1976)

As can be seen from the formulas, the computing methods of both virtual saturation vapor pressure and adsorption volume can affect the results. To determine an optimal group, correlation coefficient and residual sum of square (RSS) are the main factors for reference to evaluate the accuracy and precision of the fitting adsorption characteristic curve. The RSS, which reflects the discrete degree between the expression and the experimental data, is given as equation (7)

Moreover, the average relative error (ARE), which can describe the deviation between the experimental data and the predicted data (Shafeeyan et al., 2015; Srenscek-Nazzal et al., 2016), is given by equation (8)

In this study, the combination of the formulas is listed in Table 2.
Combination of the formulas of P0 and ρad.

Results and discussion
Adsorption isotherms of pure component
Figure 1 illustrates the adsorption isotherms of pure CO2 and CH4 on CMS at 298, 308, and 318 K, respectively. The dash lines in Figure 1 are Langmuir fitting curves. It can be concluded from the shape of the adsorption isotherms that all the curves are characteristic of type I according to the classification of IUPAC. The adsorbed amounts of the two gases increase with the pressure and tend to approach the equilibrium. The reason is mainly that the strong adsorption sites are first occupied by the gases and become less during the adsorption process, resulting in the maximum adsorbed amount. At all pressures, the adsorbed amount of CO2 is larger than that of CH4, because of its smaller dynamic diameter (CO2 0.34 nm, CH4 0.38 nm) (Song et al., 2017) and its greater quadrupole moment (while CH4 has no quadrupole moment) which cause a stronger electrostatic interaction between CO2 molecules and the adsorbents (Li et al., 2012). Moreover, for both CO2 and CH4, the isotherms measured at a higher temperature are below that at a lower temperature due to the exothermic nature of adsorption process (Álvarez-Gutiérrez et al., 2016; Singh and Kumar, 2016).

Adsorption isotherms of CO2 and CH4 on CMS at different temperatures. The dashed lines represent Langmuir fitting curves. (a) CMS-A, (b) CMS-B, and (c) CMS-C.
Determination of adsorption characteristic curves
Dubinin’s method
Dubinin’s method was widely applied to calculate the virtual saturation vapor pressure. According to this method, the characteristic curves of CO2 and CH4 on CMS-As are shown in Figure 2 and that on CMS-B and CMS-C are shown in Supporting Information (Figures S1 and S2).

Adsorption characteristic curves of CO2 and CH4 on CMS-A. (a) and (b) calculated by methods in Group 1 and (c) and (d) by methods in Group 2. The solid lines represent exponential form fitting curves. (a) CO2 characteristic curves on CMS-A, (b) CH4 characteristic curves on CMS-A, (c) CO2 characteristic curves on CMS-A, and (d) CH4 characteristic curves on CMS-A.
As can be seen, adsorption characteristic curves of not only CO2 but also CH4 obtained at all temperatures are almost the same one, indicating that the characteristic curves are unique and independent of temperature. Comparing two curves described by different calculation methods, it is clear that the data computed by Group 1 are featured by narrow distribution, while the data obtained by Group 2 are relatively dispersing. Thus, on the condition that Dubinin’s method is used to calculate the virtual saturation vapor pressure, adopting Ozawa’s method to gain the adsorbate density in adsorbed phase is more suitable, as Ozawa’s method generates more accurate characteristic curves.
In addition, the figures above show that there is a negative correlation between adsorption potential and adsorption volume. In most literature, cubic polynomial, which is expressed as equation (9), is adopted to describe this kind of relationship, namely to fit the adsorption characteristic curve (Huan et al., 2015; Su et al., 2008; Wang et al., 2010; Zhang et al., 2008)

In several literatures, logarithmic function is utilized to represent the curve (Bai, 2002), which is shown in equation (10)

Based on the variation of adsorption potential accompanying with adsorption volume, a functional exponential form expressed as equation (11) is proposed to fit the curve in this study

The three kinds of equations were applied to match the adsorption characteristic curves. Correlation coefficient r and RSS between the experimental data and predicting data, which are important to evaluate the accuracy and precision of the equations, are summarized in Tables 3 and 4.
Values of r and RSS of the three equations for adsorption characteristic curves of CO2 on CMS samples calculated by Dubinin’s method.

CMS: carbon molecular sieve; RSS: residual sum of square.
Values of r and RSS of the three equations for adsorption characteristic curves of CH4 on CMS samples calculated by Dubinin’s method.

CMS: carbon molecular sieve; RSS: residual sum of square.
It can be concluded that when calculated by method of the same group, for the adsorption characteristic curves of both CO2 and CH4, the fitting precision of exponential form is the best, due to its highest fitting correlation coefficient and lowest RSS, followed by the cubic polynomial form and then the logarithmic form. The results prove that the equation proposed in this study can express the curve more precisely with its highest fitting degree. Thus, the fitting lines of adsorption characteristic curves in Figure 2 and Figure S1-2 by exponential form (equation (11)) were given. When fitted by the same equation, contrasting the results obtained by methods of Group 1 and that of Group 2, the values of correlation coefficient and RSS differ obviously from each other and mostly the fitting degree gained by methods of Group 1 is higher than that of Group 2. This finding manifests that a proper method to calculate the adsorbate density in the adsorbed phase is of great importance for the fitting accuracy, which is inconsistent with the statement that the method to compute adsorbate density has little impact on the results (Huan et al., 2015).
The results given in Tables 2 and 3 show that when Dubinin’s method is chosen to calculate the virtual saturation vapor pressure, it is more accurate to use Ozawa’s formula to obtain the adsorbate density in the adsorbed phase. This is consistent with the results obtained from Figure 2 and Figure S1-2, in which the data obtained by Ozawa’s method distribute more intensively than that by the empirical formula.
Amankwah’s method
When Amankwah’s method is applied to get the virtual saturation vapor pressure, three kinds of functional forms, namely equations (9) to (11), were adopted to fit adsorption characteristic curves calculated by methods of Group 3 and Group 4. The variation of correlation coefficient and RSS with the parameter k of CMS-A is demonstrated in Figure 3 and that of CMS-B and CMS-C are shown in Supporting Information (Figures S3 and S4).

Variation of correlation coefficient and RSS with parameter k for characteristic curves on CMS-A. Solid points and center points show correlation coefficient and RSS, respectively. (a) CO2 characteristic curve obtained by methods of Group 3, (b) CO2 characteristic curve obtained by methods of Group 4, (c) CH4 characteristic curve obtained by methods of Group 3, and (d) CH4 characteristic curve obtained by methods of Group 4.
As illustrated in Figure 3 and Figure S3–S4, no matter if the adsorption density in the adsorbed phase is computed by Ozawa’s method or the empirical method, the exponential equation used to fit the characteristic curves has the maximum correlation coefficient and the minimum RSS though for CO2, the fitting results of exponential equation differs a little with that of cubic polynomial form. Then the cubic polynomial and logarithmic form were followed by. Taking Figure 3 as an example, comparing Figure 3(a) with Figure 3(b), it can be observed clearly that when fitted at the same condition, for the adsorption characteristic curves of CO2, the values of the correlation coefficient in Figure 3(a) are larger than that in Figure 3(b). On the contrary, the values of the RSS in Figure 3(a) are less than that in Figure 3(b). The similar phenomenon is also found for the adsorption characteristic curves of CH4 on the basis of Figure 3(c) and (d). It means that when Amankwah’s method is used to compute the virtual saturation vapor pressure, it is more precise to apply Ozawa’s equation to get the adsorbate density in the adsorbed phase, which is in accordance with the conclusion drawn in “Dubinin’s method” section.
Furthermore, based on Figure 3 when fitted by the same equation, the values of both correlation coefficient and RSS change with the parameter k, proving that there is an optimal value of k for different adsorbents. For the results obtained in this study, the optimal k, which corresponds to the maximum correlation coefficient and the minimum RSS, is not equal to 2. Similar phenomenon also can be found in Figures S3 and S4. In Figure 3, for adsorption characteristic curves of CO2, the optimal value of k is 5 by methods of Group 3 and 5.5 by methods of Group 4. While, for the curves of CH4, the best k is equal to 1.46 when analyzed by methods of Group 3 and 2.3 of Group 4.
When fitted by the three equations under the condition of the optimal k, the exponential form exhibits best fitting degree, then the cubic polynomial and the logarithmic form according to Tables 5 and 6, which is the same with the consequence when k equals to 2. Through methods in Group 2 based on Tables 3 and 4, the fitting correlation coefficients of both the exponential and the cubic polynomial equations are above 0.99 and the values of RSS of which are below 0.22. It also can be seen obviously that when the exponential equation is adopted, the values of correlation coefficient and RSS at the optimal k are a little different with that at k = 2 for the adsorption characteristic curves of both CO2 and CH4. Taking Figure 3(a) as an example, the values of r and RSS obtained by exponential equation at the optimal k are 0.99466 and 0.07751, while that at k = 2 are 0.99282 and 0.10207. The parameters fitted by the three functions of the adsorption characteristic curves of CO2 and CH4 at the optimal k are shown in Tables 5 and 6, respectively. Comparing the values of Group 3 and Group 4, it can be concluded that by the methods of Group 3 the results are more accurate and precise than that of Group 4. Thus, the fitting lines obtained by the methods of Group 3 at the optimal k and their fitting results by three equations are illustrated in Figure 4.
Parameters of different fitting equations of the characteristic curves for CO2 on CMS-A at the optimal k.

CMS: carbon molecular sieve; RSS: residual sum of square.
Parameters of different fitting equations of the characteristic curves for CH4 on CMS-A at the optimal k.

CMS: carbon molecular sieve; RSS: residual sum of square.
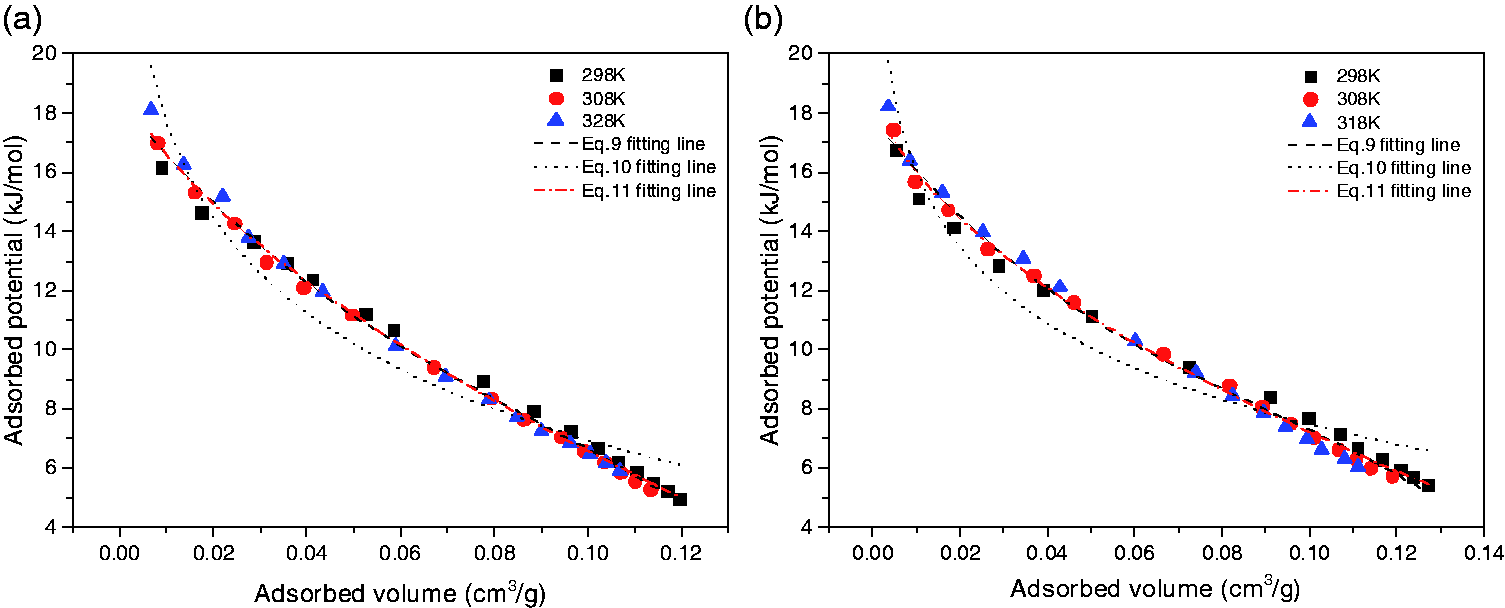
Adsorption characteristic curves obtained by methods of Group 3 and the corresponding fitting results by three different equations. (a) Adsorption characteristic curves of CO2 and (b) adsorption characteristic curves of CH4.
Contrasting methods of Group 1 and Group 3 according to Tables 3 to 6, for CO2 characteristic curves on the condition that adsorbed density is calculated by Ozawa’s method, the fitting results by the exponential formula when k is equal to 2 differ little with that when k is the optimal, the correlation coefficients and RSS of which are 0.99282 and 0.10207 for the former and 0.99413 and 0.07751 for the latter. Similarly, the results for CH4 show almost no distinction between when the value of k is 2 and the optimal, with the correlation coefficients and RSS 0.99137 and 0.1076 for the former and 0.99296 and 0.1021 for the latter. This finding is more obvious as shown in Figure 3, that is when fitted at the same condition, the values of correlation coefficients and RSS change slightly with k, with the optimal ones differing little compared with that at k = 2. Therefore, when the exponential formula is applied to fit the adsorption characteristic curves, taking the value of k = 2 is more feasible and convenient so as to simply the calculation complexity. In this study, the condition discussed below is on the basis of adopting Dubinin’s method (k = 2) to obtain the virtual saturation vapor pressure.
As shown in Figure 4, the fitting effects of the three kinds of equations are clear and definite. The fitting lines of the logarithmic form deviate from the computational data apparently, proving that the fitting effect of logarithmic form is the worst. However, the results show that the cubic polynomial and exponential form can fit the data very well. Taking the results listed in Tables 2, 4, and 5 into consideration, it is explicit to conclude that the exponential form can fit the adsorption characteristic curves best among the three at the same condition.
Prediction of the adsorption isotherms
Prediction of the single component adsorption isotherms
To take full advantage of the adsorption potential theory, the adsorption characteristic curves which were obtained according to the adsorption data of CO2 and CH4 on CMS at a certain temperature and calculated by combination of Ozawa’s method and Dubinin’s method were fitted with the three types of equations to predict the adsorption isotherms at different temperatures. And the accuracy and precision of the fitting results are estimated by ARE between the experimental data and the predicted ones.
The adsorption data of CH4 on CMS sample in Yang et al. (2014) at 343 K were adopted to obtain the adsorption isotherms at 323 K by methods of Group 1 based on the theory, and the results were fitted by three equations. The value of ARE for the cubic polynomial form is 7.64%, the logarithmic form 14.41%, and the exponential form 6.1%. The other verifications, in which the adsorption data in Song et al. (2017) were utilized, are displayed in Tables 7 and 8.
ARE of the adsorption isotherms between the experimental and the predicted ones at 308 and 318 K fitted by the three equations based on the data of CMS-1 at 298 K (Song et al., 2017).
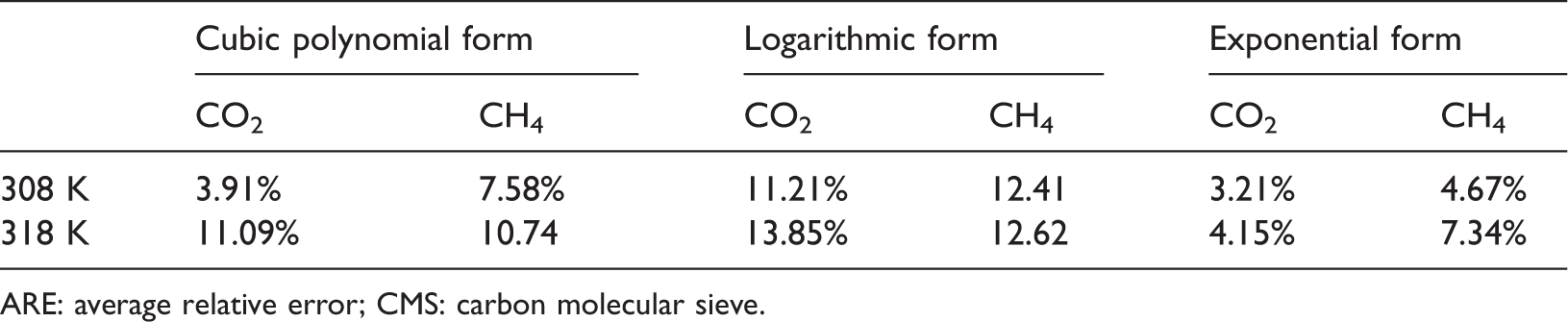
ARE: average relative error; CMS: carbon molecular sieve.
Table 7 lists the three kinds of fitting results of the adsorption isotherms at 308 and 318 K predicted by the adsorption data of CO2 and CH4 on CMS-1 measured at 298 K in Song et al. (2017). It is obvious that for both CO2 and CH4 at the temperature of 308 and 318 K, the values of ARE between the experimental data and the predictive data fitted by the exponential form are the minimum. Table 8 shows a similar phenomenon. The adsorption isotherms of CO2 and CH4 on CMS-2 of Song et al. (2017) were obtained from the characteristic curve drawn based on its adsorption uptake at 308 K. And the fitting results of the exponential form are the optimal, with their values of ARE less than that of the other two types of equations. Therefore, it can be concluded that the exponential equation is the best form to fit the adsorption characteristic curves. The comparison of the predictive isotherms and the practical ones at the optimal condition is illustrated in Figure 5.
ARE of the adsorption isotherms between the experimental and the predicted ones at 298 and 318 K fitted by the three equations based on the data of CMS-2 at 308 K (Song et al., 2017).

ARE: average relative error; CMS: carbon molecular sieve.
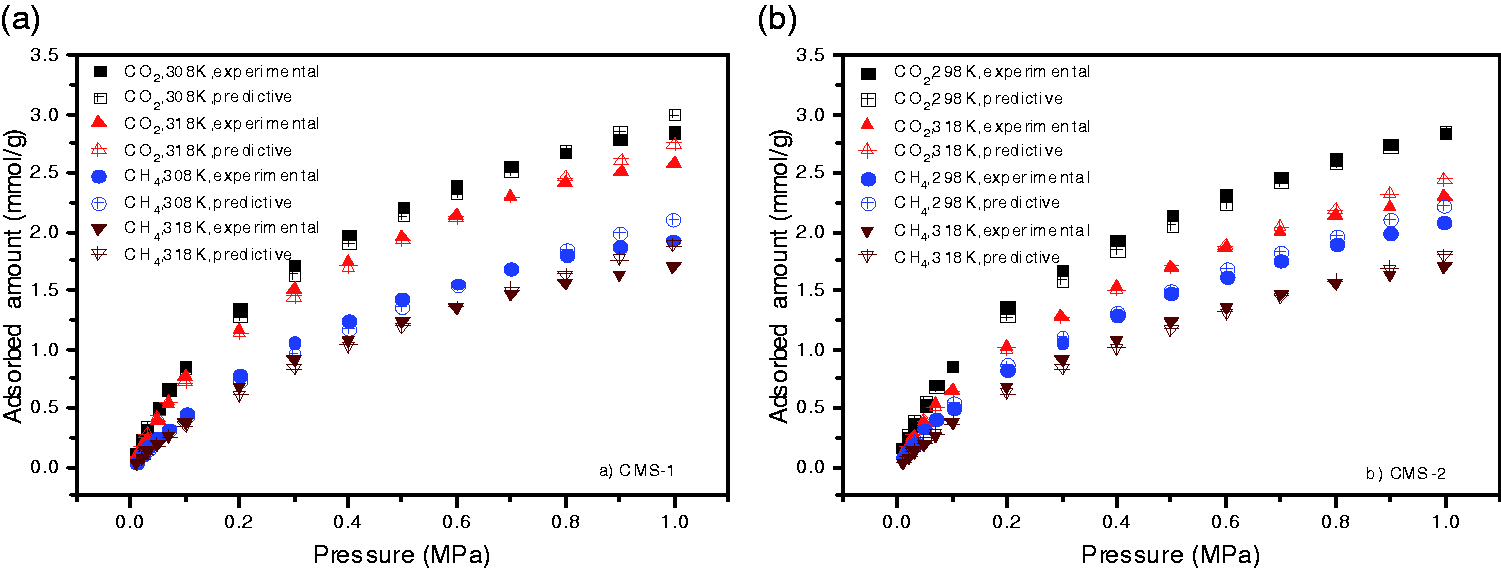
Predictive adsorption isotherms via experimental data. (a) and (b) based on the experimental data at 298 and 308 K of CMS samples in Song et al. (2017), respectively. (a) Predictive adsorption isotherms of CMS-1 (Song et al., 2017) and (b) predictive adsorption isotherms of CMS-2 (Song et al., 2017).
Consequently, it is a reliable approach to obtain the adsorption isotherms at different temperatures based on the adsorption data at a certain temperature by the adsorption potential theory. The condition is that when Ozawa’s method is used to calculate the adsorbate density in the adsorbed phase, Dubinin’s method is applied to compute the virtual saturation vapor pressure and the exponential equation utilized to fit the characteristic curve.
Prediction of multicomponent adsorption isotherms
In order to expand the adaptability of the method proposed in this paper, the adsorption isotherms of CO2 and CH4 among the multicomponent gas mixtures are predicted by combination of Ozawa’s method and Dubinin’s method and characteristic curves fitted by exponential function form. The adsorption isotherms of each component in the mixture, which were composed of H2 (75.5 mol%), CO2 (16.5 mol%), CO (4.5 mol%), and CH4 (3.5 mol%), were measured at different temperature in Yavary et al. (2016). The adsorption isotherm of CO2 and CH4 at certain temperature can be predicted by the data at one temperature based on the method. Here the adsorption data of CO2 on 308 and 318 K predicted by the adsorption isotherm at 298 K and that of CH4 on 298 and 318 K predicted by 308 K were given for a random example and the corresponding predictive and the experimental data were illustrated in Figure 6. The isotherms at other conditions were given in Supporting Information (Figure S5).

Predictive adsorption isotherms via experimental data (Yavary et al., 2016).
It can be seen that the predictive data are close to the experimental ones. And according to the values of ARE between the predictive and the experimental ones, adsorption isotherms of CO2 calculated at 308 and 318 K are 2.90 and 2.94%, and that of CH4 at 298 and 318 K are 7.83 and 8.63%, respectively. Then it can be concluded that the method proposed in this paper can also be used in predicting adsorption isotherms of CO2 and CH4 in the gas mixtures.
A new model of adsorption amount, temperature, and pressure
The attractive property of the adsorption potential theory is that the adsorption characteristic curves obtained at different temperatures are specific. Because of this advantage, it is convenient to establish a correlation between the adsorption potential and the adsorption volume, and the adsorption isotherms at any temperatures can be obtained by the adsorption data measured at a certain temperature. Therefore, a new model of adsorption amount, temperature, and pressure was proposed to predict the adsorption isotherms based on the adsorption potential theory.
The conclusion obtained above is that the exponential equation can well fit the adsorption characteristic curve drawn on the basis of experimental data. And the expression of the exponential equation is simpler than that of the cubic polynomial equation. To reduce the calculated amount, improve the accuracy, and decrease the complexity, the exponential form was adopted to express the adsorption characteristic curves. The expression of the model, which presents the correlation of adsorbed amount, temperature, and pressure, can be presented as equation (12). The deduced process is shown in Appendix 1

The model proposed in this study, the expression of equation (12), can be used to predict the adsorbed amount at any temperatures or pressures by the adsorption isotherm measured at a certain temperature, which has already been verified in “Prediction of the adsorption isotherms” section.
Conclusion
In this study, the methods to calculate the adsorption density in the adsorbed phase and the virtual saturation vapor pressure in the prediction of the adsorption isotherms of CO2 and CH4 on CMS are discussed based on the adsorption potential theory.
The method to calculate the adsorption density in the adsorbed phase has influence on the results. When adopting the same equation to obtain the virtual saturation vapor pressure, it is more accurate to utilize Ozawa’s method to gain the adsorption density than to use the empirical method.
Amankwah’s method to compute the virtual saturation vapor pressure is better than Dubinin’s. There is little distinction between the fitting precision when the parameter k of Amankwah’s method is optimal and that when k equals 2. Thus, it is more convenient to use Dubinin’s method (k = 2) to compute the virtual saturation vapor pressure.
A functional exponential form proposed in this study could describe the adsorption characteristic curve better than the cubic polynomial and logarithmic function used in the previous literature.
Based on the adsorption potential theory, a new model of adsorbed amount, temperature, and pressure, which is verified experimentally, enables a reliable way to predict the adsorption isotherms of CO2 and CH4 both in the form of both single component and multiple components at different temperatures according to the adsorption data at a certain temperature. It can reduce a large amount of measurements in evaluating the separation effect of adsorbates and selecting a suitable type for gas separation.
Supplemental Material
Supplemental material for Application of adsorption potential theory in prediction of CO2 and CH4 adsorption on carbon molecular sieves
Supplemental material for Application of adsorption potential theory in prediction of CO2 and CH4 adsorption on carbon molecular sieves by Xue Song, Li’ao Wang, Yifu Li, Yunmin Zeng, Xinyuan Zhan and Jian Gong in Adsorption Science & Technology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key Technology Support Program of China (2014BAC29B01).
Supplemental material
Supplemental material for this article is available online.
Appendix 1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
