Abstract
We report here a reliable green method for the synthesis of palladium nanoparticles supported on copper oxide as a highly active and efficient catalyst for Suzuki cross-coupling reaction. The experimental synthetic approach is based on microwave-assisted chemical reduction of an aqueous mixture of palladium and copper salt simultaneously using hydrazine hydrate as reducing agent. The catalyst was fully characterized using various techniques showing well-dispersed palladium nanoparticles. The catalytic activity and recyclability of the prepared catalyst were experimentally explored in the ligand-free Suzuki cross-coupling reaction with a diverse series of functionalized substrates. The synthesized Pd/CuO catalyst shows many advantages beside its high catalytic efficiency such as the recyclability of up to five times with negligible loss of catalytic activity, short reaction times, use of environmentally benign solvent systems, and mild reaction conditions.
Introduction
Catalysis research based on transition metal nanoparticles has been extensively investigated as potentially advanced route in several catalytic applications due to their promising properties (Beckert et al., 2015; Elazab et al., 2014, 2017a; Kumar et al., 2015; Mankarious et al., 2017; Mohsen et al., 2017; Neri et al., 2015; Radwan et al., 2017). The nanostructured materials play a vital role in the field of heterogeneous catalysis through its huge impact on several related applications especially in the few recent years (Atarod et al., 2016; Nasrollahzadeh et al., 2017, 2014a, 2014b; Nasrollahzadeh and Sajadi, 2016) The precise design of an overall particle size distribution through controlling particle size is one of the decisive key points to obtain new unique physical and chemical properties (Xie and Shen, 2009; Xu et al., 2009; Yan et al., 2009). Recently, the palladium-based nanoparticles have attracted tremendous efforts in the field of catalysis research due to its broad range of potential applications in the chemical, biochemical, and pharmaceutical industries. Palladium-catalyzed cross-coupling reactions play a crucial role in organic synthesis as they are widely used in assembly of complex organic molecules (Abdelsayed et al., 2009; El-Shall et al., 2009; Fouad and El-Shall, 2012). Those reactions also simply represent the optimum chemical route for carbon–carbon bond formation. Those types of cross-coupling reactions have been mostly adopted under homogeneous reaction conditions via using a ligand in order to achieve a remarkable enhancement of the catalytic activity and selectivity for specific reactions. Copper-based catalysts have been also tested in some sorts of important reactions such as Suzuki–Miyaura cross-coupling (Bondioli et al., 2008; Glasnov et al., 2009). Palladium has a unique ability to catalyze several chemical reactions under both homogeneous and heterogeneous reaction conditions (Chen et al., 2012; Iglesias-Juez et al., 2011; Ivanova et al., 2010; Kim and Henkelman, 2012). Recently, research developments have been reported using both metallic and bimetallic nanoparticle catalysts for a variety of several chemical transformations (Fouad and El-Shall, 2012; Moussa et al., 2011, 2012; Zedan et al., 2010). The previously mentioned research studies have revealed superior catalytic activity for both metallic and bimetallic nanoparticles using copper oxide as an ideal support (Chattopadhyay et al., 2009; Fouad and El-Shall, 2012; Nicolaou et al., 2005). The importance of this focused scientific research approach is due to the fact that C–C cross-coupling reactions are considered as one of the most relevant processes in organic synthesis (Nicolaou et al., 2005). The importance of those nanomaterials is not only covering the research area of cross-coupling reactions which are widely used in many such as cosmetic, pharmacy, agriculture, and natural products, but also covers other potential applications in sensors, catalysis, and energy conversion (Gaikwad et al., 2007; Tsvelikhovsky et al., 2008).
The bimetallic Pd-based nanoparticles were designed using several other transition metals such as Co, Ag, Pt, Au, Ni, and Cu (El-Shall, 2009). We have recently developed a series of iron, cobalt, and nickel metal oxide supported palladium nanoparticles with high catalytic activity (Elazab et al., 2014, 2017b, 2015). Currently, we are working on the development of similar catalytic systems through using one of the most promising transition metals which is copper due to its unique several advantages like abundant reserve, low cost, versatility, less harmful to the environment, and wide use in different applications (Nasrollahzadeh et al., 2015a, 2015b). There is also a main advantage of using copper oxide as a support for palladium-based catalysts which is preventing the potential agglomeration of palladium nanoparticles.
The main advantage of using those types of nano-sized particles is that they largely increase the surface area of the active ingredient of the used catalyst, hence causing a huge enhancement of the contact between reactants and catalyst to be nearly like that of the homogeneous catalysts (Chattopadhyay et al., 2009; Hoseini et al., 2017; Hosseini-Sarvari and Razmi, 2015; Mandali and Chand, 2013). This also led to some innovative ideas regarding the use of nanocatalysis for green chemistry development including the possibility of using the concept of microwave-assisted synthesis combined with nanocatalysis (Bondioli et al., 2008; Ceylan et al., 2011; Fukui et al., 2012; Glasnov et al., 2009; Gupta et al., 2011; Kirschning et al., 2012; Malewicz et al., 2009; Nishioka et al., 2011; Pourmortazavi et al., 2012; Shviro and Zitoun, 2013).
The current research efforts in most catalysis research groups concerned with this hot topic are focused mainly on how to avoid the issues associated with homogeneous catalysis. Those issues are considered as the main challenge to pharmaceutical applications due to the lack of recyclability and potential contamination from residual metals in the reaction product. Several research efforts have been reported that discuss the preparation of palladium nanoparticles supported on copper oxide in cross-coupling reactions through different synthetic routes including wet-chemical strategy, electrodeposition, and oleylamine synthesis (Nasrollahzadeh et al., 2015). Our research efforts are focused on eliminating the product contamination through investigating new catalytic systems using ligand-free heterogeneous palladium catalysis. The aim of this work is to make an enhancement to the current research efforts by development of highly active heterogeneous Pd nanocatalysts that can be easily separated from the reaction medium and recycled. The adopted microwave irradiation method in this research is simple, reliable, versatile, and rapid. It allows the synthesis of the nanoparticles with controlled size. The important remarkable advantage of the microwave dielectric heating over the convective heating is that the reactants can be added at room temperature (or slightly higher temperatures) without the need for high-temperature injection (Bondioli et al., 2008; Fukui et al., 2012; Glasnov et al., 2009; Kirschning et al., 2012; Malewicz et al., 2009). In this manuscript, we report on a green efficient method to prepare highly active palladium nanoparticles supported on copper oxide as a highly efficient catalyst for Suzuki cross-coupling. The high performance of the catalyst including both catalytic activity and recyclability of the synthesized catalytic nanoparticles is experimentally investigated in ligand-free Suzuki cross-coupling reaction using a series of substrates.
Experimental
All chemicals were used as received without any purification. Absolute ethanol (99.9%) and deionized water (D.I. H2O) were used for all experiments. Palladium nitrate (10 wt% in 10 wt% HNO3, 99.999%), copper (II) nitrate hemipentahydrate, hydrazine hydrate (80%), bromobenzene, all other aryl halides, and potassium carbonate were obtained from Sigma Aldrich. A CEM Discover microwave instrument was used for cross-coupling reactions. The reactions were performed at operator selectable power output of 250 W, 2.45 MHz.
Synthesis of Pd, CuO nanoparticles, and Pd supported on CuO nanoparticles
Synthesis of Pd supported on CuO nanoparticles (Pd/CuO)
In this experimental procedure, catalysts with different weight percents of 5, 10, 20, 30 wt% palladium loading on copper oxide were prepared. In a 250 ml beaker, a solution containing 20 ml deionized water and the relevant weight of copper (II) nitrate hemipentahydrate (347, 329, 292, 256 mg, respectively) were sonicated at room temperature for approximately 1.5 h. Then the volume 97, 194, 388, 582 µl, respectively, of the palladium nitrate solution corresponding to the previously mentioned loading percents were added using a micropipette to that previously sonicated solution and stirring was continued for another 1.5 h. At room temperature, hydrazine hydrate (1200 µl) was then added and the solution was heated in a microwave of selected power output of 250 W at 95°C for 30 s, filtered, washed with deionized water and then ethanol, and finally, the catalyst is dried in oven till constant weight of catalyst.
Synthesis of Pd nanoparticles
Palladium nitrate (1940 µl) was added using a micropipette to 50 ml deionized water, and then the solution was sonicated for 1 h. Then, the mixture was stirred for another hour. After finishing the step of stirring, 1200 µl hydrazine hydrate was added using a micropipette to the entire mixture. Then, it is heated using a microwave oven of selected power output of 250 W at 95°C for 20 s, filtered, washed with deionized water and then ethanol, and finally, dried in oven till constant weight of catalyst.
Synthesis of copper oxide nanoparticles
A total of 366 mg of copper (II) nitrate hemipentahydrate Cu(NO3)2·2.5H2O was added to 50 ml deionized water and then sonicated for 1 h. Then, the mixture was stirred for another 1 h. After finishing the step of stirring, 1200 µl hydrazine hydrate was added using a micropipette to the entire mixture. Then, it is heated using a microwave oven of selected power output of 250 W at 95°C for 20 s, filtered, washed with deionized water and then ethanol, finally, dried in oven till constant weight of catalyst.
Catalyst characterization
A JEOL JEM-1230 electron microscope was used for TEM images. GC–MS analyses were used to monitor the catalytic activity of the catalyst in selected reactions. The X-ray photoelectron spectroscopy (XPS) analysis was performed on a Thermo Fisher Scientific ESCALAB. The X-ray diffraction patterns were measured at room temperature using an X’Pert PRO PAN analytical X-ray diffraction unit.
General procedure for Suzuki cross-coupling reactions
In this experimental procedure, aryl halide (0.32 mmol, 1 eq.) was dissolved in a mixture of 4 ml H2O:EtOH (1:1) as an environmentally benign solvent system. The aryl boronic acid (0.382 mmol, 1.2 eq.) and potassium carbonate (0.96 mmol, 3 eq.) were added to this mixture. The Pd/CuO (1 mol%) was then added; then heating under microwave irradiation took place in microwave of selected power output of 250 W at different temperatures of 80, 120, and 150°C for 10 min. It was reported by Ying-Jie Zhu et al. that most of the alcohols with low molecular weights are usually with relatively low boiling points, so implementing the reactions in closed systems is the key to overcome the problem associated with the open reaction systems. This is our case in this research study as a mixed solvent system composed of water and alcohols will allow only low-temperature chemical reactions. So, using microwave assisted-synthesis approach in a closed system will allow using higher reaction temperatures. The use of this automated and calibrated microwave reactor makes it possible to obtain reliable and reproducible experimental results. After the reaction was completed, the reaction mixture was extracted and the solvent in the filtrate was removed and the solid product was further purified (Elazab et al., 2017b, 2015).
General procedure for catalyst recycling
In this typical experimental procedure, aryl halide (0.32 mmol, 1 eq.) was dissolved in a mixture of 6 ml H2O:EtOH (1:1). The aryl boronic acid (0.382 mmol, 1.2 eq.) and potassium carbonate (0.96 mmol, 3 eq.) and finally the palladium–copper oxide nanoparticles Pd/CuO (1 mol%) were then added. The test tube was then sealed with a cap and heated under microwave irradiation (250 W, 2.45 MHz) at the required temperature and time. After the reaction completed, the progress of the reaction was monitored using GC–MS analysis to an aliquot of the reaction mixture. Then, the mixture was diluted with ethanol and shaken. Hence, the solvent above the catalyst was decanted and the catalyst was removed. Ethanol washing for the catalyst was repeated for five times to make sure that all products were removed from catalyst surface. The catalyst was then transferred directly to another microwave tube and fresh reagents were added in order to start the next run. This experimental procedure of recycling the catalyst was repeated for every run and the GC–MS was used to determine the percent conversion of the product (Elazab et al., 2017b, 2015).
Results and discussion
The Suzuki cross-coupling reaction of bromobenzene and phenyl boronic acid in 50 vol% aqueous ethanol under various reaction conditions was investigated using microwave reactor of selected power output of 250 W (Scheme 1). In order to study the effect of temperature on the catalytic activity, the cross-coupling reaction was carried out in microwave of selected power output of 250 W at constant reaction time (10 min) and different temperatures (80, 120, 150°C) using the nanoparticles catalyst (1 mol%). After the reaction was completed, the mixture was diluted with ethanol and shaken. Then, the solvent above the catalyst was decanted and the catalyst was removed. Ethanol washing for the catalyst was repeated for five times to make sure that all products were removed from catalyst surface. The catalyst was then transferred directly to another microwave tube and fresh reagents were added for next run. This procedure of recycling the catalyst was repeated for every run.

Suzuki cross-coupling reactions using Pd/CuO catalyst.
The results reveal that the temperature has a great influence on the reaction yield where increasing the temperature of the reaction mixture favors higher conversion to the product with maximum conversion obtained at 150°C. This behavior was identical for all prepared Pd/CuO catalysts having different Pd loading percents on the solid support (5, 10, and 20 wt%) as seen in Table 1.
Conversion percentage for different ratios of Pd/CuO.

Also, it was found that increasing the catalyst weight percent on the solid support from 5–10 to 20 wt% enhances product conversion (at all studied temperatures) with maximum product conversion reaching 100% for 20 wt% Pd-loaded catalyst at 150°C. Surprisingly, further increase in the Pd content on the solid support (30 wt%) led to a sharp drop in the catalytic performance and lower conversion (about 65%) to the desired product was observed in Table 2.
Conversion percentage for different ratios of Pd/CuO.

It is also noteworthy to mention that repeating the cross-coupling reaction under the optimized conditions (10 min and 150°C) and replacing the 1 mol% Pd/CuO catalyst with either 1 mol% CuO nanoparticles or 1 mol% Pd nanoparticles only resulted in very low conversion to the desired cross-coupling product (less than 30%). These findings reflect on one hand the superiority of Pd metal over copper catalyzing such reactions. On the other hand, it shows the crucial role played by the copper oxide solid support (found in Pd/CuO catalyst and not in Pd nanoparticles catalyst) in minimizing the agglomeration effect of the Pd nanoparticles which is responsible for the reduction in the catalyst activity. The versatility of substrates that can be used in Suzuki cross-coupling reaction using the synthesized catalyst is explored in Tables 3 and 4.
Suzuki cross-coupling reactions with various substrates using Pd/CuO catalyst.

aAryl halide (0.32 mmol, 1 eq.), aryl boronic acid (0.382 mmol, 1.2 eq.), potassium carbonate (0.96 mmol, 3 eq.), and Pd/CuO (1 mol%) in 4 ml (H2O:EtOH) (1:1) were heated at 150°C (MWI) for 10 min.
bConversions were determined by GC–MS.
Diversity of the Suzuki cross-coupling reactions using Pd/CuO catalyst.
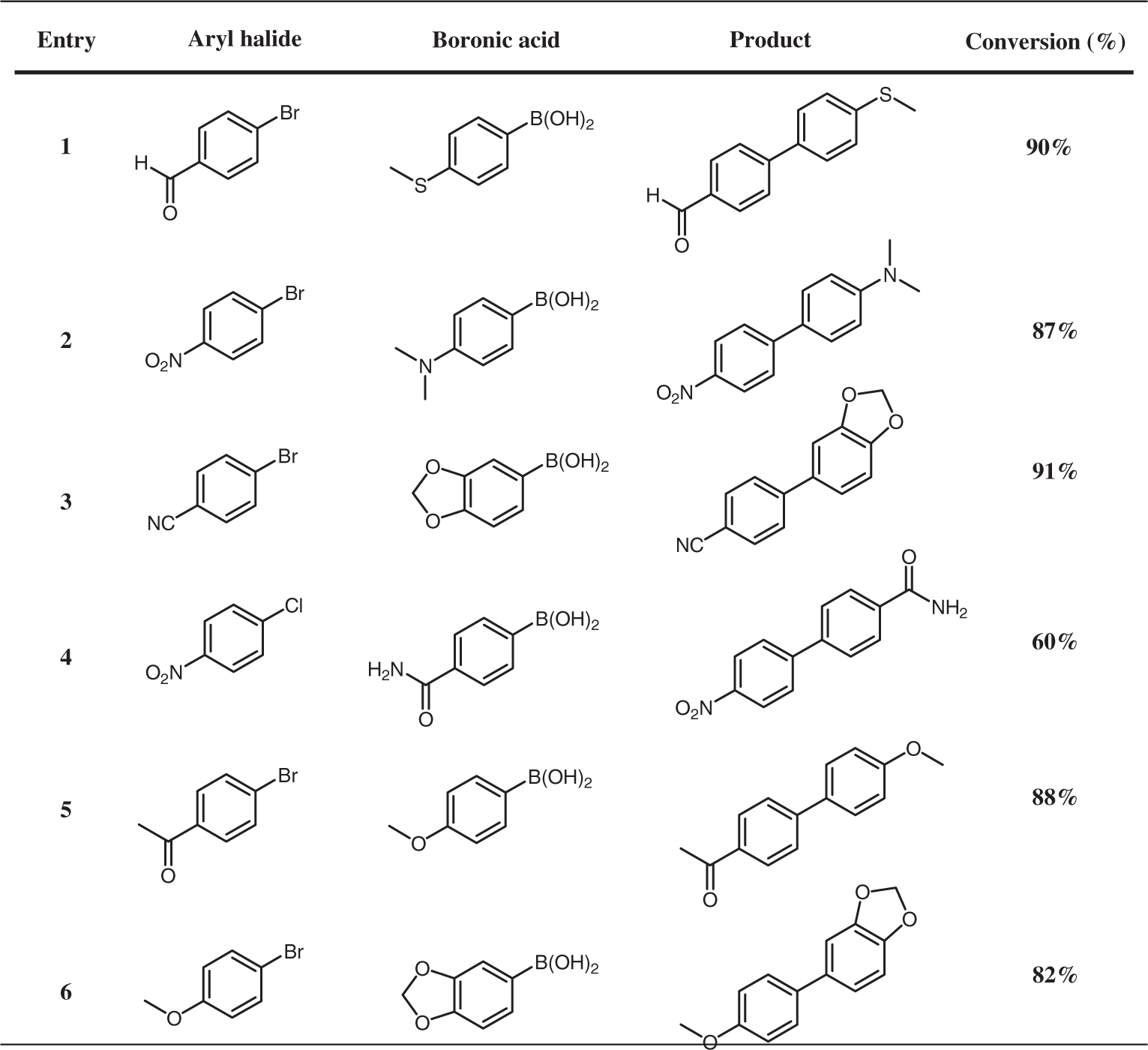
aAryl halide (0.32 mmol), aryl boronic acid (0.382 mmol, 1.2 eq.), potassium carbonate (0.96 mmol, 3 eq.), and Pd/CuO (1 mol%) in 4 ml (H2O:EtOH) (1:1) were heated at 150°C (MWI) for 10 min.
bConversions were determined by GC–MS.
From the TEM images in Figure 1, the well dispersion of palladium nanoparticles of average size (20–40 nm) is obviously noticed as in Figure 1(a) and the histogram shown in Figure 2. The TEM images here can be used as an evidence of the high catalytic activity in case of prepared Pd/CuO when compared with the same catalyst after the fifth run which is probably due to the negative effect of the agglomeration of the particles that took place after reaction as shown in Figure 1(b).

TEM images of Pd/CuO. (a) Before the reaction, (b) after fifth run of the reaction.

The corresponding histogram of Pd/CuO nanoparticles.
Figure 3 displays the XRD diffraction pattern of palladium supported on copper oxide that was prepared by microwave method. The exact palladium content in microwave synthesized Pd/CuO catalysts prepared with different palladium loading weight percent was determined by means of inductively coupled plasma optical emission spectrometry (ICP-OES) and it was found to be 3.5, 6.5, 10, 18 wt% Pd corresponding to prepared 5, 10, 20, 30 wt% of Pd/CuO catalysts, respectively. Further characterization of the microwave synthesized palladium supported on copper oxide catalyst (Pd/CuO) was achieved by XRD pattern of catalyst sample as seen in Figure 2. A sharp diffraction peak was also easily noticed at 2θ = 40° which is characteristic to palladium. The XRD reflections of CuO match that of JCPDS no. 48-1548 corresponding to monoclinic structure (Nasrollahzadeh et al., 2015a, 2015b). The diffraction peaks are ascribed to the (110), (111), (112), (202), (112), and (113) planes of copper oxide NPs as shown in Figure 2 (Elazab et al., 2014, 2017b, 2015).

XRD pattern of Pd/CuO nanoparticles.
The XPS technique is widely used as a more accurate and reliable technique for the chemical analysis of surface oxides than XRD. In Figure 4(a) and (b), samples reveal the existence of copper oxide. The XPS shows that the binding energy of Cu 2P3/2 was located at 933.1 eV and the binding energy of Cu 2P1/2 was located at 953.1 eV, showing that copper was found as Cu2+. There is also shake-up satellite peaks located at 941.9 and 961.7 eV. Also, the binding energy of Pd 3d3/2 was 340.1 eV and Pd 3d5/2 was 334.8 eV, as evidence that the Pd was present as Pd0. Also, the binding energy of Pd 3d5/2 was 336.23 eV and Pd 3d3/2 was 341.38 eV, showing that the Pd was found as PdO (Pd2+) (Elazab et al., 2014, 2017b, 2015).
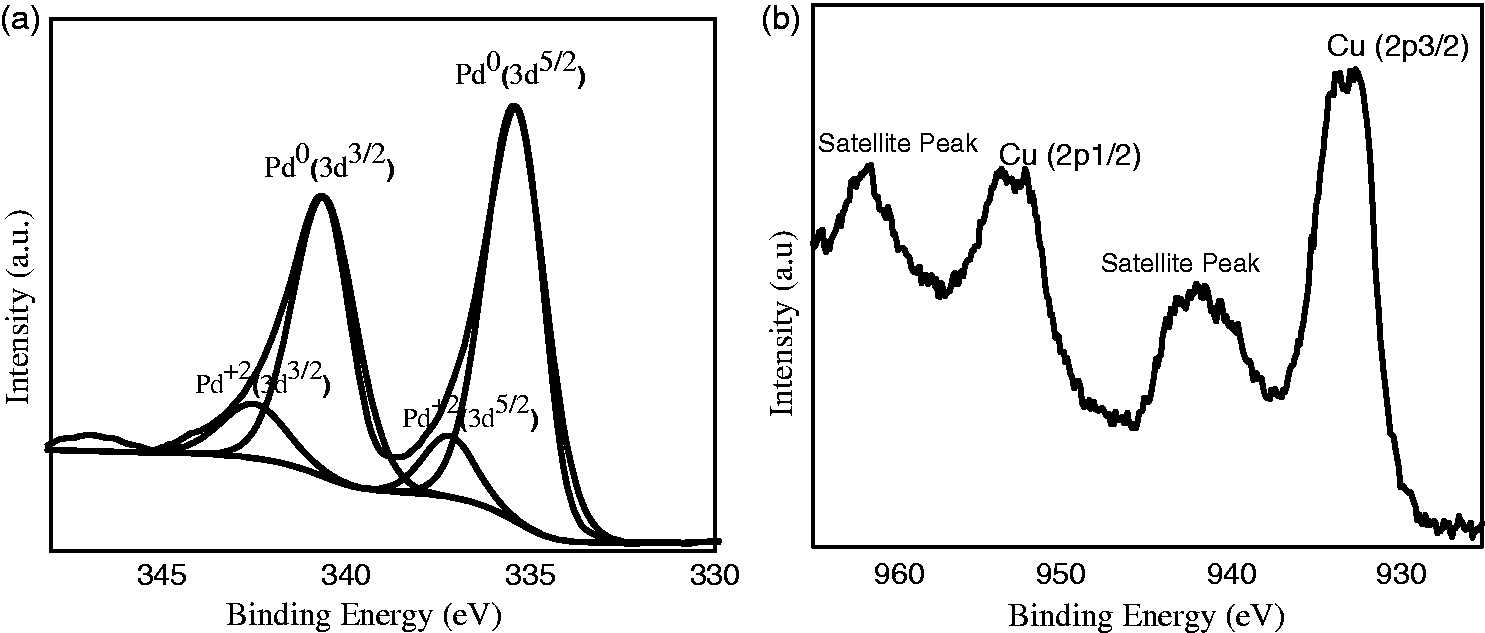
(a) XPS (Pd3d) and (b) XPS (Cu2p) of Pd/CuO.
The results of catalyst recycling were previously discussed in “General procedure for catalyst recycling” section and are summarized in Table 5. As seen in Table 5 and Figure 5, that catalyst has an excellent catalytic activity. For example, a catalyst with 0.5 mol% could be recycled up to three times with nearly 100% conversion. Also, by using 1 mol%, the catalyst was recycled up to five times with high conversion near 100%.
Recycling experiments for Pd/CuO catalyst using a concentration of 0.5, 1 mol%. a

aBromobenzene (50 mg, 0.32 mmol), boronic acid (47 mg, 0.382 mmol, 1.2 eq.), potassium carbonate (133 mg, 0.96 mmol, 3 eq.), and Pd/CuO (0.5 mol%) or (1 mol%) in 4 ml (H2O:EtOH) (1:1) were heated at 150°C (MWI) for 10 min.
bConversions were determined by GC–MS.

Recycling experiments of 0.5 mol% Pd/CuO catalyst for Suzuki cross-coupling reaction heated at 150°C (MWI) for 10 min. Note: Bromobenzene (50 mg, 0.32 mmol), boronic acid (47 mg, 0.382 mmol, 1.2 eq.), potassium carbonate (133 mg, 0.96 mmol, 3 eq.), and Pd/CuO (0.5 mol%) in 4 ml (H2O:EtOH) (1:1) were heated at 150°C (MWI) for 10 min. Conversions were determined by GC–MS.
TEM images of Pd/CuO can support the opinion that the deactivation process that took place after the fifth run is clearly due to the negative effect of agglomeration and accumulation of both Pd and CuO nanoparticles on the surface of the catalyst as shown in Figure 1(b). The leaching of palladium from the catalyst was tested by performing the reaction in the presence of 0.5 mol% catalyst at 150°C for 10 min under microwave-assisted synthesis heating conditions. Once the reaction is completed, the Pd content was determined in the filtrate to be 184 ppm based on the ICP-MS analysis.
In general, the results reported here are in good agreement with many reported data; however, our research findings have some advantages including using a simple synthetic protocol, shorter reaction time, mild reaction conditions, and environmentally benign solvent system if compared with other research reports (Chattopadhyay et al., 2009; Hoseini et al., 2017; Hosseini-Sarvari and Razmi, 2015; Mandali and Chand, 2013; Nasrollahzadeh et al., 2015a, 2015b).
Conclusions
In summary, we developed a simple and efficient synthetic protocol to highly active Pd nanoparticle catalysts supported on copper oxide matrix using microwave irradiation. The adopted MWI approach represents a simple and fast route to implement a smooth synthetic process where no high temperature or high pressure is needed. The synthesis of the catalyst is based on the chemical reduction of the corresponding aqueous mixture of palladium nitrate and copper nitrate salts using hydrazine hydrate as reducing agent. The synthesized Pd/CuO bimetallic catalyst was fully characterized and found to have average size of 20–40 nm. In addition, its catalytic performance was examined in catalyzing Suzuki cross-coupling reactions in 50% aqueous ethanol as green solvent using microwave heating and the results were compared to that of the unsupported Pd nanoparticles. The superior catalytic activity of Pd/CuO catalyst emphasizes the crucial role played by the CuO solid support in preventing nanoparticles agglomeration. The prepared Pd/CuO catalyst was found to be stable showing excellent conversion percents within 15 min at 150°C. Furthermore, it could be simply recovered and recycled up to five times with negligible loss in performance or catalytic activity under batch reaction.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We gratefully express our deep gratitude to The British University in Egypt (BUE) for supporting this work through Young Investigator Research Grant (YIRG-2016) awarded to Dr Hany A Elazab.
