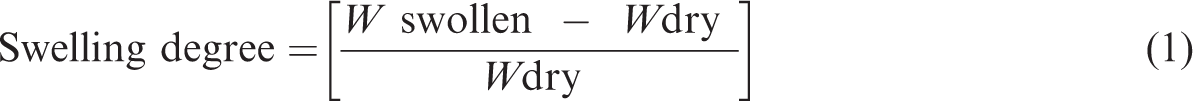Abstract
The present study describes an easy and efficient procedure for the purification of horseradish peroxidase from horseradish roots. For this purpose, supermacroporous cryogels having Concanavalin A were prepared by photosensitive cross-linking polymerization. Horseradish peroxidase binding and elution from the prepared cryogels were carried out changing various parameters such as initial peroxidase concentration and pH. The best binding performance was obtained at pH 7.0. The maximum horseradish peroxidase binding of the cryogels was found to be 3.85 mg g−1 cryogel. Horseradish peroxidase purification from crude extract resulted in 115.1-fold. SDS-PAGE analysis and circular dichroism measurements indicated that the horseradish peroxidase purification from horseradish roots was successfully carried out.
Introduction
Horseradish peroxidase (HRP, E.C. 1.11.1.7) is a plant enzyme that is responsible for the catalysis for oxidation of various substrates. HRP is widely used in different application areas such as removal of phenolic contaminants from wastewater (Dalal and Gupta, 2007; Kim et al., 2006; Lai and Lin, 2005; Song et al., 2003; Tonegawa et al., 2003), dye decolorization (Jadhav et al., 2009), peroxide removal from food samples (Hamid and Rehman, 2009). Some other reported applications of HRP in industry have also been demonstrated (Krieg and Halbhuber, 2003; Veitch, 2004). Horseradish (Armoracia rusticana) roots contain the most abundant peroxidase and therefore it is the primary source of industrial HRP production.
HRP purification has been reported by several research groups using different techniques such as two-phase partitioning (Miranda et al., 1995, 1998), reverse micelles (Regalado et al., 1996), ion exchange chromatography (Gautério et al., 2015), dialysis and gel filtration chromatography (Khurshid et al., 2012). However, all these traditional approaches have some limitations such as less selectivity, multi-step processes and high cost. Thus, the development of rapid and cost effective processes and technologies with higher selectivity and less process steps is needed.
On the other hand, microbeads are used in conventional packed-columns which have considerable disadvantages such as slow diffusional mass transfer and low flow-rates. Cryogels, a type of interconnected supermacroporous gel materials prepared at sub-zero temperatures, can overcome these problems by providing high flow rates and low back pressure due to the continuous large pores (Arvidsson et al., 2003; Bereli et al., 2013; Çimen et al., 2016; Ersöz et al., 2014; Lozinsky et al., 2001, 2003).
ANADOLUCA approach used in this work is a conjugation and cross-linking technique by using photosensitive amino acid monomers (Say, 2011; Say et al., 2011, 2012). The most important advantages of this approach, compared to traditional immobilization techniques are the elimination of chemical activation of affinity support and ligand immobilization. All of these support activation techniques suffer from some disadvantages such as: (i) need of anhydrous conditions, (ii) toxic chemicals are used and (iii) immobilization process is time consuming. In addition, activation of the support and ligand immobilization increase the total cost of the protein purification process.
In ANADOLUCA method, there is no Concanavalin A (Con A) leakage due to cross-linking of protein to cryogel matrix through photosensitive monomers. In addition, the developed method in this study is facile, straightforward, cheap and does not require toxic chemicals. ANADOLUCA has already been used for the purification of proteins and antibodies such as albumin and immunoglobulin G from human plasma (Uzun et al., 2013).
Con A is a lectin purified from the jack bean. Unlike the other lectins, Con A is a metalloprotein and specifically binds glycoproteins such as HRP through mannosyl and glucosyl residues of the protein. This binding requires calcium ions and a transition metal such as manganese. Each subunit of Con A binds one Ca2+ ion and one Mn2+ ion. The binding of Ca2+ and Mn2+ ions to the subunits of Con A (unlocked form) leads to the re-organization of the ion binding site of the subunits. The conformation change from unlocked to locked form accompanying the ion-bound Con A is governed by a trans-to-cis isomerization of the Ala-207:Asp-208 peptide bond (Bouckaert et al., 2000). This leads to increase in glycoprotein binding activity of Con A. So far, several reports on HRP purification have been published (Fraguas et al., 2004; Guo and Ruckenstein, 2003; Mohamed et al., 2011), but until now there has been no reported study on the photosensitive cross-linking of Con A to the cryogel materials for the purification of HRP.
In this study, Con A cross-linked supermacroporous cryogels for HRP purification from horseradish roots were prepared by using ANADOLUCA photosensitive cryo-polymerization. The prepared cryogels were characterized and HRP binding to the cryogels was analyzed and discussed.
Materials and methods
Materials
Horseradish peroxidase, Con A, Acrylamide, N,N′-methylenebisacrylamide, tris(hydroxymethyl)aminomethane, 2-hydroxyethyl methacrylate (HEMA), Sephadex G-25, sodium chloride, calcium chloride, magnesium chloride, manganese chloride, Coomassie Brilliant Blue R-250 and all organic solvents were supplied by Sigma-Aldrich (Steinheim, Germany). Sodium phosphate dibasic, citric acid, hydrochloric acid, glycine, and glycerol were purchased from Merck (Darmstad, Germany). Sodium dodecyl sulfate, H2O2 and (NH4)2SO4 were purchased from Panreac (Barcelona, Spain).
Farm cultivation of horseradish as plant material was carried out in Eskisehir, Turkey.
Instrumentation
Elemental analyses were performed using Vario El III elemental analyzer and NMR analyses were carried using Bruker Avance III 500 MHz spectrometer. FT-IR analyses were carried out using a Perkin Elmer Spectrum 400 spectrometer. SEM analyses were performed using Zeiss ULTRA plus SEM and SDS-PAGE analyses were carried out using Peqlab PerfectBlue Dual Gel Twin S Electrophoresis System.
Synthesis of the photosensitive functional monomer bis(2,2-bipyridyl) MATyr-MATyr-ruthenium (II)) (MATyr-Ru(bipyr)2-MATyr)
For this purpose, firstly, the methacryloylamido tyrosine (MATyr) monomer was synthesized applying the published procedure (Yavuz et al., 2007). Then, the functional monomer MATyr-Ru(bipyr)2-MATyr was synthesized using a reported recipe (Evans et al., 1973) briefly described as follows:
0.1 g of RuCl2(bipyr)2 (1 eq.) was added in 10 mL deionized H2O. Then, triethylamine and 0.1 g of MATyr in 10 mL deionized H2O were slowly added in the mixture and allowed to stir for 30 min. The final solution was stirred for 26 h at 80°C. The obtained functional monomer was filtered off, washed with EtOH, H2O and dried under vacuum. M.p.: >200°C. The structure of the functional monomer MATyr-Ru(bipyr)2-MATyr is shown in Figure 1.

The structure of the functional monomer MATyr-Ru(bipyr)2-MATyr.
Preparation of Con A cross-linked poly(HEMA-co-Con A) cryogels
Con A as co-monomer cross-linked macroporous cryogels poly(HEMA-co-Con A) was synthesized by photosensitive cryo-polymerization. For this purpose, 650 µL of HEMA was added to the solution of 0.15 of N,N′-methylenebisacrylamide in 5 mL of water and 1 mg Con A in 3 mL H2O was slowly added into the mixture. Then, 75 µL of MATyr-Ru-MATyr photosensitive monomer was added to the mixture and allowed to stir for 3 h at N2 atmosphere. After addition of APS (10 mg) and TEMED (12.5 µL), the final solution was placed into a 10 mL plastic disposable syringe with closed outlet and allowed to polymerize at −20°C for 17 h. Then, the frozen mixture is thawed at room temperature. Finally, the synthesized cryogel was washed with EtOH and deionized H2O to remove undesired impurities and stored at +4°C. The schematic depiction of the poly(HEMA-co-Con A) cryogels is shown in Figure 2.

Schematic depiction of poly(HEMA-co-Con A) cryogel.
A control cryogel without Con A was prepared using the same protocol.
Characterization of the photosensitive functional monomer and poly(HEMA-co-Con A) cryogels
The synthesized photosensitive functional monomer was characterized by elemental analyses and NMR spectroscopy. The prepared cryogels were dried at 50°C in the oven before characterization tests. The characterization of the synthesized cryogels was carried out by FT-IR, SEM and swelling experiments. For FT-IR analyses, the dried cryogel (ca 10 mg) was placed on the surface of ATR crystal and the spectrum was then recorded in ATR mode.
For the SEM analyses, the dried cryogels were covered with a 20 nm gold to provide conductivity. Then, SEM pictures were obtained.
The swelling of the synthesized cryogels was also tested since it is a crucial factor for the binding performance of the cryogels towards the target enzyme HRP. For this purpose, the dried cryogels were interacted with deionized H2O in a 25 mL-baker at room temperature for 3 h. Then, the swollen cryogels were taken out of the baker, wiped with filter paper and weighed. The swelling degree of the prepared cryogels was determined using equation (1):
Preparation of crude extract
32.5 g of horseradish root was cut into slices and homogenized in 100 mL of 20 mM Tris–HCl buffer, pH 7.0. Then, the homogenates were centrifuged at 14,000 r/min for 45 min at +4°C. After filtration of the supernatant using a 0.45 µm membrane filter, the filtrate was stored as crude extract in 0.01% NaN3 at +4°C.
Ammonium sulfate precipitation
The precipitation of the proteins in the crude sample was carried out by using (NH4)2SO4 at +4°C. For this purpose, 47 g of (NH4)2SO4 was slowly added to 99 mL of crude extract and stirred for 2 h at +4°C. The precipitate was collected after centrifugation at 15,000 r/min for 30 min at +4°C and dissolved in a least volume of 20 mM Tris–HCl buffer, pH 7.0. The remaining (NH4)2SO4 in the medium was removed by Sephadex G-25 gel permeation column. For this purpose, a certain amount of Sephadex G-25 was suspended in 20 mM Tris–HCl buffer, pH 7.0 (bed volume: 20 mL per g) and allowed to swell for 5 h at 90°C. Then, 2 mL of protein sample was pumped to the column at 2 mL min−1 flow rate and protein fractions were collected.
HRP binding and elution studies
HRP binding end elution experiments were performed using a column system. The prepared cryogel was washed with 40 mL of deionized H2O and then equilibrated with loading solution (20 mM Tris–HCl buffer, pH 7.0). The HRP solution was then pumped to the column at 1 mL min−1 flow rate for 30 min. The HRP binding was spectrophotometrically determined by measuring the decrease in protein concentration at 280 nm. Then, the cryogel was washed with 50 mL of loading solution. The interaction between Con A and HRP is competitively inhibited by using a sugar such as mannose. Thus, the bound HRP was completely eluted from the cryogels using 30 mL of 0.5 M mannose. The effect of pH, initial HRP concentration on binding capacity and reusability of the cryogels were also studied.
Peroxidase activity studies
Peroxidase activity assay was performed by using the slightly modified procedure (Shindler et al., 1976) as described in the following:
The activity assay was carried out by using the substrate 0.5 mM ABTS in 50 mM acetate buffer pH 4.5, 10 µL of HRP and 2.9 mM H2O2 in a final volume of 150 µL. Peroxidase activity was determined by the monitoring the increase in absorbance at 405 nm. One unit of peroxidase activity (IU) is defined as the enzyme amount which catalyzes the oxidation of 1 µmol of ABTS/min.
SDS-PAGE analysis
The purity of lyophilized HRP purified by using poly(HEMA-co-Con A) cryogels was checked by SDS-PAGE using the reported protocol (Laemmli, 1970) as described in the following:
Analyses were performed by using 15% resolving gel and 5% stacking gel under denaturing and reducing conditions. Protein staining was performed with Coomassie Brilliant Blue.
Circular dichroism (CD) spectroscopy measurements
CD measurements of commercial and purified HRP were performed using an Applied Photophysics Chirascan CD Spectropolarimeter. For this purpose, 500 ppm HRP solution in 20 mM Tris–HCl buffer, pH 7.0 was prepared. Spectropolarimetric analyses were performed using a quartz cuvette with a 1 mm path length. Each spectrum was scanned two times at 1 nm intervals with an integration time of 0.5 s and the average spectrum was obtained.
Results and discussion
Characterization of the photosensitive functional monomer MATyr-Ru(bipyr)2-MATyr and poly(HEMA-co-Con A) cryogels
Elemental composition of the synthesized photosensitive functional monomer was determined by elemental analysis. As can be seen from the obtained results in the following, the experimental values are compatible with the theoretical values.
On the other hand, NMR spectroscopy was also used for the characterization of the functional monomer MATyr-Ru(bipyr)2-MATyr. According to the obtained NMR signals as given in the following, the doublet signal observed at 8.56 ppm confirms the bipyridyl group in the structure. The doublet signal at 5.3 ppm indicates the terminal methylene (–CH2) group of the MATyr. The triplet signal observed at 1.8 ppm confirms the protons of neighbor –CH group and the doublet signals observed at 7.18 ppm and 7.1 ppm confirm the protons exist on the tyrosine ring of the MAT.
1H NMR (500 MHz, CDCl3), ppm: 9.7 (d, 4H,3J = 5.08 Hz), 8.56 (d, 4H,3J = 7.92 Hz), 7.92 (t, 4H,3J = 7.82 Hz), 7.3 (t, 1H,3J = 7.4 Hz), 7.23 (t, 2H,3J = 7.47 Hz), 7.2 (d, 2H,3J = 7.46 Hz), 7.1 (d, 4H,3J = 6.30 Hz), 5.3 (d, 1H,2J = 1.52 Hz), 1.8 (t, 1H).
Figure 3 shows FT-IR spectra of the prepared cryogels. As can be seen from the figure, all cryogels showed very similar patterns due to the similarity of the cryogel backbone. The peaks due to C=O group stretching (∼1720 cm−1), C–H stretching (∼2935 cm−1) and C–N stretching (∼1035 cm−1) were observed and the peaks at ∼1090 cm−1 and ∼750 cm−1 confirmed the presence of benzene ring in the obtained spectra.

FT-IR spectra of the cryogels.
The SEM images of the poly(HEMA-co-Con A) cryogels are shown in Figure 4. As shown in the figure, the cryogels have macropores (up to 100 μm) which provide continuous interconnected macropores for the solvent to flow through and convective migration of the HRP molecules. These macropores also provide lower back pressure compared to conventional monolithic materials.

SEM images of poly(HEMA-co-Con A) cryogel.
The swelling degrees of the cryogels were found to be as 9.17 g H2O/g cryogel and 10.25 g H2O/g cryogel for the poly(HEMA-co-Con A) and poly(HEMA) cryogels, respectively.
HRP binding studies
The effect of pH on binding
Determination of the optimum pH was performed at different pH values in the range of 3–9. The effect of pH on HRP binding to the poly(HEMA-co-Con A) cryogels is shown in Figure 5. The maximum binding of HRP was achieved at pH 7. Therefore, the pH 7 was chosen as the optimum value for the further studies. Con A consists of identical subunits of molecular weight 26.5 kDa and each subunit has a single binding site for a glycoprotein. The pH significantly affects association of these subunits.

Effect of pH on HRP binding. CHRP: 100 ppm, mcryogel: 175 mg. Flow rate: 1 mL min−1.
Studies on the three-dimensional structure of Con A (Wang et al., 1971) indicate that it exists as a tetramer form at pH 7 with four binding sites that exhibit strong affinity towards glycoproteins and the major contribution to organized structure is to be the β conformation. At lower pH values, Con A dissociates into dimer form that has less binding sites compared to tetramer form. Above pH 7, the conformation of Con A approaches the random coil or unordered form (Zand et al., 1971).
The effect of initial HRP concentration on binding
Figure 6 shows the effect of initial HRP concentration on the binding of HRP onto the poly(HEMA-co-Con A) cryogel and the control cryogel poly(HEMA). The amount of bound HRP onto the cryogels increased when HRP concentration increased and a saturation value was achieved at 1.0 mg mL−1 HRP which shows the saturation of the binding groups of the cryogels. The maximum binding capacity of the poly(HEMA-Con A) cryogels towards HRP was found to be as 3.85 mg g−1.

Effect of initial HRP concentration on binding. mcryogel: 175 mg, pH: 7.0. Flow rate: 1 mL min−1.
Reusability of the cryogels
The reusability of the poly(HEMA-co-Con A) cryogels was also tested. For this purpose, binding and elution cycle for HRP was repeated 20 times. The results from the reusability experiments of the cryogels are shown in Figure 7. As seen from Figure 7, the cryogels are stable after 20 cycles.

Reusability of the cryogels. CHRP: 1.5 mg mL−1, mcryogel: 175 mg. pH: 7.0. Flow rate: 1 mL min−1.
HRP purification from horseradish roots
The outcomes of the overall HRP purification from horseradish roots are shown in Table 1. As shown in the table, HRP was purified by using poly(HEMA-co-Con A) cryogels with a 115.1-fold purification which is much higher than other results in previously reported studies as shown in Table 2 and a 17.27 units.mg−1 protein specific activity.
Purification of peroxidase from a crude extract of horseradish (Armoracia rusticana) roots.

Comparison of peroxidase purification from horseradish (Armoracia rusticana) roots by different research groups.

The purity of HRP eluted from poly(HEMA-co-Con A) cryogels was checked by SDS-PAGE analysis. SDS-PAGE image of purified HRP from horseradish roots and commercial HRP is given in Figure 8. As seen from the image, the eluted HRP from the cryogel migrated as a single band (lane 1).

SDS-PAGE image of purified HRP from horseradish roots (lane 1) and commercial HRP (lane 2).
CD measurements of the free and purified HRP were also carried out to get information about the conformation of the HRP backbone. Figure 9 shows the CD spectra of free and purified HRP. The positive peak at 192 nm which indicates the primary structure of HRP enzyme is corresponded to the π–π* transition of the amide groups of HRP, the peaks at 208 and 222 nm belong to the π–π* and n–π* amide transition of peptide chain in HRP, respectively, and assigned to α-helix secondary conformation of HRP (Chang et al., 1978; Myer, 1968). The spectra in Figure 9 confirmed that secondary structure of purified HRP enzyme has not been destroyed during the purification process.
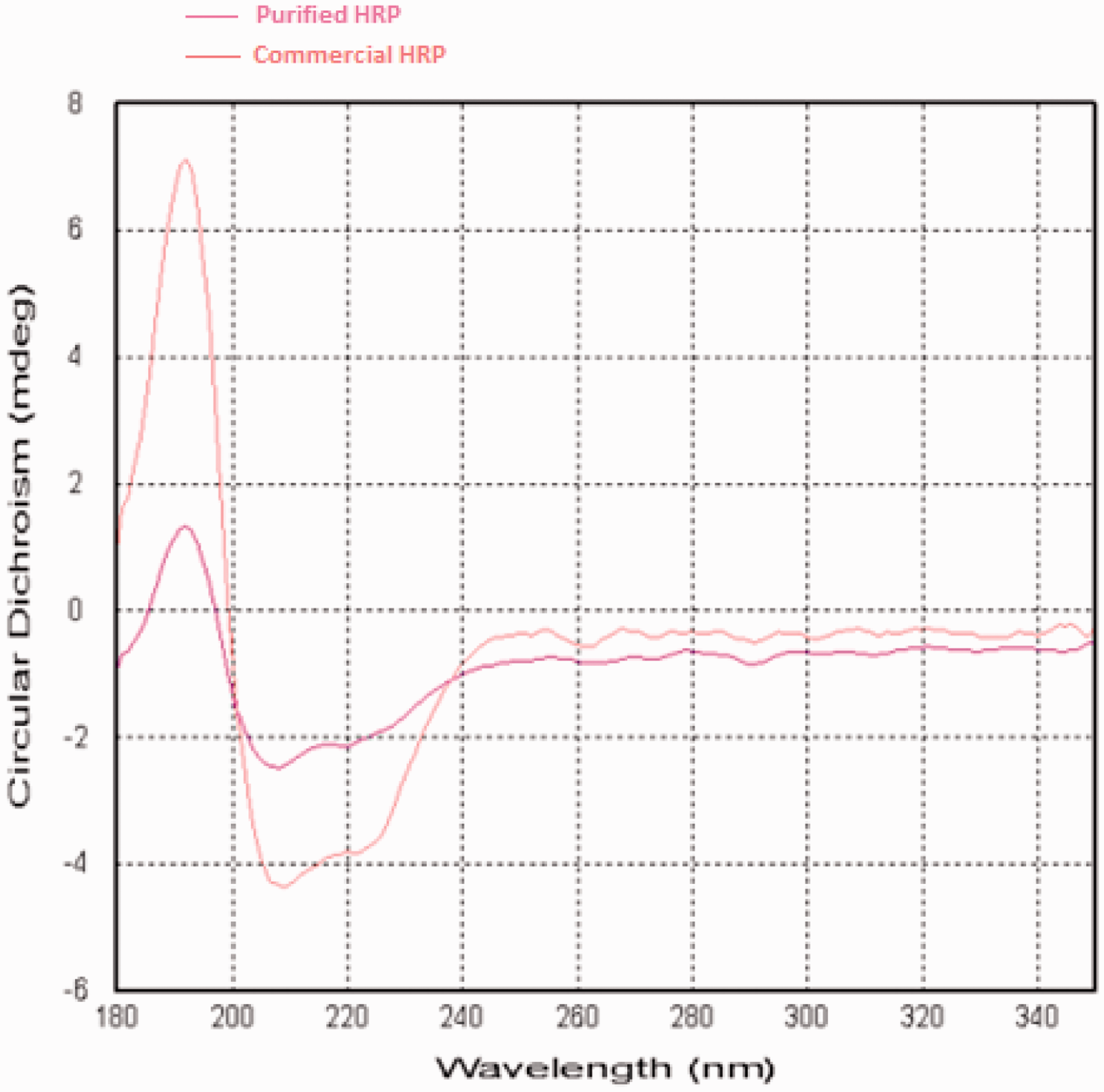
CD spectra of purified HRP and commercial HRP. CHRP commercial: 0.5 mg mL−1, CHRP purified: 0.2 mg mL−1.
Conclusions
In this work, we have described a facile and effective approach for selective purification of HRP from horseradish roots. The obtained results showed that the prepared poly(HEMA-co-Con A) cryogels exhibited high selectivity towards HRP without any potential leakage of monomer and maintained their stability even after 20 cycles. The prepared cryogels have also considerable advantages such as low back pressure, high flow rate through the interconnected pores and short diffusion path compared to traditional packed-bed chromatography materials. The developed method in this study is straightforward, cheap and does not require toxic chemicals. As a concluding remark, the prepared Con A cross-linked cryogels are efficient affinity materials for the purification of HRP from horseradish roots.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Scientific and Technological Research Council of Turkey (TÜBİTAK) (Project Number: 1130019).