Abstract
Herein, we demonstrate a simple and cost-effective method to prepare the new hierarchically Ni-doped porous CaCO3 monoliths in a large scale by mineralizing finger citron residue templates with a calcium acetate precursor. The morphology, microstructure, and element composition of as-prepared adsorbents are characterized by Scanning Electron Microscope (SEM), X-ray Photoelectron Spectroscopy (XPS), Transmission Electron Microscopy (TEM), and N2 adsorption, respectively. Adsorption performance of anionic dye Congo red was investigated in a batch experiment. The results showed that pseudo-second-order kinetic model and Langmuir adsorption isotherm matched well for the Congo red adsorption. Compared with previously reported adsorbents, due to positive and negative charge effect between Congo red and Ni, Ni-doped porous CaCO3 monoliths demonstrated a superior Congo red dye adsorption capability. The results of the present study substantiate that Ni-doped porous CaCO3 monoliths is a promising adsorbent for the removal of the anionic dyes from wastewater.
Introduction
Nowadays, preparation of functional porous and micro-sized materials has received significant attention due to their applications in both of energy and environmental applications (Boyjoo et al., 2014; Peng et al., 2014). The porous- or micro-sized particles not only offer significantly high surface area and volume for better mass and energy transfer but also provide opportunity for chemical reactions in a controlled manner (Fang et al., 2016). Moreover, the material can be dispersed in small quantities into other materials' matrices so as to improve their chemical and physical properties. As one of the most common and abundant minerals on the earth, calcium carbonate (CaCO3) nanoparticles have been widely studied in many fields (Colfen, 2003). For instance, it can act as important fillers/modifiers in paints, plastics, rubber, and paper industry (Guo et al., 2006). In medicine, it not only can act as a host matrix for the construction of highly sensitive biosensors (Shan et al., 2007) but also is taken as a therapeutic agent carrier for drug delivery (Ueno et al., 2005). In particular, in tissue chemical engineering, porous CaCO3 has been considered as one of potential adsorbents for environmental pollution control (Yu et al., 2011). Especially, meso structures are advantageous to for pollutants with large molecular sizes because of their relative large pore sizes (2–50 nm) and high surface areas and pore volumes (Jiang et al., 2016). Therefore, it is a challenging project for obtaining mesoporous CaCO3 materials at different sizes and with exotic morphologies.
Nature creates a diversity of biologically exquisite micro/nanostructures that has long attracted great interests for scientists (Xu et al., 2007). These fascinating structures with hierarchical and porous characters have been employed as templates to reproduce biomorphic functional materials (Zhang et al., 2008). Currently, a large variety of biological species such as butterfly wings (Losic et al., 2007), diatoms (Yang et al., 2010), bacteria (Hu et al., 2012), proteins, etc. (Zoe et al., 2010) have been used as templates. However, collecting and treating these templates are rather intricate, and sometimes require some special training. Plant leaf, possessing a feature of hierarchical and porous structure, as a kind of renewable and accessible biomass, has been used as template to synthesize metal carbide (Li et al., 2009), metal oxide (Huang et al., 2014), and noble metals (Hu et al., 2013).
In this study, finger citron residue (FCR) was selected as a template for the synthesis of hierarchically porous CaCO3 materials in this work. Finger citron (Scheme S.I. 1), a subtropical plant, grows widely in Fujian, Zhejiang, Guangdong, and Sichuan provinces of China (Gong et al., 2013). A lot of FCRs are produced during the finger citron beverage processing. As waste materials, the FCRs have to be disposed as garbage, which can also sometimes cause another environment problem (Gong et al., 2013). Given its porous texture and the fact that it contains various organic compounds, FCR is an optimal precursor for the preparation of porous material and it can also be potentially applied in environmental protection. On the other hand, as one of the typical anionic dyes, Congo red (CR) is a benzidine-based anionic disazo dye (Hu et al., 2014). The discharge of CR into the aquatic ecosystem has generated much concern due to its reported genotoxic, mutagenic, teratogenic, and carcinogenic effects (Gong et al., 2013; Yu et al., 2015). Therefore, hazardous anionic dyes such as CR containing effluents have to be adequately treated before they are discharged into the environment.
In the present study, FCR as template for the first time, we describe a simple and cost-effective method for the controlled synthesis of hierarchically porous CaCO3 monoliths (PCMs) via a biomimetic method. The mesoporous structure of PCM is further activated with nickel ions for removal of CR dye from aqueous solutions. The equilibrium and kinetic data of adsorption studies were modeled using the adsorption isotherm models Langmuir, Freundlich, and the adsorption kinetic models (pseudo-first-order, pseudo-second-order, and intra-particle diffusion models). The possible mechanism of the adsorption process is also discussed in this study.
Materials and methods
Materials
FCRs were collected from the Jinshoubao Biological Technology Co., Ltd, Jinhua, China. Nickel nitrate (Ni(NO3)2) was purchased from Sigma Chemical Company (St. Louis, MO, USA). High purified nitrogen (99.99%) was provided by Gas Supplier Center of Datong Co., Ltd, Jinhua, China. All the other reagents (Ca(CH3COO)2, HCl, NaOH, etc.) were purchased from the Sinopharm Chemical Reagent Co., Ltd.
PCMs preparation
The FCRs were first washed with deionized water to remove dirt from their surface and were then dried overnight in an oven at 378 K. The dried husk was cut and sieved to desired mesh size (∼150 µm). Then the sample (50 g) was washed by using NaOH (1 M) and HCl (1 M), respectively. The resulting samples were subsequently immersed in the Ca(CH3COO)2 solution for 12 h. Then, the sample was dried at room temperature. The calcination was carried out at different temperatures for 3 h. Incipient wetness technique was applied to impregnate PCMs with an aqueous solution of nickel nitrate. The amounts of Ni(NO3)2used for modification were precalculated to achieve the loading of nickel equal to 20, 30, or 40 wt%. After impregnation, the adsorbents were successively dried at 373 K for 5 h and heated nitrogen atmosphere for 3 h at 973 K. The samples obtained were denoted as Ni20@PCM, Ni30@PCM, and Ni40@PCM, respectively
Material characterization
X-ray diffraction (XRD) data were recorded from a Bruker D8 Advance X-ray Diffracto meter with CuKα radiation (
Batch sorption investigations
A series of CR solutions with concentrations ranging from 50 to 1200 mg/l were prepared by dissolving different amounts of the dye in deionized water. In each batch experiment, 20 mg of adsorbents were suspended in 50 ml of the dye solution. At first the time dependence of CR adsorption onto all adsorbents was investigated to determine the time required for adsorption equilibrium. After certain contact time, the resulting mixture was continuously shaken in a shaking bath at a constant temperature (298 ± 1 K). Finally, the samples were filtered and the residual concentrations of CR in the filtrate were analyzed by a UV–Visible spectrophotometer (Thermo Fisher Evolution 300 PC) at maximum wave lengths of 497 nm. It was found that the calibration curves were very reproducible and linear relation between the concentration and absorbance over the concentration range were found in this work. The adsorbed amount of CR at equilibrium, 
Results and discussion
Material characterization
The nitrogen adsorption isotherms and the pore size distribution curves of Ni@PCMs series are shown in Figures 1 and 2 with the related parameters listed in Table S.I.2, respectively. The isotherms of the Ni@PCMs samples confirm the nature of mesopore and are of type IV isotherm with two condensation steps in the mesopore range according to the IUPAC (International Union of Pure and Applied Chemistry) classification (Deng et al., 2012; Li et al., 2014). It exhibits a H1 hysteresis loop with the capillary condensation at a relative pressure (P/P0) ≈ 0.45–0.90. With the Ni(II) amount increasing, the BET surface area and total pore volume of the Ni30@PCM (353 m2/g and 0.34 cm3/g) increased compared with Ni20@PCM (315 m2/g and 0.24 cm3/g), however decreased compared with Ni40@PCM (182 m2/g and 0.25 cm3/g), respectively. As follows from these data (Table S.I.2), the average pore diameters of Ni20@PCM, Ni30@PCM and Ni40@PCMare 3.07, 3.84 and 5.48 nm, respectively. The mesoporous structure after nickel(II) doping increased, which can suggest nickel (II) nanoparticles can probably expand the micropores of the samples into mesopores leading to the mesopores features in Ni@PCM series samples. In addition, nickel (II) nanoparticles can also probably block the pores leading to severe reduction of the surface area measured.
N2 adsorption isotherms of Ni20@PCM, Ni30@PCM, and Ni40@PCM at 77 K, respectively. Ni@PCM: Ni-doped porous CaCO3 monoliths. Pore size distribution of Ni20@PCM, Ni30@PCM, and Ni40@PCM, respectively. Ni@PCM: Ni-doped porous CaCO3 monoliths.

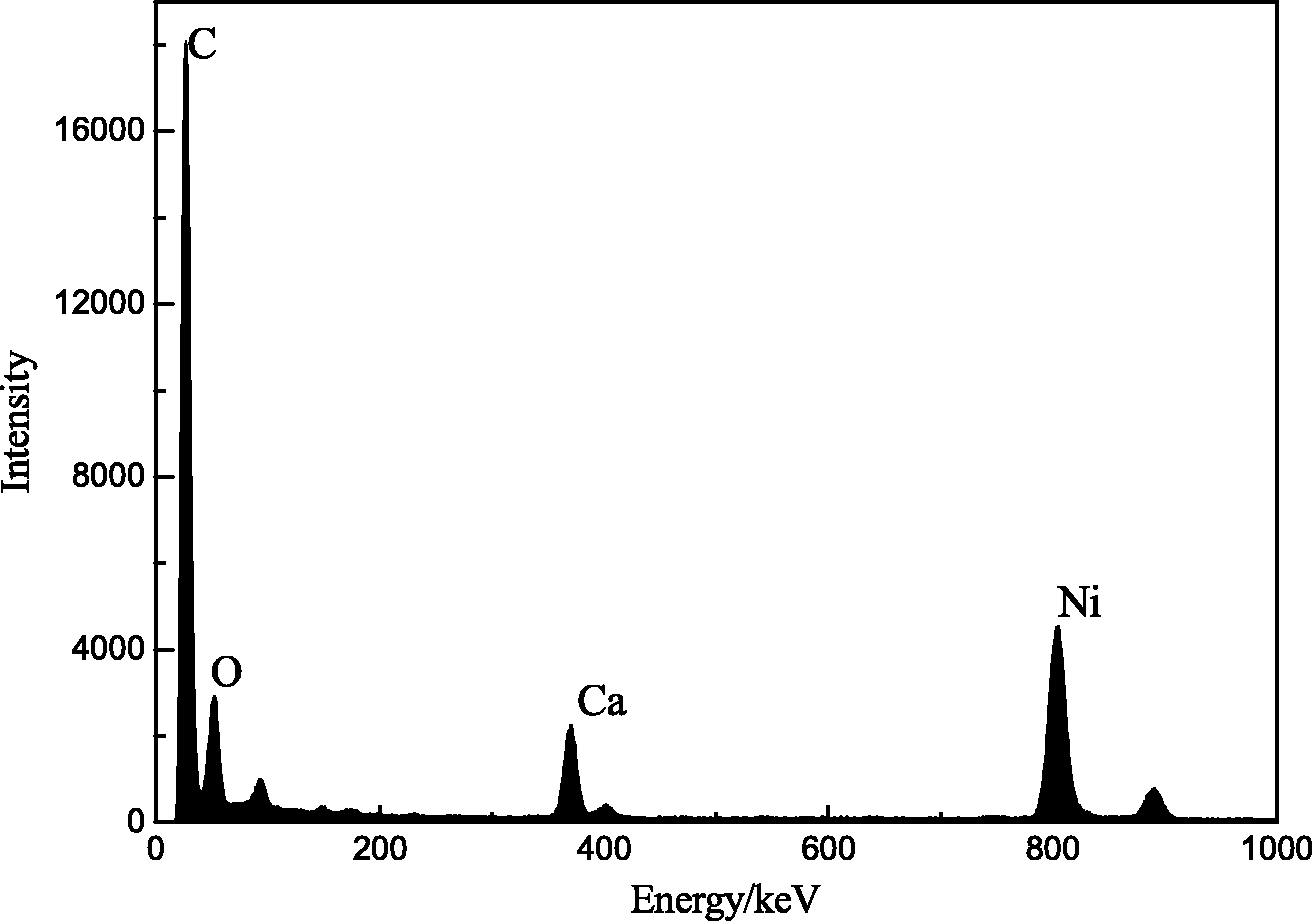
The SEM micrograph of Ni30@PCM is presented in Figure 3(a) to analyze its surface shape. The surface of Ni30@PCM particles showed cavities, pores and more rough surfaces due to the Ni(II) activation process. The surface area of the Ni30@PCM will be enhanced by the presence of more porosity, which can hold more CR dye molecules from solution during adsorption. In addition, Ni(II) can be observed to disperse onto the surface of Ni@PCM as shown in the TEM image of Ni30@PCM (Figure 3(b)). Only four elements, C, O, Ca and Ni, could be foundfrom the EDS results (Figure 4). No other impurities were detected in the nanostructures.
SEM and TEM image of Ni30@PCM sample. (a) SEM and (b) TEM. Ni@PCM: Ni-doped porous CaCO3 monoliths; SEM: Scanning Electron Microscope; TEM: Transmission Electron Microscopy. EDS image of Ni30@PCM. EDS: Energy Disperse Spectroscopy; Ni@PCM: Ni-doped porous CaCO3 monoliths.
XRD spectra (Figure 5) were used to confirm the crystal line phase of the samples, which indicates atypical crystalline character of calcite. The most intensive peak at 2 XRD of Ni30@PCM sample. Ni@PCM: Ni-doped porous CaCO3 monoliths; XRD: X-ray diffraction.
In order to obtain the surface information of Ni(II), i.e. location and distribution of nickel phases on PCMs, XPS analysis has been carried out (Figure 6). As shown in Figure 6, the XPS survey spectra of Ni30/PCM indicated the presence of three distinct peaks, which is due to nickel, oxygen, and carbon, respectively. The Ni 2p3/2 peaks centered at 855 and 856 eV can be ascribed to NiO and Ni(OH)2 (Yu et al., 2016). Presence of satellite peaks demonstrated that nickel atoms were present in oxygen environment.32 The peak at 861.6 eV was the satellite peak of Ni(OH)2.33 The survey XPS spectra corresponded to Ni 2p1/2 further confirmed the existence of element Ni.
XPS of Ni30@PCM sample. Ni@PCM: Ni-doped porous CaCO3 monoliths; XPS: X-ray Photoelectron Spectroscopy.
Adsorption isotherms
The experimental data at equilibrium between the amount of adsorbed CR ( Adsorption isotherms of CR on Ni20@PCM, Ni30@PCM, and Ni40@PCM, respectively (pH = 7, dose = 0.4 g/l, T=298 K). Ni@PCM: Ni-doped porous CaCO3 monoliths.
The linear form of Langmuir's isotherm model is given by the following equation

The well-known logarithmic form of Freundlich model is given by the following equation

In this work, adsorption isotherm experiments were carried out at initial CR concentrations of 50–1200 mg/l. The value of
The values of
It was found that after loading more nickel, the CR uptake capacity of Ni30@PCM was significantly increased in comparison with the Ni20@PCM, which is due to interaction forces between nickel and CR (anionic dye). Further increasing the nickel doping level increases the amount of the active nickel sites, which results in the better CR capacity of Ni30@PCM than Ni20@PCM. While if more nickel were introduced into the support, it may block the pore and leads to the increase of the diffusion resistance during the mass transfer reactions, which can decrease the amount of accessible active nickel sites, and thus the sorbent with higher nickel loading (Ni40@PCM) shows a smaller CR uptake capacity than Ni30@PCM. However, interaction forces other than pore properties could also contribute to the adsorption process. Thus, the CR uptake capacity of Ni40@PCM is better than that of adsorbent Ni20@PCM.
In addition, the adsorption capacity of CR on Ni@/PCM in this work is superior to some previously reported adsorbents (Table S.I.4) (Abbas and Razieh, 2010; Bhattacharrya and Sharma, 2004; Chou et al., 2001; Emrah et al., 2008; Kadirvelu et al., 2003).
Adsorption kinetics
The kinetics of adsorption is important because it controls the efficiency of the process and the time to reach equilibrium. It also describes the rate of adsorbate uptake on adsorbents. In order to identify the potential rate controlling steps involved in the process of adsorption, three kinetic models were studied and used to fit the experimental data. These models are the pseudo-first-order, pseudo-second-order, and intra-particle diffusion models. These models (Yu et al., 2016) can be expressed as



In order to quantitatively compare the applicability of different kinetic models in fitting to data, a normalized standard deviation, Δ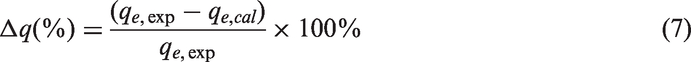
The effect of contact time on CR adsorption capacities of the Ni20@PCM, Ni30@PCM, and Ni40@PCM is shown in Figure 8. It shows that the adsorption capacities for CR increase with increasing contact time, and the adsorption reaches equilibrium within about 10 min. The saturation capacities of CR onto Ni20@PCM, Ni30@PCM, and Ni40@PCM are 99, 289, and 160 mg/g under the condition of 298 K, 1200 mg/l initial concentration, 7 pH value, and 0.4 g/l adsorbent dose. The capacity is constant when all the parameters are fixed, and with the evolution of time, the uptake or adsorbed amount may change. The fast adsorption at the initial stage may be due to the availability of the uncovered surface area and the remaining active sites on the adsorbent.
Effect of contact time on the adsorption capacities of CR on Ni20@PCM, Ni30@PCM, and Ni40@PCM, respectively (C0 = 450 mg/l, pH = 7, dose = 0.4 g/l, T=298 K). CR: Congo red; Ni@PCM: Ni-doped porous CaCO3 monoliths.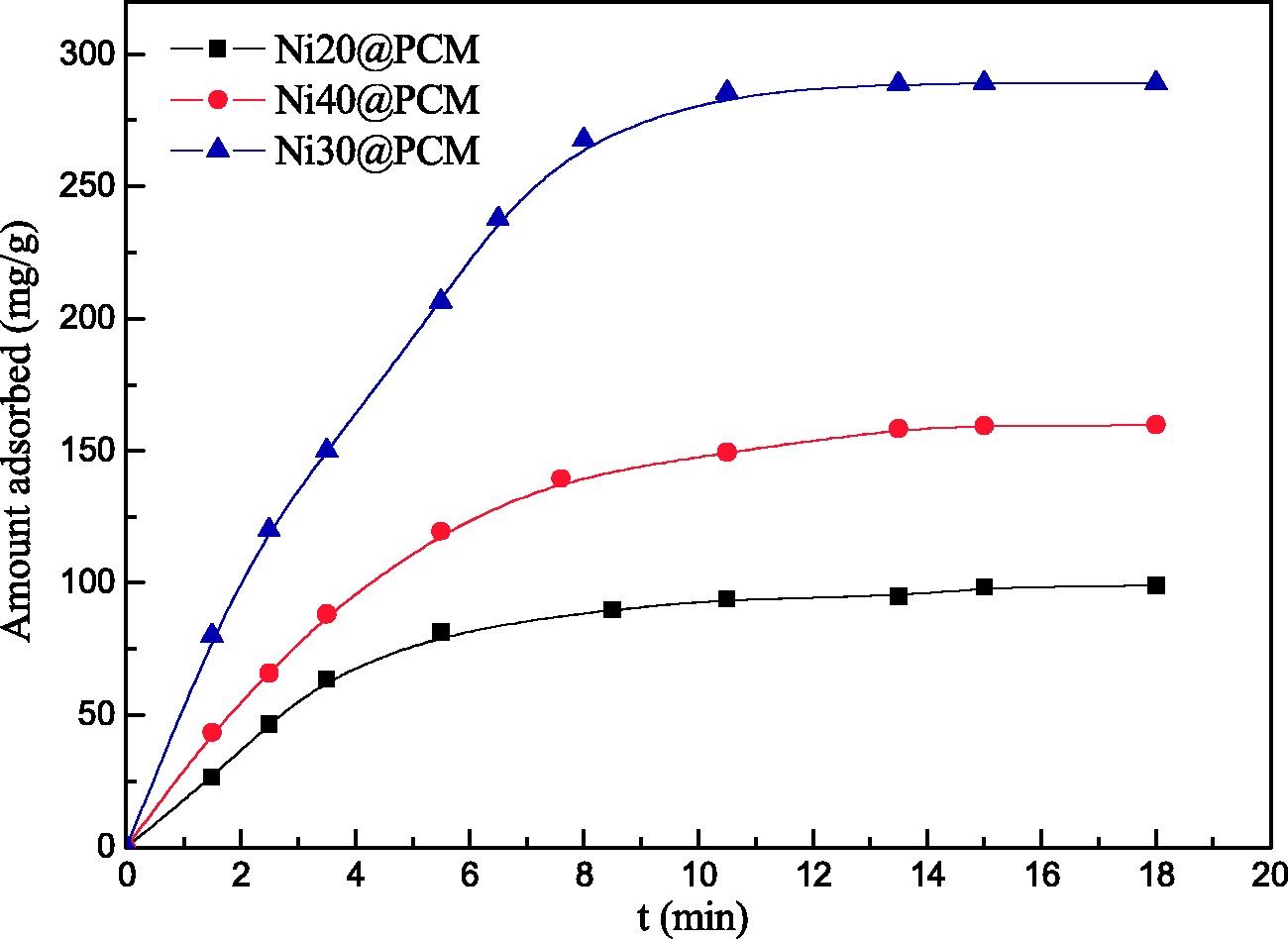
The experimental kinetic data of CR, calculated from equations (4), (5), and (6), were correlated by three kinetic models: pseudo-first-order, pseudo-second-order, and intra-particle diffusion models. The calculated constants of the three kinetic equations along with Weber–Morris intra-particle diffusion plots for the adsorption of CR on Ni20@PCM, Ni30@PCM, and Ni40@PCM, respectively. CR: Congo red; Ni@PCM: Ni-doped porous CaCO3 monoliths.
Regeneration studies
The recyclability of Ni@PCMs and the desorption efficiency of dyes are important in pollution control and environmental protection. Based on the solvent elution procedures of the literatures (Yu et al., 2015, 2016), the saturated 50Ni/PCM, which is retaining the CR dye and obtained after the adsorption experiments, was washed by NaOH and ethanol solutions, respectively. Column experiments to collect the adsorption and desorption curves were performed in a vertical quartz column of length 150 mm and inner diameter of 5 mm with a glass grid for supporting the adsorbents. The testing CR dye solution was pumped up with a mini creep pump (BT01-YZ1515, Zhejiang Nade Company, China). Prior to adsorption measurement, the adsorbent was activated by being heated to 473 K and maintained for 2 h in nitrogen stream. After cooling to room temperature (298 K) in a nitrogen stream, the sorbent was consolidated by light tapping. The adsorption experiments were carried out at 298 K and atmospheric pressure. First, the fixed bed was flushed downward with deionized water at a flow rate of 0.5 cm3/min for 30 min, and then the feed was switched to the CR dye solution with unchanged flow rate. Samples were taken regularly to examine the CR content in the solutions until saturation was reached. Adsorption–desorption curves were obtained by plotting the transient CR dye concentration versus the cumulative solution volume. The concentration was normalized with the total CR dye content in the feed, and the cumulative solution volume was normalized with the volume of the adsorbent bed. The normalized CR dye adsorption capacity of the Ni30@PCM was calculated with the following equation (8)

As shown in Figures 10 and 11, the CR desorption efficiency of Ni30@PCM was as high as 95.02% in the first adsorption–desorption cycle, which still maintained 92.02% in the fifth cycle. Meanwhile, the adsorption capacity of 5Ni30@PCM was 261 mg/g in the fifth cycle, which maintained 90.23% of the maximum CR adsorption capacity. The results demonstrated that the Ni@PCM has good recycling ability for the removal of CR anionic dye.
The adsorption capacity and desorption efficiency of CR onto regenerated Ni30/PCM. CR: Congo red; Ni@PCM: Ni-doped porous CaCO3 monoliths. Adsorption–desorption curves of CR dye over the regeneration of Ni30/PCM. CR: Congo red; Ni@PCM: Ni-doped porous CaCO3 monoliths.

Conclusion
In summary, we report a simple and cost-effective approach to prepare hierarchically porous CaCO3 monoliths by using the FCR as template. The novel hierarchically CaCO3 monoliths (Ni@PCMs) are evaluated by choosing the CR as the model adsorbate. The adsorption capacity increases with increasing initial CR concentration and percentage content of Ni(II) chloride, which indicates that both physisorption and chemisorption takes place during this adsorption process. Adsorption parameters of the Langmuir and Freundlich isotherms were determined and the equilibrium data were found to be the best described by the Langmuir isotherms. The adsorption kinetics can be successfully fitted to the pseudo-second-order kinetic model. This study highlights the potential of well-designed Ni@PCMs as highly efficient adsorbent for the removal of anionic dyes from wastewater. Besides metal carbonate, we believe that hierarchically porous metal carbide as well metal oxide can be prepared by adopting the same template and judiciously selecting calcinations conditions.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Zhejiang Provincial Natural Science Foundation of China under Grant No. LY16B060002 and National Undergraduate Innovative Training Project of China (2017022).
