Abstract
Mechanochemical modification of low-dispersed SnO2 and high-dispersed SnO(OH)2 powders with specific surface area 6 and 198 m2 g−1 have been studied. The crystal, porous, and electronic structure of obtained products, as well as their photocatalytic activity under visible light in the process of dyes degradation has been investigated. It has been established that high-dispersed powder is porous and poorly crystalline. Its specific surface area decreases after milling. On the contrary, low-dispersed powder is almost nonporous and well crystallized. Its specific surface area increases but degree of crystallinity reduces as a result of milling. Milling of both powders in water leads to formation of meso-macroporous structure. The narrowing of band gap and increase of adsorption of visible light occur after modification of low-dispersed sample. As a result, photocatalytic activity of milled powders under visible irradiation significantly increases. Maximal activity showed high-dispersed sample milled in air and water.
Keywords
Introduction
Tin(IV) oxide and hydroxide are important versatile materials, which have high chemical stability and excellent physical–chemical properties. For example, they are applied as adsorbents (Misak et al., 1992; Nilchi and Dehaghan, 2013), catalysts (Sergent et al., 2002), materials for gas sensors (Saraladevi et al., 1999), etc. Besides, SnO2 as semiconductor oxide has been used as photocatalyst for degradation of organic compounds, including dye pollutants of water. The applications of SnO2, including as photocatalyst, depend on specific surface area (particle size), porous and crystal structure, electronic properties, particularly band gap value. Mechanochemical treatment (MChT) is a perspective technique for regulation of these characteristics. For example, MChT enables to improve photocatalytic properties of oxides through introduction of “intrinsic” defects into their structure (Baláž et al., 2013; Boldyrev, 2002; Buyanov et al., 2009; Lamelas, 2004; Šepelák et al., 2012). But this aspect is insufficiently studied for tin dioxide (Al-Hamdi et al., 2014; Cukrov et al., 2001; Kersen, 2002; Kersen and Sundberg, 2003; Lamelas, 2004; Manzato et al., 2014; Senna et al., 2015; Yang et al., 2004; Ye et al., 2014). The listed works are devoted to study of other physical–chemical properties and applications of tin mono- and dioxide. The changes in specific surface area, porosity and electronic parameters, photocatalytic activity are not investigated here. Therefore, the aim of this work is investigation the possibility for regulation of porous and crystal structure, electronic characteristics of SnO2 powders via their milling. The study of photocatalytic properties of milled powders under visible irradiation is other important aim.
Experimental
Two commercial powders were used: high-dispersed porous powder SnO(OH)2 (series I) with specific surface area S = 198 m2 g−1 and low-dispersed nonporous powder SnO2 (series II) with specific surface area S = 6 m2 g−1. These samples were subjected to mechanochemical treatment in air and water at 300, 600, and 850 r min−1 for 0.5 h using a planetary ball mill “Pulverisette-7” (“Fritsch,” Germany).
Physical–chemical parameters of initial and modified samples were determined using the following techniques. XRD analysis was done on a diffractometer Philips PW 1830 (Netherlands) with CuKα-radiation. Curves of differential thermal analysis (DTA–TG) were obtained using apparatus Derivatograph-C (“F. Paulik, J. Paulik, L. Erdey,” Hungary) in the range of temperature 20–800℃ at the heating rate 10° min−1. Parameters of porous structure were calculated from isotherms of low-temperature nitrogen adsorption which were measured with the help of analyzer ASAP 2405N (“Micromeritics Instrument Corp,” USA): specific surface area S, micropores volume Vmi, mesopores volume Vme using BET, BJH, and t-methods, respectively. Sorption pore volume Vs was determined at relative pressure of nitrogen close to 1.0 and total pore volume by means of impregnation of samples with liquid water. Mesopore size d was calculated from pore size distribution curves. UV–Vis spectra of powders were registered on Lambda 35 UV–Vis spectrometer (Labsphere RSA-PE-20 diffuse reflectance and transmittance accessory, “Perkin-Elmer Instruments,” USA). The testing of photocatalytic activity in visible region was carried out using degradation of safranin T (ST) and rhodamine B (RhB) in aqueous medium (0.5·10−5 mol l−1 solution, Philips LED Cool daylight lamp, 100 W, Netherlands). The calculation of photodegradation rate constants Kd was based on the concentration change of the dyes solution after establishment of adsorption–desorption equilibrium and next illumination for 10–600 min.
Results and discussion
DTA–TG curves for initial sample of series I are presented in Figure 1. According to these data, the initial sample corresponds to composition SnO(OH)2. The mass loss Δm in the temperature range 20–200℃ was attributed to physically adsorbed water release. At the same time, Δm in the range 200–800℃ corresponds to removal of OH groups. Experimental value Δmexp is 10.31% w/w for second stage. On the other hand, the theoretical mass loss Δmtheor according to equation
DTA–TG curves for SnO(OH)2 initial. DTA–TG: differential thermal analysis and thermogravimetry. The data of DTA–TG for milled powders SnO(OH)2. DTA–TG: differential thermal analysis and thermogravimetry; MChT: mechanochemical treatment.

Sample of low-dispersed powder (Series II) has composition close to SnO2 since the mass of the sample remains almost constant after heating to 800℃ (no mass loss on the curve TG). Its chemical composition does not change after milling.
The diffractograms for samples of both series before and after milling in air and water at 850 r min−1 are depicted in Figure 2. The patterns at 2θ = 26.7°, 33.9°, 51.9° from (110), (101), and (211) planes are observed for all samples. These reflexes are attributed to tetragonal modification of cassiterite/rutile (JCPDS N 41-1445). As can be seen, SnO(OH)2 powders have poorly crystalline structure but SnO2 is well crystallized. The decrease of intensity for all reflexes Ihkl and their broadening take place as a result of milling. Thus, I110 for SnO(OH)2 modified samples is reduced by 3–4 times compared with initial sample but crystallite size D110 increases from 2.6 to 3.6 nm. In turn, reflexes intensity also decreases 1.2–2.9 times for modified SnO2 powders, but the size of crystallites is also reduced. For example, D110 is equaled 47.4 and 22.8 nm for initial SnO2 and that milled at 850 r min−1 in air, respectively (Table 2). Therefore, MChT causes aggregation of crystallites for high-dispersed SnO(OH)2 and dispersion of crystallites for low-dispersed SnO2.
XRD for SnO(OH)2 initial (1) and after MChT in air at 850 r min−1 (2), in water at 850 r min−1 (3), SnO2 after MChT in water 850 r min−1 (4). MChT: mechanochemical treatment; XRD: X-ray diffraction. The data of XRD for milled powders SnO(OH)2 and SnO2. MChT: mechanochemical treatment; XRD: X-ray diffraction. I(110)—intensity (a.u.). D(110)—average crystallite size in the direct of (110) crystallographic plane (nm). β—full width at half maximum (degree).

The obtained isotherms of nitrogen adsorption–desorption relate to different types: they are close to type I and V for samples of series I and II, respectively. They contain hysteresis loop of H3 type in the region of high P/P0 values in the second case (Figure 3) which corresponds to filling of large pores. Increasing of the mesopores volume after MChT of low-dispersed sample indicates this (Table 3). Other parameters of porous structure for samples of both series are also presented in Table 3. Thus, total pore volume VƩ and sorption pore volume Vs for initial SnO(OH)2 are equaled 0.10 cm3 g−1 and adsorption isotherm indicates a high content of micropores Vmі. Dry milling causes decrease of VƩ and VS as well as Vmі and consequently specific surface area S. At the same time, the magnitudes of these parameters almost do not change after milling in water. However, formation of secondary porosity represented by macropores occurs in the latter case. Excess of VΣ over Vs shows this.
Isotherms of nitrogen adsorption–desorption and curves of PSD (insert) for samples SnO(OH)2: initial (1), and after MChT in air at 300 r min−1 (2), 600 r min−1 (3), 850 r min−1 (4) and in water at 300 r min−1 (5), 600 r min−1 (6), 850 r min−1 (7). MChT: mechanochemical treatment; PSD: pore size distribution. Tin (IV) oxide and oxo-hydroxide porous structure parameters. MChT: mechanochemical treatment.
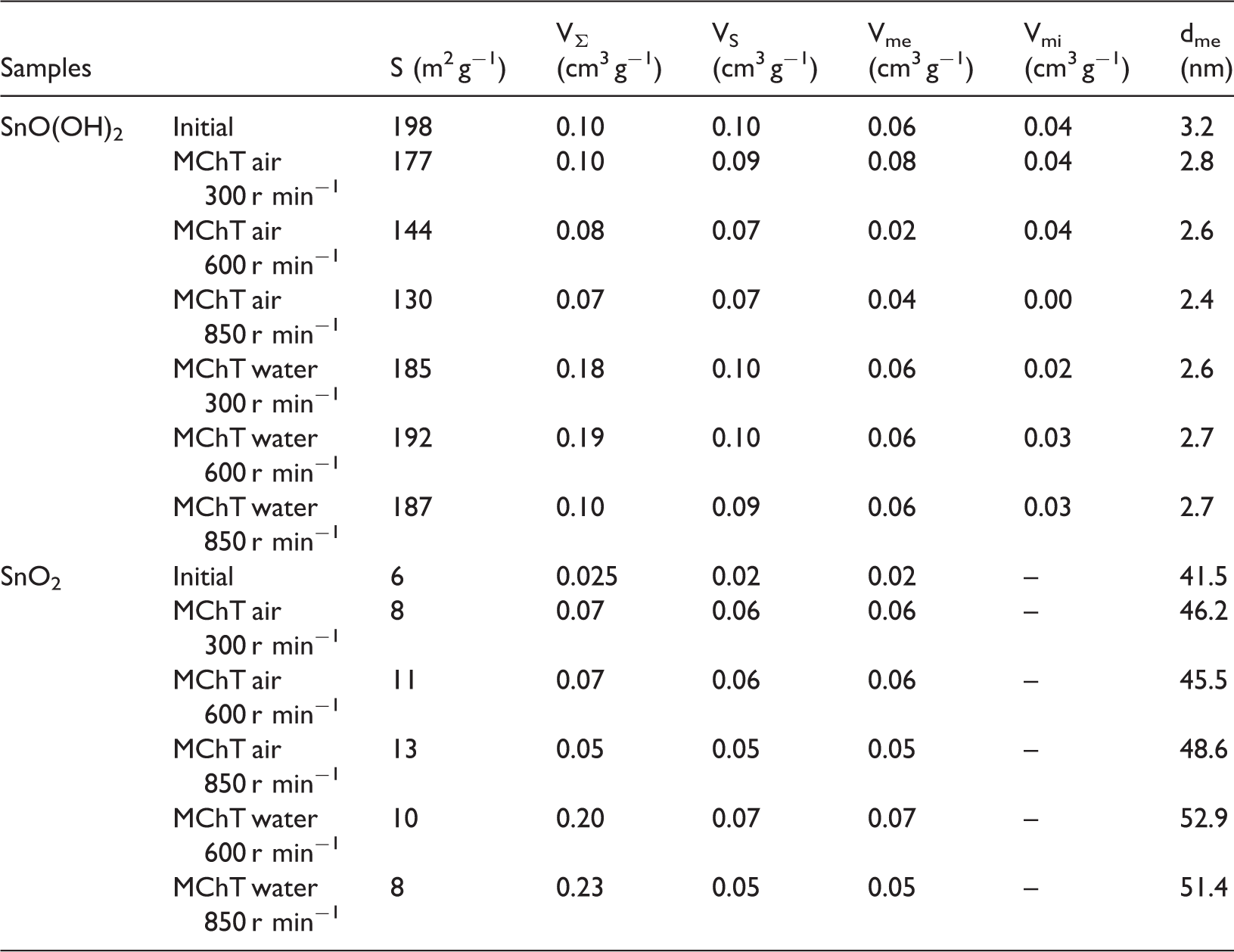
The isotherms of adsorption–desorption for samples of series II are also similar to each other. However, isotherms obtained for milled samples contain hysteresis loop shifted toward lower P/P0 values (Figure 4, curves 2, 3). This indicates the formation of mesopores, which is obviously realized through consolidation of powder particles due to mechanical action. As a result, increase of sorption and total pore volume after treatment in water additionally occurs. The latter shows the formation of secondary porosity in the form of macropores just as under milling in water for samples of series I. Thus, mesoporous and meso-macroporous structure are formed due to the dry and wet milling, respectively. In both cases, an increase in the specific surface area (Table 3) and mesopore size d (Table 3) is observed. It should be noted that set trends in specific surface area changes for both series are correlated with crystallite size calculations carried out using XRD data (Table 2) and described above.
Isotherms of nitrogen adsorption–desorption and curves of PSD (insert) for samples SnO2: initial (1) and after MChT in air at 850 r min−1 (2), in water at 850 r min−1 (3). MChT: mechanochemical treatment; PSD: pore size distribution.
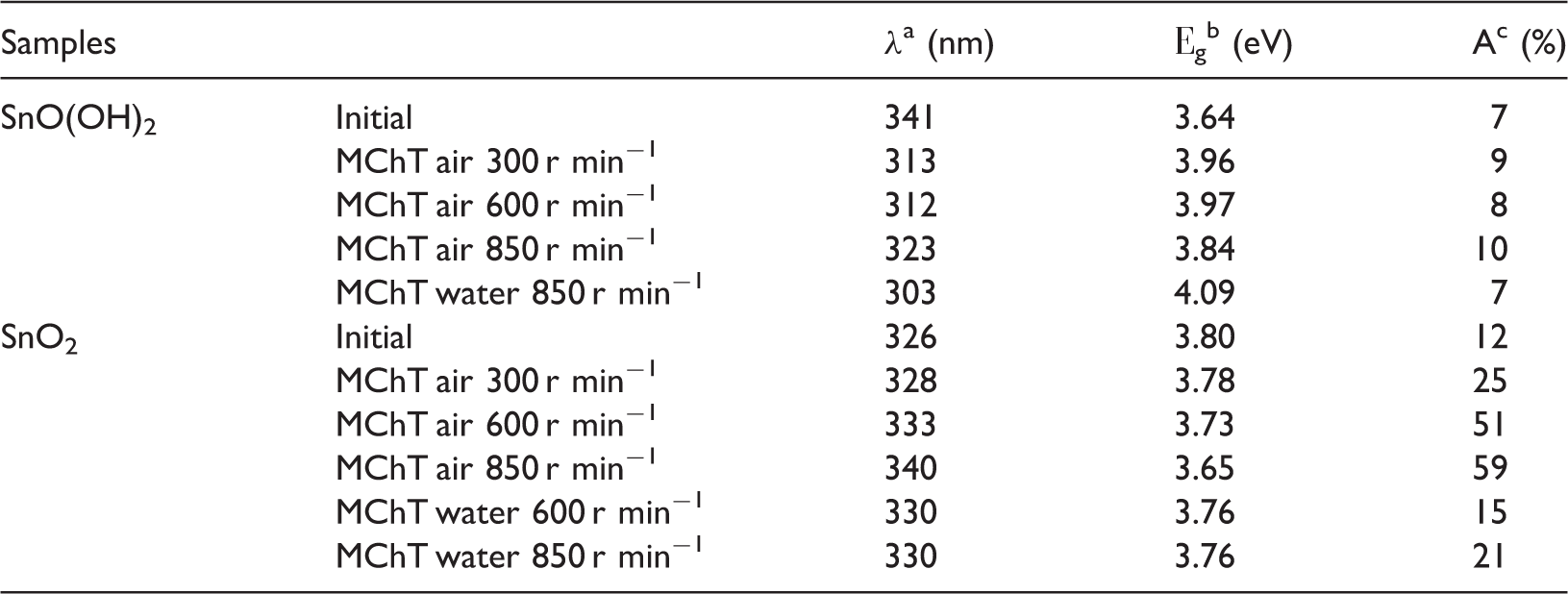
UV–Vis spectra show (Figures 5 and 6) that absorption in the visible region (> 380 nm) is almost unchanged as a result of MChT of SnO(OH)2 powders and equals about 7–10% at 500–600 nm (Table 4). At the same time, its value significantly increases for SnO2 with the growth in intensity of the dry milling: from 12% for initial sample to 59% for that milled at 850 r min−1. The bands at 265–270 nm that are characteristic of tin dioxide (He and Zhou, 2013; Sergent et al., 2002) are observed in the UV region. Their position is slightly changed after MChT (Figures 5 and 6). Besides, hypsochromic shift of absorption edge λ is observed for milled samples (Kryukov et al., 2013; Yuan and Xu, 2010): for example, from 341 nm (initial) to 303 nm (milled in water at 850 nm). The latter results in widening of band gap Eg from 3.64 to 4.09 eV (Table 4). On the contrary, a slight bathochromic shift of absorption edge occurs after milling of SnO2 powders. The narrowing of band gap from 3.80 to 3.65 eV takes place for sample milled in air at 850 r min−1. Hypsochromic shift of absorption edge in the first case can be associated with partial transformation SnO(OH)2 → SnO2 recorded with the help of DTA–TG (see above). At the same time, bathochromic shift observed for milled SnO2 can be explained by formation of structure defects (Baláž et al., 2013; Lamelas, 2004; Šepelák et al., 2012).
UV–Vis spectra of samples SnO(OH)2: initial (1), and after MChT in air at 300 r min−1 (2), 600 r min−1 (3), 850 r min−1 (4), and in water at 850 r min−1 (5). MChT: mechanochemical treatment; UV–Vis: ultraviolet–visible. UV–Vis spectra of samples SnO2: initial (1), and after MChT in air at 300 r min−1 (2), 600 r min−1 (3), 850 r min−1 (4) and in water at 600 r min−1 (5), 850 r min−1 (6). MChT: mechanochemical treatment; UV–Vis: ultraviolet–visible. Electronic properties of tin(IV) oxide and oxo-hydroxide. MChT: mechanochemical treatment. λ—absorption edge (nm). Еg—band gap (eV). A—light absorption in the visible region (%).

It is well known that photocatalytic activity, among other factors, depends on specific surface area, presence of structure (intrinsic) defects. Therefore, we studied influence of indicated factors on activity of tin dioxide in process of RhB and ST degradation in aqueous medium. RhB photodegradation in the presence of tin dioxide can occur by two pathways (Sangami and Dharmaraj, 2012; Zhu et al., 2014): as deethylation process in a stepwise manner (with the formation of the three intermediates from RhB to Rh110) or as a direct cleavage of the chromophore rings. The gradual blue shift in λmax on spectrum of RhB is observed in the first case and the reduction in the absorbance without shift takes place in the second case. It should be noted that only one work is devoted to the use of tin dioxide for RhB degradation under visible light (Wang et al., 2006).
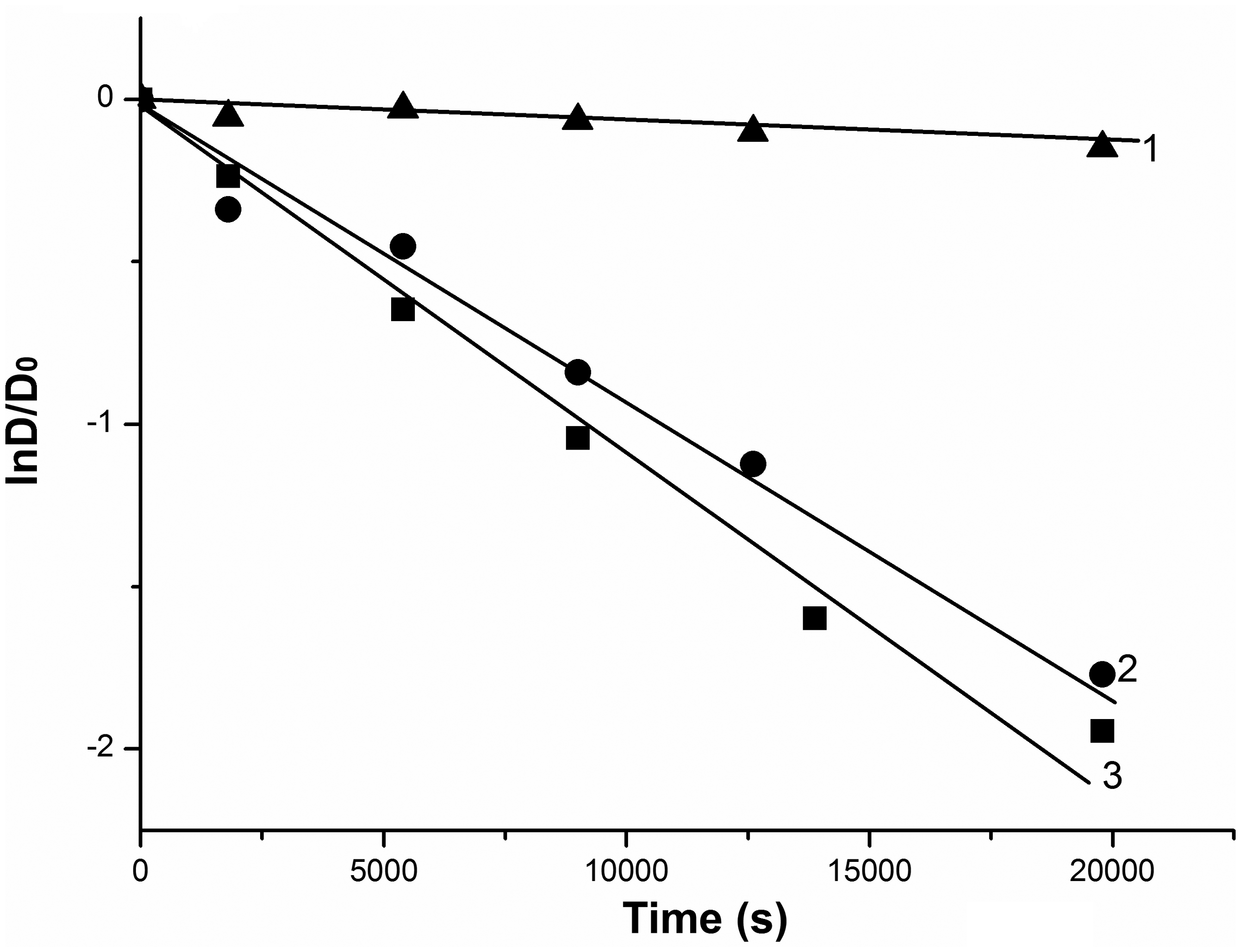
Initial low-dispersed SnO2 causes removal of only two ethyl group for 6 h and three for 10 h. Milled at 300–600 r min−1 samples somewhat accelerate this process: transformation of RhB into Rh110 (completely deethylated RhB) ends within 10 h. The samples milled at 850 r min−1 catalyze direct degradation for the first hour of illumination. Corresponding kinetic curves are presented in Figure 7. The rate constant of this process Kd is 7.3·10−5 and 6.4·10−5 s−1 for samples milled in air and water, respectively. Then (for 2–10 h), complete deethylation of undegraded RhB occurs (Merka et al., 2011). The acceleration of both deethylation and direct degradation for milled SnO2 can be associated with increase of specific surface area as well as appearance of structural defects and corresponding narrowing of band gap (bathochromic shift) and increasing of visible light absorption. The latter, as well known, is a consequence of mechanochemical processes (Lamelas, 2004).
Kinetic curves of photocatalytic degradation of RhB solutions under visible irradiation: without catalyst (1), with SnO2 after MChT in water at 850 r min−1 (2) and in air at 850 r min−1 (3). MChT: mechanochemical treatment.
As expected, high-dispersed SnO(OH)2 shows a significantly higher photocatalytic activity. The mixture of intermediates is formed for 10 min (maxima at 529 and 506 nm on spectrum, Figure 8) when sample milled in air at 850 r min−1 is used as catalyst. RhB deethylation is completed within 20 min. The degradation of Rh110 takes place on the second stage. Corresponding kinetic curves are presented in Figure 9. The Kd values for this process are equaled 1.0·10−4 and 8.3·10−5 s−1 for samples milled in air and water, respectively.
UV–Vis spectra of RhB aqueous solution after its photodegradation in the presence of SnO(OH)2 after MChT in air at 850 r min−1. MChT: mechanochemical treatment; UV–Vis: ultraviolet–visible. Kinetic curves of photocatalytic degradation of RhB solutions under visible irradiation: without catalyst (1), with SnO(OH)2 after MChT in water at 850 r min−1 (2) and in air at 850 r min−1 (3). MChT: mechanochemical treatment.
Conclusions
Milling of low- and high-dispersed SnO2 is accompanied by formation of meso-macroporous structure, introduction of defects in crystal structure, and increasing of absorption of visible light. Under these conditions, specific surface area for low-dispersed sample increases but for high-dispersed sample, on the contrary, decreases. Photocatalytic properties of SnO2 powders are improved in both cases. The main reason of photocatalytic activity growth is increase in specific surface area (for samples of both series). Narrowing of band gap and increase in absorption of visible illumination causes additional increment of activity for low-dispersed SnO2.
Footnotes
Authors’ Note
This article has been presented at the 15th Ukrainian–Polish Symposium on Theoretical and Experimental Studies of Interfacial Phenomena and their Technological Applications, Lviv, Ukraine, 12–15 September 2016.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

