Abstract
In the present work adsorption of selected phthalates from the water samples on mesoporous carbon material was investigated. Carbon material was obtained by soft-templating method in the presence of citric acid. Adsorption process of phthalates was carried out from water solution using static method with sonication for process acceleration. Phthalates extraction from water phase, after adsorption on carbon material, was carried out using solid phase microextraction method. Extracted samples were analyzed using gas chromatography coupled with mass spectrometry method. Considering obtained experimental results it can be indicated that almost 100% of phthalates were adsorbed from water solutions. The efficiency of adsorption decreases with increasing molecular weight of phthalates.
Introduction
Phthalic acid esters (PAEs) are widely used as additives in the production of plastics to make them flexible. For example, bis(2-ethylhexyl) phthalate is one of the main phthalates used as plasticizer in production of polyvinylchloride and other polymers, described by Cao (2008). Worldwide production of PAEs and their common application in different products of everyday use has resulted in their widespread presence in environment and foods (Riosa et al., 2010).
The aim of present work was the investigation of adsorption process of six phthalates: dimethyl phthalate (DMP), diethyl phthalate (DEP), di-
Experimental
MOC was obtained by soft-templating method in the presence of citric acid (C-KC) applying synthesis procedure described by Choma et al. (2013). Basic, standard parameters characterizing the mesoporous structure of studied carbon were determined on the basis of experimental, low-temperature (−196℃) nitrogen adsorption–desorption isotherms. Obtained adsorbent C-KC has the following parameters: specific surface area Scanning electron microscopy (SEM) image of mesoporous-ordered carbon.
Phthalates standard from Supelco in the form of 1 cm3 ampule containing six phthalates diluted in methanol was used. Concentration of each phthalate in standard was 2000 µg/cm3.
Carbon adsorbent was crushed mechanically and fraction of 0.40–0.63 mm was sieved. Then suitable amounts of carbon material were transferred into 15 cm3 glass vials with phthalate solutions and placed in the incubator set to 25℃ for 60 min. The platform with vials in the incubator was shaking, performing 150 round moves per minute. Adsorption process, on C-KC material, was carried out using 50, 100 and 150 mg of carbon in phthalates water solutions. Concentrations of each phthalate were 0.03/0.25 mg/dm3 and they were at low level concerning phthalates solubility, so the solutions of phthalates were not saturated.
Phthalates extraction from aqueous phase, before and after adsorption on carbon material, was carried out using solid phase microextraction method (SPME), described by Wideł et al. (2015). The 65 µm film of stationary phase polydimethylsiloxane/divinylbenzene (PDMS/DVB) was used for microextraction. Adsorption and thermal desorption, in microextraction procedure, were performed for 45 min and 2 min, respectively.
Adsorption process of selected phthalates was controlled by gas chromatography-mass spectrometry (GC-MS) method (Clarus 600 from PerkinElmer). The compounds were separated on Elite-5MS 30 m × 0.25 mm × 0.25 µm capillary column, using following temperature program: 60℃ held for 1 min, then increased at rate of 15℃/min up to 250℃ and held for 5 min. Total analysis time was approximately 19 min. Injection port temperature was 250℃, helium carrier gas ( ≥ 99.9999% purity) was maintained at a constant flow rate of 1 cm3/min. The ion source and transfer line temperature was set at 250℃. The electron ionization was 70 eV. Analysis was carried out in selected ion recording (SIR) mode (
Results
The efficiency of adsorption decreases with increasing molecular weight of phthalate (Figure 2). The exception is decreasing adsorption between DEHP and DOP despite the same molecular weight of both phthalates. It can be explained by different structure of DEHP and DOP molecules. The DOP molecule has longer chain substituent groups and this can be the reason of lower adsorption on carbon material.
Efficiency of phthalates adsorption in percentage. Time of adsorption is 60 min.
The optimum adsorption time of all studied phthalates on MOC is greater than 60 min. The exemplary adsorption kinetic curve for DBP is shown in Figure 3.
Adsorption kinetics of di-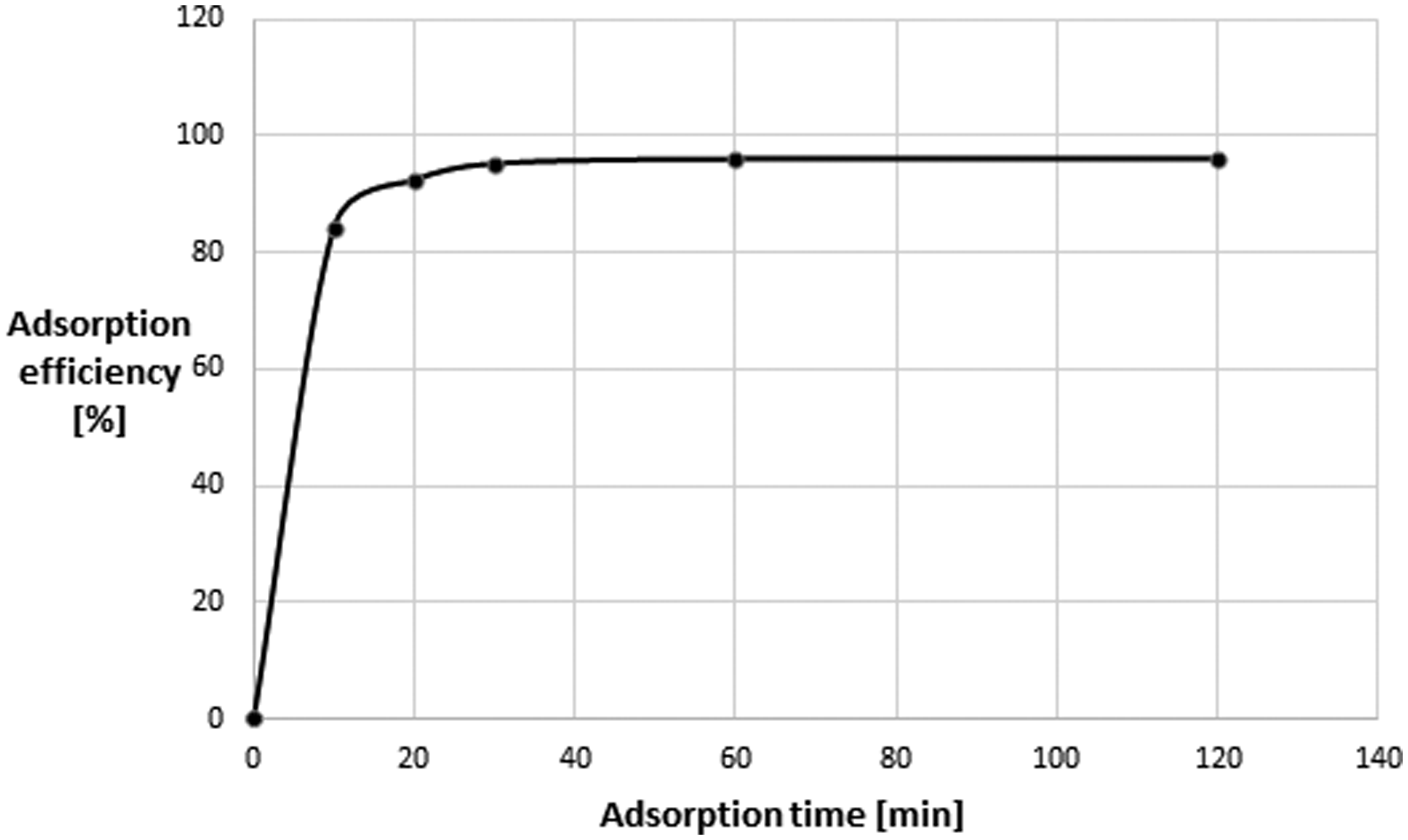
Filling the pores with DBP occurs through micropores followed by mesopores (Figure 4).
The isotherm of DBP adsorption on mesoporous-ordered carbon. Temperature 298 K. Adsorption (mg/g) vs. concentration at equilibrium state (mg/dm3).
The amount of adsorbate adsorbed at equilibrium per unit mass of mesoporous carbon material ‘
Adsorbed amount of selected phthalates on mesoporous carbon adsorbent were shown in Figure 2. The efficiency of adsorption decreases with increasing molecular weight of phthalate.
The kinetics of adsorption of DBP on mesoporous carbon adsorbent was shown in Figure 3.
According to Wang (2015), the kinetic data were further analyzed using the pseudo-second-order equation which can be expressed as

Integrating equation (2) for the boundary conditions 
Isotherm adsorption of DBP on mesoporous carbon adsorbent is shown in Figure 4. The shape of the adsorption isotherms shown in Figure 4 can provide multilayer adsorption of DBP above 0.07 mg/dm3 of equilibrium concentration.
According to Wang (2015), the linearized form of Freundlich (
The linearized Langmuir [L] equation is


The value of
The following equation indicates the Dubinin–Radushkevich [D–R] isotherm


The mean free energy of adsorption (
The magnitude of
The adsorption isotherm parameters of DBP on mesoporous carbon.

Conclusions
The efficiency of adsorption decreases with increasing molecular weight of phthalate. The kinetic data were described by the pseudo-second-order equation. The equilibrium data fitted with Freundlich isotherm are better than Langmuir and D–R isotherms. The D–R isotherm indicated a physi-sorption process of DBP adsorption through micropores filling of carbon adsorbent. However data shown in Table 1 consider the calculations for equilibrium concentrations of DBP up to 0.058 mg/dm3 and these data are related to the process of filling the adsorbent’s monolayer. Filling the pores with DBP occurs through micropores can be followed by mesopores. Mesoporous carbon adsorbent (approx. 68% of mesoporosity) was applied because it can accelerate the diffusion processes during the adsorption.
Footnotes
Acknowledgements
Authors acknowledge The National Science Centre (Poland) for support of this research under Grant DEC-2012/05/N/ST5/00246 and The Ministry of Science and Higher Education as research project BS 612 490.
Authors’ note
This study was first presented at the 15th Ukrainian-Polish Symposium on Theoretical and Experimental Studies of Interfacial Phenomena and their Technological Applications, Lviv, Ukraine, 12–15 September 2016.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
