Abstract
In this work, the adsorption of platinum (II, IV) chloride complexes from acidic solutions on silica gel modified with quaternary ammonium salts (QAS) was studied. The uptake of the platinum chloride complexes is caused by the formation of ionic (QAS+)2[PtCl x ]2− (x = 4, 6) associates on the surface of silica gel. The isotherms of adsorption are fitted by the Langmuir model. The maximum capacity for [PtCl4]2− and [PtCl6]2− is 0.99 and 1.13 mmol/g, correspondingly. The respective adsorption constants KL = 6.8 and 10 × 105 l/mol prove the high affinity of the adsorbates to the QAS-modified surface. Platinum metal nanoparticles supported on the surface of the silica gel were prepared by reducing the adsorbed platinum (II, IV) complexes. Such nanoparticles functioning at the moderate temperature regime have demonstrated a reasonable catalytic activity for the hydrogen and oxygen recombination, and an excellent stability over 35 cycles of the reaction.
Keywords
Introduction
Sorbents, in particular, modified with reagents contained N-donor atoms are today used for solid phase extraction and pre-concentration of platinum metals from acid solutions (Tarkovskaya et al., 1997). Sorbents on the base of silica gel (SG) modified with highmolecular weight quaternary ammonium salts (QAS) are found to be the most efficient for the uptake of metals in the form of the acidic complex (Zaporozhets et al., 2000). Such sorbents are stable in the acid medium due to strong electrostatic/dispersion interactions between QAS and silica surface groups. Earlier, the QAS-SG sorbents were successfully applied for the solid-phase extraction and pre-concentration of cobalt, iron, and bismuth acidic complexes before their spectroscopic determination (Zaporozhets et al., 1998, 1999). From our point of view, such ion-exchange sorbents should have an affinity to the acidic complexes of platinum metals that form in the hydrochloric acid solutions. Besides their analytical application, the sorbents can be of interest for the preparation of platinum metal nanoparticles supported on the surface of SG. Platinum catalysts are known to be required in converters recombining a gas mixture of air oxygen (O2) and leaked hydrogen (H2). Emitting from the tanks fed the power engines or fuel cells (Folkson, 2013), the hydrogen accumulates in confined spaces giving rise to fire and explosion risks (Butler et al., 2009; Molokov and Bragin, 2015). No doubt, the catalytic technology can keep the H2 level at values compatible with safety requirements (Safety of Hydrogen as an Energy Carrier [HYSAFE], 2009), and so prevents the oxyhydrogen formation (Bradley et al., 2013; Hajji et al., 2015). To enhance converters efficiency, one can use the advanced catalysts (Yatsimirsky et al., 2000; Yatsimirsky and Boldyreva, 2003) that satisfy the operation requirements influenced by the mixture composition and temperature (Zarko et al., 1999). In this context, supported platinum metal nanoparticles could be effective in this field. The synthesis of platinum catalysts includes the stage of reduction of the platinum surface complexes adsorbed on QAS-SG.
Taking all this into account, here we report on the adsorption of QAS on SG, and solid-phase uptake of [PtCl4]2− and [PtCl6]2− using QAS-SG. The activity for hydrogen and oxygen (H2–O2) recombination found for the catalysts prepared is also discussed.
Materials and methods
Quaternary ammonium salts (QAS, TSI, Japan): tetradecyl ammonium nitrate (QAS1) and bromide (QAS2) were dissolved in chloroform (Merck, 98% w/w). The static mode adsorption experiments were conducted in chloroform–hexane 1:9 (v/v) mixture: 1.000 g of silica gel (Merck SG-60, SBET = 490 m2/g) was mixed with 2.5 ml of 0.01 M of QAS chloroform solution and 37.5 ml of hexane (Sigma, 98% w/w) and then stirred for 15 min. After that, the modified sorbent (QAS-SG) was decanted and dried. The equilibrium concentration of QAS was measured by absorption at 620 nm of (QAS)2[CoII(SCN)4] extracted with chloroform–hexane.
The platinum adsorption experiments carried out at 285 K and 293 K were disclosed below. From 10.0 to 100.0 ml of 2 mg/ml of Pt (II, IV) in 2 M HCl solution was prepared by sequential dilution of standard H2PtCl6 and K2PtCl4 solutions. These solutions were prepared from H2PtCl6·6H2O and K2PtCl4 (Aldrich, A.r. reagents).
To find adsorption dependence against acidity, pH was adjusted with either 0.1 M NaOH or HCl. The ionic strength of 0.1 M was fixed with 1 M NaCl solution. The resulted mixture was stirred for 1–20 min with 0.05–0.10 g of QAS-SG. Thereafter, the adsorbate was centrifuged and dried in air. The uptake percentage (R), the capacity of sorbent (a), and the separation coefficient (D) were calculated as
The sorbents containing 0.1, 0.3, and 0.5 wt% Pt were converted into the supported metal catalysts by treating in hydrogen stream (10 vol% H2 in Ar; 50 ml/h) at below 503 K for 2 h. The catalyst activity in the H2–O2 recombination reaction was measured in a flow reactor operating at atmospheric pressure. A gas mixture of 1:20 H2/O2 (v/v) was diluted with argon and fed through a catalyst fixed-bed at the rate of 50 ml/h. The catalyst loading was ∼0.5 g/1 cm3 and the grain size was 1–2 mm. Programmed temperature regime of 273–393 K was controlled with digital thermopiles inside the reactor. The mixture composition was analyzed at the reactor outlet by gas chromatography (GC) using a Shimadzu GC 2100 instrument with a thermal conductivity detector. The kinetic studies were performed as reported earlier (Lesnyak et al., 2009). To compare the activity, a commercial 0.1 wt% Pt/Al2O3 catalyst was used as the reference. Single-point surface area (SPSA) was measured by nitrogen adsorption at 77 K. Transmission electron microscopy (TEM, JEM 1400 microscope) and scanning electron microscopy (SEM, JSM 7700 F microscope combined with energy dispersive spectrometer [EDS, Oxford Energy 350]) were used for the morphology imaging and element mapping.
Results and discussion
H-type isotherms are best characterized by the strong interaction between the SG surface and the long-chain aliphatic QAS (Figure 1). The isotherms initial branches are well plotted in Langmuir coordinates.
The isotherms of quaternary ammonium salts (QAS) adsorption for SG-60, V/m, 800 ml/g (1), 100 ml/g (2).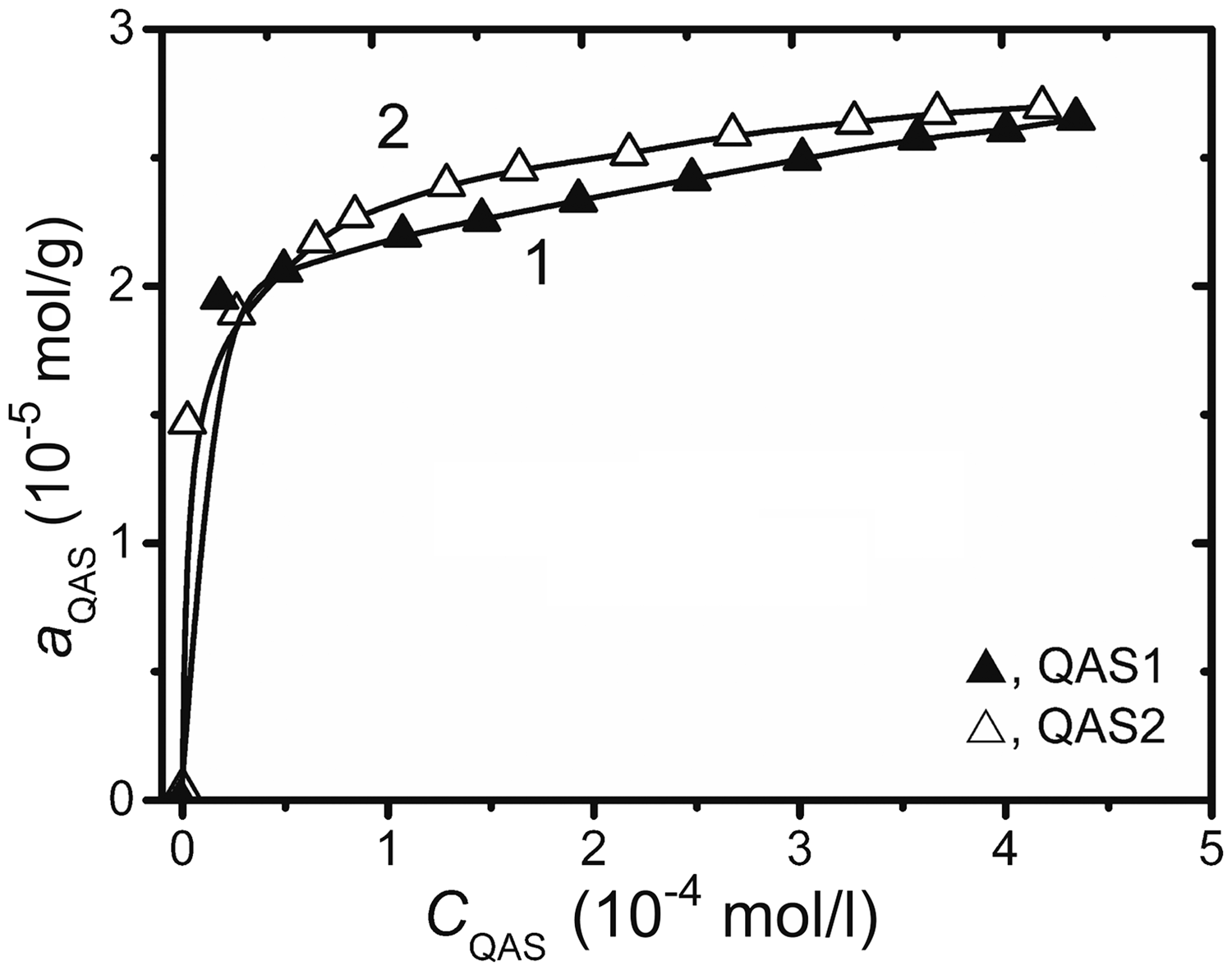
The capacity of SG for all QAS, with respect to chemisorption area of a single QAS molecule, is ∼0.1 mmol/g. And, probably, the counter ion of QAS could not affect the platinum ions uptake. The monolayer capacity of SG for QAS1 and QAS2 is practically the same, of ∼0.025 mmol/g. That is why both of the prepared samples further will be mentioned as QAS-SG. The quantitative uptake of [PtCl4]2− is reached between 2.5 ≤ pH ≤ 6.0, while [PtCl6]2−—at pH ≤ 6.0 (Figure 2).
Influence of pH (1, 2) and time (3, 4) at 293 K on adsorption of 10−5 M [PtCl4]2− (1, 3) and [PtCl6]2− (2, 4) on SG-QAS, V/m = 200 ml/g, aSG–QAS = 0.025 mmol/g, T, 285 K (1, 2), 293 K (3, 4), τc = 20 min (1, 2). SG-QAS: silica gel-quaternary ammonium salts.
Caused the partial hydrolysis of the chlorides, the optimal pH value is found to be 2.0 ± 0.1 for [PtCl4]2− and 3.0 ± 0.1 for [PtCl6]2−. The adsorption equilibrium at the optimal pH is reached for 3 and 15 min, correspondingly.
Figure 3 shows the quantitative uptake of the platinum in the form of acidic complexes (R > 90%) at V/m ∼ 1000.0 ml/g. Under this condition, the D value is ∼1.0 × 104 ml/g. Therefore, further research was conducted with 0.1 g of sorbent at the sample volume of 100 ml. The adsorption isotherms of [PtCl4]2− and [PtCl6]2− belong to high-affinity H1 and H2 types, correspondingly, the saturation is reached at aQAS:aPt ∼ (2:1). The H-type isotherms and high KL values (Table 1) indicate the strong binding of the chloride complexes of platinum.
Adsorption isotherms and uptake against V/m for 5 µmol [PtCl4]2− and [PtCl6]2− solutions, pH: 2 (1, 3) and 3 (2, 4), correspondingly, time, 3 min (3), 15 min (4). Adsorption isotherms fitting parameters. QAS: quaternary ammonium salts.

Perhaps, the platinum complexes uptake caused the formation of the surface ion associates (QA+)2[PtCl
x
]2−, where x is 4 or 6. To confirm the hypothesis, we compared the normalized and extrapolated spectra of the heterochromatic ionic associates that adsorbed on SG and extracted in the chloroform–hexane 1:4 (Figure 4).
Comparison of normalized UV-Vis absorption spectra of extracts: (QA+)2[PtCl4]2− (1), (QA+)2[PtCl6]2− (2), aqueous solutions: [PtCl4]2− (3), [PtCl6]2− (4); diffuse reflection (DR) spectra of silica gel-quaternary ammonium salts (SG-QAS) treated with [PtCl4]2− (5) and [PtCl6]2− (6); pH: 2.0 (1, 3, 5), 3.0 (2, 4, 6); V/m, 250 ml/g (5, 6); time, 2 min (5), 15 min (6), CNaCl = 0.1 M, ΔA = AI − A600.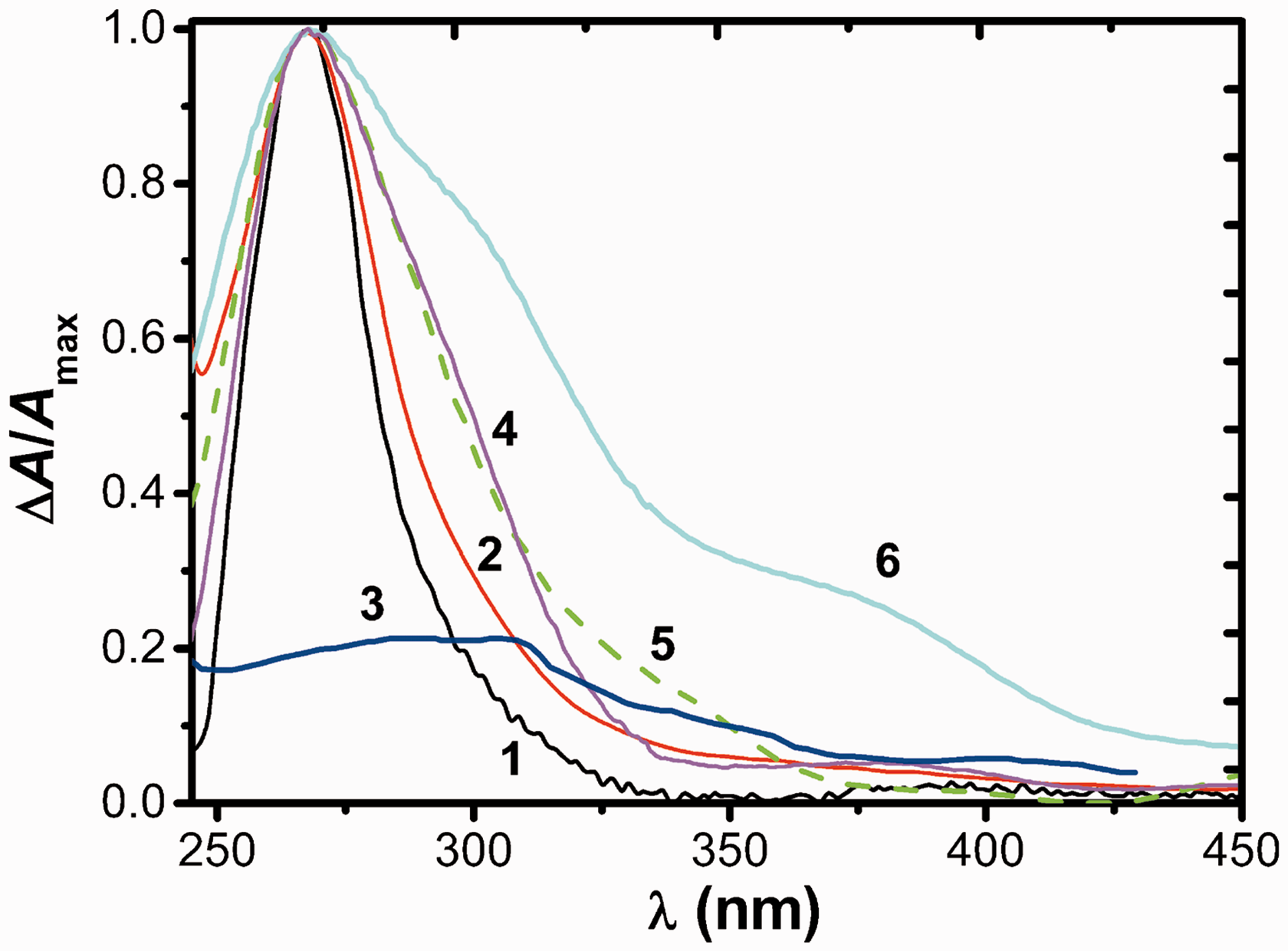
This comparison shows a good agreement between the spectra proving that QAS forms the ion associates with the platinum chloride complexes. Obviously, the pure SG does not uptake the platinum metal ions in the form of the chloride platinum complexes.
The partial destruction at the mild reduction of the surface (QA+)2[PtCl
x
]2− complexes supports the dispersed platinum metal over SG (Figure 5(a) and (b)). The samples prepared contain from 0.1% to 0.5% (by weight per total catalyst weight) of the platinum metal loadings. This shows the ability of the prepared ion-exchange substrates (QAS-SG) pre-concentrating the platinum chloride complexes from very dilute acidic solutions. Such retain of metal complexes could be a valuable technique in order to support platinum, in the form of nanoscale metal particles, to be future used for catalysis needs.
Distribution evaluation of platinum by scanning electron microscopy–energy dispersive spectrometer (SEM–EDS) element map (a) and (b) insert transmission electron microscopy (TEM) photo of 0.5 wt% Pt/silica gel-quaternary ammonium (SG-QA) catalyst; (c) H2 conversion vs. reaction temperature over Pt/SG-QA catalysts and (d) concentration dependence of reaction rate (r) over 0.1 wt% Pt/SG-QA catalyst, filled/unfilled points are the data collected at heating/cooling mode.
SPSA of the resulted SG solids with supported platinum metal is ∼480–490 m2/g, maintaining a large surface area-to-volume ratio of the initial carrier. Prepared catalysts exhibit a high efficiency in the oxygen-rich H2/O2 gas mixture. The conversion of H2 about 50% is reached in the temperature range from 310 to 350 K (Figure 5(c)). The catalysts show lowering the temperature at the conversion of 50% with an increase in the metal loading. The thermal hysteresis is clearly seen on the H2 conversion plots. This effect may be due, among other possible explanations collected in (Marin and Yablonsky, 2011; Rubtsov, 2016), to the passage of heterogeneous–homogeneous reactions involving free radicals. Typically, the branching radical chain reactions of H- and O-containing species are highly exothermic (Rubtsov, 2016). Radicals, such as •OH and •HO2, can form on the platinum catalysts surface and can desorb in the near-catalyst gas volume (Golodets and Vorotyntsev, 1984). This situation is observed at the initial stages of the reaction. At the same time, the high conversion of hydrogen at a low temperature and atmospheric pressure was registered. Obviously, the realization of rapid stages at the radical chain oxidation of H2 numerously increases the temperature at the catalyst surface and heats the near-surface volume of the reacting gas phase (Rubtsov, 2016). All this causes a subsequent increase in the number of the radicals giving ∼100% conversion. In this regime, the reaction can pass in the absence of the external heating. Consequently, at the cooling regime, the conversion maintains a certain time. This time is enough to provide an explanation of the reason for the presence of the hysteresis loop seen in the stated temperature range. It is noteworthy that the effective formation of radicals was observed at relatively low temperatures even in the case of more high-temperature oxidation reactions, such as the catalytic oxidation of carbon monoxide over complex oxides (Ishchenko et al., 2005).
H2–O2 recombination kinetic parameters.

k2: the reaction constant; Ea: the reaction activation energy; Dav.: the average diameter of supported platinum metal; SG-QA: silica gel-quaternary ammonium sorbent.
Clearly, the highly dispersed platinum metal over silica, which is the most effective catalyst, can be obtained under proposed technique. Perhaps, the high effective catalyst is formed as a result of agglomeration of platinum from the island structures of (QA+)2[PtCl x ]2− that after the reduction with H2 transforming into submicron or nm particles (Figure 5(a) and (b)).
Conclusions
In summary, the adsorption studies prove the effective uptake of platinum, in a form of chloride complexes, from acidic solutions. At the quaternary amine monolayer, the surface ionic associates of (QA+)2[PtCl x ]2−, where х = 4 or 6, are formed. Arguably, the catalysts prepared from the used sorbents reduction are highly active in the H2–O2 recombination. This caused the effect of average platinum particle size that is smaller than in the traditional, supported aluminum–platinum catalyst, 0.1 wt% Pt/Al2O3. Besides pre-concentration, the modified silica with adsorbed platinum chloride complex can be applied to prepare efficient catalysts for the total oxidation reactions.



