Abstract
Mechanochemical and microwave-assisted hydrothermal (MicroWave Treatment [MWT]) procedures were applied to prepare crystalline CaCO3. Mechanochemical process was carried out at different speeds of rotation (500 or 850 rpm/min), different duration times (30 or 60 min) and in the aqueous suspensions or in dry state. MWT synthesis was conducted in a saturated water vapour or under the layer of water. The crystalline and porous structures of the prepared samples as well as their morphology were investigated using N2 adsorption, X-ray diffraction and scanning electron microscopy methods. As a result, the calcium carbonate samples in the form of calcite were obtained. The materials prepared by the mechanochemical route performed in the aqueous suspension are characterized by smaller crystallite sizes as compared to those obtained without the addition of water. The samples obtained hydrothermally have the largest size of crystallites. Powders prepared by energetical milling possess higher values of specific surface area in relation to the parameter for those synthesized hydrothermally. In the process of hydrothermal treatment, macroporous structure of the prepared materials is created. With the increasing specific surface area of the sample, the size of the crystallites decreased.
Keywords
Introduction
Calcium carbonate (CaCO3) is a substance widely distributed in nature. It is estimated that 7% of the Earth’s crust, such as marble and limestone, is composed of calcium carbonate. It occurs in three crystalline forms, i.e. calcite (rhomboeder), aragonite (needles) and vaterite (polycrystalline spheres) of different morphology and different shapes of crystals, wherein calcite is the most stable (Peric et al., 1996; Tai and Chen, 1998). This substance has many applications in different branches of industry, among others, as a polymer filler (Gorna et al., 2008), as a pigment (Walton et al., 1973; Yoo et al., 2009), as a template for the encapsulation of bioactive compounds (Sukhorukov et al., 2004) or as an adsorbent for the removal of heavy metals from water (Ahmad et al., 2012; Ma et al., 2012a, 2012b). Its use is defined by a large number of specific parameters, such as crystalline form, morphology, particle sizes and their distribution, specific surface area, chemical purity and the like.
There are many methods of production of calcium carbonate. Commonly, it is prepared in the recarbonization of natural calcium carbonate to the calcium oxide with a subsequent reaction with water to form calcium hydroxide, which then creates a pure precipitated CaCO3 in the reaction with CO2. Also an interesting example of calcium carbonate preparation is using the eggshell waste as a source (Yoo et al., 2009). The authors used recovered calcium carbonate powder as an ingredient of coating suspension for ink-jet printing paper.
Nowadays, the main aim of chemistry is to design chemical products and processes that will limit or eliminate the use and generation of hazardous substances and consume the lowest possible energy by applying alternative energy sources, e.g. microwaves, ultrasounds and mechanical energy. It is also important to replace the toxic organic substances with water (hydrothermal processes) or carrying out the processes without the use of solvents. The field of chemistry that deals with those aspects is the ‘green chemistry’. Utilizing the water/ethylene glycol system with the addition of surfactants and microwave energy for the production of different morphologies calcium carbonate in the form of vaterite is a good example (Qi and Zhu, 2006). The authors applied microwave heating in the stage of CaCO3 nuclei formation. By the use of different heating times (from 8 to 30 min), they obtained the samples with various forms of vaterite. The authors of the article (He et al., 2005) successfully used ultrasound energy to synthesize monodispersed nanoparticles of calcium carbonate. They obtained CaCO3 in the form of calcite and stated that the use of the batch carbonation method with ultrasonication leads to formation of particles of smaller size compared to the product prepared without ultrasonication. In the article (Hosoi et al., 1997), the hydrothermal method of solidified calcium carbonate preparation was presented. The authors studied the transformations of different crystalline forms of CaCO3 (aragonite-calcite mixture) during hydrothermal hot pressing in the autoclave at a pressure 40 MPa and a temperature 140–160℃. These experimental results presented a low temperature solidification method for the synthesis of calcite compact bodies. In the article (Tsuzuki et al., 2000), the synthesis of calcite using mechanical energy is reported. The particle size of products was controlled by varying the ratio of the reactants. The authors used NaCl as a solid diluent. As a result, it was stated that the resulting particle size was dependent on the amount of salt used as diluent.
Therefore, the aim of this study was to obtain crystalline CaCO3 applying two alternative energy sources, i.e. mechanochemical treatment in the planetary mill and microwave-assisted hydrothermal synthesis.
Novelty of this study is that in a simple manner, in a short time and with high yield, the target product can be obtained in the crystalline form. The applied procedure did not use the additional factors in the form of organic templates or inorganic salts as thinners. The target product was obtained in one step without further heat treatment. Hydrothermal treatment in a microwave reactor was used as the method which allows modification of the forms created during high-energy milling.
Experimental
Materials
The starting substrates in the synthesis were CaCl2 and Na2CO3 (POCh, Poland). The powder mixture in the molar ratio of 1:1 (10.5 g Na2CO3 and 11.1 g CaCl2) was closed in a vessel made of Si3N4 having a volume of 80 cm3 with 25 balls made of the same material having a diameter of 10 mm. Mechanochemical (MChT) processes were carried out in a planetary mill (Pulverisette 7, FRITSCH, Germany) at different speeds of rotation (500 or 850 rpm), different duration times (30 or 60 min) and in dry state(s) or in the aqueous suspensions (liq) (10 cm3 of water per one portion of mixture). The MChT process at 500 rpm in a dry state (MChT500−30s) was performed for 30 min for the initial activation of the starting mixture of reactants before the next microwave-assisted hydrothermal treatment (MWT). Milling at 850 rpm in water suspension or in the solid state during 30 or 60 min was performed to synthesize directly the target product (MChT850−30liq; MChT850−60liq; MChT850−60s). After the reaction, the mixture was subjected to washing in order to remove formed NaCl. The reaction proceeded in the following manner:

The hydrothermal treatment of the sample MChT500−30s was conducted in a high pressure microwave reactor, power 300 W (Plazmatronika, Poland) in a saturated water vapour (vap) or under the layer of water (liq). The weighed sample (2 g) was put to the quartz thimble, which was placed in the microwave reactor. In the case of the saturated water procedure (vap), 20 cm3 of water was poured on the bottom of microwave reactor under the quartz thimble. But in the case of modification under the liquid water layer (liq), 10 cm3 of water was put into the quartz thimble and 10 cm3 on the bottom of the microwave reactor. The reaction time was 60 min. The resulting parameters of temperature and pressure were 180℃ and 90 atm. In such a way, there were prepared the two samples MChT500−30sMWTvap and MChT500−30sMWTliq.
Methods
Parameters of porous structure, i.e. specific surface area SBET, sorption pores volume Vp and diameter of pores Dp (Gregg and Sing, 1982) were determined from the isotherms of low-temperature nitrogen adsorption measured using the analyzer ASAP 2405N (‘Micromeritics Instrument Corp’). The total volume of pores VΣ was determined by intrusion of liquid (methanol) (McDaniel and Hottovy, 1980). The volume of macropores Vmacro was calculated by subtracting the value Vp of VΣ.
The X-ray diffraction (XRD) analysis was done on a diffractometer Empyrean (PANalytical, Netherlands) with CuKα-radiation. The surface topography and character of particles were studied using High resolution Scanning Electron Microscope Quanta 3D FEG, FEI (USA). Before taking scanning electron microscopy (SEM) pictures, the samples were fixed to the aluminium tape and then sprayed with gold and platinum.
Results and discussion
In Figure 1, the nitrogen adsorption-desorption isotherms and differential pore volume distribution functions for the studied samples are presented. The shape of isotherms corresponds to type II with hysteresis loop H3 of the IUPAC classification (Gregg and Sing, 1982; Rouquerol et al., 1994), corresponding to the textural porosity of aggregates of nonporous nanoparticles. The hysteresis loop shape indicates dominant contribution of mesopores. Comparing the relative position of the curves in Figure 1(a), it can be seen that the hydrothermal modification (MWT) causes a decrease in porous structure parameters as the isotherms for these samples are positioned below the adsorption isotherm of the sample MChT500−30s. Moreover, the hysteresis loop for the sample prepared in the liquid phase (MChT500−30sMWTliq) is shifted to higher p/p0. This results in the shift of the maximum peak in Figure 1(a) (right picture) in the direction of higher values of the pore diameter compared to the other samples. In the case of isotherms for the materials synthesized mechanochemically (Figure 1(b)), it can be seen that elongation of milling process causes drop of the specific surface area and the sorption volume of pores, but the position of the main peak observed in Figure 1(b) (right picture) for all samples is the same.
Low-temperature nitrogen adsorption/desorption isotherms (left pictures) and differential pore volume distribution functions (right pictures) for all calcite samples.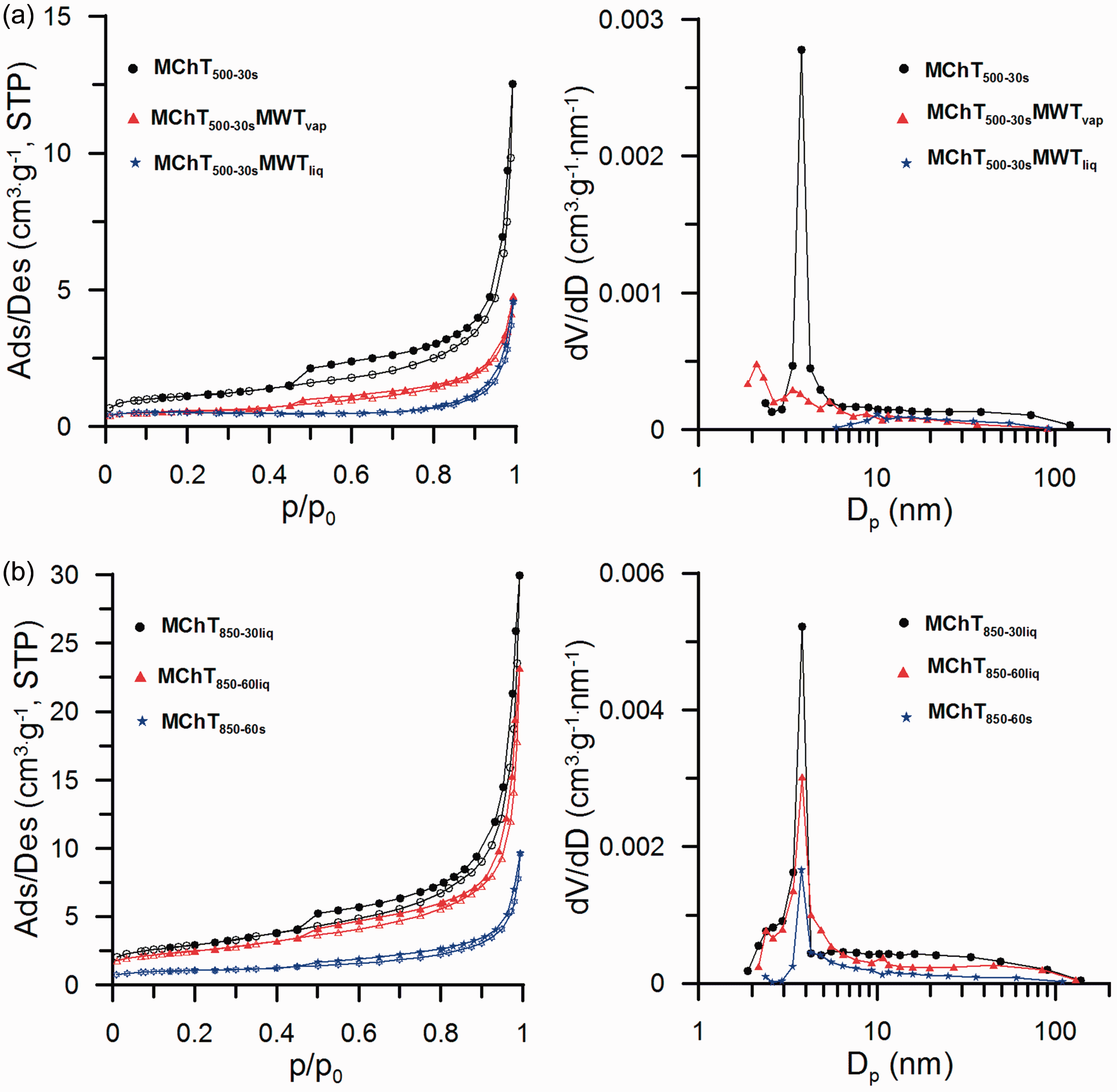
Parameters of porous structure of studied samples.

Comparing the values of the sorption pore volumes (Vp, Table 1), it can be seen that the MChT procedure allows to synthesize samples with higher Vp in comparison to MWT. For the samples prepared by MChT in the liquid phase (MChT850−30liq; MChT850−60liq), these values are about 5 times higher compared to the hydrothermally treated ones (MChT500−30sMWTvap, MChT500−30sMWTliq). In turn, the values of the total pore volumes VΣ and consequently the volumes of macropores Vmacro are higher for the samples treated hydrothermally. Based on this, it can be stated that in the process of hydrothermal treatment, a macroporous structure of the prepared materials is created.
On the basis of the XRD analysis, it was found that all materials contained the desired product (CaCO3) in the form of calcite (JCPDS card No. 01-078-3262) and the concentration of this phase was 100%. The patterns at 2θ = 22.95°; 29.3°; 39.3°; 43.06°; 48.4°; 57.3° from (012), (104), (113), (202), (116), (122) planes characterized for calcite are observed for all samples. Figure 2 shows the diffractogram of the sample MChT850−60liq as an example because the XRD patterns for the other samples were the same, they differed only in the intensity of peaks (Table 2). From the comparison of the data in Table 2, it can be observed that the highest peak intensities (37,909 and 36,424 counts) are found for the samples prepared under hydrothermal conditions both in the saturated water vapour (MChT500−30sMWTvap) or under the layer of liquid water (MChT500−30sMWTliq). These samples are also characterized by the largest crystallite sizes, i.e. 2844 and 3420 Å, respectively. In Table 2 these values were marked in bold.
XRD pattern for sample MChT850−60liq. Parameters of crystallographic structure. D: average crystallite size in the direction of (104) crystallographic plane; d: distance between the planes; Imax (104): intensity of highest peak at 2Θ = 29.3.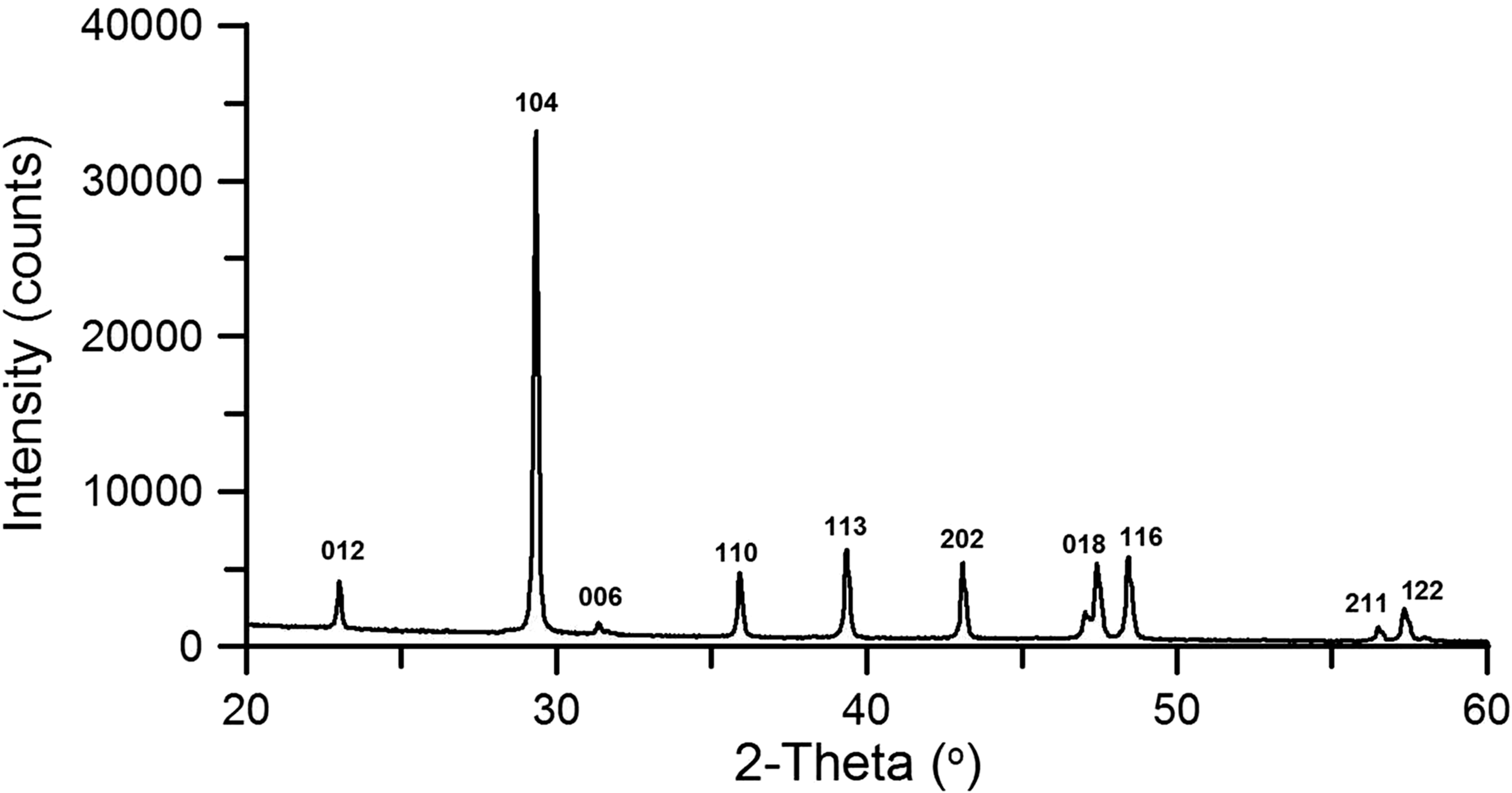
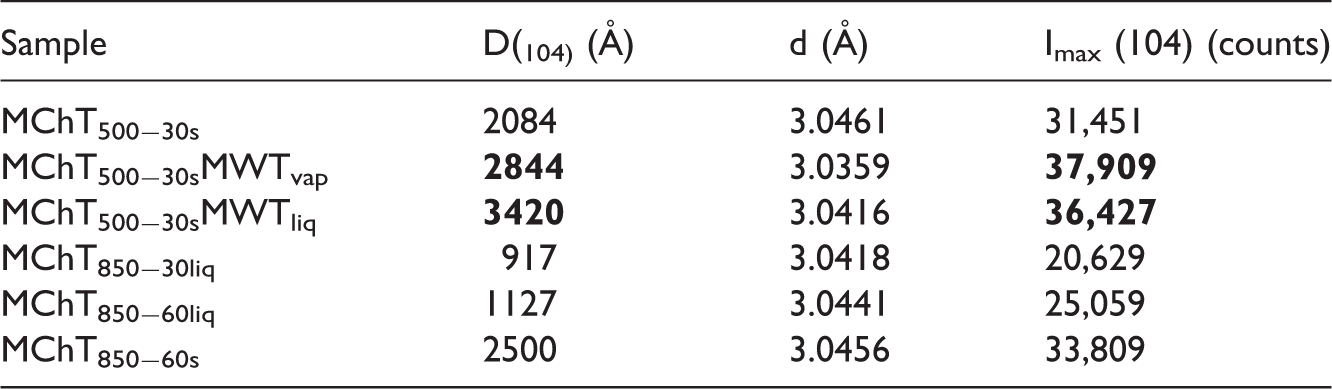
The analysis of the data for the preparations synthesized mechanochemically (Table 2) shows that the samples obtained in the aqueous suspension (MChT850−30liq, MChT850−60liq) are characterized by smaller crystallite sizes (917 and 1127 Å) as compared to those obtained without the addition of water, i.e. 2084 Å (MChT500−30s), and 2500 Å (MChT850−60s). Analyzing the values of d spacings for all materials, it can be generally stated that these values do not differ substantially from each other.
The dependence of crystallite sizes on the specific surface area for all samples is presented in Figure 3. It is interesting that for most of them, with the exception for MChT500−30s and MChT500−30sMWTliq, this relationship is rectilinear. It means that with the increasing specific surface area of the sample, the size of the crystallites decreases.
Dependence of crystallite sizes on the specific surface area for all studied calcite samples: MChT500−30s (a), MChT500−30sMWTvap (b), MChT500−30sMWTliq (c), MChT850-30liq (d), MChT850−60liq (e) and MChT850−60s (f).
Figure 4 presents the exemplary SEM images. Here it can be observed that MChT processes performed in the solid state (samples MChT500−30s and MChT850−60s, Figure 4(a) and (b)) gave materials with crystallites of higher sizes than the samples prepared in the water suspension (MChT850−30liq and MChT850−60liq, Figure 4(c) and (d)). This is consistent with the data in Table 2. Meanwhile treatment under the hydrothermal conditions (MChT500−30sMWTvap, MChT500−30sMWTliq, Figure 4(e) and (f)) makes that the edges of the calcite particles become smooth. Probably, it is a result of dissolving processes in steam under high pressure (MWT). It can also be observed that the particles of studied materials tend to agglomerate (Figure 4). This trend can be also seen in other publications (Pesenti et al., 2008; Qi and Zhu, 2006; Sukhorukov et al., 2004).
SEM images of all calcite samples: MChT500−30s (a), MChT850-60s (b), MChT850−30liq (c), MChT850−60liq (d), MChT500−30sMWTvap (e) and MChT500−30sMWTliq (f). Magnification – 25,000×.
Conclusions
MChT and microwave-assisted procedures allow to prepare calcium carbonate in the form of calcite. The samples prepared in the aqueous suspension are characterized by smaller crystallite sizes as compared to those obtained without the addition of water. Higher values of the specific surface area are displayed by the samples prepared by means of energetical milling in relation to this parameter for those synthesized hydrothermally. In the process of hydrothermal treatment, a macroporous structure of materials is created.
Footnotes
Acknowledgements
This article was first presented at the 15th Ukrainian Polish Symposium on Theoretical and Experimental Studies of Interfacial Phenomena and their Technological Applications, Lviv, Ukraine, 12–15 September 2016.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the European Community (FP7/2007-2013) under the Marie Curie International Research Staff Exchange Scheme (IRSES), Project NANOBIOMAT, Grant No. 612484. The research was carried out with the equipment purchased thanks to the financial support of the European Regional Development Fund in the framework of the Polish Innovation Economy Operational Programme (contract no. POIG.02.01.00-06-024/09 Centre for Functional Nanomaterials).
