Abstract
In this study, nano-sized particles of manganese oxyhydroxide (MnO(OH)) are synthesized. Sorption properties of this material towards 90Sr and 137Cs radionuclides in the pH range from 2 to 9 at a temperature of 293.15 K solution are studied. The time of sorption achievement equilibrium in the radionuclide sorbent system depends on the nature of sorbed cation, but does not exceed 60 min.
Keywords
Introduction
Since the explosion in the nuclear power station in Chernobyl over 30 years ago, the interest of many researchers has been focused on monitoring and determination of the physicochemical properties as well as the behavior of migration of anthropogenic radionuclides in the environment. The results obtained allow us not only to identify the various factors that influence the behavior of radionuclides in these systems, but also became the basic for the development of new methods for removing radionuclides such as 90Sr and 137Cs.
Various sorption materials are widely used for the extraction of radionuclides from water solvents. These are natural and synthetic ion exchangers, complexing agents, composites and other types of sorbents (Krasnopyorova et al., 2007, 2008, 2013; Lujaniene et al., 2008; Mokhodoeva et al., 2011; Myasoedova, 2005; Odnovolova et al., 2014; Ozverdi and Erdem, 2006; Sofronov et al., 2013a; Wu et al., 2008). During the preparation of the sorption materials, much attention is paid on obtaining the particles with large specific surface, which increases the sorption capacity of the materials (Wu et al., 2008). One of the promising sorption materials is manganese oxyhydroxide MnO(OH). Its advantages are the simplicity of synthesis and low cost (Odnovolova et al., 2015a, 2015b, 2015c). In our work, the nano-sized particles of MnO(OH) are synthesized first and are studied regarding the sorption properties of this material towards 90Sr and 137Cs radionuclides at different values of pH of the solution.
Experimental
Nano-sized particles of manganese oxyhydroxide MnO(OH) were produced by reacting KMnO4 with MnCl2 in an alkaline solution (Odnovolova et al., 2015b). For this material we performed an X-ray photography analysis by powder diffractometer Siemens D500 in copper radiation with graphite monochromator at secondary beam. IR spectra of MnO(OH) are measured in tablets KBr by infrared Fourier-spectrometer Spectrum One (Perkin Elmer) (Sofronov et al., 2013b). The investigation of the morphology of the surface of the obtained powder was performed by scanning microscope (SEM) JSM-6390LV (Sofronov et al., 2013b). The sorption ability of MnO(OH) to 90Sr and 137Cs radionuclides was studied by the limited volume method in the pH range from 2 to 9 at temperature of 293.15 K. Radionuclides of 90Sr (
In order to determine the sorption equilibrium in 0.5, 1, 2, 3, 5 hours, the aliquot of solution was taken and the radioactivity was measured. Stability of two successively taken samples showed that sorption equilibrium was attained in a system. Time that corresponds to achievement of equilibrium in solution was determined graphically according to the dependence of solution activity on the time of the solution contact with the sorbent.
Results and discussion
Powder of brown color was obtained as a result of synthesis. Its diffraction pattern is represented in Figure 1(a). Microphotos of obtained particles are represented in Figure 1(b). Independent of the synthesis conditions, the large agglomerates are formed, which consist of particles of size no more than 150 nm.
The diffractogram of the sample prepared at 80℃ (a), the micrograph of the synthesized particles (b).
Figure 1(a) shows an X-ray diffraction pattern of the sample synthesized from a manganese solution. The diffraction pattern shows weak reflections that can be attributed to MnO(OH) and MnO2. In infrared spectrum (Figure 2), the absorption bands are observed at 443 and 505 cm–1, which correspond to the vibrations of the Mn-O bond in MnO(OH) (Odnovolova et al., 2015b). Absorption bands at 3400 cm−1 and 1627 cm−1 are related, respectively, to the valence and deformation vibrations of the water molecules adsorbed on the surface of the fine particles.
IR spectrum of the sample deposited at T = 80℃.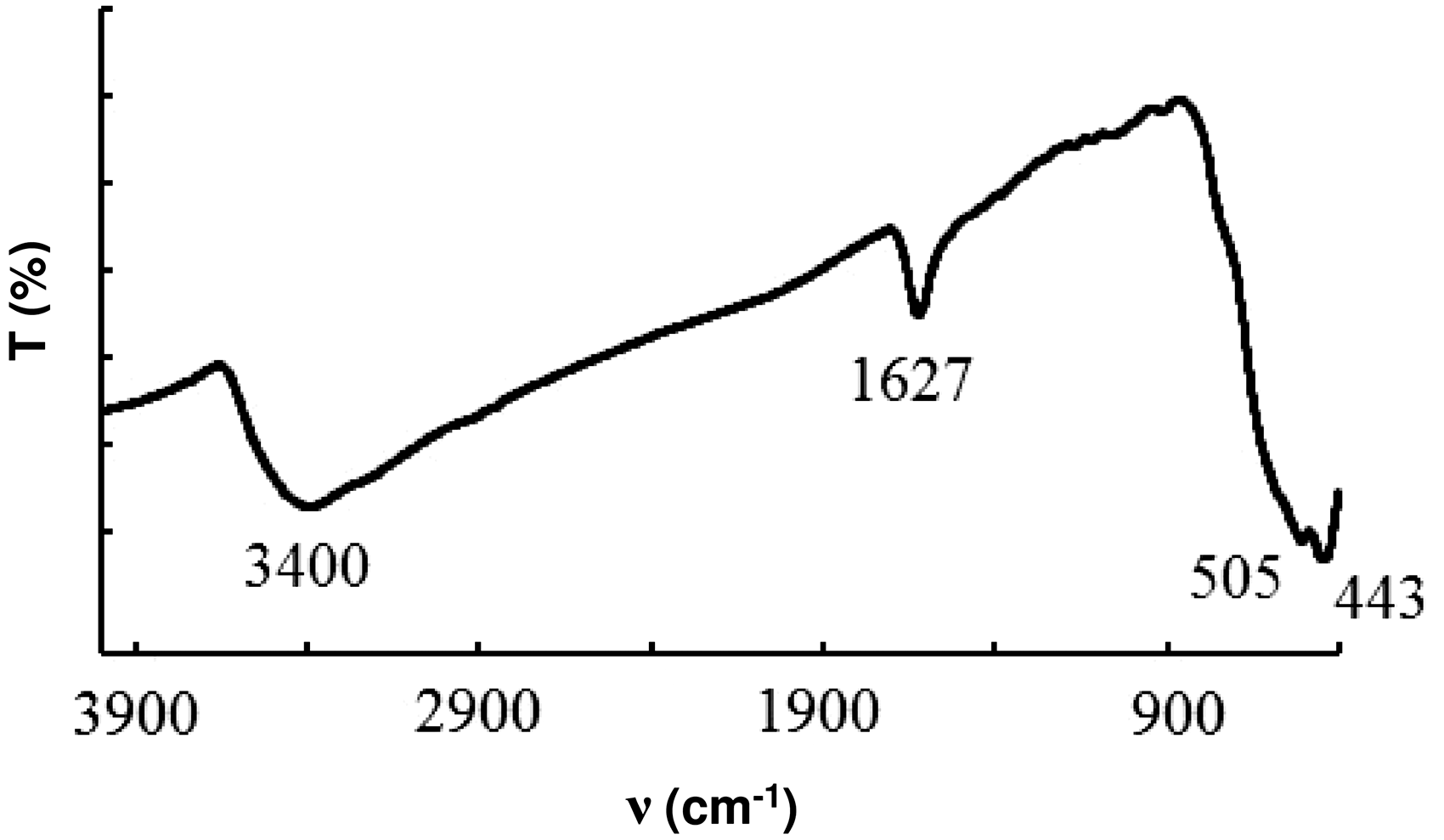
The analysis of the obtained kinetic curves of radionuclide sorption on MnO(OH) at various pH values demonstrates that the sorption equilibrium in radionuclide-sorbent systems is attained at different time intervals. The time of sorption equilibration in the radionuclide sorbent system depends on the nature of sorbed cation, but does not exceed 1 hour (Figure 3).
Kinetic curves of radionuclides sorption on MnO(OH) at pH 7.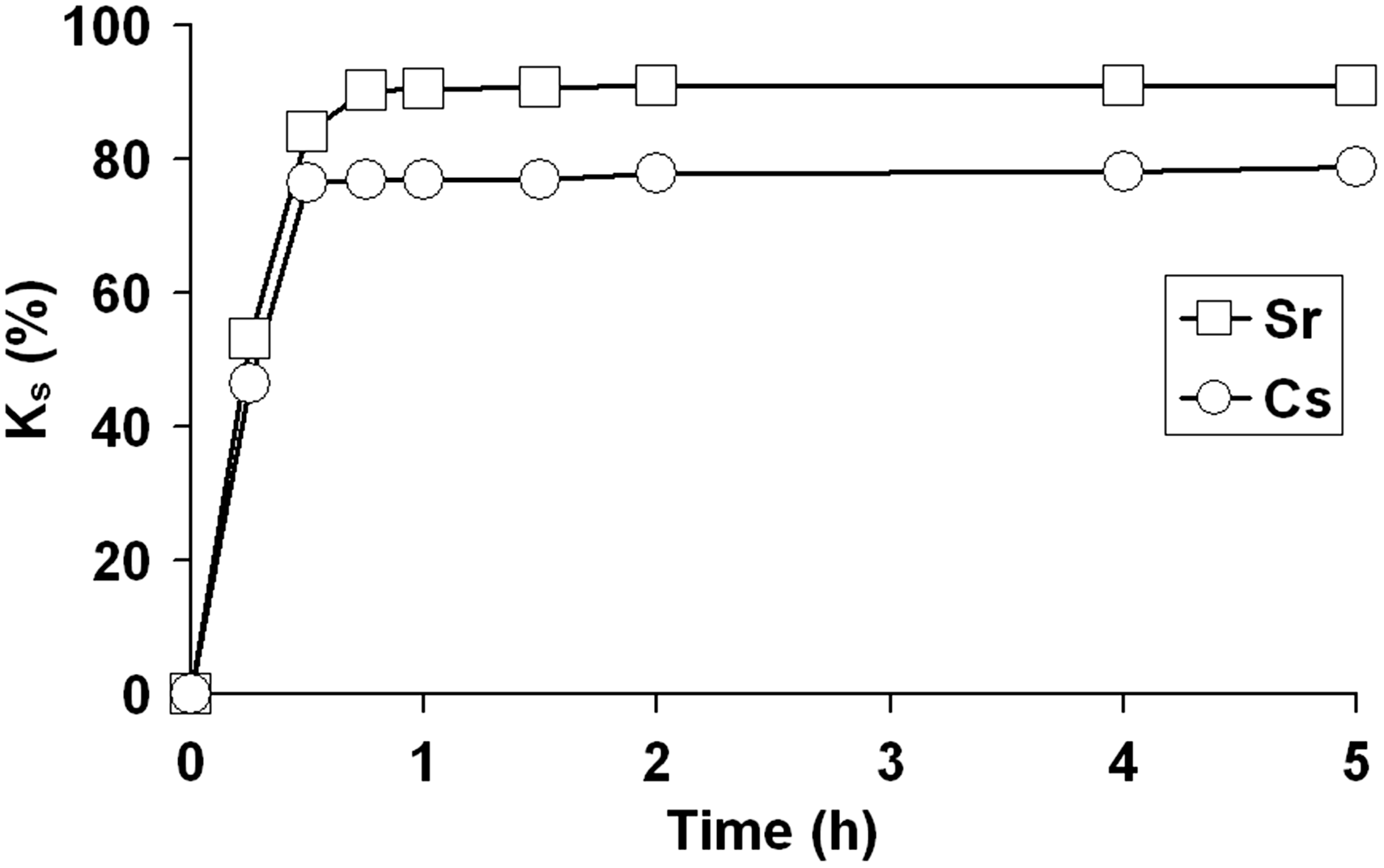
Quantitative characteristics of sorbent interactions with radionuclides were determined according to partition coefficient (Kd, cm3/g), and sorption coefficients (Ks,%), which were calculated according to the formulas (Krasnopyorova et al., 2008)
Partition coefficient Kd and sorption coefficients K s values are different for various radionuclides 90Sr and 137Cs on MnO(OH).

The pH value of the solution, as well as the nature of sorbed cation, affects significantly the efficiency of radionuclide extraction by manganese oxyhydroxide (Figure 4). By its nature, oxyhydroxide is a weakly acidic ion exchanger that contains hydroxyl groups. That is the reason why its sorption properties depend on the pH of the solution. Radionuclides sorption is minimal at pH 2. With an increase of pH, this sorption rises significantly up to a maximal value at pH 7. At the same time it should be noted that the sorption of 90Sr at all pH values is significantly higher than 137Cs sorption. At the same time it should be noted that the sorption of bivalent and hydrated 90Sr ( Sorption coefficients Ks for radionuclides 90Sr and 137Cs on MnO(OH) from pH.
Conclusion
Nano-sized particles of manganese oxyhydroxide MnO(OH) were produced by reacting KMnO4 with MnCl2 in an alkaline solution. For this material, the X-ray photography analysis was performed and IR spectra were measured. The surface morphology of the resulting powder was studied. Irrespective of conditions of synthesis, large agglomerates are formed, which consist of particles of size no more than 150 nm. Sorption ability of MnO(OH) to 90Sr and 137Cs radionuclides was studied by the limited volume method in the pH range from 2 to 9 at temperature of 293.15 K. The pH value of the solution as well as the nature of sorbed cation affects significantly the efficiency of radionuclides extraction by manganese oxyhydroxide.
Footnotes
Acknowledgement
This article was first presented at the 15th Ukrainian-Polish Symposium on Theoretical and Experimental Studies of Interfacial Phenomena and their Technological Applications, Lviv, Ukraine, 12–15 September 2016.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


