Abstract
Ethanol adsorption on sulfonic resins of different morphological types, such as gel, macroporous sulfonic resin, mixed sample (1:1 mixture of gel-type sulfonic resin with aerosil) and sulfonic resin loaded on wide-porous mineral carrier was studied using the methods of quasi-equilibrium thermal desorption and quartz crystal microbalance. It has been observed that the faster ethanol adsorption proceeds on the macroporous sulfonic resin and the slower ethanol adsorption on the gel-type one. It is due to different swelling ability. It was found that the transfer of the ethanol molecules through the gel phase of sulfonic resin proceeds by means of the adsorption–desorption mechanism. The ratio of ethanol adsorption on sulfonic resin is one molecule per one acid site. During the ethanol adsorption on the gel and mixed sulfonic resin, one type of adsorbed complex is formed, whereas on the macroporous and loaded samples two adsorbed complexes with different energies are formed. This is probably because of the different localization of the acid sulfonic groups.
Introduction
Ethanol produced from biomass is a perspective renewable energy source. Currently, there are many potential reactions to produce valuable chemical products from ethanol (Sun and Wang, 2014), such as steam reforming to hydrogen (Haryanto et al., 2005; Rass-Hansen et al., 2007), dehydration to ethylene (Bedia et al., 2011; Bi et al. 2010; Zhang and Yu, 2013), conversion to butadiene (Angelici et al., 2015; Baylon et al., 2016), oxidation to acetaldehyde (Enjamuri et al., 2016), and synthesis of alkyl tert-butyl ethers (Aiouache and Goto, 2003; González-Rugerio et al., 2012; Umar et al., 2008, 2009; Vlasenko et al., 2009).
The industrial production of ethyl tert-butyl ether (ETBE) proceeds by the reaction of ethanol with isobutylene over bulk sulfonated resin catalysts, especially Amberlyst 15. Macroporous resins consist of agglomerates of the polymer gel particles that form macropores. The functional groups are present both on the outer surface of the gel microparticle and on the inner surface of the pores (Corain et al., 2001) Thus, morphology is the relevant factor; underlining that accessibility of acid sites is crucial and provides high catalyst activity. This is due to its influence on the diffusion length of the reactants and products, exposure degree of active sites of active sites, etc. (Zhang et al., 2014).
Swelling process is a result of absorption of a liquid by a polymer. Therefore, it mainly depends on the nature of both the liquid and the polymer. The process is also affected by other factors like: density of the polymer cross-linking, the particle size and the particle size distribution, and the density of the polymer and specific surface area of the particles. The rate of absorption depends on the maximum amount of liquid that can be absorbed, which provides the driving force for the swelling process (Chee and Gan, 2009). Ethanol is a polar molecule which can penetrate into the polymer matrix of sulfonic resin that leads to its swelling. This makes it possible the access of non-polar reactants (e.g. olefins) inside the polymer matrix. That is why ethanol adsorption is crucial for the reactions of its interaction with non-polar substances, for example, etherification processes.
Langmuir–Hinshelwood (Slomkiewicz, 2004) as well as Eley-Rideal (Zhang et al., 1997) mechanisms of ETBE synthesis are considered. A key step in any case is the activation of ethanol at acid sites of catalyst (Goodwin et al., 2002). It was also established that before the etherification process in a flow reactor, ethanol vapor must be supplied to a fixed bed catalyst, and only then isobutylene is added to the reaction mixture. This procedure is necessary in order to avoid blocking the acid sites with isobutylene. It was found that the optimal ratio of ethanol to isobutylene is 1.1–1.5 (Li et al., 2003; Weber de Menezes and Cataluña, 2008).
The purpose of the work was to study the adsorption of ethanol as a key reagent of ETBE synthesis, over sulfonated resins of various morphologic types, with various location of acid sites, namely, organic: gel-type (KU-2-8) and macroporous (Amberlyst 15), and mineral-organic: mixed with aerosil (KU-2-8/A-300) and loaded sulfonated resins on silica-gel KCK (LR).
Experimental
Physicochemical properties of sulfonic resins.

Polymer loading was determined by weight loss of the solids after their calcination at 500℃ during 4 h.
Single-point BET surface area of solids was obtained by nitrogen adsorption at −196℃ using a Gazometer GKh-1 (Khromatograph, Russia).
[КU 2-8 GOST 20298-74].
QETD of ammonia.
The adsorption–desorption of ethanol on sulfonic resins was studied using vacuum unit with quartz balances of the McBain type and Quartz crystal microbalance method (QCM).
When using the unit with McBain type quartz balances, the experiments were performed in two different modes. When studying the dynamics of adsorption of ethanol, vapors at 20℃ have supplied into the reactor and monitored the weight change over time.
To determine the adsorption characteristics of solids, the Quasi-Equilibrium ThermoDesorption (QETD) method was used (Vlasenko et al., 2006, 2009) that gives stepwise ethanol thermodesorption under the condition of reaching adsorption equilibrium at each step. Before measurements, a sample was evacuated at a temperature of 150℃ till reaching constant weight (typically 1 h). Then the temperature was lowered to 30℃ and ethanol vapors were allowed into sample until no change of weight was observed. Then the temperature was raised stepwise (5℃ min−1 between steps, step size 20℃) with holding at constant temperature until constant weight was achieved (typically 15–25 min).
QCM method (Filippov, 2012; Filippov et al., 2014) was used to study the ethanol adsorption and diffusion within gel phase of sulfonic resins. The experiments were carried out in a flow cell (electronic nose (EN) type) in a helium stream. Finely dispersed adsorbents (30–50 nm) were applied on the electrodes of the piezoelectric quartz resonator (PQR). Finely pounded (30–50 nm) styrene–divinylbenzene co-polymer (St-DVB) was used as a coating for inertialess reference sensor (Filippov et al., 2014). The temperature of the evaporator (tEv) in all experiments was 100℃. At the stationary mode, the flow rate of the carrier gas (helium; vHe) was from 20 to 80 cm3/minute; at the pulsed mode, the flow rate was 100 cm3/minute. The adsorption isotherms were obtained in the stationary mode with a step change in concentration of alcohol vapor in the gas phase at temperatures of 40–120℃ and p/ps <0.04.
For the investigation of diffusion dynamics within the adsorbent grain, the same approach as in previous article (Filippov et al., 2014) was used by applying the model developed earlier (Crank, 1975; Timofeyev, 1962). The effective diffusion coefficient Def was evaluated from desorption dynamics at low filling degree in the absence of adsorbate vapors in gas phase (Cτ = 0). In accordance to the model, the degree of desorption 
Results and discussion
The ethanol adsorption dynamics on sulfonic resins of different morphology was studied using the methods of QETD and quartz-crystal microbalance.
Figure 1 shows the time dependences of the amount of ethanol adsorbed at room temperature, on sulfonic resins of different morphological types.
Ethanol adsorption dynamics (at 20℃) on samples of bulk sulfonic resins of different morphology and КU-2-8/A-300 composite.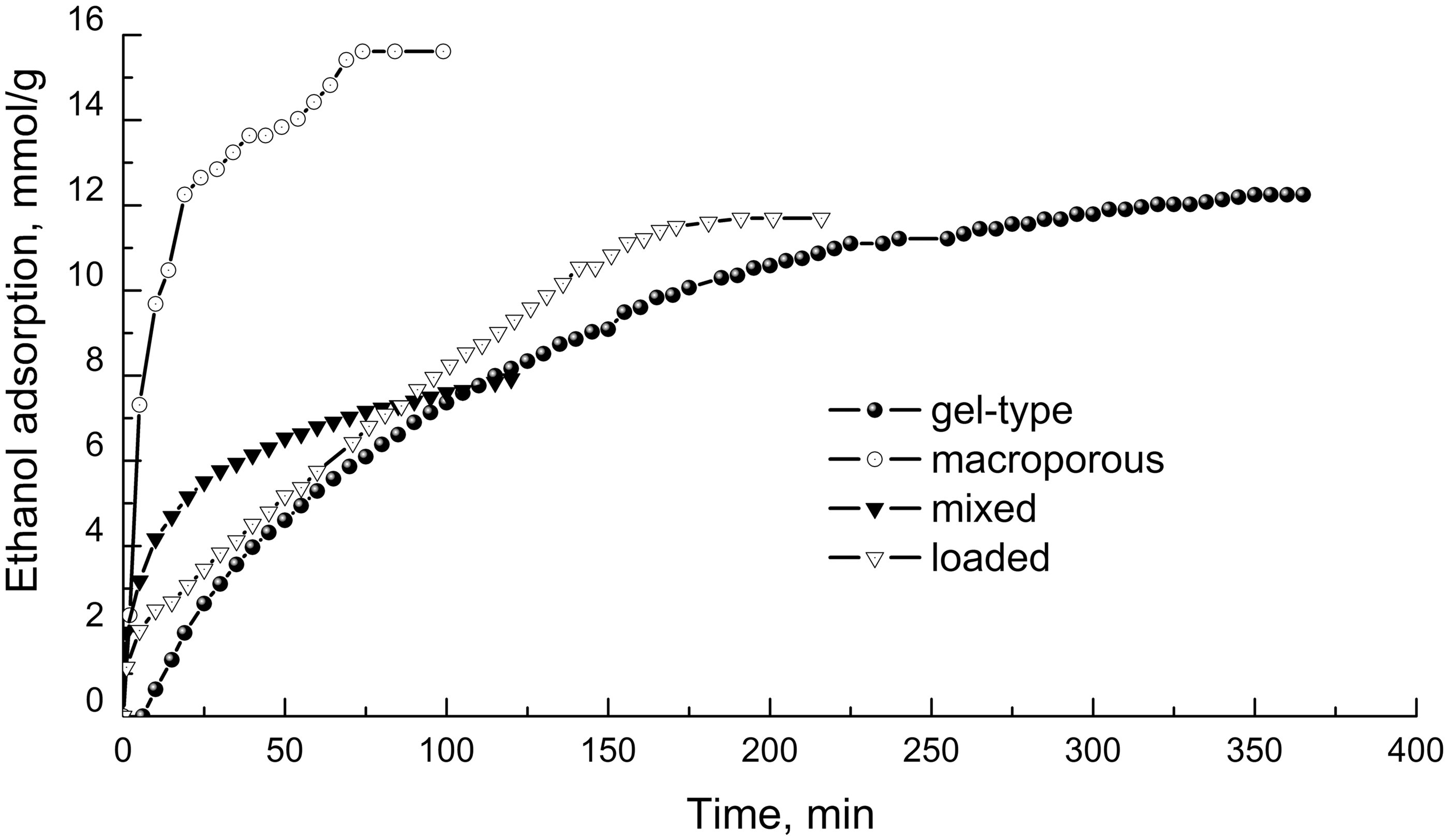
On gel-type sulfonic resin, KU-2-8 adsorption does not begin immediately (induction period is over 6 min) and extended to a long time. Even after 6-h, this process is not completed. The loaded sulfonic resin within the first 3–5 minutes adsorbs a large amount of ethanol rapidly, and then the adsorption takes place at a constant rate. The adsorption equilibrium is established within 175–180 min. In the initial period of time, ethanol adsorption on the mixed sample KU-2-8/A-300 occurs quickly. But after 30–40 min, it slows down and then occurs as slowly as in gel-type sulfonic resin. The fastest adsorption of ethanol occurs on the macroporous sulfonic resin Amberlyst 15 where the adsorption equilibrium is established within 70–80 min. This sulfonic resin was selected to study the adsorption–desorption dynamics of ethanol with the use of QCM method.
The dependences of the frequency change of PQR coated with Amberlyst 15 on the ethanol vapor concentration in a step mode at temperatures of 40–120℃ were obtained. Figure 2 shows the corresponding plot for 100℃ (a) and obtained from this plot the isotherm of ethanol adsorption (b). On the slope of the initial section of adsorption isotherms in a stationary mode (in equilibrium), the value of the adsorption constants Henry (KH) was calculated.
The dependence of frequency change of PQR coated with Amberlyst 15 on the ethanol vapor concentration in a step mode (a) and ethanol adsorption isotherm (b) at 100℃.
Figure 3 shows the responses of the inertialess reference sensor (St-DVB) and the PQR covered by Amberlyst 15 to the pulse of the alcohols vapours. Similar data have been obtained at 40–120℃.
The responses of the PQRs of the inertialess reference sensor (St-DVB) and the PQR covered by Amberlyst 15 to alcohol vapour pulses. Liquid ethanol dose 1 µl, tEN = 100℃, VHe = 100 cm3/min.
Figure 3 shows that while the response of the inertialess sensor had stopped (ethanol vapor disappeared in the cell), the response of Amberlyst 15 covered PQR still continued. This indicates that desorption process on Amberlyst 15 continues under zero concentration of ethanol vapors in the cell. Under such conditions (zero vapor concentration in the gas phase), an approximation described by equation (1) is applicable for the desorption from Amberlyst 15.
Figure 4 shows the dependence (at 100℃) of Linearization of desorption part of response presented in Figure 3 in the absence of ethanol vapour in the gas phase.
The values of Henry adsorption constant and effective diffusion coefficient for ethanol in the gel phase of Amberlyst 15.

As is evident, the values of the effective diffusion coefficient of ethanol are of the order 10−13–10−15 cm2/s. This is in good agreement with the literature data. Thus, the value of the effective diffusion coefficient of methanol inside the microspheres of Amberlyst 15 according to Oktar et al. (1999) amounts to 5.6·10−16, and it was considered to take place in the adsorbed state. In considering the sorption of alcohols by solids with bidisperse pore structures, the value of Def = 10−6–10−15 cm2/s was found (Ruckenstein et al., 1971).
Representing the temperature dependence of effective ethanol diffusion coefficient and Henry adsorption constants in logarithmic coordinates (Figure 5(a) and (b)), it is possible to calculate the value of ethanol adsorption heat and activation energy of ethanol diffusion.
Arrhenius plots of Henry constant (a) and effective ethanol diffusion coefficient (b) on Amberlyst 15.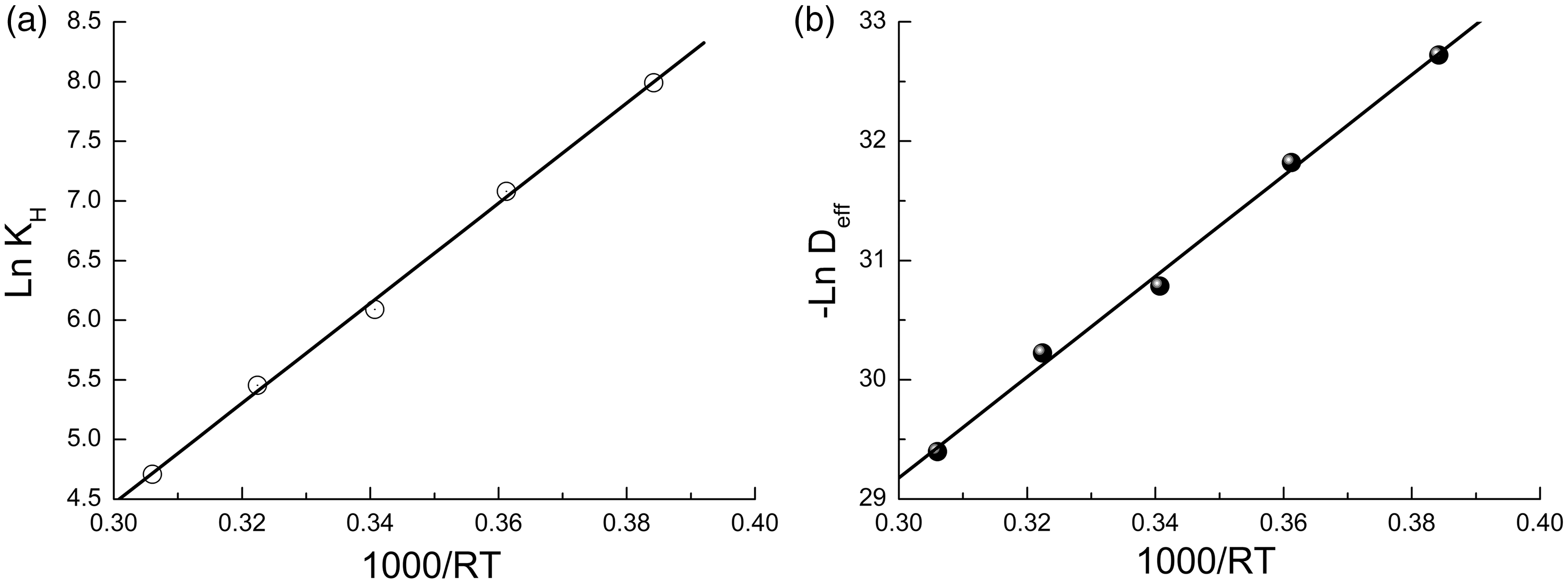
As a result, the adsorption heat of ethanol is determined as ΔH = (43.2 ± 0.8) kJ/mol and activation energy for its transfer in gel phase of Amberlyst 15 is Eact = (43.1 ± 1.1) kJ/mol. The comparison shows that these values are identical. It allows to conclude that the transfer of the ethanol molecules through the gel phase of Amberlyst 15 proceeds by means of adsorption–desorption mechanism.
The data obtained from the QETD of ethanol provide information on the adsorption capacity of the acid sites of sulfonic resins of different morphological types.
Figure 6 shows the integral curves of ethanol thermal desorption on sulfonic resins of different morphologies. It can be clearly seen that at room temperature, the adsorption stoichiometry amounts to one ethanol molecule per one acid site. Thus, it can be assumed that even within the gel granules of sulfonic resin, the ethanol molecule is not “stretched” between multiple sulfonic groups and activated at the unit acid site.
Integral curves of ethanol thermal desorption from sulfonic resins of different types.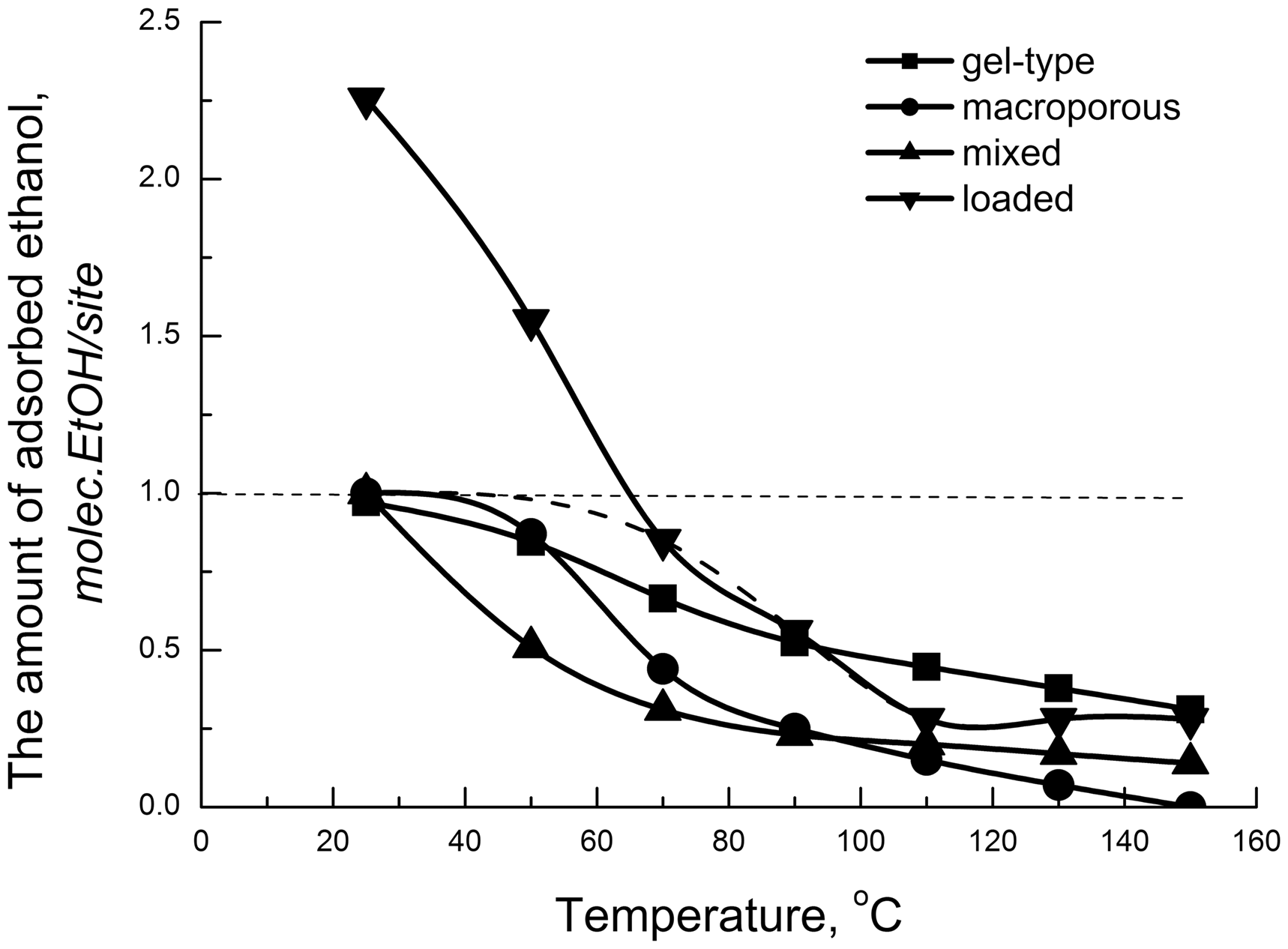
At room temperature, each acid site of the bulk and mixed sulfonated resins adsorbs one molecule of ethanol (Figure 3). At the same time, the number of ethanol molecules adsorbed on an acid site of the loaded sulfonated resin exceeds 2. Possibly, some of the ethanol is adsorbed on the carrier. The basis for this assumption gives the trend of QETD-curve which shows two inflections – at 70 and 110℃. Extrapolation of the integral curve for the loaded sulfonated resin (Figure 1, dashed line) gives the value of ethanol adsorption close to 1 molecule per 1 acid site at a temperature of 40–50℃.
Figure 7 presents the differential curves of QETD-ethanol for sulfonated resins of various morphological types.
Differential curves of ethanol thermal desorption for sulfonic resins of different types.
Whereas for both the gel-type and the mixed sulfonated resins the curves have one maximum, for macroporous and loaded sulfonated resins two peaks are presented. For the bulk macroporous and loaded sulfonated resins, they are probably due to different localization of the adsorbed complexes – in cavities of macropores and in the volume of gel grains.
It should be noted that at the limit temperature of the adsorption experiment (150℃) complete desorption of ethanol can only be achieved on the macroporous Amberlyst 15. On the other samples along with the adsorbed complexes fixed by the differential curve, a certain amount of strongly bounded ethanol remains. It amounts 0.31 and 0.14 mmol/site for KU-2-8 and KU-2-8/A-300, respectively (see Figure 6). This ethanol is adsorbed on strong acid sites located within the microgranules of sulfonic resin. It is not possible to fix such sites by thermal desorption methods, since the experiment is limited by the thermal stability of samples, losing sulfonic groups above 150℃. However, acid sites with ammonia adsorption heat above 90 kJ/mol are recorded by calorimetric method (Siril and Brown, 2006).
Conclusions
It was found that the ethanol adsorption over sulfonic resins is largely determined by their morphology. Themorphology determines the rate of adsorption–desorption of ethanol and its diffusion in polymeric matrix. An important factor is the swelling ability of sulfonic resins, limiting the rate of adsorption. As a result, the adsorption of ethanol on acid sites inside the swelling gel grain of KU-2-8 occurs slowly. The highest rate of adsorption is reached on macroporous sulfonic resin Amberlyst 15. This is due to the presence the intergranular space wherein acid sites are localized. These sites are accessible to specific sorption of ethanol without swelling of polymer matrix. However, even in this case, the rate of adsorption–desorption defined the mass transfer stages within the gel microgranules. This is evident from a comparison of the values of ethanol adsorption heat and activation energy of diffusion: 43.2 ± 0.8 kJ/mol and 43.1 ± 1.1 kJ/mol, respectively.
Depending on the localization of acid sites on sulfonic resins of different morphology, one or two types of adsorbed ethanol complexes with different energy can be formed. In the gel-type sulfonic resin, the majority of the acid sites are localized within the gel grains. The homogeneity of structure causes the same type of adsorbed ethanol complexes on sulfonic resin KU-2-8 and mixed sample KU-2-8/A 300. At the same time, on the surface of macroporous Amberlyst 15 and loaded LR 6.9, wherein the sulfonated polymeric phase formed on the wide-porous carrier, two types of adsorbed complexes are present. Probably, they correspond to ethanol adsorbed in mesopores and within the polymeric phase. In any case, the stoichiometry of ethanol adsorption on sulfonic resins corresponds to the ratio of 1 ethanol molecule per 1 acid site.
Footnotes
Acknowledgement
This paper was first presented at the 15th Polish–Ukrainian Symposium on Theoretical and Experimental Studies of Interfacial Phenomena and their Technological Applications, Lwov, Ukraine, 12–15 September 2016.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
