Abstract
Aluminum-free zeolite silicalite-1 and the ordered mesoporous silicates Mobile Crystalline Material No. 48 (MCM-48) were prepared and functionalized with 3-aminopropyltriethoxysilane (APTES) and 3-mercaptopropyltrimethoxysilane (MPTMS) for the enhancement of adsorption capacity. Functionalization via post synthesis grafting method was adopted and the functionalized silicate systems were denoted as silicalite-1-NH2, silcalite-1-SH, MCM-48-NH2 and MCM-48-SH. Functionalization, that was confirmed by XRD, FT-IR and surface area measurements, indicated no structural changes on the silicate materials. The adsorption of Pb(II) ions into these modified silicates was investigated in aqueous solutions with optimized pH at 5.5 where adsorption influencing factors including contact time, adsorbent dose and metal ion initial concentration were studied. Adsorption experimental data for silicalite-1-NH2, MCM-48-NH2 and MCM-48-SH showed satisfactory correlation with Langmuir and Freundlich models. According to Langmuir isotherm, the maximum capacities for the above three modified silicate systems, for 100 ppm Pb(II) dose, are 43.5, 75.2 and 31.2 mg/g and with Kf constant values of 16.9, 44.4 and 12.0 L/mg from Freundlich isotherm, respectively. The three modified silicate systems exhibited complete sequestration of Pb(II) ion concentrations in the range 0.48–1.7 ppm from samples collected from Zarqa River in four seasons of the year 2013.
Introduction
Persistent, toxic and non-biodegradable heavy metals are discharged from anthropogenic sources into the environment (Bradl, 2005; Das et al., 2011), particularly the aquatic and ecosystems (Das et al., 2011; Schipper et al., 2008), causing them to be accumulated where different life species including humans are exposed to these metals by different routes including water and the food chain (Smith et al., 1999). Pollution with heavy metals imposes serious health problems related to bones, cardiovascular, nervous system and diseases like cancer, with special concern to lead as an abundant element with very low LD50 (Fergusson, 1990). Water pollution with heavy metals and their accumulation mandates all the time remediation technologies for aquatic systems and wastewater plants. Different techniques are available to remove heavy metals from water such as precipitation (Espinoza et al., 2012), membrane filtration (Barakat and Schmdit, 2010), reverse osmosis (Benit and Ruiz, 2002), coagulation (Ghurye et al., 2004) and ion exchange (Mahmoud and Hoadley, 2012). Most of these processes show some disadvantages owing to the disposal of sludge, low efficiency, high cost and applicability to a limited range of pollutants (Samiey et al., 2014). Simple, low-cost adsorption methods utilizing novel adsorbents such as zeolites (Babel and Kurniawan, 2003), activated carbon (Uzun and Guzel, 2000) and clays (Koppelman and Dillard, 1977) have been used. Unfortunately, most of these adsorbents have inherent limitations due to their wide distribution of pore size, heterogeneous pore structure and their relatively weak interactions with metallic ions (Fu and Wang, 2011). Some of these limitations were overcome by using biosorption (Zewail and El-Graf, 2010) and bioaccumulation methods (Arar et al., 2015). A promising class of synthetic multipurpose organic–inorganic polymers utilizing crystalline zeolites or crystalline mesoporous silicates as support demonstrated their applicability in catalysis, drug delivery and adsorbents for toxicants (Zhang et al., 2012). Mesoporous silicate materials were recognized for their large surface area with high density of silanol groups, narrow uniform pore size distribution, open framework pore structure, potentially high adsorption capacity and generation in acidic media (Al Othman, 2012). With these attractive characteristics, mesoporous silicate materials have been widely employed for the removal of heavy metal ions from aqueous solutions (Lam et al., 2006). Among the mesoporous silicate systems that have been extensively studied for the adsorption of heavy metal ions are MCM-41 (Mobil Crystalline Materials no. 41) and SBA-15 (Santa Barbara Amorphous-15). Amino-functionalized MCM-41 was investigated for the removal of Cu(II), Co(II), Cd(II) and Pb(II) from aqueous solutions (Faghihian and Naghavi, 2014). Thiol-functionalized MCM-41 was tested for the adsorption of Cu(II), Pb(II), Ag(I) and Cr(III) (Wu et al., 2009). Micro and nano amino-functionalized MCM-41 were studied for the removal of Ni(II), Cd(II) and Pb(II) from a ternary aqueous solution (Heidari et al., 2009). On the other hand, amino-functionalized SBA-15 was extensively studied for the removal of divalent metal ions including Pb(II) (Ezzeddine et al., 2015; Giraldo and Moreno-Piraján, 2013; Hernandez-Morales et al., 2012). Monoamino-, diamino- and triamino-functionalized SBA-15 were used to remove Pb(II) from aqueous solutions. Unexpectedly, monogroup-functionalized SBA showed the highest adsorption capacity (Li et al., 2015b).
Recently, MCM-48 has attracted the attention because of its three-dimensional (3D) network of channel system. Unfunctionalized MCM-48 was investigated for the removal of Pb(II) from aqueous solutions (Li et al., 2015a).
In this study, we report a revised procedure for the synthesis and functionalization of the aluminum-free microporous zeolite, silicalite-1 (Ahmad et al., 1996) and the mesoporous silicate system MCM-48 (Wang et al., 2001). Both systems have the same structure (MFI) with 3D network of channels. In contrast to most previous reports dealing with systems with 2D network of channels (MCM-41 and SBA-15), the present is based on 3D microporous and mesoporous silicate systems. The 3D silicate systems were surface modified via 3-aminopropyltriethoxysilane (APTES) and 3-mercaptopropyltrimethoxysilane (MPTMS) via post synthesis grafting method, to enhance adsorption capacity (Athens et al., 2009). The functionalized silicate materials were denoted as silicalite-1-NH2, silcalite-1-SH, MCM-48-NH2 and MCM-48-SH. The silicate materials with modified surfaces were used to study the adsorption of Pb(II) ions in aqueous solutions, where after that representative samples from polluted Zarqa river near Jerash (48 km north of Amman) were tested for Pb(II) ion contamination.
Experimental
Materials
Cetyltrimethylammonium bromide (Aldrich), tetraethylorthosilicate (Aldrich) and sodium metasilicate nonahydrate (Aldrich), tetrapropylamonium bromide (Acros Organics) and lead standard solution (Acros Organics), sodium hydroxide pellets (SD fine-Chem), HCl (36%, ANALYT), HNO3 (72%, SD fine-Chem), NaCl (Carlo Erba), isopropanol (GCC), 3-aminopropyltriethoxysilane (Acros), 3-mercaptopropyl-trimethoxysilane (Acros) were used as purchased. Toluene was dried over anhydrous CaCl2 for two days, refluxed for an hour and then distilled.
Instrumentation
X-ray powder diffraction spectra were obtained using XRD-7000 Shimadzu diffractometer (Cu-Kα radiation source at λ = 1.5418 Å) at a scan rate of 2°/min. FT-IR spectra (4000 to 400 cm−1, 4 cm−1 spectral resolution, KBr pellets) were measured using a Thermo Nicolt NEXUS 670 FT-IR spectrometer. Specific surface areas were estimated through BET modeling using a nitrogen adsorption/desorption instrument (Nova 2200e). Thermal gravimetric analysis (TGA) was conducted using a Netzsch Sta 409 PC instrument (NETZSCH-Ger) in the temperature range of 25℃–1000℃ at a heating rate of 20℃/min. Elemental analysis was carried out using Euro Vector Elemental Analyzer. The morphologies of the solid samples were monitored by scanning electron microscopy (SEM) using FEI-FEG INSPEC F50 instrument. Lead metal ion concentrations were determined by Flame atomic absorption spectroscopy (FAAS) using Varian Spectra Plus spectrophotometer at λ = 261.5 nm.
Synthesis of microporous and mesoporous silicate materials
Silicalite-1
Silicalite-1 was synthesized according to a previously published procedure (Ahmad et al., 1996) with some modifications. These modifications include: the optimization of the molar ratio of template to silica source to reduce template residue and prevent the formation of carbon black in the product. The pH of the hydrogel was adjusted to 10.5 instead of 11.0 to facilitate the recovery of the gel. The aging temperature at 170℃ was modified to aging at 150℃ for 48 h and then at 170℃ for the next 24 h, to prevent quartz formation with silicalite-1 (for the detailed synthesis of silicalite-1, see online data supplement in the supplemental material file).
MCM-48
MCM-48 was prepared following published literature (Wang et al., 2001). In a typical procedure, 8.70 g of cetyltrimethylammonium bromide (CTAB) was added to a solution of sodium hydroxide (0.96 g) with constant stirring. After that, 10.50 g of tetraethylorthosilicate (TEOS) was added drop wise with vigorous stirring till the addition was complete. The solution was further stirred for 1 h at room temperature. It was then transferred to a Teflon-coated stainless autoclave and heated for three days at 100℃. The autoclave was allowed to cool to room temperature and the pH of solution was adjusted at pH = 7 using 6 M HCl. The mixture was further heated in the autoclave at 100℃ for one day. The white solid product was filtered, washed with deionized water (3 × 50 mL) and dried at 110℃ for 12 h. Finally, it was calcined at 550℃ for 6 h in air to remove the template.
Functionalization of porous silicate materials
Post-synthesis grafting of silicalite-1 with amine functional group
Functionalization was accomplished by adopting published method (Athens et al., 2009; Delacôte et al., 2009). One gram of silicate material was dried at 110℃ overnight to remove adsorbed water. The silicate sample was dispersed with stirring in 50.0 mL toluene and then APTES (10 mmol) was added to the suspension. The mixture was heated under reflux for 12 h under nitrogen. The resulting product was filtered, washed with toluene (3 × 50 mL) and then washed with isopropanol (3 × 50 mL). The functionalized silicate material was dried at 60℃ for 12 h under vacuum. The optimized ratios for reaction precursors mentioned above were beneficial for increasing the percentage of functionalization. The functionalized products were coded as silicalite-1-NH2.
Post-synthesis grafting of MCM-48 with amine functional group
The preparation of the modified MCM-48 coded as MCM-48-NH2 was done using the procedure outlined in section “Post-synthesis grafting of silicalite-1 with amine functional group” but using 5 mmol of APTES for each gram of MCM-48.
Post-synthesis grafting of silicalite-1 and MCM-48 with thiol functional group
Functionalized of silicalite-1and MCM-48 with thiol functional groups (coded as silicalite-1-SH and MCM-48-SH) was conducted via the procedure outlined in section “Post-synthesis grafting of silicalite-1 with amine functional group” but using 3-mercaptopropyltrimethoxysilane (MPTMS).
Aqueous Pb(II) batch adsorption influential parameters
Pb(II) initial concentration
Stock solutions of Pb(II) ions with concentrations 100, 150, 200, 250 and 300 ppm were prepared by dilution with deionized water using lead standard solution of 1000 mg/L. Samples of 50 mg of functionalized silicate were added to solutions of Pb(II) (25 mL) with different concentration levels. The solutions were agitated at 200 r/min using orbital shaker at 25℃ for 2 h allowing the adsorption to reach equilibrium. The pH value in all solutions was adjusted at 5.5 using 0.1 M NaOH and 0.1 M HNO3 solutions. The adsorbent was separated by filtration and the final Pb(II) concentrations in the solutions were measured using FAAS. The optimum initial Pb(II) concentration for each modified silicate that causes active sites saturation was determined according to the percent uptake of Pb(II) according to equation (1). The results of measurements were determined according to a previously constructed calibration curve from Pb(II) solutions in deionized water adjusted to pH = 5.5 in the range 0–100 ppm.

Effect of contact time
The uptake of lead metal ion by silicalite-1-NH2, MCM-48-NH2 and MCM-48-SH was carried out at different contact time intervals (1, 2, 3, 4, 5, 10, 20, 30, 40, 60, 80, 100 and 120 min) in a 150 mL Erlenmeyer flasks containing 25 mL of 100 mg/L Pb(II) ions initial concentration at 25℃, adsorbent dose of 50 mg and pH = 5.5. The residual Pb(II) aqueous ions were measured by FAAS.
Effect of adsorbent dose
The adsorption of Pb+2 into silicalite-1-NH2, MCM-48-NH2 and MCM-48-SH with different modified sorbent doses (0.01, 0.02, 0.03, 0.04, 0.05, 0.06, 0.08 and 0.1 g) in a 150-mL Erlenmeyer flasks containing 25 mL of 100 mg/L Pb(II) ions was studied. The pH of solutions was adjusted to pH = 5.5 at 25℃.
Adsorption isotherms
Solutions with initial Pb(II) concentrations of 100, 150, 200, 250 and 300 mg/L were adjusted to pH = 5.5 at 25℃ and were treated with Silicalite-1 (parent), silicalite-1-NH2, MCM-48 (parent), MCM-48-NH2 and MCM-48-SH for 120 min contact time. The equilibrium concentration of Pb(II) for each solution was measured using FAAS after adsorbent filtration. The obtained data were used to construct mathematical models of Langmuir and Freundlich.
The linear equations of Langmuir (equation (2)) (Freundlich, 1906) and Freundlich (equation (3)) (Langmuir, 1916) were used in data correlation and obtaining isotherm constants.


Pb(II) remediation in Zarqa River water samples
Four representative water samples were collected from the Zarqa River near Jerash, 48 km north of the Amman in four different seasons in 2013. The samples were acidified and stored for analysis and coded as sample-1 for the sample collected on 15 February, sample-2 for 15 May, sample-3 for 15 August and sample-4 for 15 November. Portions of 25 ml of the river samples were transferred into 150 ml Erlenmeyer flasks where the pH of the samples was adjusted to 5.5 then the Pb(II) concentration for each portion was measured by FAAS. Similar portions were treated as above and the flasks were subjected to treatment with silicalite-1-NH2, MCM-48-NH2 and MCM-48-SH for 2 h. The flasks were kept at 25℃ by means of water bath with agitation. The equilibrium concentrations of Pb(II) were measured for each portion after sorbent filtration. The results of measurements were determined against a calibration curve Pb(II) solutions in deionized water adjusted to pH = 5.5 in the range 0–3 ppm.
Results and discussion
Characterization of silicalite-1and MCM-48 and their functionalized derivatives
The silicate systems and their functionalized derivatives were characterized by XRD, FT-IR, surface area measurements and TGA analysis. The X-ray diffraction pattern of the prepared silicalite-1 (Figure 1) showed the characteristic peaks of the ZSM-5 structure which is in agreement with its diffractograms according to International Zeolite Association (IZA-SC). Similarly, silicalite-1-NH2 and silicalite-1-SH showed similar X-ray patterns to unfunctionalized silicalite-1 but with a decrease in peak intensity accompanied by a slight shift to lower values of 2θ, indicating that the zeolite structure remained intact after functionalization (Aguando et al., 2009; Sepeherian et al., 2009). The X-ray diffraction pattern of the mesoporous MCM-48 was also in good agreement with the reported pattern of MCM-48 (Schmidt et al., 1995). Similar to the silicalite-1 system, amine or thiol functionalization does not affect the structural features of MCM-48 but caused a slight shift and lowering in intensity for the main peak.
XRD spectra of (a) silicalite-1, (b) silicalite-1-NH2, (c) silicalite-1-SH and (d) MCM-48.
The FT-IR spectra of silicalite-1 and MCM-48 show the characteristics bands of porous silicate materials which are consistent with that reported in the literature (Marzouqa et al., 2012; Venkatachalam et al., 2012). Upon functionalization, the Si-OH related peak at about 3743 cm−1 in the FTIR spectrum of unmodified disappeared and new peaks for the propyl backbone (νC-H) appeared at about 2930 cm−1. The stretching bands due to νNH2 which are expected to appear in the range 3200–3500 cm−1 were masked by the broad band of adsorbed water (Figure 2) (for the FT-IR spectra of unfunctionalized silicalite-1 and amine-functionalized silicalite-1 see online data supplement in the supplemental material file).
Representative FT-IR spectra of (a) Unfunctionallized MCM-48, (b) amine-functionalized MCM-48 and (c) thiol-functionalized MCM-48.
The adsorption/desorption curves for silicalite-1 and MCM-48 correspond to type I and type IV isotherms, respectively. The isotherm of silicalite-1 is characteristic of ordered microporous material, whereas that of MCM-48 is characteristic of ordered mesoporous material (Figure 3). The measured surface area, pore volume and pore diameter of silicalite-1 and MCM-48 and their amine-functionalized derivatives are listed in Table 1. The surface area values are in good agreement with reported data (Petushkov et al., 2009; Schumacher et al., 1999). As shown in Table 1, an appreciable decrease occurred on the surface area and pore volume upon functionalization. This could be due to partial blockage of the pores by the functional groups, especially in case of silicalite-1.
Nitrogen adsorption/desorption isotherm of (a) silicalite-1 and (b) MCM-48 (squares represent adsorption and triangulars correspond to desorption). Specific surface area (BET), pore volume and pore diameter of MCM-48, MCM-48-NH2, silicalite-1 and silicalite-1-NH2. The adsorption/desorption instrument used in this study does not give accurate values for the pore diameter of the a micro porous material. BET: Brunauer–Emmett–Teller.

The micrographs obtained by the scanning electron microscope (Figure 4) for solid powders showed hexagonal prismatic particles for silicalite-1 and irregular spherical particles for MCM-48.
SEM micrographs of (a) silicalite-1 and (b) MCM-48.
The thermal stability of silicalite-1 and MCM-48 crystals was tested over the temperature range of 25℃–1000℃. The mass loss in the temperature range 25℃–300℃ was attributed to desorption of water residing within the pores plus water H-bonded to surface silanol groups. The mass loss in the range 300℃–700℃ was attributed to the decomposition of the grafted organic functional group. As shown in Figure 5, the mass loss in the range 300℃–700℃ for amino-functionalized silicalite-1 and amino-functionalized MCM-48 are about 6.5% and 10.2%, respectively, indicating higher functionalization for MCM-48. Similar trend has been observed for thiol-functionalized silicalite-1 and MCM-48. The TGA results are in agreement with the elemental analysis data (Table 2).
TGA thermographs of (a) unfunctionalized MCM-48, (b) MCM-48-NH2 and (c) MCM-48-SH. Elemental analyses for the functionalized materials silicalite-1-NH2, silicalite-1-SH, MCM-48-NH2 and MCM-48-NH2 and the percent of functionalization in mmol/g of adsorbent. Calculations are based on % nitrogen. Calculations are based on % sulfur.

Aqueous Pb(II) batch adsorption influential parameters
Pb(II) initial concentration
The results for the effect of initial concentration on the percent Pb(II) uptake are presented in Figure 6. The maximum percentage of removal occurred at the minimum concentration of Pb(II) used (100 ppm), where beyond that concentration metal uptake decreased due to the saturation of active sites and buildup of repulsion forces among adsorbed Pb(II) ions and free ions in solution (Das et al., 2011). The optimum pH of 5.5 was chosen to ensure that Pb(II) ions are in the soluble form (preventing the formation of insoluble lead oxide) and also to prevent the domination of H3O+ at lower pH values leading to competition with Pb(II) ions on the basic amine coordinating sites (Heidari et al., 2009).
Effect of initial Pb(II) concentration on the percent removal efficiency. MCM-48-NH2 (▪); silicalite-1-NH2 (♦); MCM-48-SH (▴).
Effect of contact time
For all silicate systems, percentage Pb(II) uptake increased with contact time till saturation is reached and with adsorption rates in the order: MCM-48-NH2 > MCM-48-SH ∼ silicalite-1-NH2 (Figure 7). For MCM-48-NH2, the percent Pb(II) uptake reached 96.9% within a contact time of 20 min, whereas for MCM-48-SH and silicalite-1-NH2 the values for percent Pb(II) uptake and contact time are 50% at 40 min; 60% at 40 min, respectively. In general, the results indicate that MCM-48 is superior to silicalite-1 in removing Pb(II) ions, which could be due to the higher degree of functionalization or/and the blockage of silicalite pores by the functional group. The results also indicate that amine functionalization is more efficient in removing Pb(II) ions than the corresponding thiols.
Effect of contact time on the uptake of Pb2+ onto MCM-48-NH2 (▪); silicalite-1-NH2 (♦); MCM-48-SH (▴) (temperature = 25℃, adsorbent dose = 50 mg, pH = 5.5, initial Pb2 +concentration =100 ppm).
Effect of adsorbent dose
For the silicate systems under investigation, there is an increase in the percent Pb(II) uptake with the increase of adsorbent dose, at an initial concentration of Pb(II) = 100 ppm (Figure 8). However, the maximum percent Pb(II) uptake and adsorbent dose (at maximum percent Pb(II) uptake) vary significantly. These values for the three systems are: silicalite-1-NH2 (76.6 %, 0.1 g); MCM-48-NH2 (100 %, 0.06 g); MCM-48-SH (57.5 %, 0.08 g). The proportionality in the results is expected because increasing the adsorbent dose provides more surface area and more active sites to interact with the metal ion (Giraldo and Moreno-Piraján, 2013). Achieving values less than 100% Pb(II) uptake, at adsorbent dose of 0.06 g, in case of MCM-48-SH and silicalite-1-NH2 could be explained in view of the weak interaction with the thiol groups and low accessibility to the amine groups, respectively. However, in the above both cases, there was a slight increase in percent metal uptake with increasing the adsorbent dose (Figure 8).
Effect of adsorbent dose on the uptake of Pb2+ onto MCM-48-NH2 (▪). silicalite-1-NH2 (♦). MCM-48-SH (▴) (temperature = 2℃, contact time = 2 h, pH = 5.5, initial Pb2+ concentration = 100 ppm).
Adsorption isotherms
The adsorption capacity for Pb(II) by the unfunctionalized silicates MCM-48 (0.0 mg/g) and silicalite-1 (2.5 mg/g) in addition to the thiol-functionalized silicalite-1-SH (10.0 mg/g) are low enough to exclude them from the current study. The low affinity to uptake Pb(II) by the unfunctionalized silicates is due to the very weak interactions of Pb(II) ions and the silanol groups. However, in case of silicalite-1-SH, there are two factors affecting the adsorption process: first, the small pore diameter of the microporous silicate of about 0.56 nm, which is close to the size of the hydrated Pb(II) ions with a diameter of 0.52 nm; and second, the low percentage of functionalization in case of silicalite-1-SH. Practically, the incorporation of the thiol group into the silicalite-1 had minor effect on the adsorption capacity of silicalite-1.
The adsorption isotherm data for silicalite-1-NH2, MCM-48-NH2 and MCM-48-SH are in good agreement with Langmuir (Figure 9) and Freundlich (Figure 10) isotherms as indicated in Table 3. The maximum adsorption capacities (qmax) for silicalite-1-NH2, MCM-48-NH2 and MCM-48-SH according to Langmuir were 43.5, 75.2 and 31.2 mg/g, respectively, where qmax = (1/slope) based on equation 2. The higher adsorption capacity of MCM-48-NH2 as compared with silicalite-1-NH2 could be explained in view of the higher functionalized area that provides more active amine groups to interact with Pb(II) ions, in addition to the larger pore diameter that can provide more access to the hydrated Pb(II) ions. On the other hand, MCM-48-SH has the lowest adsorption capacity compared with MCM-48-NH2 and silicalite-1-NH2, although it has a higher percentage of functionalization than silicalite-1-NH2. These results suggest that Pb(II) ions have weaker interaction with thiol than with amine groups. The results also indicated that the rates of adsorption of Pb(II) ions into these modified surfaces were in the ascending order MCM-48-NH2 > silicalite-1-NH2> MCM-48-SH. The amine-functionalized materials investigated in this study show good adsorption capacity compared with other silicate systems studied previously (Table 4).
The linearized form of Langmuir isotherm equation for MCM-48-NH2 (▪). silicalite-1-NH2 (♦). MCM-48-SH (▴) (temperature = 25℃, contact time = 2 h, pH: 5.5, qmax = 1/slope). The linearized form of Freundlich isotherm equation for silicalite-1-NH2 (♦), MCM-48-NH2 (▪) and MCM-48-SH (▴) (temperature = 25℃, contact time = 2 h, pH = 5.5, 1/n = slope and antilog intercept = Kf). Adsorption parameters of Langmuir and Freundlich isotherms at room temperature for the adsorption of Pb2+ onto silicalite-1-NH2, MCM-48-NH2 and MCM-48-SH. Comparison between published adsorption capacities of amine-functionalized silicate systems and adsorption capacities in this study. Functional moiety contains two amine groups.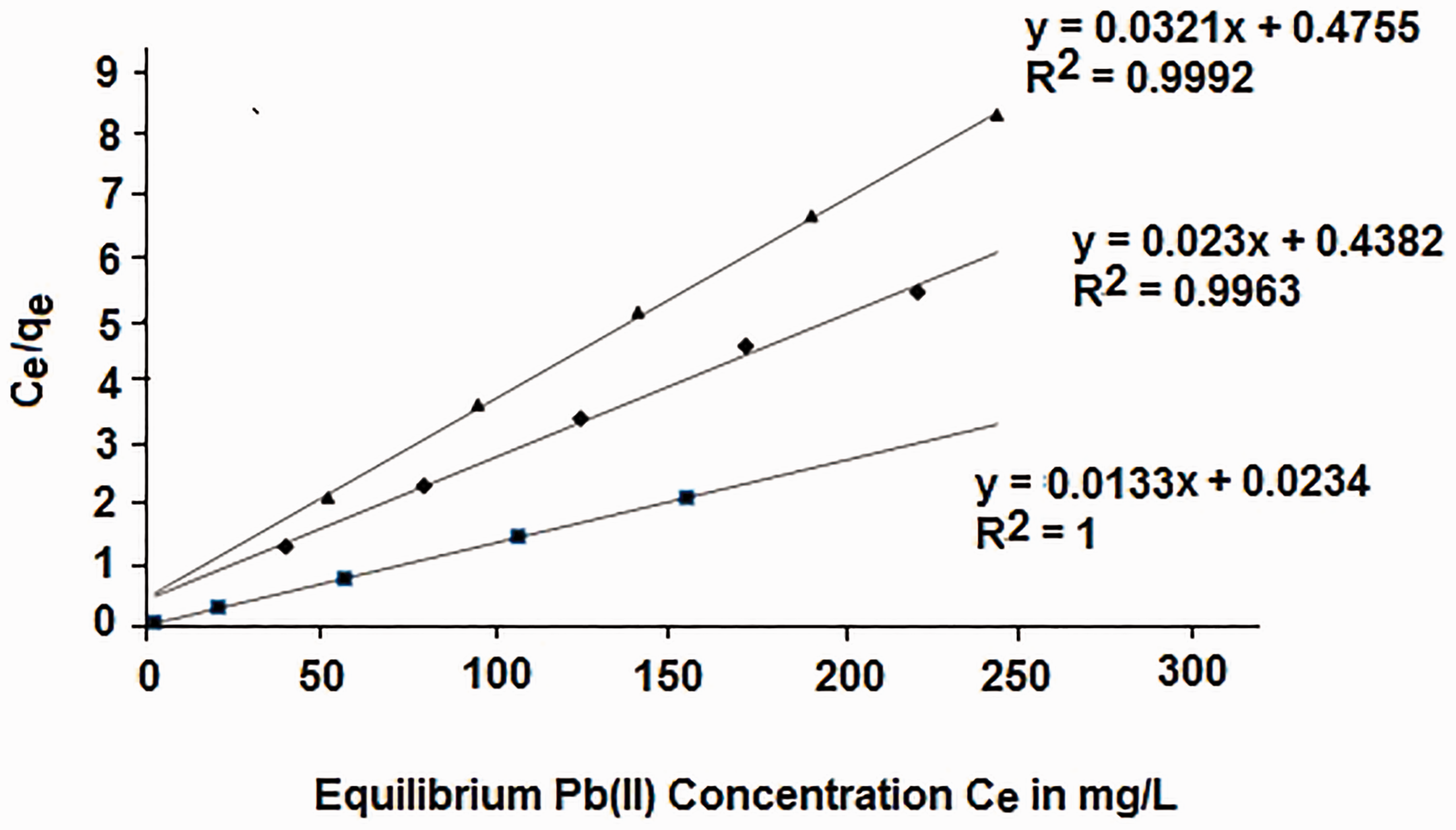



Pb(II) remediation in Zarqa River water samples
Concentration of Pb2+ (ppm) ion in water samples from Zarqa River before treatment.

Calibration equation y = 0.038x + 0.0015, R2 = 0.9991.
Conclusion
The surface-modified materials and silicalite-1-NH2, MCM-48-NH2 and MCM-48-SH showed high potential as remediation sorbents for Pb(II) in polluted aquatic systems, where total removal of Pb(II) was achieved. The amine functionalized MCM-48-NH2 showed the highest adsorption capacity for Pb(II) ion concentrations up to 300 ppm, due to the relatively high density of silanol groups and high percentage of functionalization. In addition, its pore diameter enables high accessibility of hydrated ions leading to better interaction in solution. A comparison between the amine and thiol-fuctionalized MCM-48, MCM-48-NH2 shows higher percent uptake of Pb(II), thus indicating stronger interaction with the amine group versus the thiol group. Accordingly, amine-modified silicate systems can be used as sorbents for effluents of industrial plants contaminated with Pb(II) and other heavy metals waste in concentrations reaching up to 100 ppm or more. The efficiency for Pb(II) removal could be further enhanced by modification of the MCM-48 with bi- or tri-dentate amino groups which will have a large impact on increasing the value of Kf. On the other hand, producing silicalite-1 particles in the nano-scale is expected to increase the surface area and the density of Si-OH groups and hence enhancing the percentage of functionalization leading to better metal uptake.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The financial support from Deanship of Scientific Research/The University of Jordan and Scientific Research Support Fund, Amman, Jordan is gratefully appreciated.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
