Abstract
In this study hydrothermal method was used to synthesize MIL-53(Al) (MIL stands for Materials Institute of Lavoisier). Plackett–Burman (P–B) as an experimental design method was applied to investigate the effect of synthesis and activation conditions on specific surface area, relative crystallinity, and production yield of MIL-53(Al) synthesis. Some parameters such as ligand-to-metal molar ratio, synthesis time, synthesis temperature, calcination temperature, and calcination time were selected as the variables. The Brunauer–Emmett–Teller (BET) technique was used in order to estimate the specific surface area of samples while the relative crystallinity of the samples was estimated by comparing their X-Ray Diffraction (XRD) pattern. The morphology of the samples was investigated by field emission scanning electron microscopy. The yield of final products was determined based on organic ligands. The results revealed the significant effect of synthesis temperature on BET surface area, particle size, yield, and crystallinity. The calcination temperature has significant positive effect on BET and crystallinity. Also, the negative significant effect of molar ratio on yield was concluded from the results. However, negligible effect of synthesis and calcination time on the properties of prepared materials were observed. Furthermore, separation capability of a selected sample for carbon dioxide (CO2) and methane (CH4) was measured. Pure gas adsorption data were successfully fitted to Langmuir, Sips, and Toth models. The selected sample provided high adsorption capacity for both gases. The binary adsorption of gases was also investigated based on extended Langmuir equations and the ideal adsorbed solution theory (IAST) models. Comparing the experimental and models data indicated good agreement between the IAST model and experiments. Finally, high CO2/CH4 selectivity of 7.6 was obtained experimentally for the CO2/CH4 molar ratio of 0.2/0.8.
Introduction
The existent of CO2 in biogas and natural gas will not only directly lead to global warming but also reduce the energy content and has corrosion effect on transportation system in the presence of water (Finsy et al., 2009; Wang et al., 2011). Thus, capture of CO2 is an urgent challenge in processing technologies. The established technologies for CO2 separation such as cryogenic distillation and amine-based absorption has disadvantages of high energy lost. The adsorption-based process is emerging as an energy-efficient alternative. The adsorption process is a cost effective and scalable method to capture CO2 (Sun et al., 2015). The selection of proper adsorbent is one of the key parameters in the adsorption technique and also can be considered as heart of this technology (Choi et al., 2009; Grande, 2011).
An emerging new class of crystalline porous solids called metal-organic frameworks (MOFs) has recently been investigated to use as an adsorbent in many studies (Farha and Hupp, 2010; Furukawa et al., 2013; Janiak and Vieth, 2010; Kuppler et al., 2009; Li et al., 2009, 2012; Liu et al., 2012; Ma, 2009; Mueller et al., 2006; Snurr et al., 2004; Yaghi et al., 2003). They comprise metal ions (or metal cluster) and organic ligand that link by coordination bonds. The investigation about the application of MOF adsorbents in gas separation is still in early steps (Czaja et al., 2009; Eddaoudi et al., 2001; Férey 2008; Rowsell and Yaghi, 2004; Zhao et al., 2011).
Some methods such as solvothermal/hydrothermal, microwave, sonication, and mechanochemical synthesis have been currently used for MOF synthesis (Dey et al., 2014). Hydrothermal technology as a conventional method for synthesis of zeolite is a simple technique and environmentally friendly while there is no need to redevelop this method (Čejka et al., 2010). However, there are several challenges in optimization of the reaction conditions which can be led to proper MOF with high yield and crystallinity. It has been reported that the temperature and time of synthesis, solvent compositions, reagent ratios, reagent concentrations, metal salt, and pH play key role in MOFs’ synthesis (Biemmi et al., 2009; Li et al., 2011; Stock and Biswas, 2012). Accordingly, slight change in any of these parameters can increase network connectivity and adversely affect gas storage and separation application. Therefore, large number of experimental trials are required to discover the new desired MOFs in which the reaction parameters are systematically varied.
MIL-53 (MIL stands for Materials Institute of Lavoisier) is one of the most well-known MOFs which is involved of MO4(OH)2 corner-sharing octahedral chains (M=Al, Cr, Fe, Ga, Sc, In) linked by terephthalate groups to form a three-dimensional building (Kozachuk et al., 2013). MIL-53(Al) provides several advantages compared with other MOFs such as high thermal stability, cheap, and available raw materials. It is also moisture resistant and has relatively high surface area which makes it an attractive MOF candidate for adsorption process (Alhamami et al., 2014; Burtch et al., 2014; Camacho et al., 2015; Heymans et al., 2012; Lukose, 2012; Mowat et al., 2011; Patil et al., 2011).
In general, the required parameters for MOF production scale-up are as follows:
Raw material availability and cost; Space–time–yield of synthesis (STY): kilograms of MOF product per cubic meter of reaction mixture per day, kg/m3 day; Time and amount of solvent for filtration/washing; Drying and activation condition; Stability; Safety and environmental issues.
STY, as the most important parameter for scale-up, should be as high as possible. For maximization of STY, the yield of final product must be high and the total time of production should be low as possible (Ma and Zhou, 2010; Stock and Biswas, 2012; Yilmaz et al., 2012). In addition to former parameters, surface area of adsorption should be high to create high capacity for separation or purification process (Thomas and Crittenden, 1998). Walton and Snurr (2007) reported that Brunauer–Emmett–Teller (BET) method can be applied for characterization of MOFs and calculation of their specific surface area.
Despite of many studies on kinetics and thermodynamics of MOF synthesis (Bernini et al., 2010; Forster et al., 2004, 2005; Lee et al., 2006; Mahata et al., 2008; Stavitski et al., 2011; Tong et al., 2004; Yeung and Cheetham, 2014), there are few researches and publication on the thermodynamic and kinetic synthesis of MIL-53 synthesis (Haque et al., 2010). To the best of our knowledge, there is no work on the identification of main synthesis and activation parameters of MIL-53(Al) (i.e. the factors that have governing impact on the product formation) by considering both of them. In this work, the effect of synthesis and the activation parameters on final products were investigated using design of experiments (DOE) in order to minimize the number of trials. The response for DOE is an objective function which is representative for STY (the factor for scale-up) and BET surface area (the factor for separation applicability). It should be noted that CO2 adsorption-based separation at low pressure range (like flue gas) is mainly controlled by surface functionality of the adsorbent surface while at high pressure range (like biogas), it is mostly influenced by surface area of the adsorbent which is this discussion issue. In the other hand, the surface functionality of the synthesized materials was not changed in the experiences. Therefore, in this research, BET surface area, as a key factor for the adsorbent capacity, was investigated to evaluate the adsorbent performance for CO2 separation.
The objective function was considered as BET surface area multiplied by yield of product divided by total time of production. In other words, the target is to maximize BET surface area and yield at the minimum required time for final product (MIL-53(Al)) formation. Organic ligand-to-metal salt molar ratio, temperature, and time of synthesis as well as temperature and time of calcination (activation) were selected as variables for this DOE.
Also, equilibrium adsorption isotherm of the selected sample was measured for CO2 and CH4. Well-known Langmuir, Sips (Langmuir and Freundlich), and Toth models were applied to fit the experimental data (Foo and Hameed, 2010). Finally, the selectivity of CO2 over CH4 was predicted based on extended Langmuir (EL) and the ideal adsorbed solution theory (IAST) models (Myers and Prausnitz, 1965) and then compared with experimental data.
Experimental
Materials and methods
In this work, hydrothermal method as a well-known method was used to synthesize MIL-53(Al) samples (Férey et al., 2003; Lieder et al., 2010; Liu et al., 2008; Loiseau et al., 2004; Thuy et al., 2008). For this purpose, 13 g Al(NO3)3·H2O (Riedel-de Haen AG, 98%) and 2.88/6.91 g 1,4-benzenedicarboxylic acid (BDC) (Sigma Aldrich, 98%) were mixed with 50 cm3 deionized water and then placed in a Teflon-lined steel autoclave and heated up. The working volume of reactor was approximately 100 cm3 with 40 mm internal diameter and height of 80 mm. At the end of reaction time, the autoclave was cooled down to room temperature and the formed white powder was filtered and washed with deionized water until the pH of filtrate reached about 7. After overnight drying of wet powder at 353 K, dried powder was calcined in hot air stream in order to obtain buff-colored powder.
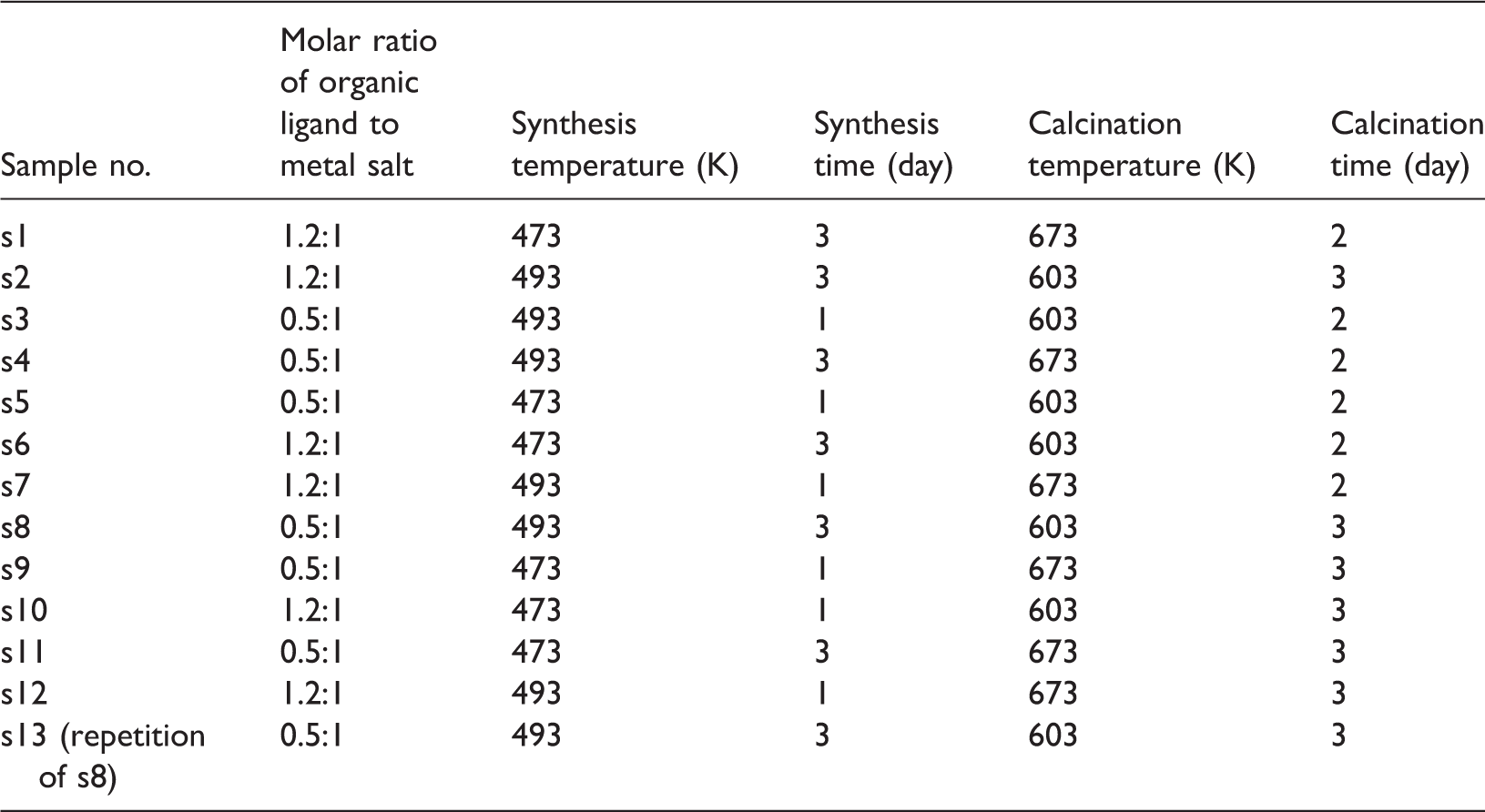
Plackett and Burman method (P–B design) with 12 runs was applied to identify those variables that are most influential on the response of DOE. Many factors can be studied using P–B design with minimum number of trials. In this method, all main effects are estimated with the same precision and major variables impacting on the response can be derived (Antony, 2014).
The response of DOE should be quantitative, singular, universal, and measurable (Lazic, 2004). Hence in this study, an objective function (product of yield and BET-specific surface area divided by total production time) was selected as the main response of DOE. In addition, BET-specific surface area, relative crystallinity, and product yield, separately, were selected as responses.
Synthesis and calcination conditions of trials.
The BET technique (in the relative pressure range from 0.05 to 0.2) was used to estimate the surface area of samples using N2 sorption at 77 K up to 1 bar.
The results were analyzed using Minitab statistical software (release 14, Minitab Inc.) to identify the main effective variable on the responses. It should be noted that repetition of run 8 (s13) was carried out just for repeatability evaluation. Adsorption isotherm measurements of pure CO2 and CH4 were performed with an in-house built apparatus by the volumetric method. Binary mixture adsorption data were also measured by volumetric method and chromatographic method (Keller et al., 1999). The composition of mixture gas, after reaching equilibrium, was also analyzed using gas chromatography (GC-HP 5890, Agilent Technologies, Basel, Switzerland).
The schematic diagram of the experimental setup is demonstrated in Figure 1. The apparatus consisted of an adsorption cell, a holder (reference) cell, two pressure transmitters (one for high and another one for low pressure range), two vacuum pumps (one for imposing vacuum and another one for circulation), an electrical heater equipped with thermocouple, a temperature indicator and controller, some valves, and a check valve. At first, the adsorption column was loaded with 0.5 g of the adsorbent. After outgassing and activation of the adsorbent at 493 K in situ for 4 h under vacuum pressure, dead volume of the adsorbent was calculated using the helium gas measurement while it is assumed that helium is not adsorbed on the adsorbent. The sorption tests were performed using the volumetric method by changing pressure of the holder cell, which is isolated from adsorption cell by a valve. After opening the isolating valve, the pressure of adsorption cell changed until reaching the equilibrium condition. By knowing the exact volume of different parts of the apparatus, the numbers of the gas moles adsorbed could be calculated according to the initial and equilibrium pressures.
Schematic diagram of the experimental setup.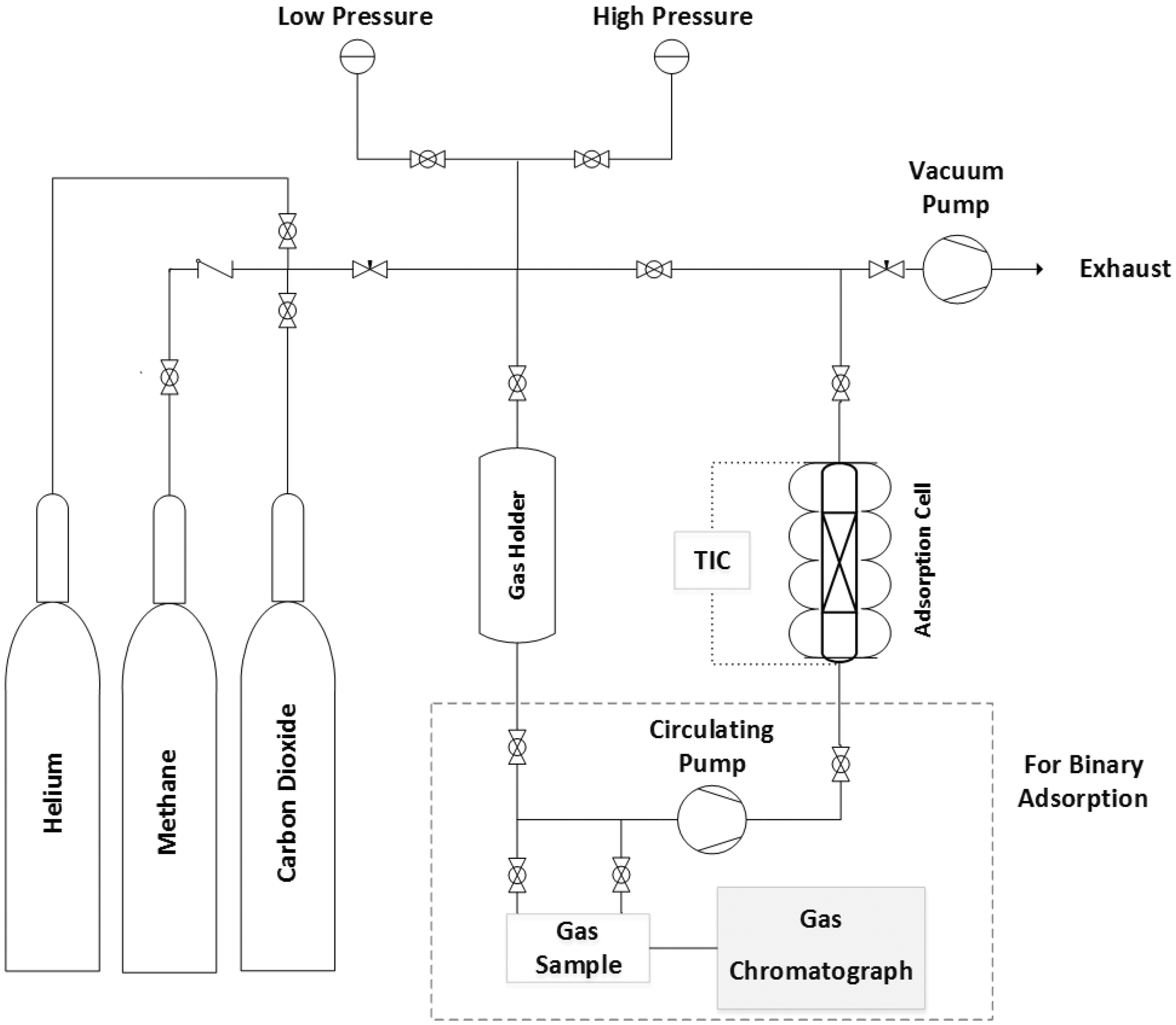
For binary adsorption, in addition to the equilibrium pressure, the equilibrium composition should be known. Therefore, the volumetric-chromatographic method was applied. Final composition of the gas mixture was analyzed with a GC. A pump was applied to circulating gas mixture to accelerate the equilibrium and avoid concentration gradient. The experimental setup was placed in a room equipped with a ventilation system which regulated the room temperature and kept the ambient temperature constant about 298 K.
Characterization
As the XRD pattern is the fingerprint of crystalline material (Gustafsson, 2012; McCusker and Baerlocher, 2007), all synthesis materials were characterized by X-ray powder diffraction analysis at ambient temperature using a PHILIPS PW-1840 X-ray diffraction machine with Co lamp.
By using Micromeritics TriStar II, N2 adsorption–desorption isotherm at 77 K up to 1 bar pressure was measured.
The morphology of the samples was investigated by field emission scanning electron microscopy (FESEM, HITACHI S4160).
Results and discussion
Relative crystallinity
XRD pattern of run s4 as a selected sample is shown in Figure 2. The XRD pattern of all samples (as shown in Figure s1 of supplementary material) provided a good agreement with the literature (Loiseau et al., 2004) which confirms successful synthesis of MIL-53(Al) in all runs regardless of the synthesis conditions. Average crystallite size of the samples was estimated in the range of 40–100 nm using the Scherrer equation (Harold and Klug, 1974). The strong and narrow diffraction peaks indicate a high purity and good crystallinity of the synthesized materials. Relative crystallinity was defined as the ratio of the sum of the major peaks intensities of each sample to the best crystallinity one (Li et al., 2009). This method is similar to calculation of relative crystallinity of zeolite (ASTM D3906). Therefore, the relative crystallinity was calculated by comparing the diffraction intensities of the representative major peaks of the samples at the range of 2θ=0–30. The results are provided in Table 2.
XRD pattern of s4. XRD: X-Ray Diffraction. Relative crystallinity, crystallite size, and particle size of trails.

Since at high crystallinity the pores are easy to reach, the kinetic rate of adsorption is estimated to be high. Therefore, high crystallinity is desirable in adsorption processes due to improvement of kinetic rate of adsorption. Results confirmed that high relative crystallinity (s4, s7, and s12) was obtained at high synthesis and calcination temperature.
The effect of main variables on crystallinity is shown in Figure 3. The Pareto chart exhibits the absolute magnitude of the effects and draws a reference line. Each effect which passes the reference line in Pareto plot is considered as the most important variable for the response (Antony, 2014).
The effect of the main variables on relative crystallinity. (a) Pareto chart of the standardized effects (response is relative crystallinity, α = .05) and (b) main effects plot (data means) for relative crystallinity.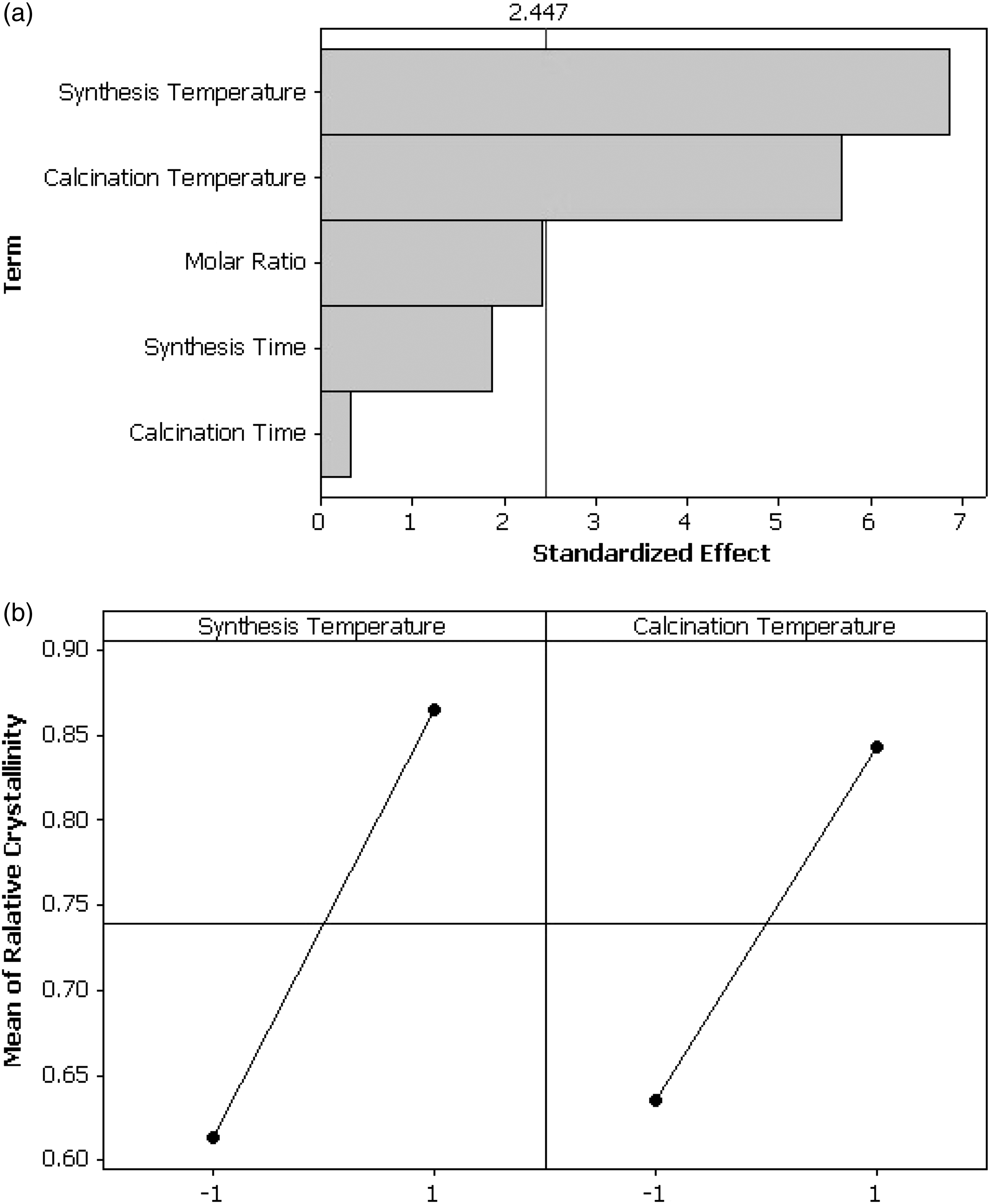
The results indicated that the calcination temperature and the synthesis temperature are the most effective parameters on the crystallinity of the final products. The calcination temperature had positive effect due to the higher impurities removal at high temperature. It should be noted that the presence of disordered terephthalic acid, as a major impurity, reduces the relative crystallinity of the final product because of differences in crystal structure of MIL-53(Al) before and after BDC removal. Loiseau et al. (2004) reported that heating to 548–693 K can remove the unreacted BDC from the framework of MIL-53(Al) and decomposition of MIL-53(Al) occurs at temperature above 773 K. At high level of calcination temperature (673 K), which is near the sublimation temperature of BDC (675 K) (Lucchesi and Lewis, 1968), hot air stream removed most of the unreacted terephthalic acid from structure of the synthesis material without structural collapse of the framework.
It has been shown that the rate of nucleation and crystal growth obeys Arrhenius law (Cubillas and Anderson, 2010; Haque et al., 2010). Therefore, a rise in the synthesis temperature will increase both the nucleation rate and the crystal growth. As a consequence, the crystallinity of the samples increases by increasing the synthetic temperature. Forster et al. (2004, 2005), Lee et al. (2006), Mahata et al. (2008), and Yeung and Cheetham (2014) investigated the kinetic and thermodynamic of MOF synthesis. They reported that temperature is a more important factor for the MOF synthesis than reaction time which is in agreement with the results of this work.
The results indicated that molar ratio, synthesis time, and calcination time have no significant effect on crystallinity, compared to other variables (synthesis and calcination temperatures).
The SEM images of the samples are depicted in Figure s2 of supplementary material. All FESEM images indicate that the particles have nonuniform size (up to 5 µm). This nonuniformity can be explained by simultaneous process of the nucleation and crystal growth (Haque et al., 2010). The particle size of the samples obtained from FESEM images along with the crystallite size is presented in Table 2. As this table indicates, the same trend for particle size and crystallite size can be observed for the samples. The impact of the main parameters on the particle size is illustrated in Figure 4. The synthesis temperature was found to be the most major influence on the particle size of MIL-53(Al).
The effect of the main variables on particle size. (a) Pareto chart of the standardized effects (response is relative crystallinity, α = .05) and (b) main effects plot (data means) for relative crystallinity.
The particle size is affected by the factors such as nucleation, crystal growth, and supersaturating. The crystal growth is effected more than the nucleation since the activation energy of the crystal growth of MIL-53(Al) is higher than the nucleation (Haque et al., 2010). Also, increasing of the synthesis temperature leads to reducing the supersaturation of the solution by increasing the solubility and as a result, large particles were obtained.
The results of the crystallite size of the samples agreed well with the results of relative crystallinity. It means that the increase of the crystallite size can explain the rise of the relative crystallinity of a sample and vice versa.
The results implied that the other synthesis and calcination parameters have no significant effect on particle size, compared to synthesis temperatures.
BET surface area
The adsorption and desorption isotherms of N2 at 77 K are depicted in Figure 5. According to Figure 5, N2 sorption isotherms were reversible and did not show any hysteresis upon desorption. Obtained isotherms of all the samples could be classified as type-I according to IUPAC classification (Thommes et al., 2015). The major uptake of N2 in the adsorption/desorption isotherms occurred at relatively low relative pressure and reaches the plateau. The obtained results from the N2 sorption isotherms are presented in Table 3. As it is observed in Table 3, BET-specific surface area of the samples was between 572 and 1203 m2/g which was below the theoretical BET surface area of perfect crystal (1632.8 m2/g) (Mounfield and Walton, 2015). The minimum BET surface area, approximately was close to BET surface area of the conventional zeolite adsorbent, 13X.
Isotherms of nitrogen adsorption and desorption at 77 K up to 1 bar. Results of nitrogen sorption at 77 K up to 1 bar. BET: Brunauer–Emmett–Teller.

The effect of the main variables on BET surface area is shown in Figure 6. The results indicated that calcination temperature and synthesis temperature had the most significant effect on BET surface area. The highest BET surface area (1203 m2/g) was obtained for the sample synthesized at 493 K and calcined at 673 K. This value for BET surface area is higher than the reported value (1140 m2/g) by Loiseau et al. (2004) which was synthesized at the same temperature but calcined at lower temperature (603 K). The temperature of 603 K might be not enough for perfect removal of impurities. Rallapalli et al. (2010, 2011) reported an enhancement of surface area for MIL-53(Al) to 1235 m2/g by the solvent extraction method. This enhancement may be related to higher removal of impurities by extraction method.
The effect of the main variables on BET-specific surface area. (a) Pareto chart of the standardized effects (response is BET surface area, α = .05) and (b) main effects plot (data means) for BET surface area. BET: Brunauer–Emmett–Teller.
A possible explanation for enhancement of BET surface area as a result of increasing of synthesis temperature could be an increasing of nucleation and crystal growth rate and consequently high crystallinity which implies largely accessible empty pores and higher specific surface area.
Yield
Yield of final products.

The effect of the main variables on the yield of the final product is presented in Figure 7. The influence of the synthesis temperature and the molar ratio were found to be the most significant on yield of the final product. From Figure 7, it can be seen that increasing the molar ratio of BDC:Al significantly led to reduction of yield; however, increasing of the synthesis temperature led to higher yield. The other variables had no significant impact on the product yield.
The effect of the main variables on yield of final product. (a) Pareto chart of the standardized effects (response is yield, α = .05) and (b) main effects plot (data means) for yield.
As mentioned previously, higher synthesis temperature results in higher nucleation and crystal growth rate, which is responsible for increasing the yield. Increasing the organic ligand-to-metal molar ratio causes terephthalic acid to be in excess that leads to yield decrement.
Objective function
High BET-specific surface area and yield and low synthesis duration time are considered as desirable issues for MOF synthesis. Hence, maximization of the product of BET surface area by yield divided by total time of production was considered as objective function. The effect of main variables on the objective function (BET*yield/time) is depicted in Figure 8. Results demonstrated that synthesis temperature and molar ratio were the most important variables that affect the response. Recently, Sun and Sun (2014) reported that the temperature is one of the key parameters among reaction conditions which is in consistent with findings in this work.
The effect of the main variables on response (BET*yield/time). (a) Pareto chart of the standardized effects (response is BET*yield/time, α = .05) and (b) main effects plot (data means) for BET*yield/time.
The results disclosed that reduction of synthesis time from 72 to 24 h and calcination time from 72 to 48 h implies negligible impact on objective function. It means that STY can be increased as a result of reducing of the required time for preparation of MIL-53(Al) from six to three days.
It was observed that the increasing of the calcination temperature from 603 to 673 K had negligible effect on the objective function.
Adsorption experiments
Pure gas adsorption of CO2 and CH4 on the selected sample (s4) was measured by volumetric method at 298 K and up to 20 bar pressure. This sample was selected due to its highest crystallinity and BET surface area among the samples. The obtained isotherms are shown in Figure 9. A step (at about 5–8 bar pressure) was observed in CO2 adsorption isotherm which was not regular for microporous materials. This behavior could be attributed to MIL-53 structural transition. MIL-53 series exist in two forms (but same topology) including narrow pore (NP) with almost square shape pores (0.85 nm × 0.85 nm), and large pore (LP) with lozenge shape pores (0.26 nm×1.36 nm). They exhibit reversible structural transformation from LP form to NP form upon adsorption of some guest molecules which is called “breathing effect” (Alhamami et al., 2014). This phenomenon has been observed previously for MIL-53 with polar molecules such as CO2 with significant quadrupole moment (Li et al., 2009).
The adsorption/desorption isotherm of CO2 and CH4 on s4 up to 20 bar at 298 K.
It has been reported that the structure of MIL-53 upon adsorption of CO2 at a pressure from atmospheric pressure up to about 5 bar and more than about 8 bar is NP and LP, respectively. There is no intermediate structure between 5 and 8 bar for MIL-53 and a combination of both forms coexists at the same time; however, by increasing the pressure more than 5 bar, the fraction of LP increases (Hamon et al., 2009). Because of stepwise behavior in the adsorption isotherm of CO2, it was divided into two sections for fitting appreciate model for each section. One model for the low pressure region (NP) and another one for the high pressure region (LP) was fitted to the experimental data.
A little increase was observed in the CO2 and CH4 adsorption capacity of s4 comparing with previously reported data (Bourrelly et al., 2005).



pressure of gas at equilibrium with adsorbed phase (bar)
Langmuir, Sips, and Toth fitting parameter for CO2 and CH4 adsorption on s4 at 298 K.
LP: large pore; NP: narrow pore; SSE: Sum of the Square of Errors.
adsorbed amount per mass of adsorbent (mmol/g)
saturation or maximum capacity of adsorbent (mmol/g)
affinity coefficient (1/bar)
adsorbate–adsorbent interaction (surface heterogeneity) parameter
Sum of the square of errors (SSE) as the most commonly used error function (Sreńscek-Nazzal et al., 2015) was considered as an objective function for fitting these isotherms to experimental values. R-square or coefficient of determination, denoted as R2, and SSE of each fitting are provided in Table 5. According to R2 and SSE values for all models, a good agreement between experimental adsorption data and model results was observed. Maximum adsorption capacity of the pure gases (qs), which was estimated from models, was in close agreement with each other according to Table 5. In all pressure ranges and in all three models, the affinity of MIL-53(Al) for CO2 was observed to be so much higher than CH4. The affinity of MIL-53(Al) for CO2 adsorption at high pressure (LP region) was less than one at low pressure (NP region) due to the weaker interaction of CO2 molecules within the LP structure (Hamon et al., 2009; Ramsahye et al., 2007). The maximum adsorption capacity (Qs-estimated by the Toth model) for the selected sample was found to be about 15% higher than industrial powder MIL-53(Al) (Basolite®, A100) (Heymans et al., 2012). The breathing effect has not been reported in A100 which could be related to the use of DMF as the solvent for its synthesis (Heymans et al., 2012; Mounfield and Walton, 2015). Therefore, the industrial sample affinity for CO2 adsorption (b-estimated by the Toth model) has been reported to be equal at low and high pressure which is near to the affinity of the selected sample at high pressure (LP) for CO2 in this study. However, the affinity of the selected sample at low pressure (NP) was about seven times more than it for A100.
By using Matlab software, EL and IAST models, based on fitted parameters of Langmuir equation for pure CO2 and CH4 (due to its simplicity), were used to predict the selectivity of binary mixture adsorption. The separation of CO2/CH4 mixtures with CO2 mole fraction ranging from 0.2 to 0.5 was investigated at the pressure between 1 and 8 bar.
The constituents of biogas depend on the decomposed material; however, usually the composition of CO2 in biogas is in the range of 0.15–0.6. Consequently, partial pressure of CO2 is less than 5 bar in conventional pressure swing adsorption (PSA) process, due to typical operating adsorption pressure (Lestinsky et al., 2015). It has been reported that structural deforming of MIL-53(Al) upon adsorption of mixture of CO2/CH4 is related to partial pressure of CO2 (except for low CO2 content) and MIL-53(Al) exists in the narrow form at pressures of 1–7 bar and CO2/CH4 ratios more than 0.2/0.8 (Ortiz et al., 2012; Coudert et al., 2009). Hence adjustable parameters of Langmuir model at low pressure region were used in binary mixture models. The obtained selectivities from IAST and EL models for three different compositions in the pressure range of 1–7 bar are depicted in Figure 10. As it was expected, the EL selectivity of CO2/CH4 was constant (equal to 10.75) for all gas compositions and over entire pressure ranges. On the other hand, the IAST selectivity factors were variant with both total pressure and composition. At low pressure region, the selectivity calculated based on EL equation and IAST was almost similar. The IAST model results indicated that by increasing the total pressure from 1 to 7 bar for a particular gas composition, the selectivity decreases. In low pressure region, the selectivity is mostly controlled with the interaction of the adsorbent with the adsorbate. Hence, CO2 molecules were mostly adsorbed by adsorbent due to high affinity of MIL-53 for them. Whereas, the CO2 molecules needed to compete with CH4 molecules for available adsorption sites as the total pressure was increased.
IAST and EL predicted and experimental selectivity toward CO2/CH4 as a function of pressure and composition at 298 K.
By increasing CO2 molar composition from 0.2 to 0.5, the selectivities, resulted from IAST model, decrease. For example at 7 bar, the selectivity decreases from 5.7 to 4.6 as the composition of CO2 increases from 0.2 to 0.5 in the mixture of CO2/CH4. The reasons for these phenomena could be explained by changing of CO2 adsorption mechanism. At low CO2 content, CO2 molecules interacted strongly with hydroxyl groups of MIL-53 (two hydroxyl group per unit cell) and selectively adsorbed while at higher CO2 content, they must to be adsorbed on remaining hydroxyl sites and other available site (in nonspecific way) which leads to lower selectivity.
To examine the models, three different binary mixtures of CO2/CH4 with molar ratio of 0.2/0.8, 0.35/0.65, and 0.5/0.5 (equimolar) were tested at total pressure of 5 bar. The obtained experimental selectivity values were about 7.6, 6.8, and 6.1, respectively. The experimental selectivity for equimolar mixture of CO2/CH4 was found to be greater than that previously reported for MIL-53(Al) and MIL-53(Cr) which was determined from breakthrough curves (Finsy et al., 2009; Rallapalli et al., 2011) that are in consistent with results of commercial MIL-53(Al) (Basolite® A100) (Heymans et al., 2012). These obtained values were slightly greater than the values calculated from IAST model (5.32, 5.82, and 6.84, respectively) but also smaller than EL selectivity value (10.75). The IAST in conjunction with Langmuir equation showed better results than EL theory for prediction of binary mixture adsorption of CO2/CH4. The difference between the selectivity values of experiments and the results obtained from IAST model could be attributed to the steric effect of NP of MIL-53(Al) which may exclude more CH4 than CO2 molecules due to higher kinetic diameter of CH4 than CO2. The significant difference between experimental selectivity of this work and the results obtained by Finsy et al. (2009) and Rallapalli et al. (2011) may be related to the experimental procedures for the measurement of selectivity. In breakthrough experiments which were done by mentioned researchers, thermodynamic equilibrium was not reached due to the short contact time while CH4 molecules could be trapped in NP form. Another possible reason for this observation could be related to higher BDC removal from s4 sample which decrease the LP portion in structure of MIL-53(Al). It has been shown that the residual unreacted BDC retains the structure of MIL-53(Al) in LP form (Loiseau et al., 2004). As mentioned before, the affinity of CO2 for MIL-53 increased as the LP to NP transition occurred. Consequently, s4 sample with high NP portion possess high CO2/CH4 selectivity.
It should be noted that selectivity is the key factor for evaluating a new adsorbent. The minimum acceptable value of selectivity factor for PSA application was reported to be 2 (Tagliabue et al., 2009). Hence, the obtained MIL-53(Al) is a proper adsorbent for CO2 separation.
Adsorption capacity and selectivity of MIL-53(Al) for CO2/CH4 were comparable to those reported for 13X zeolite (Silva et al., 2014), therefore it can be considered a promising material for CO2 separation from CH4.
Conclusions
The results exhibited that molar ratio, calcination time, and synthesis temperature were the most effective parameters on the relative crystallinity, BET surface area, morphology, and yield of MIL-53(Al).
It can be highlighted that MIL-53(Al) can be synthesized in one day instead of three days with no significant decrement in BET surface area, crystallinity, and yield. As-synthesized MIL-53(Al) can be calcined in two days instead of three days. It means that the total time for preparation of MIL-53(Al) was reduced from six to three days and time saving leads to higher STY. Also, it was found that increasing the organic ligand-to-metal molar ratio remarkably reduced the yield of final products.
Calcination of MIL-53(Al) at 673 K for two days resulted in higher crystallinity and BET surface area compared to the regular calcination method (at 603 K for three days) as well as higher CO2/CH4 selectivity was obtained.
The obtained remarkable CO2 adsorption capacity and selectivity confirmed that the synthesized MIL-53(Al) can be appropriately applied in a PSA process for CO2 separation from biogas.
Also, it was concluded that IAST model can properly predict the binary mixture adsorption of CO2/CH4 in the usual pressure region of the PSA process with a biogas feedstock.
In the usual pressure region of PSA process for CO2 capture from biogas, IAST model can properly predict the binary mixture adsorption of CO2/CH4.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors wish to thank Research Institute of Petroleum Industry of Iran for the financial support of this study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
