Abstract
Diffusional mass transfer models with and without external resistance were applied to represent the adsorption of Acid Red 357 (AR357) and Acid Black 210 (AB210) by tannery solid wastes. The mass transfer parameters, such as, external mass transfer coefficient (k f ), surface diffusion coefficient (D s ), and Biot number (B i ) were estimated and interpreted. It was found that the two models agreed with the experimental data and, very similar values of the parameters were obtained. This indicated that the external mass transfer mechanism can be neglected. Then, the model without external resistance, which is simpler, can be used. The D s values ranged from 5.01 × 10−13 to 1.30 × 10−12 m2 s−1 for AR357 and from 3.19 × 10−14 to 5.38 × 10−14 m2 s−1 for AB210. The high values of the B i number confirmed that the adsorption of AR357 and AB210 on tannery solid wastes was controlled by surface diffusion.
Introduction
Colored effluents are a direct result of the dyes production, and also, consequence of its use in textile and related industries (Noroozi and Sorial, 2013). It is estimated that about 6–12 L of colored effluents are generated per kilogram of wet blue leather, during the wet finishing process (Piccin et al., 2012). The inadequate release of these colored effluents in aqueous ecosystems is esthetically unpleasant and leads to a reduction of sunlight penetration, which in turn, decreases the photosynthetic activity, dissolved oxygen concentration, and water quality. Furthermore, had acute toxic effects on aquatic flora and fauna, causing severe environmental problems worldwide (Saratale et al., 2011). This scenario shows the importance of the treatment of dye containing effluents prior to their final discharge into the environment.
Several technologies for dyes removal from effluents are presented in the literature (Gupta and Suhas, 2009; Mezohegyi et al., 2013; Saratale et al., 2011; Srinivasan and Viraraghavan, 2010). Among all of these, adsorption has been preferred due to its cheapness and high-quality of the treated effluents, especially for well-designed processes (Dotto et al., 2014; Filipkowska and Rodziewicz, 2012; Jin et al., 2012; Samarghandi et al., 2014). The main adsorbent used for dye removal is activated carbon, but, the use of cheap and eco-friendly materials have been studied as alternative (Ali, 2012; Ali et al., 2012). Recently, tannery solid wastes (TSWs) were successfully employed for dyes adsorption, providing some advantages, for example, the reuse of treated water in leather industry and an extended survival of such waste before reaching its final destination (Piccin et al., 2012, 2013). Moreover, the reduction in color, applying TSW to treat colored tannery wastewater, is able to reduce significantly the presence of organic and inorganic pollutants in wastewater, as well as, toxicity. Also, TSW can be applied to equipments already existing in this type of industrial plant (Gomes et al., 2016; Piccin et al., 2016). However, more detailed studies about the mass transfer mechanism of dyes adsorption onto TSWs are still necessary.
In solid–liquid adsorption systems, the mass transfer mechanism occurs by external mass transfer, intraparticle diffusion (pore volume diffusion, surface diffusion, or a combination of both mechanisms) and adsorption on the active sites (Ruthven, 1984; Suzuki, 1990). The diffusional mass transfer models are constructed on the basis in the above mentioned steps (Dotto et al., 2014; Ocampo-Pérez et al., 2010, 2011, 2012). In this way, these models can be used as realistic physico-mathematical tolls to elucidate the dyes adsorption onto TSWs. It should be highlighted that, interpretations about the mass transfer mechanism of dyes adsorption onto TSWs, based on diffusional models, were not performed yet. This information is fundamental for scale up and design purposes.
This work aimed to develop diffusional mass transfer models with and without external resistance to represent the adsorption of AR357 and AB210 by TSWs. The models were based on the mass transfer principles and used to represent the adsorption decay curves under different temperatures (288, 298, and 308 K). The values of k f , D s , and B i were accurately estimated and its dependence with the temperature was studied. The rate limiting step of the process was elucidated.
Materials and methods
Obtainment of the TSW
TSWs were obtained from chromium-tanned leather shaving operation in a local tannery (Portão-RS, Brazil). These residues were dried under vacuum conditions (−500 mmHg) at 60℃ for 48 h (Piccin et al., 2012). TSW were cut in rectangular shape (3.31 mm × 3.31 mm) with thickness of 0.50 mm.
Characterization techniques
TSW were characterized according to the moisture content (ASTM D3790-79) solid density (ρ s ) (picnometry), void fraction (ε p ), surface area, and pore size distribution (S BET ) (BET method) (Quantachrome Instruments, New Win 2, USA) (Dotto and Pinto, 2012). Scanning electron microscopy (SEM) (Jeol, JSM–6060, Japan) was used to observe the TSW surface. The main elements on the TSW surface were identified by energy dispersive X-ray spectroscopy (Jeol, JSM 5800, Japan). The main groups on the TSW surface were identified by Fourier transform infrared spectroscopy (FTIR) analysis (PerkinElmer, Spectrum 1000, USA) (Piccin et al., 2012).
Dyes
Acid Red 357 (AR357) (azo-dissulphonated Cr organic-complex dye, molecular weight 956.7 g mol−1, CAS 57674-14-3, λmax = 494 nm) and Acid Black 210 (AB210) (amine dissulphonated triazo organic dye, molecular weight 965.9 g mol−1, CAS 99576-15-5, λmax = 459 nm) were supplied by the Business Leather Unit of Lanxess Company, with purity higher than 95%. The optimized three-dimensional structural formula of the dyes was obtained from MarvinSketch software, version 14.9.22.0, and is presented in Figure 1. Distilled water was used to prepare all solutions. All reagents were of analytical-grade.
Optimized three-dimensional structural formula of the dyes: (a) AR357 and (b) AB210.
Adsorption experiments
The adsorption assays were carried out in batch conditions at different temperatures (288, 298, and 308 K) using a thermostated shaker (Nova Ética, 218 MBD, Brazil). Two hundred fifty milligrams of TSW (dry basis) were added to 10 mL of citric acid/sodium diphosphate buffer (0.1 mol L−1) at pH 2.5. After 10 min, 50 mL of dye aqueous solution (with desired concentrations) were added and the system was stirred at 150 r/min under controlled temperature. Aliquots were withdrawn in preset time intervals and the dye remaining concentration was determined by spectrophotometry (Varian, Cary 100, USA) at the maximum wavelength for each dye. The maximum wavelength used to quantify the dyes was obtained by a spectrophotometric scanning in the visible region. For Acid Red 357, λmax was 494 nm and for Acid Black 210, λmax was 459 nm. All adsorption experiments were performed in duplicate (n = 2) and blanks were performed. The experimental conditions were based on preliminary tests and literature (Piccin et al., 2012, 2013). The mass of dye adsorbed per gram of TSW at any time (q(t), mg g−1), and the mass of dye adsorbed at equilibrium (q
e
, mg g−1) were calculated by the equations (1) and (2), respectively
Model development
Main characteristics of TSW adsorbent.

Mean ± standard error for n = 3.
At first, the mass transfer model with external resistance was developed according to the mass balances in the liquid phase, describing the variation of dye concentration along the time (equation (3)), and in the solid phase, describing the variation of dye concentration along the time and in the solid position (equation (4)) (Ruthven, 1984; Suzuki, 1990). The initial and boundary conditions are presented in the equations (5), (6), and (7)





The equations are connected through the external mass transfer from the bulk liquid phase to the solid surface. The liquid concentration at surface C
S
was considered to be in equilibrium with the adsorbed phase concentration at solid surface. The equilibrium relations are defined according to the Langmuir isotherm (equation (8)) for Acid Red 357 and BET model (equation (9)) for Acid Black 210. The values of q
m
, k
L
, q
BET
, k1, and k2 for different temperatures can be found in Piccin et al. (2012).


After, the above model (equations (1) and (3) to (7)) was simplified considering the external mass transfer very fast (model without external resistance). In this case, k
f
is very high and C
S
is equal to C(t). Then, the equation (7) is reduced to equation (13). Equations (4), (5), and (6) that describe the solid volume are not changed and are rewritten in equations (10), (11), and (12)




The partial differential equations were discretized with respect to z coordinate through finite difference method and, the resulting system of ordinary differential equations was solved using is an integrator based on an adaptive explicit Runge–Kutta of fourth and fifth orders, by a Dormand–Prince pair (Dormand and Prince, 1980).
In order to allow the models evaluation at different temperatures simultaneously, D
S
was considered a function of the temperature (T), according to the equation (14) (Schwaab and Pinto, 2007)

In equation (14), D S,Tref (m2 s−1) is the surface diffusion coefficient at reference temperature Tref (in this work, Tref value was equal to 298 K), B is a parameter that describes how surface diffusion coefficient varies with temperature T. The dependence of k f with T was found to be negligible and a constant value for all temperature was estimated.
The parameters D
S
,
Tref
, B, and k
f
were estimated through minimization of the nonlinear least squares function presented in equation (15)

The error minimized in equation (15) was the sum of squared residuals, for example, the sum of squared difference between an observed value (experimental value) and the fitted value (model predicted value) provided by a model that is nonlinear. The minimization of equation (15) was performed by the Nelder–Mead simplex algorithm (Lagarias et al., 1998). The computer used for the parameters estimation has an Intel® Core™ i7–3770 with 3.40 GHz processor and 12 GB of RAM memory, running with the Windows 7 64 bits operating system. The software Matlab® was chosen to parameter estimation and model simulation.
Results and discussion
TSW characteristics
The SEM image, N2 adsorption isotherm, BJH pore size distribution and FTIR spectra of TSW are shown in Figures 2, 3, 4 and 5, respectively. The main characteristics of TSW adsorbent are presented in Table 1. Figure 2 reveals a complex, nonporous, and heterogeneous surface of TSW. Some interlaced fibers can be observed. This interlacement suggests a lower void fraction and surface area, as demonstrated by the BET analysis (Table 1 and Figure 3). The values of length, width, and thickness of TSW ensure that the mass transfer can be considered mainly in z direction (Table 1).
SEM image of TSW adsorbent. N2 adsorption/desorption isotherms of TSW adsorbent. BJH pore size distribution of TSW adsorbent. FTIR vibrational spectrum of TSW adsorbent.



The N2 adsorption/desorption isotherm (Figure 3) presented a type II sigmoid curve (Thommes et al., 2015). The BJH pore size distribution data (Figure 4) revealed that TSW has pores ranging from 100 to 1100 Å. The majority of pores are from 200 to 400 Å. These pore dimensions are extremely desirable for dyes adsorption, since these compounds have large molecular size (Figure 1). Also, comparing the pore size and dye molecules dimensions, it can be proved the diffusion mechanism. In the FTIR analysis (Figure 5), bands were observed near to 3000 cm−1, representing the symmetric stretching vibration of O−H group, which overlaps the NH vibration. Absorption bands at 1960 cm−1 are from the stretching vibration of C−H. Moreover, characteristic bands of the proteinaceous material structure can be seen at 1627 cm−1 (C=O), 1536 cm−1 (C−N−H), 1233 cm−1 (C−N), and 1078 cm−1 (C−O). On the basis of these structural characteristics, it can be inferred that the large dye molecules are transferred mainly by surface diffusion in the solid phase.
Equilibrium isotherms
In order to find the equilibrium relations according to the Langmuir isotherm (equation (8)) for Acid Red 357 and BET model (equation (9)) for Acid Black 210, equilibrium isotherms were constructed. The experimental isotherms were obtained in the same way of Adsorption experiments section, but, the solutions were stirred until the equilibrium. The equilibrium isotherms at 298 K are presented in Figure 6 (similar isotherms were found in the other temperatures). The isotherm parameters, q
m
, k
L
, q
BET
, k1, and k2 for different temperatures were estimated by the fitting of the models ((equations (8) and (9)) with the experimental data and are shown in Table 2. Based on the high values of R2 presented in Table 2, it can be seen that the Langmuir (equation (8)) and BET models (equation (9)) were able to explain the equilibrium adsorption of Acid Red 357 and Acid Black 210, respectively.
Equilibrium isotherms for the adsorption of Acid Red 357 (AR357) and Acid Black 210 (AB210) onto TSW at 298 K. Source: adapted from Piccin et al. (2013). Adsorption isotherm parameters of tannery dyes adsorption by TSW. Source: adapted from Piccin et al. (2013).

Table 2 shows also the thermodynamic parameters, which were estimated by the Van't Hoff's plot (Piccin et al., 2013). The negative ΔG values indicated that the dyes adsorption onto TSW was a spontaneous and favorable process. The negative ΔH values indicated that the dyes adsorption onto TSW was exothermic in nature. Another fact in Table 2 is that the adsorption capacity for AR357 was higher than the adsorption capacity for AB210. This can be occurred because AR357 is a Cr organic-complex dye and its structure is organized around the Cr, becoming more ordered and easy to adsorb. On the contrary, the AB210 molecule is long and its adsorption is hindered by the steric hindrance.
Model validation and parameters estimation
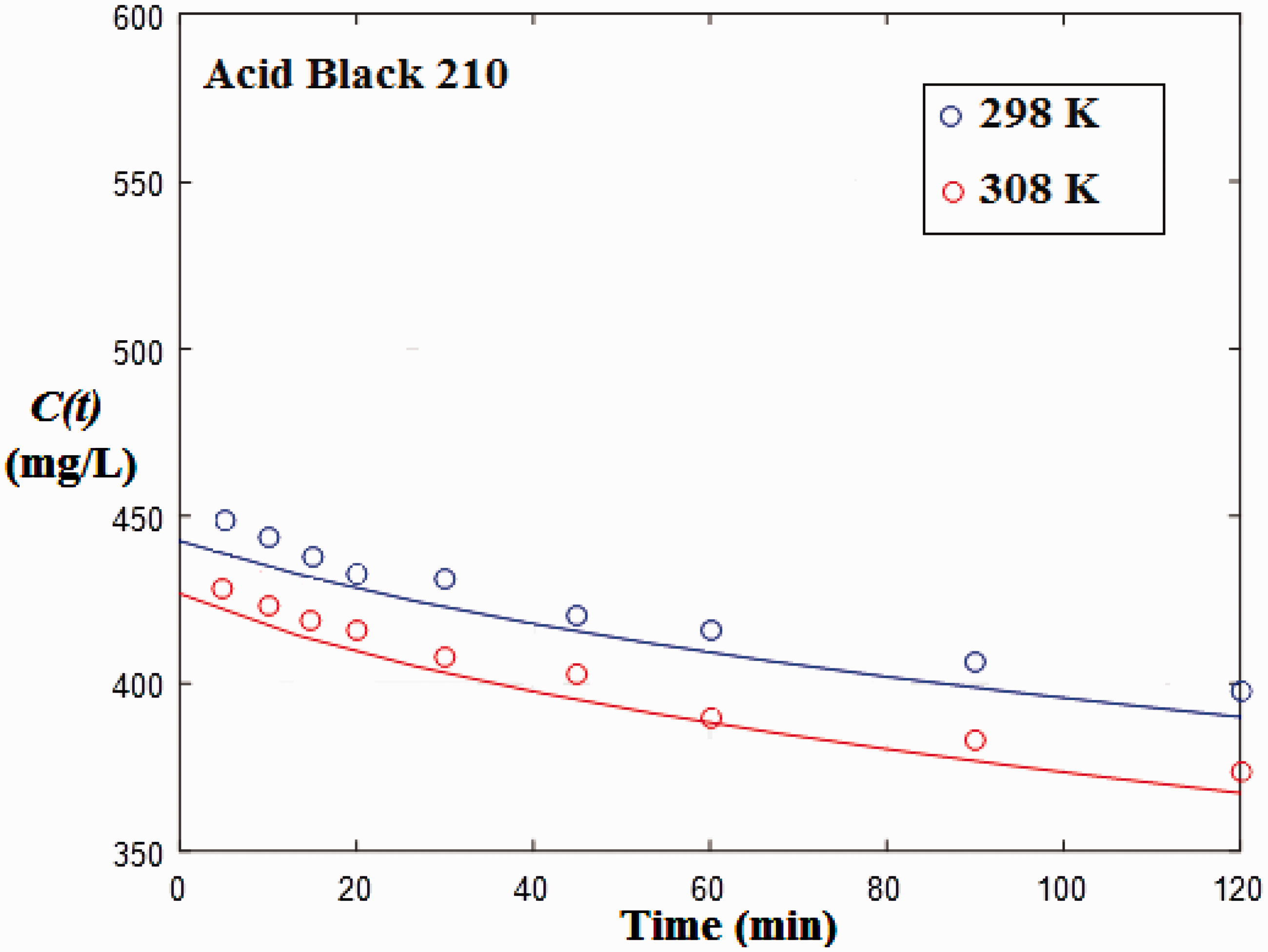
The mass transfer models with and without external resistance were used to represent the experimental data of the adsorption decay curves of Acid Red 357 and Acid Black 210 at different temperatures. During the models resolution and parameters estimation of Acid Black 210, it was observed that the experimental data at 288 K present a strange behavior, since adsorption rate was higher than the values observed at 298 K. For this reason, adsorption data of Acid Black 210 at 288 K were not considered. Figures 7 and 8 show the experimental and modeled data for the adsorption of Acid Red 357 and Acid Black 210 on TSW. It was found that the experimental data presented a typical behavior, with C(t) starting from C0 and tending to C
e
. The models agreed very well with the experimental data. It was also verified that the modeled curves were overlapped. This shows that, in this case, the external mass transfer is very fast and this mechanism can be neglected. As consequence, the model without external resistance, which is simpler, can be used.
Model fit for the adsorption decay curves of Acid Red 357 at different temperatures. Model fit for the adsorption decay curves of Acid Black 210 at different temperatures.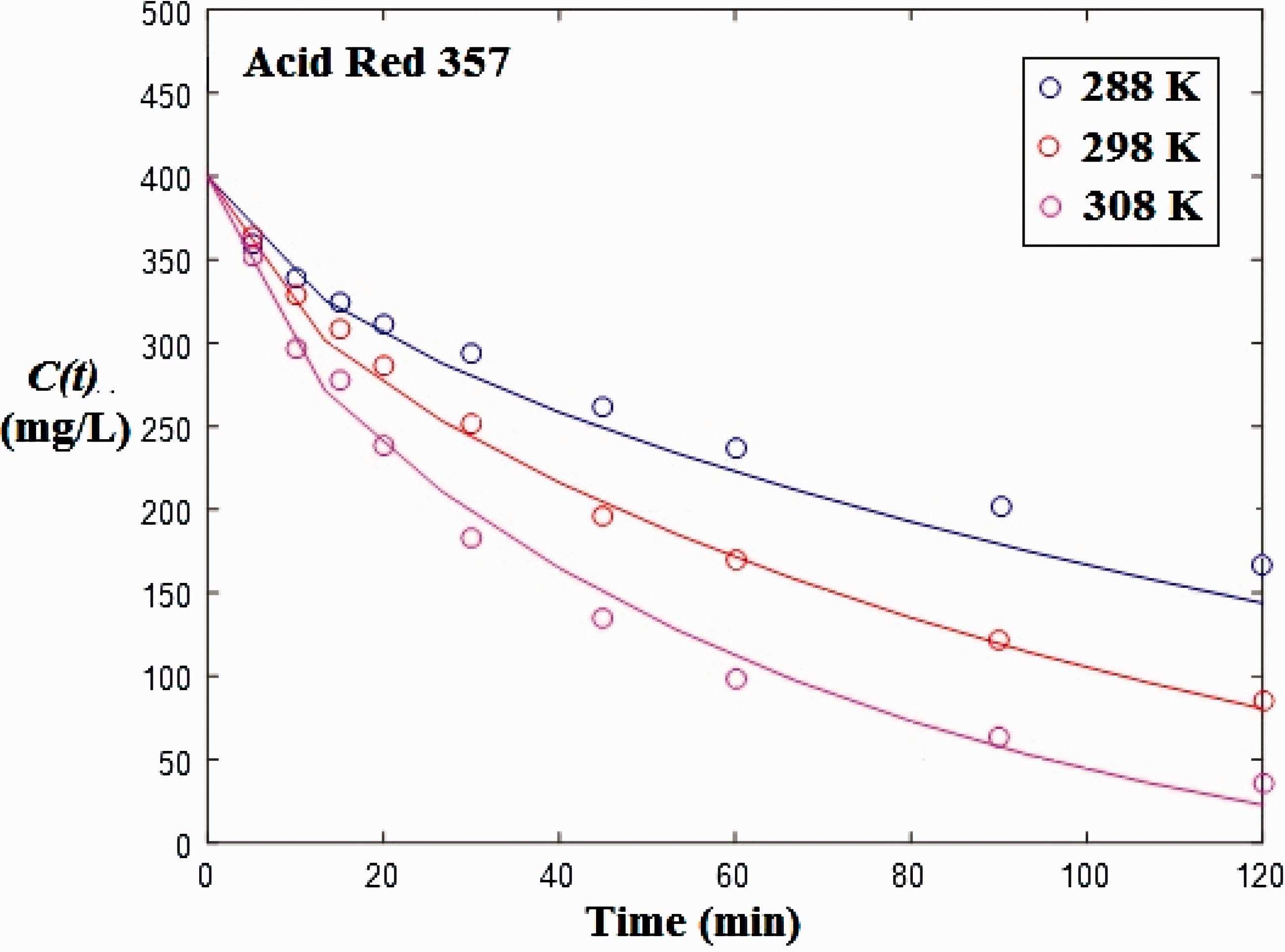
Estimated parameters with intervals of 95% of confidence for the adsorption of both dyes.

Student's t-test for C(t).

Note:
Fisher's exact test for C(t).

Note: Lower limit <
In general lines, it can be affirmed that both models (with and without external resistance) were suitable to represent the experimental data. However, the external mass transfer mechanism can be negligible, and then, the model without external resistance, which is simpler, is sufficient to represent the experimental data. From the statistical viewpoint, the parameters D S,Tref , B, and k f were significant (95% of confidence) and consistent, allowing the correct physical interpretation of the mass transfer phenomena that occurs in the adsorption of Acid Red 357 and Acid Black 210 on TSW.
Interpretation of the mass transfer parameters
Mass transfer parameters for the adsorption of Acid Red 357 and Acid Black 210 on TSW at different temperatures.

Table 6 shows that for both dyes, the surface diffusion coefficient increase with the temperature. The mass flux due surface diffusion occurs when one adsorbed molecule is desorbed from an active site and then, is diffused in the solid surface to another adsorption site (Medved and Cerný, 2011). So, the dependence of Ds against T can be explained as follows: At lower temperatures, the adsorbed dye molecules had lower internal energy, so, desorption and diffusion to another site is hindered. When the temperature was increased, the adsorbed dye molecules had higher internal energy. Then, the dye molecules required less energy to be desorbed from a site and diffused to another site. Similar trend was found by Ocampo-Pérez et al. (2010) in the adsorption of pyridine onto granular activated carbon. They found that the surface diffusion coefficient increased from 4.6 × 10−12 to 6.9 × 10−12 m2 s−1, when the temperature increased from 288 to 308 K.
The surface diffusion modified B
i
was calculated by the equation (16) in order to verify the predominance of surface diffusion in relation to the external mass transfer (Weber and Di Giano, 1995)

It is accepted that, for B i < 0.5, it exists a complete dominance of the external mass transfer, while for B i > 10 a considerable dominance of the surface diffusion exists (Costa and Rodrigues, 1985). The B i values obtained in this work are presented in Table 6. It was found that the B i values were higher than 200, confirming that the surface diffusion is the rate limiting step in the adsorption of Acid Red 357 and Acid Black 210 on TSW. This statement is in accordance with the model assumptions and TSW characteristics.
Comparison with other adsorbents
The maximum adsorption capacities obtained in this work were 239.9 and 138.1 mg g−1, for Acid Red 357 and Acid Black 210, respectively. For these specific dyes, consistent comparisons were not found. However, for dyes, in general, the adsorption capacities using waste materials as adsorbents can be from 0.75 to around 1500 mg g−1, depending of the experimental condition (Anastopoulos and Kyzas, 2014; Salleh et al., 2011; Yagub et al., 2014). In this way, it can be concluded that TSW has adequate adsorption capacity for the studied dyes.
Conclusion
In this work, mass transfer models were used to represent the adsorption data of Acid Red 357 and Acid Black 210 dyes onto TSWs. Experiments were performed at three different temperatures. Diffusional models with and without external resistance were employed to fit the experimental data. The results showed that external mass transfer resistance is negligible. In this way, the model without external resistance, which is simpler, can be used. The estimated values for surface diffusion coefficient ranged from 5.01 × 10−13 to 1.30 × 10−12 m2 s−1 for AR357 and from 3.19 × 10−14 to 5.38 × 10−14 m2 s−1 for AB210. These values are in accordance with literature and were dependent of the dye and temperature. The B i numbers were higher than 200, indicating that the internal mass transfer due surface diffusion was the rate limiting step in the adsorption of Acid Red 357 and Acid Black 210 onto TSW.
Footnotes
Acknowledgements
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Coordination for the Improvement of Higher Education Personnel (CAPES) and National Council for Scientific and Technological Development (CNPq).


