Abstract
In this present study, response surface methodology was applied for the optimization and evaluation of different heavy-metal sorption processes. Poly(vinylpyrrolidone) and poly(vinylpyrrolidone-co-methylacrylate) hydrogels were used as pH-responsive adsorbents to investigate their efficiency in the uptake of Cu2+, Ni2+, Zn2+ and Cd2+ metal ions from aqueous solution in a batch reactor system. The effect of operational parameters such as initial pH and contact time were studied using central composite design. Analysis of variance depicted the central composite design model was significant for Cu2+, Ni2+, Zn2+ and Cd2+ metal ions removal by the investigated hydrogels. A comparison between the model results and experimental data showed that poly(vinylpyrrolidone-co-methylacrylate) possessed higher metal ions binding affinity than poly(vinylpyrrolidone) hydrogel. The heavy-metal ions sorption kinetic well fitted the pseudo second-order kinetic model. Evaluation of the intraparticle diffusion model suggested the sorption process occurred in two phases: surface sorption and intraparticle diffusion. Isothermic results revealed that Freundlich and Temkin isotherms best described the adsorbate–adsorbent interactions. Maximum sorption capacities were observed for Cu2+ ion and were determined as 86.66 mg/g and 98.53 mg/g for poly(vinylpyrrolidone) and poly(vinylpyrrolidone-co-methylacrylate) hydrogels, respectively.
Introduction
High effluent discharge by the industrial sector such as metal finishing, welding, alloy manufacturing, plating and mining serves as vital pollutant sources of heavy metals in the soil and water. This has remarkably contributed to the increase in the concentration of toxic heavy-metal ions in waters which represent a potential source of several hazardous disadvantages on aquatic flora, animals and even on humans (Gonzalez-Gomez et al., 2014; Jadhav et al., 2014). Unlike some other organic pollutants, heavy metals are nonbiodegradable, highly toxic and carcinogenic. In addition, due to their high solubility, they can easily accumulate in the food chain thus causing serious health-threatening problems. Heavy-metal ions such as Cu2+, Ni2+, Zn2+ and Cd2+ classified as toxic metals have demonstrated to be associated with several health problems. For example, nausea and stomach ache which are gastrointestinal diseases are caused by Cu ion poisoning. Also, consumption of high levels of Ni ion in water may cause significant harm to the kidneys, lungs and the skin (Jamnongkan et al., 2014; Kalantari et al., 2014). Therefore, great attention towards the treatment of industrial effluents is eminent. Various treatment techniques have been investigated for the removal of heavy-metal ions from aqueous solutions. Among these techniques, sorption method has proven to be the most widely applied due to its simplicity, less time consuming and cost effective (Mahmoud, 2013). This method of treatment has demonstrated significant benefits attributed to the high-level loading capacity of heavy-metal ions for the used adsorbent. In the midst of used adsorbents, hydrogels have been classified as one of the most suitable material for the removal of heavy-metal ions from aqueous solutions (Antic et al., 2015; Soto et al., 2015; Wang and Li, 2015). Hydrogels, as previously defined in the literature, are 3D crosslinked polymeric material with the ability to retain high level of water and remain insoluble due to the existence of physical or chemical crosslinking in the material’s network structure (Erdem et al., 2016; Kamal et al., 2016; Yildiz et al., 2010). Stimuli-responsive hydrogels have shown great applicability in different areas of research such as sensors materials, drug delivery, tissue engineering and wastewater treatments. This is due to their unique capability to change in volume or shape in response to surrounding environmental signals. This stimuli response can either be physical or chemical such as temperature, pH, electric field and enzymes or light (Eswaramma et al., 2016; Koetting et al., 2016; Ozay, 2014).
Response surface methodology (RSM) is an assembly of mathematical and statistical analysis used to evaluate the relative significance of different experimental factors by approximating the shape of the response surface and determining optimum conditions within a limited number of experiments (Daneshvar et al., 2015; Xu et al., 2015). As one of the practical RSM models, central composite design (CCD), a second-order model, is widely used to estimate the true response surface (Alslaibi et al., 2014a). This second-order model minimizes the experimental points and maintains high accuracy and precision of prediction compared to the one factorial design. This methodology is favourable and widely applied in the optimization of sorption processes (Ghosh et al., 2014; Soleymani et al., 2015).
The objective of the present study was to optimize and evaluate the effect of initial pH and contact time on the sorption of Cu2+, Ni2+, Zn2+ and Cd2+ metal ions by RSM modelling using two types of pH-responsive hydrogels composed of poly(vinylpyrrolidone) (PVP) and poly(vinylpyrrolidone-co-methylacrylate) (P(VP-co-MA)). The optimum conditions for elimination of the heavy-metal ions were determined with RSM using CCD modelling. A comparison between the hydrogels as adsorbents in heavy-metal ions sorption was performed for estimating their performance by determining the best fit for the sorption system using different sorption kinetic and isotherm models.
Materials and methods
Materials
Vinylpyrrolidone (VP) and methylacrylate (MA) were purchased from Merck. Analytical grade 4,4′-dicyano-4,4′-azovaleric acid (Fluka), poly(ethylene glycol) (Merck) and 4-vinylbenzyl chloride (Sigma–Aldrich) were used as monomers for synthesizing the macroinimer (MIM) (Yildiz et al., 2010). Chemicals of Cd(NO3)2·4H2O, CuCl2·2H2O, ZnCl2·2H2O, NiCl2·2H2O, NaOH and HCl were supplied by Merck and used without further purification.
Synthesis of hydrogels
Preparations of PVP and P(VP-co-MA) hydrogels (Figure 1) were performed based on the detailed described procedure in previous study (Yildiz et al., 2010). Via bulk polymerization at 70℃, 2 g of VP was mixed with varying ratios of MIM (Table 1). The mixture was then allowed to stir for 5 min under argon gas and after 3 h, the tubes containing the different samples were then immersed in distilled water for 24 h. The obtained PVP samples were then dried under vacuum at 50℃ until constant weight. Synthesis of different P(VP-co-MA) samples was also performed as described above. Varying amounts of VP and MA (Table 1) were mixed in the presence of same MIM (0.30 g) amount and then the obtained hydrogel samples were dried. The prepared PVP and P(VP-co-MA) hydrogels were then characterized accordingly using various techniques as reported by our previous study (Yildiz et al., 2010), and then used for further applications and optimization.
Depicting structural formation of (a) MIM-initiated PVP and (b) MIM-initiated P(VP-co-MA) hydrogel. MIM: macroinimer; PVP: poly(vinylpyrrolidone); P(VP-co-MA): poly(vinylpyrrolidone-co-methylacrylate). Synthesis composition of monomers. MA: methylacrylate; MIM: macroinimer; PVP: poly(vinylpyrrolidone); VP: vinylpyrrolidone; PV: poly(vinylpyrrolidone) hydrogel sample; PVM: poly(vinylpyrrolidone-co-methylacrylate) hydrogel sample.

Preparation of aqueous metal ions solutions
Stock solutions of 1000 ppm concentration for Cu2+, Ni2+, Zn2+ and Cd2+ metal ions were prepared using CuCl2·2H2O, NiCl2·2H2O, ZnCl2·2H2O and Cd(NO3)2·4H2O, respectively. The sorption properties of PVP and P(VP-co-MA) hydrogels for the different metal ions were then tested under non-competitive conditions by immersing the hydrogels in the different metal ions solutions. The sorption experiments were performed by agitating a given amount of hydrogel in 50 mL of solution containing a metal-ion concentration of 5 ppm at varying pH of 2, 5 and 8 for a time period of 24 h. The pH value of each solution was adjusted by either 0.1 M HCl or 0.1 M NaOH. The metal-ion concentrations in the solutions were quantified before and after the hydrogel immersion by dilution to an appropriate concentration with distilled water and measured with atomic absorption spectrometer (Perkin-Elmer Analyst 800). Readings were conducted at wavelengths of 324, 232, 213.9 and 228.8 nm for copper, nickel, zinc and cadmium ions, respectively. Absorption readings were collected in duplicates, and the average value was determined as the final value.
Batch sorption investigations
Binding capacities of PVP and P(VP-co-MA) hydrogels for Cu2+, Ni2+, Zn2+ and Cd2+ metal ions were investigated under agitation at 200 r/min in 50 mL of varying metal-ion concentrations solutions (5–15 ppm), pH (2–8) and contact time (1–24 h) to reach sorption equilibrium. In order to minimize error, experimental runs were performed in duplicates, and then average results were obtained. The equilibrium sorption uptake for the heavy metal ions was calculated using the equation below:

Design experiment and optimization of sorption

Independent parameters and their levels used in the CCD experimental design.

CCD: central composite design; P(VP-co-MA): poly(vinylpyrrolidone-co-methylacrylate); PVP: poly(vnylpyrrolidone).
Results and discussion
Batch sorption optimization
In order to determine the most efficient of PVP and P(VP-co-MA) varying hydrogel samples, batch sorption investigations were carried out at predetermined conditions of pH 8, 5 ppm initial metal concentration, 50 mg hydrogel dosage, 25℃ temperature and contact time 6 h. The results obtained as represented in Figure 2 showed that maximum sorption capacities were observed in PV3 and PVM5 samples of PVP and P(VP-co-MA) hydrogels, respectively. On the other hand, P(VP-co-MA) hydrogels demonstrated greater efficiency in the removal of the metal ions compared to PVP hydrogels. This was attributed to the additional binding affinity via the incorporation of the methacrylate monomer in the hydrogel network structure. The removal efficiency of the metal ions by PVP and P(VP-co-MA) hydrogels, respectively, was observed to be in the magnitude of Cu2+ > Zn2+ > Ni2+ > Cd2+ and Cu2+ > Ni2+ > Zn2+ > Cd2+.
Equilibrium sorption capacities of different (a) PVP and (b) P(VP-co-MA) hydrogels for the sorption of varying metal ions. P(VP-co-MA): poly(vinylpyrrolidone-co-methylacrylate); PVP: poly(vinylpyrrolidone).
RSM model and analysis
Obtained results of response-surface CCD.
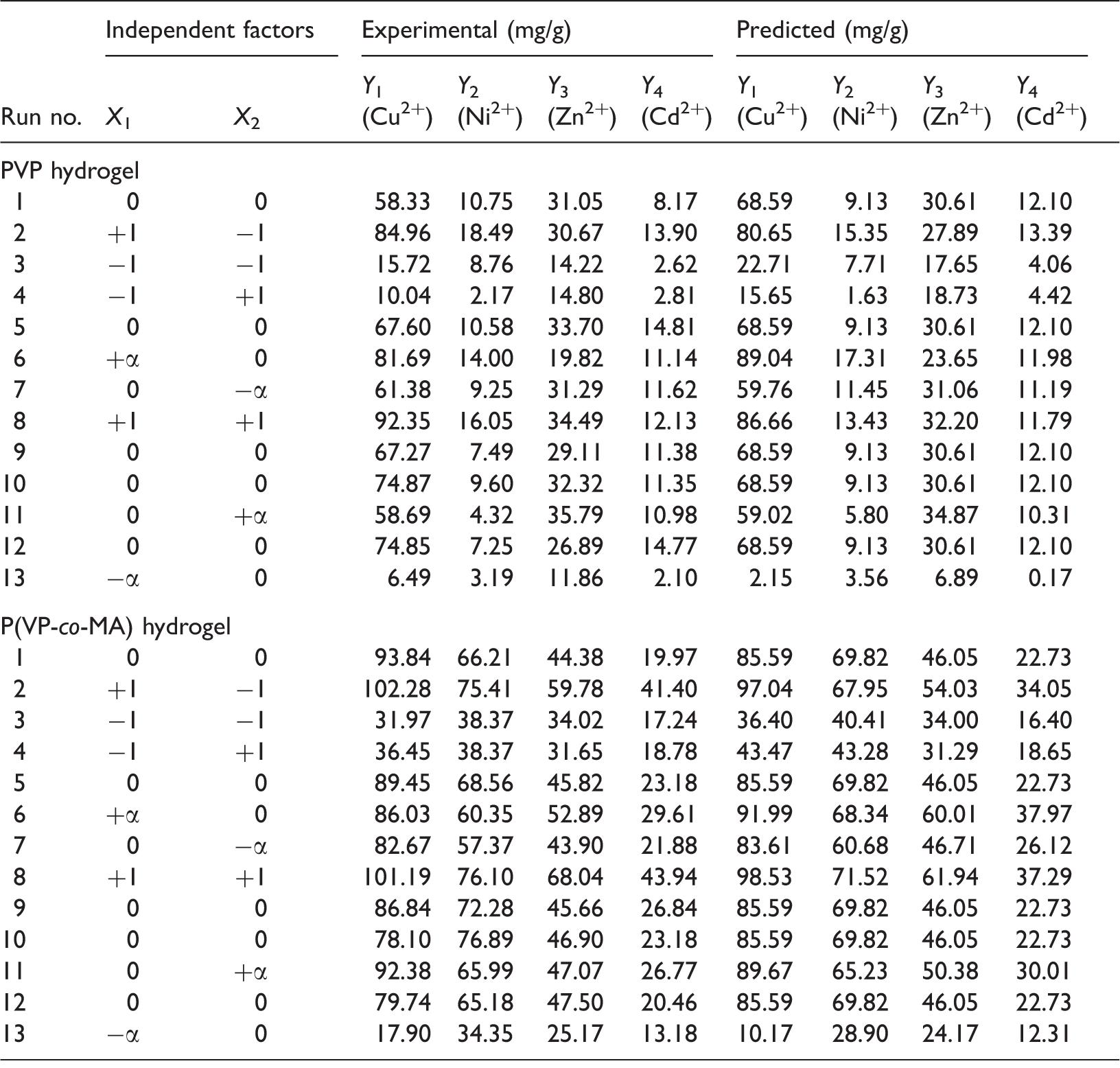
CCD: central composite design; P(VP-co-MA): poly(vinylpyrrolidone-co-methylacrylate); PVP: poly(vinylpyrrolidone).
Second-order polynomial equations for the investigated heavy metals.

P(VP-co-MA): poly(vinylpyrrolidone-co-methylacrylate); PVP: poly(vinylpyrrolidone).
A negative effect of a factor indicates that the response is not significantly affected when the factor level increases and vice versa for a positive effect (Bajpai et al., 2012; Bingöl, 2011).
Therefore, from the obtained equations, X1 for both PVP and P(VP-co-MA) hydrogels has a positive effect on the heavy metals sorption. On the other hand, X2 negatively affects the sorption of the heavy-metal ions using PVP hydrogel while a positive effect is observed for P(VP-co-MA) hydrogel. In other words, increasing initial pH increased sorption efficiency while increase in contact time respectively decreased and increased sorption efficiency of PVP and P(VP-co-MA) hydrogels.
Statistical analysis of the model
Analysis of variance (ANOVA) for RSM quadratic model.

CV: coefficient of variance; P(VP-co-MA): poly(vinylpyrrolidone-co-methylacrylate); PVP: poly(vinylpyrrolidone); RSM: response surface methodology.
The acceptability of the applied mathematical model was analyzed by establishing diagnostic plots of normal probability versus studentized residuals for the uptake of Cu2+, Ni2+, Zn2+ and Cd2+ metal ions on PVP and P(VP-co-MA) hydrogels. Figure 3 depicts normal probability plot of the studentized residuals for the heavy metal ions sorption. From Figure 3, it was observed that the residual behaviours of the various metal ions adsorbed by PVP and P(VP-co-MA) hydrogels followed a linear and normal distribution. This demonstrates that the model was adequate for the removal Cu2+, Ni2+, Zn2+ and Cd2+ metal ions.
Normal probability plots of studentized residuals. (a) PVP and (b) P(VP-co-MA) hydrogels for Cu2+, Ni2+, Zn2+ and Cd2+ metal ions sorption process.
Effect of initial pH and contact time on heavy-metal ions sorption
To best describe the interactive effect of two variables on the responses, three-dimensional response surface plots were constructed to analyze the effects of initial pH and contact time on the efficacy of sorption using PVP and P(VP-co-MA) hydrogels at initial metals concentration of 5 ppm, adsorbent dosage of 50 mg and temperature of 25℃. Figure 4 shows the response surface plots of initial pH and contact time on metal ions sorption. As observed from Figure 4, the metal ions sorption efficiency increased by increasing contact time from 1 to 6 h for PVP and P(VP-co-MA) hydrogels. This is explained based on the binding of the adsorbates with increasing contact time and finally saturation of adsorbent sorption active site (Bajpai et al., 2012). The effect of initial pH is an important parameter for evaluating the potency of sorption capacity of an adsorbent due to its influence on the adsorbent functional groups. Figure 4(a) illustrates the sorption efficiency of Cu2+, Ni2+, Zn2+ and Cd2+ metal ions using PVP hydrogel. It was observed that increasing the pH from 2 to 8 enhanced the binding of the metal ions, and maximum metal ions uptake was obtained at solution pH 8. This was attributed to the behaviour of the N-atoms on the PVP hydrogel surface. At low pH, protonation of the N-atoms occurs which leads to electrostatic repulsion with the existing metal ions in solution. However, increasing solution pH leads to ionization of the N-atoms, which induces an increase in sorption of the metal ions (Faisal, 2016; Li et al., 2008). The binding capacities of the PVP hydrogels were observed in the magnitude of Cu2+ > Zn2+ > Ni2+ > Cd2+ with obtained maximum sorption capacities of 86.44, 32.04, 13.12 and 12.90 mg/g, respectively. The sorption capacity of P(VP-co-MA) hydrogel for the different metal ions was also investigated and the obtained plots represented in Figure 4(b). Based on theoretical concept, addition of MA in the PVP matrix increases metal-ion binding efficiency of the hydrogel. Comparatively, the obtained sorption capacities of the copolymer hydrogel for Cu2+, Ni2+, Zn2+ and Cd2+ metal ions were significantly higher than that of the homopolymer hydrogel. This was due to the presence of additional oxygen atoms from methylacrylate which coordinated more metal ions thereby increasing the metal ion binding capacities (Milosavljevic et al., 2011). The sorption capacity of P(VP-co-MA) hydrogel for the different metal ions was in the order of Cu2+ > Ni2+ > Zn2+ > Cd2+ with obtained maximum sorption capacities of 98.84, 73.21, 61.28 and 36.88 mg/g, respectively. However, the higher sorption capacities of Cu2+ metal ion obtained using PVP and P(VP-co-MA) hydrogels was attributed to the Jahn–Teller effect which states ‘For any non-linear molecule in an electronically degenerate state, distortion must occur to lower the symmetry, remove the degeneracy and lower the energy’ (Justi et al., 2005; Yildiz et al., 2010). In addition, Cu2+ is known as one of the easiest metal ions to form complexes with ligands containing nitrogen and oxygen atoms.
3D plots showing effect of pH and contact time using (a) PVP and (b) P(VP-co-MA) hydrogels for the sorption of Cu2+, Ni2+, Zn2+ and Cd2+ metal ions.
Optimization of sorption conditions
Optimum values of the sorption parameters for the sorption of metal ions onto PVP and P(VP-co-MA) hydrogels.

Validation of the regression model
Parametric values for statistical model validation.

P(VP-co-MA): poly(vinylpyrrolidone-co-methylacrylate); PVP: poly(vinylpyrrolidone).
Error functions


Kinetic parameters for the sorption of metal ions onto PVP and P(VP-co-MA) hydrogels.

MSE: mean standard error; P(VP-co-MA): poly(vinylpyrrolidone-co-methylacrylate); PVP: poly(vinylpyrrolidone); SSE: sum of squared errors.
Sorption kinetic models
To investigate the transport behaviour of the adsorbate molecules in relation to time and controlling mechanism of the sorption process, the reaction-based and diffusion-based models were employed to test the experimental data obtained for Cu2+, Ni2+, Zn2+ and Cd2+ metal ions sorption onto PVP and P(VP-co-MA) hydrogels. The three models applied were pseudo first-order, pseudo second-order and intraparticle diffusion kinetic models.
The pseudo first-order kinetic model was first suggested by Lagergren for the sorption of solid–liquid systems (Evren et al., 2014). The model is expressed using the equation below.

The pseudo second-order kinetic model was suggested by Ho and McKay. This model is mostly applicable in predicting the sorption behaviour over the whole range and is in agreement with chemical sorption occurring as the rate-controlling step (Karadağ et al., 2015). The model is represented using the formulation below.

Table 8 shows the values of the kinetic models parameters of Cu2+, Ni2+, Zn2+ and Cd2+ metal ions sorption onto PVP and P(VP-co-MA) hydrogels. The validity of the investigated models was examined by the regression correlation coefficient (R2) and close agreement between qe,exp and qe,cal values. Based on the regression coefficient, the pseudo second-order kinetic model best described the overall sorption process of the metal ions onto the studied hydrogels, which confirmed chemical sorption occurred as the rate-controlling step. Also, the close agreement between the obtained qe,exp and qe,cal values, and low values of SSE and MSE determined for the different metal ions as represented in Table 8 confirmed the pseudo second-order best fitted the sorption process.
The sorption mechanism of the metal ions onto the hydrogels follows three steps which are film diffusion, pore diffusion, and intraparticle diffusion (Shoueir et al., 2016). The effect of intraparticle diffusion on sorption is determined based on the relation of sorption capacity and time, which is represented by the formulation below.

Figure 5 represents the intraparticle diffusion plots for different metal ions sorption onto PVP and P(VP-co-MA). As observed in Figure 5, the sorption process followed two phases with the resulting fitting passing through the origin. The existence of two phases in the intraparticle diffusion plot suggests that the sorption process occurred by surface sorption and the intraparticle diffusion. On the other hand, the intercept C of the plots reflected the boundary layer effect which occurred to be higher for P(VP-co-MA) compared to PVP hydrogel (Table 8). And the high R2 values (≥0.9) depict the suitability of this model for expressing the experimental data which may confirm that the rate-limiting step is the intraparticle diffusion process.
Intraparticle diffusion kinetic plots for Cu2+, Ni2+, Zn2+ and Cd2+ sorption onto (a) PVP and (b) P(VP-co-MA) hydrogel.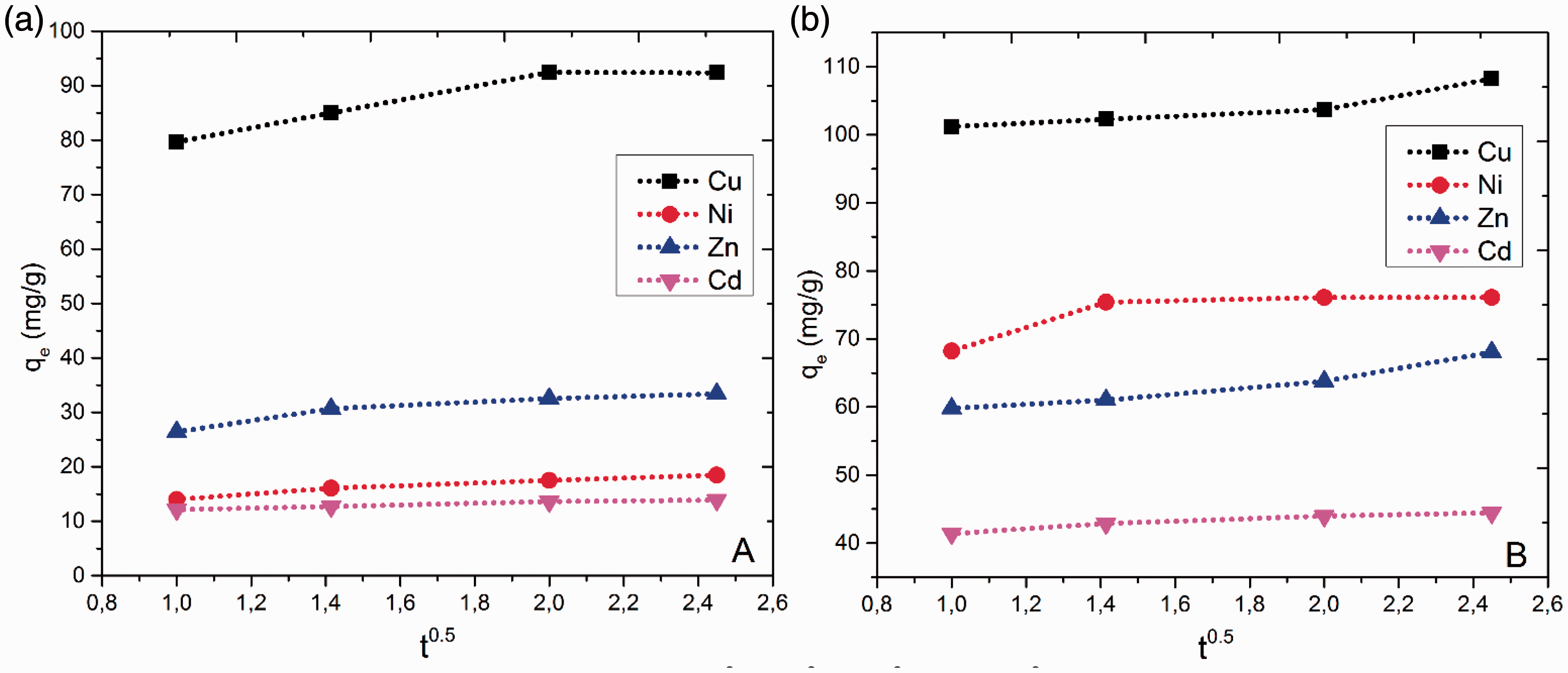
Sorption equilibrium isotherms
Several studies (Kumar et al., 2008; Paulino et al., 2011) have reported drawbacks involved with linearization of isotherm models, which may affect the normality assumptions of the curves thereby violating the theories behind the isotherms. So to avoid such hitches, non-linear regression equations were employed in this study for two-parameter isothermic models of Langmuir, Freundlich and Temkin, which enabled the comparison study of isotherm more reliable. These models are the most commonly used in the investigation of sorption of heavy metal species onto hydrogels because they described the adsorbate–adsorbent interaction but also reflects thermodynamic data providing information about sorption spontaneity and stability towards sorption affinity of the metal ion molecules (Piccin et al., 2011). The Langmuir isotherm theory is based on the assumption that adsorption occurs at specific homogeneous sites inside and sorption sites possess equal sorption affinity for adsorbate. In addition, once a metal ion molecule resides a site, no additional sorption can take place there (Kamal et al. 2016; Ngwabebhoh et al., 2016). The isotherm is represented be the formulation below.



Isothermic parameters of metal ions sorption onto PVP and P(VP-co-MA) hydrogels.

P(VP-co-MA): poly(vinylpyrrolidone-co-methylacrylate); PVP: poly(vinylpyrrolidone).
Conclusions
This study aimed at a comparative optimization and evaluation of the sorption efficiency of synthesized homopolymer PVP and copolymer P(VP-co-MA) hydrogels for the uptake of Cu2+, Ni2+, Zn2+ and Cd2+ metal ions from aqueous solution in a batch reactor system. The obtained sorption experimental data revealed that P(VP-co-MA) hydrogel possessed greater binding capacity for investigated metal ions compared to PVP hydrogel. CCD proved to be a valuable tool for the determination of the optimal conditions for the sorption process via response surface analysis. The results showed that the sorption capacity increased with increasing the initial pH of solution and contact time. Optimum operational condition were determined at pH 8 and contact time of 6 h. Highest binding capacity was obtained for Cu2+ metal ion for PVP and P(VP-co-MA) hydrogels. The validation experiments affirmed the CCD model was suitable for optimizing the sorption process. The experimental data for the metal ions sorption well fitted the pseudo second-order kinetic model which predicts that chemical sorption occurred. The sorption process was best described by Freundlich model which suggested heterogeneity at the sorption sites of the hydrogels. The results of this study demonstrates that P(VP-co-MA) hydrogels were most suitable for the sorption of the heavy metals from aqueous environments due to high binding capacity and ease of separation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
