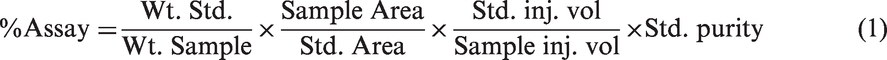Abstract
Alpha-tocopherol or vitamin E is an essential food ingredient. Due to high purity requirement in food applications, it is majorly produced in the acetate form through chemical routes. However, the isolation of food grade vitamin E acetate (VEA) is critical due to the co-generation of close characteristic impurities in the production processes. The work undertaken here provides a new kinetic chromatography method for production of food grade VEA. In this work, suitable adsorbent for chromatography separation was selected based on shallow-bed binding–elution characteristics. Loading conditions were identified by frontal adsorption behavior. Kinetic parameters and bed height for the purification was predicted by establishing the relationship between bed height, purity and the relative separation factor (RSF). The RSF of 2.8 and corresponding bed height is predicted from the established relationship for the separation of VEA from impurities. The bed height of 170 cm with isocratic mobile phase of 2% (wt/wt) water in methanol provides 98 wt.% purity of VEA with recovery of 90%. This work has highlighted methodology based on kinetic parameters for the separation of close characteristic impurities from target molecule. This work also provided the application of simple handy tools like RSF and dual wavelength detection for the rapid development of preparative separation process for VEA and other such valuable components from unknown impurities.
Keywords
Introduction
Vitamin E is considered valuable for immune system modulation in the cell membrane due to its anti-oxidative properties (Siegel et al., 2001). Vitamin E exists in eight different forms (alpha-, beta-, gamma- and delta-tocopherols and tocotrienols). Alpha-tocopherol has the highest biological activity among all the related homologues of vitamin E. This alpha-tocopherol is most stable in its ester or acetate form; therefore, it is generally prepared and stored in acetate form. Due to the highest activity and productivity, alpha-tocopheryl acetate is commonly considered and marketed as vitamin E acetate (VEA).
Various natural feed stocks, in particular, vegetable oils or deodorized distillate (DOD) obtained during refining of vegetable oils, serves as a rich source of tocopherol homologues (Cert et al., 2000; Marks, 1988). Therefore, production of natural tocopheryl acetate first involves isolation of mixed tocopherols from DOD followed by chiral chromatographic resolution of alpha-tocopherol from its associated homologues and then converted into acetate form (Ueda, 1993). As the generation of tocopheryl acetate or VEA from natural resources is a multistep cost-intensive process, it is usually produced synthetically to circumvent the recovery complications as well as the deactivation associated with native alpha-tocopherol (Lee et al., 1991; Lowack et al., 1996; Shimada et al., 2000). The easy reduction of acetate back to the native form provides an added advantage of this synthetic VEA. In cosmetic applications, this acetate form hydrolyzes easily once it gets absorbed by the skin and regenerates native tocopherol. Therefore, synthetic VEA (C31H52O3) is considered reliable, stable and popular anti-oxidant in market than alpha-tocopherol itself. The chemical structure of VEA is shown in Figure 1.
Structure of tocopheryl acetate or VEA. VEA: vitamin E acetate.
One reported production method for VEA involves cyclocondensation of trimethylhydroquinone with phytol or isophytol in the presence of an acid catalyst and an inert solvent under supercritical conditions (Lowack et al., 1996). But this method is rather expensive and is not so popular due to the requirement of co-solvent and supercritical conditions during cyclocondensation. Other semi-synthetic way for the production of VEA involves reaction of tocopherol, acetic anhydride and sodium acetate (Kulkarni et al., 2008). Downstream isolation of VEA by this method is critical due to film distillation with partial reflux under high vacuum followed by crystallization using nitrogen at low temperature (Bulychev, 1998; Bulychev et al., 2005; Kulkarni et al., 2008). Further filtration involves loss of purified liquid VEA with solid sterols, salts and other impurities solidifying during crystallization. Currently, new catalyst circulating method involving condensation of trimethylhydroquinone and isophytol in polar solvent mixture in presence of protonic acid and acid hallide catalyst is found useful for the synthesis of VEA (Krill et al., 2001).
Although the synthesis method is well established, the major challenge encountered in the production of VEA is the downstream processing or purification of it from the close characteristics impurities generated in the final reaction mass. The reported methods employ high pressure column chromatography, high speed counter current chromatography, selective deacylation followed by chromatography and nano-liquid chromatography using capillary column (Foster, 1986; Foster and Cross, 1984; Shinomiyam et al., 2000; Wan et al., 2008) for the purification of synthetic VEA. Most of these reported methods involve specialized chromatography techniques or energy intensive distillation operation for the isolation of VEA. Also prolonged processing of VEA due to heating in distillation or multiple polishing steps causes decomposition and deterioration of quality of VEA.
The regulatory norms for a high purity and the high demand of VEA in the anti-oxidant market necessitate new downstream purification methods. In the present investigation, the need of robust, scalable and cost-effective method of isolation of VEA from synthetic mass is addressed by using kinetic one-step low pressure reverse phase chromatography process. To our knowledge, no research on reverse phase chromatographic purification of VEA has been previously reported. Therefore, this work provides a new chromatographic method by exploiting the dynamic parameters of reverse phase column chromatography principle. Suitable adsorbent for the separation process was selected on the basis of selective binding and elution performance shown by VEA in shallow-bed experiments. Breakthrough binding studies were performed on the selected adsorbent for the establishment of loading and elution conditions. The poor binding strength of VEA indicated that kinetic and dynamic parameters are required for the development of purification process. The relative separation factor (RSF), based on maximum retention time of impurity (mixture) peak and VEA, was used as a measure of degree of separation in the present work. Experiments were devised by changing dynamic parameter bed height for the prediction of RSF factor required for 98 wt.% purity of VEA. Optimization of mobile phase composition was conducted for the reduction of total solvent consumption and height recovery using the predicted bed height. A comprehensive analysis of the combined effect of bed height and modulators concentration in mobile phase produces selective elution of 98 wt.% pure VEA with recovery as high as 90%. The method adopted here provides high purity product in single step at ambient temperature conditions with maximum productivity. Isocratic mobile phase conditions of the process enhance its favourability to convert this single column process to continuous simulated moving bed operation.
Material and experimental
Chemicals, adsorbents and crude VEA sample
High performance liquid chromatography (HPLC) grade methanol (99.9 wt.%) was purchased from Merck India Ltd. The other commercial grade solvents like hexane, heptane, ethyl acetate, acetone, methanol, isopropyl alcohol (IPA) and ethanol were purchased from Chem Udyog Ltd., India. The de-ionized (DI) water required for the preparation of washing and elution composition was produced in the lab using the Millipore system.
Normal phase silica gel was purchased from Hi Media, India. The adsorbents namely SEPABEADS EB-HG, SEPABEADS SP700, SEPABEADS SP207 and Diaion HP20 involved in selection and separation experiments were obtained as a gift sample from Resindiaion SRL., Italy.
The synthetic mixture containing 78–83 wt.% VEA was a gift sample from Merck India Ltd., Goa, India. Standard VEA sample of purity 99.9 wt.% for assay estimation was purchased from Sigma, India.
Columns, pumps and tubing
Five glass columns of 1.0 cm× 15 cm, 1.0 cm× 50 cm, 1.0 cm× 100 cm, 1.0 cm× 150 cm and 1.0 cm× 200 cm (diameter × length) dimensions were purchased from Omega glass works, Mumbai. Organic solvent compatible SS316 adaptors of 25 cm length were designed in the lab and further fabrication was made through the local vendor. The packed bed of specific height in these glass columns was prepared by using these adaptors. An eight-roller MasterFlex peristaltic pump was used for the external loading of feed sample on to the column. Silicon and Teflon tubing used for pumping the crude samples and solvent by external pump were purchased from local market.
Preparative chromatography instrument
Biologic Duo flow Path finder system was used for conducting all column experiments, i.e. for breakthrough curve, bed height and mobile phase optimization experiments. It is a kind of preparative chromatography system of Bio-Rad Ltd., USA. The system is equipped with two piston-type pumps for the generation of linear or step gradients of different compositions, quad-tech type detector for detection at four different wavelengths and online pH and conductivity meters. Therefore, it is very handy for the selection and online modification of elution phases and their strength for purification process optimization in minimum number of experiments. External columns of varying sizes were connected through AVR 7-3 valve to the system for the development of purification process. The system also facilitates collection of fractions according to the detector response by fraction collector at downstream end of the column.
HPLC analysis
Various methods are reported for the analysis of tocopheryl acetate. Some of these reported methods are based on normal phase chromatography principle and involve multiple solvents for running mobile phase preparation (Iwase, 2000; Lee et al., 2006). Therefore, current work devised the single solvent reverse phase method for VEA analysis.
Method for the analysis of alpha-tocopheryl acetate employed 4 mm× 250 mm Zorbax SB C18 column packed with 5 -µm-size particles. This column was purchased from Agilent technologies Ltd., USA. The mobile phase was 1 wt.% water in methanol and detection was done at 215 nm wavelength. The flow rate of the mobile phase was 1.2 mL/min. Twenty microlitres of sample was injected for the analysis. The total run time of the HPLC protocol was 40 min.
Assay method
The assay method for the estimation of VEA purity in elution fractions using the HPLC chromatogram and analysis was developed. The method involved weighing specific quantity of unknown sample/fraction containing VEA. Weighed sample was then subjected to vacuum distillation at 90℃ to remove the solvent. After the complete removal of volatile components and solvent from the sample, the remaining residue (light yellow to colourless viscous liquid) was again weighed. After this step, ethanol of known volume (10 mL) was added in to the sample. This unknown residue in ethanol was analyzed using HPLC method by injecting 20 µL of sample and the area corresponding to VEA peak was recorded. The same volume of standard VEA (Sigma) sample of known concentration (10 mg/mL) was also injected into HPLC and corresponding area was determined. The assay purity of the unknown sample was estimated as:
Miscibility study
Miscibility studies were carried out by mixing 25 mL of 78 wt.% assay VEA sample with 25 mL of solvent or solvent composition under continuous stirring conditions for 1 h at 30℃. After 1 h, the mixture was allowed to stand for 0.5 h; if two distinct phases were formed, then the sample phase and solvent phase were separated and measured. Further, miscibility of crude VEA reaction mass with added solvent was measured by equation (2):
Unsteady state kinetic study for the selection of chromatography type
All the unsteady state experiments were carried out by using 10 mL of adsorbent sample in a baffled tank with a four-bladed turbine impeller. The adsorbent was contacted with 70 mL of VEA sample of concentration 54.7 mg/mL in the selected solvent conditions depending on chosen chromatography principle. The added adsorbent was pre-equilibrated for 90 min in respective solvent. The initial optical density of solution at 284 nm was recorded before addition of adsorbent. The concentration corresponding to this was considered as initial concentration (C0) of the sample. After the addition of adsorbent in VEA solution, the absorbance of the solution decreased with time due to adsorption of VEA on added adsorbent. As the initial rate of adsorption was fast, change in the absorbance (concentration) of the supernatant liquid was first measured after every 1 min. As the time progressed, the rate of adsorption decreased. Therefore, change in the liquid phase concentration was then monitored after every 2 min and then after 5 min till the equilibration concentration.
Experiments for reverse phase principle were carried out for Diaion HP20 adsorbent using polar solvents like methanol, ethanol and iso-propanol (IPA). Experiments for normal phase principle were performed using acetone with EB-HG adsorbent and non-polar hexane with silica as an adsorbent.
Chromatography type for purification of VEA was selected on the basis of distribution characteristics estimated from the unsteady state profiles for the solvent–adsorbent systems under investigation.
Selection of adsorbent for purification
Selection of the adsorbent was done by shallow-bed column adsorption/desorption studies. Experiments were carried out by making the bed of 10 cm height in 1.0 cm× 15 cm dimension glass column to achieve a total bed volume (BV) of 8 mL. The column was equilibrated with 2 BV of methanol, followed by 0.1 BV loading of dilute VEA sample (54.7 mg/mL), which was then washed with 2 BV of methanol. Further, it was eluted with 2 BV of 1.0 wt.% IPA in methanol phase. The procedure was repeated for various matrices under investigation.
Selection of suitable adsorbent from the matrices under investigation was carried out on the basis of its elution specificity in terms of purity of fractions collected during elution.
Designing of loading conditions
Breakthrough experiments were carried out in 35 mL (1.0 cm× 45 cm) column packed with selected adsorbent to study the effect of loading conditions on purfication performance. In this study, the solute sample was fed continuously onto the adsorbent until the available area for adsorption got exhausted and the solute began to appear in the mobile phase collected at the column outlet. Variation of solute concentration at the column outlet as a function of time/volume up to the plateau concentration is known as breakthrough curve (Chase, 1984). The experimental process involved column equilibration with 2 BV of methanol. After equilibration, VEA crude of specific concentration was loaded till the concentration at the inlet was equal to the outlet concentration. Once this condition was achieved, the column was washed to remove unadsorbed components by the equilibration solvent. The bed was then regenerated by using 100 wt.% IPA. This provides information about the binding capacity and broadening of adsorption zone. The above procedure was repeated for (a) concentrated loading ‘as-is’ sample of concentration 930 mg/mL and (b) dilute loading conditions, by diluting ‘as-is’ sample by methanol to a concentration of 54.7 mg/mL.
Purification of VEA from 78 wt.% assay crude reaction mass
Purification process for VEA was developed by packing selected adsorbent and online detection of elution components by connecting column to Biologic duo flow system. Bed height required for the desired purity and mobile phase composition of VEA was optimized. Initial trials in this regard were performed in 1.0 cm× 45 cm bed height column. To improve the degree of separation between impurities and VEA, further experiments were performed in 1.0 cm× 60 cm, 1.0 cm× 75 cm and 1.0 cm× 172 cm bed height columns.
For all the trials, equilibration was done with 2 BV by choosing constant water–methanol composition. Concentrated sample with a loading capacity of 40 (mg VEA)/(mL adsorbent) was kept constant for the runs carried out in different bed height columns. Optimum height for desired resolution was predicted from RSF. Maximum retention time of major impurity and VEA required for RSF estimation was measured on the basis of dual wavelength detection methodology. This dual wavelength detection was crucial because major polar impurity of synthetic mass did not show detection at λmax (284 nm) of VEA. Fine optimization of washing and elution phase compositions was carried out by two pumps of the system to achieve maximum productivity in the optimum bed height selected for desired resolution. Numerous fractions were collected for each run and analyzed by assay method for VEA purity. Fractions containing >98 wt.% VEA were pooled together and analyzed for pooled purity and recovery of purified VEA. In all the runs, the flow rate of 1.2 mL/min was kept constant.
Regeneration of the column was carried out by using 1.5 BV of IPA. It was followed by 0.5 BV of methanol as a part of regeneration step for the avoidance of IPA–water azeotrope.
Results and discussion
Miscibility characteristics and selection of chromatography type
Study of solubility characteristics of crude VEA sample.
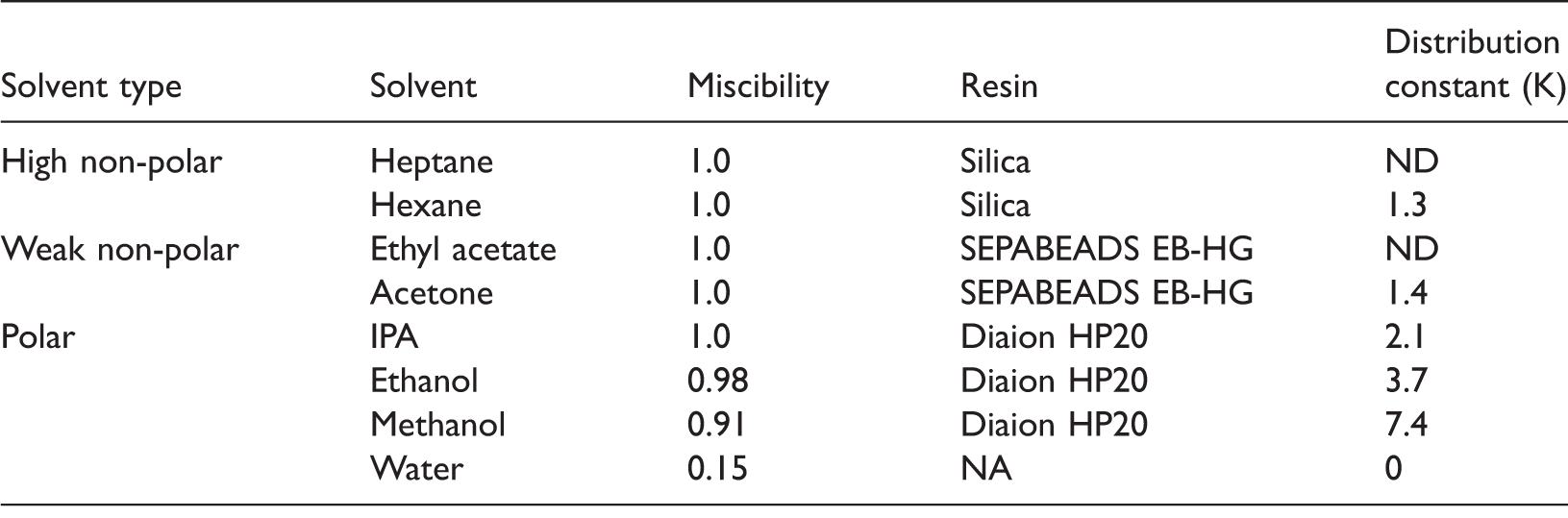
ND: no data; VEA: vitamin E acetate.
Depending on miscibility characteristics, the mobile phase conditions for chromatographic purification were applied. Also, depending on the selected solvent, different chromatography principles and adsorbents were exploited using unsteady state kinetics for the selection of chromatography type for VEA purification. The experimental unsteady state curves for the variation of dimensionless VEA concentration with time for both normal and reverse phase adsorbents in respective solvent system are shown in Figure 2. The essential parameter for purification, i.e. initial rate of adsorption and value of distribution constant obtained for specific adsorbent was collectively used for the evaluation of interaction of VEA and adsorbent. The values of liquid phase equilibrium concentration and solid phase equilibrium concentration obtained from the saturated region of unsteady state curves were used for the estimation of distribution constant (Jiang et al., 2014).
Unsteady state adsorption kinetics for VEA on various resins using different chromatography principles.♦ Methanol–HP20; ▪ Ethanol–HP20; ▴ Isopropyl alcohol–HP20; × Acetone–EB-HG; • Hexane–Silica). VEA: vitamin E acetate.
Normal phase silica with hexane as diluents and EB-HG with acetone as diluents showed poor binding and equilibration characteristics. In contrast to this, reverse phase principle showed favourable binding of VEA with HP20 adsorbent for methanol and ethanol as diluents. This is because of high distribution of crude VEA towards adsorbent due to dominant hydrophobic interactions within polar solvents, especially with lower alcohols. It is evident from Table 1 that the distribution constant (K) values are two to three times higher for methanol than for other solvents used in this study.
Favourable initial adsorption kinetics and high distribution constant values using polar solvent on HP20 media indicated that reverse phase chromatography can be considered as a favourite method for VEA purification. Another advantage with the reverse phase principle is the availability of media having different pore size, surface area and hydrophobicity index. Therefore, on this basis, reverse phase method was selected for the preparative scale purification of VEA from reaction mass.
Selection of adsorbent for purification of VEA
Selection of media for purification is a crucial task, subsequent to the decision of chromatography type to be employed. Usually binding–elution performance of small column provides an indication of specificity in binding and selectivity in elution for preparative separations (Rathore and Velayudhan, 2003). Therefore, adsorbent selection becomes more realistic compared to batch equilibrium studies. Again, the reverse phase analytical method supports this adsorbent selection strategy by recognizing the unknown impurities on the basis of their elution sequence relative to the product (VEA). In the HPLC analysis, the impurities which showed low retention time than VEA were considered polar, while those eluted after it were considered as non-polar. Considering this, in the present work, a shallow-bed binding and elution studies were performed using favourable binding conditions for the selection of a suitable adsorbent. The selectivity of matrices was measured in terms of specificity by estimating percentage assay of VEA in elution fractions. The adsorbent which showed loss of product during washing with polar impurities was considered poor, and the adsorbent which showed maximum enrichment of VEA with only one class (either polar or non-polar) was considered as a choice for the resolution of VEA.
Selection of matrix for the purification of VEA.

P: polar; NP: non-polar; NA: not applicable; × : poor conditions; √: desirable conditions; VEA: vitamin E acetate.
The unsteady state kinetic curves of VEA with various matrices indicated the importance of kinetic parameters for its separation from associated impurities. These kinetic parameters are largly governed by the pore structure and high surface area of 1200 m2/g for SP700 than 590 m2/g for SP207 and HP20 (Adachi and Isobe, 2003). Thus, optimum hydrophocity characteristics and high surface area collectively resulted in selective desorption of VEA from SP700 surface than other matrices. Also, its narrow pore-size distribution enhances retention volume. Therefore, taller bed of SP700 adsorbent can provide large number of equilibrium stages for resolution of VEA from close characteristic impurities to achieve purity as high as 98 wt.%. Therefore, SEPABEADS SP700 was selected as a stationary phase for the purification of VEA.
Loading conditions and kinetic parameters
Figure 3 provides the comparison of breakthrough profiles for concentrated and dilute loading conditions on SP700 adsorbent. Shallow breakthrough profile was observed for dilute loading, while steep breakthrough curve was obtained for concentrated loading. Depending on the breakthrough profiles, the kinetic parameters estimated for concentrated and dilute loading conditions are shown in Table 3 (Kawakita et al., 1991; Michaels, 1952). Concentrated loading showed nearly 2.3 times (228 mg/mL) higher binding capacity than dilute loading (98 mg/mL) conditions. It was also observed that 77–80 wt.% of the loaded sample gets washed away during the washing step for both the conditions. This indicates multilayer adsorption of crude components, mainly due to the weak binding strength or weak interaction in comparison to on–off-type binding. This weak binding was confirmed from the dissociation constant values estimated for both the dilute and concentrated conditions. Due to weak binding conditions, the resolution of target molecule by loading up to the breakthrough capacity was nearly impossible. Therefore, desired purity of VEA from the given crude sample was only possible by exploiting kinetic and dynamic parameters with considerably low loading than breakthrough capacity using concentrated loading conditions.
Selection of loading conditions for the purification of VEA by reverse phase chromatography (- -concentrated VEA loading, … … … dilute VEA loading, ——— conductivity). VEA: vitamin E acetate. Binding capacity and kinetic parameters for concentrated and dilute loading conditions.

Bed height for the resolution of close characteristic impurities from VEA
Trial for purification was first carried out in 45-cm bed height (in 1.0 cm× 50 cm) column with isocratic methanol conditions. The online preparative chromatograms of the trial is shown in Figure 4. The chromatogram shows advantage of dual wavelength detection in terms of single major elution peak of impurities (detected at 210 nm) with finite resolution with the long rectangular peak of VEA (detected at 280 nm). The HPLC analysis of fractions corresponding to this first single peak confirmed the presence of the mixture of polar impurities only. The rectangular peak of preparative chromatrogram shows majorly VEA with few polar impurities. This arises mainly due to the comparable binding strengths of the polar impurities and VEA (Table 3). Therefore, both VEA and associated impurities did not exhibit a kind of ‘all-or-nothing’ sort of interaction with adsorbent and hence moved with definite retention in the applied mobile phase. This implies the importance of kinetic factors and number of plates or bed height required to achieve the desired degree of separation of close characteristics impurities and VEA.
Visual differentiation of effect of column height on the resolution of impurities and VEA by detection at two different wavelengths (a) 45 cm bed height column; (b) 60 cm bed height column; (c) 75 cm bed height column (▪ H = 45 cm (210 nm, impurity elution); ▪ H = 45 cm (280 nm, VEA elution) ▴ H = 60 cm (210 nm, impurity elution), ▴ H = 60 cm (280 nm, VEA elution), • H = 75 cm (210 nm, impurity elution), • H = 75 cm (280 nm, VEA elution)). VEA: vitamin E acetate.



Variation of RSF with bed height for the enrichment of purity of VEA.

RSF: relative separation factor; VEA: vitamin E acetate.

Purity as function of bed height and RSF. (a) Effect of increase of bed height on RSF. (b) Impact of RSF on the purity of VEA. RSF: relative separation factor; VEA: vitamin E acetate.
Combining equations (4) and (5), a new correlation for purity was devised as presented by equation (6). This correlation incorporates the collective effect of VEA–adsorbent interaction with collective effect of kinetic characteristics in terms of the number of equilibrium stages required to achieve the desired purity.

Using equation (6), the bed height of 172 cm was predicted for the achievement of desired purity of VEA. When trial was conducted in 172 cm column, it produced 98 wt.% purity VEA in few fractions with lower yields.
Optimization of mobile phase conditions
The trial using 172 cm bed height (1.0 × 172 cm) column with isocratic methanol conditions showed slight loss of VEA with polar impurities during washing. This highlighted the importance of polarity of binding and desorption conditions. Therefore, further trials in same bed height were carried out by modifying isocratic methanol of equilibration and washing steps. These steps were modified by the addition of 1–4% (wt./wt.) water in methanol by keeping constant loading of 40 mg of crude/(mL adsorbent) with the objective of no loss of VEA with polar impurities or selective removal of polar impurities during washing. Effectiveness of applied solvent system was evaluated by keeping 2.0 BV of equilibration. Elution conditions were designed for optimum solvent consumption per weight of purified VEA recovered.
Comparison of effect of mobile phase conditions on the recovery of purified VEA and amount of solvent required for elution is shown in Figure 6. Isocratic conditions of 1.0 wt.% water in methanol caused stronger binding and milder elution of VEA than isocratic methanol conditions. This resulted in the improvement of recovery of purified VEA from 81 wt.% to 85 wt.% with total solvent requirement of 1.9 BV per gram of purified VEA. Further trial with the equilibration and washing using 2 wt.% water in methanol showed selective removal of polar impurities during washing. The trial with the above-modified conditions reduced the loss of VEA during the washing phase. The elution followed by this produced 98.27 wt.% pooled purity VEA. The recovery corresponding to this was 91.2 wt.% with mariginal increase, i.e. 0.2 BV/ (g of VEA) than that of 1.0 wt.% water in methanol conditions. Further, experiments for the enhancement of recovery and purity of VEA were performed by using 3 wt.% and 4 wt.% water in methanol as equilibration and washing step. However, it was observed that instead of improvement, recovery was hampered due to the strong binding of VEA and impurities. It also resulted in the requirement of extensive washing and elution for the desorption of VEA. This extensive washing caused band broadening and responsible for the the movement of some polar impurites with VEA in elution fractions. The above washing reduced the recovery below 80 wt.% and even resulted in lowering down the purity of VEA to 96 wt.% in numerous fractions for 4 wt.% water in methanol composition (Figure 6). This indicated that complete separation of polar impurities from VEA can only be possible by the increasing the number of stages with equilibration by 2 wt.% water in methanol phase. Therefore, the water:methanol ratio of 2:98 (wt./wt.) proportion with bed height of 172 cm (for volume = 135 mL) was selected as appropriate equilibrium and washing solvent system with optimum height for VEA binding and its selective desorption.
Selection of binding and washing conditions (black colour – % purity of VEA; gray colour – % recovery; ▴ total BV/g of VEA). Error in %purity is ± 2% and in % recovery is ± 3%.
Kinetic reverse phase chromatographic process for high purity VEA
The operating protocol for the purification of VEA was established using the data generated from the bed height and the mobile phase optimization. On the basis of bed height and equilibration conditions, the protocol was further fine-tuned to reduce the solvent consumption and avoid formation of azeotropic mixtures. The protocol with the optimum (10.6 BV) total solvent requirement with desired purity and recovery involved (i) packing of selected adsorbent up to 172 cm bed height, (ii) 2.0 BV equilibration of adsorbent bed with 2:98 (wt./wt.), water:methanol phase, (iii) followed by loading of concentrated VEA sample with a capacity of 40 mg of crude/(mL adsorbent), (iv) 2.0 BV washing by 2:98 (wt./wt.), water:methanol phase, (v) 1.5 BV of 1:99, water:methanol as first elution step and (vi) 2.5 BV of methanol as a second elution step. A separate two step strategy with solvent consumption of 2 BV for regenration was deviced. The online preparative chromatogram corresponding to the optimized conditions is shown in Figure 7. It clearly demonstrates the advantage of dual wavelength detection for visual differentation of resolution of VEA from associated impurities. The fractions obtained from the process were pooled together, evaporated and assayed for the purity and recovery of VEA. The comparison of HPLC chromatograms of the crude and purified VEA samples are shown in Figure 8(a) and (b), respectively. Assay purity of pooled elution fractions of VEA shown in Figure 8(b) was found to be 98.2 wt.% much improved than the 78 wt.% of crude reaction mass with recovery of 90.34%.
The online chromatogram of optimized VEA purification process. VEA: vitamin E acetate. Comparison of HPLC chromatograms of (a) crude VEA of 78% purity and (b) purified VEA of 98.6% purity (detection: 210 nm wavelength). VEA: vitamin E acetate.

Conclusions
In this work, we demonstrated the usability of miscibility and unsteady state kinetics for the selection of suitable chromatography type for separation of valuable components from unknown feed stocks. This work further explained the impact of designing of loading conditions for the purification of VEA. From our work, we demonstrated that it is possible to design crucial loading conditions by studying the breakthrough characteristics of target molecule under different loading conditions. The work defined a parameter called RSF and used it successfully through developing the correlation of bed height and purity for the prediction of height required to achieve desired targeted purity of VEA. Critical designing of equilibration, washing and elution conditions are discussed in detail for the development of kinetic separation process to generate VEA of purity >98 wt.% in one step with recovery of 90%. Thus, the developed purification method is typical and one of its kinds as compared to the conventional processes in terms of yield and operational simplicity for the production of 98 wt.% VEA from reaction mass. Single-step purification and low solvent compositions increase favourability of the method for further scale-up.
Footnotes
Acknowledgements
The authors would like to thank Mr S.M. Firke (Merck India Ltd) for providing crude sample of Vitamin E acetate.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.