Abstract
Hexavalent chromium is one of the major heavy metals. As the environmental legislation becomes increasingly strict, looking for new technology to treat hexavalent chromium is becoming more and more important. In this research, 2-mercaptobenzothiazole was used to blend polyvinylidene fluoride to prepare a modified membrane that could be applied to adsorb and purify water containing hexavalent chromium. The membrane performance was characterized by infrared spectroscopy (Fourier transform infrared spectroscopy) and water contact angle measurements. The experimental results showed that the pure water flux of the membrane was 255.57 l/m2 h, the contact angle was 80.1°, and the adsorption capacity of chromium ion was 241 µg/cm2. The polyvinylidene fluoride-modified membrane presented better adsorption abilities for chromium ion than that of the traditional polyvinylidene fluoride membrane.
Introduction
The emission of heavy metals into the aquatic and marine water systems is of great concern today because these emissions create environmental issues that are associated with human health. Large quantities of wastewater containing heavy metals are generated from industries, including textiles, leather tanning, electroplating, and metal finishing, as well as in the mine tailings. Chromium ion is one of the major toxic heavy metals, especially hexavalent chromium. The methods of dealing with chromium ion wastewater include chemical reduction precipitation (Cronje et al., 2011), electrolytic process (Golder et al., 2007), biological method (Carlos et al., 2012), adsorption method (Wenga et al., 2008), and ion exchange method (Dabrowski et al., 2004). However, all of the above methods have disadvantages to a certain degree, such as high cost, using toxic compounds, large space for installation, generation of secondary pollutants, and so on.
Membrane separation technique as an emerging technology in the 21st century (Kozlowski and Walkowiak, 2005) is witnessing an era of rapid growth due to continuous research and development in both the academic and private industry (Padaki et al., 2014). Additionally, the membrane technology has also been introduced as an efficient technique for the treatment of chromium-containing wastewater. Membrane separation technology includes microfiltration, ultrafiltration (UF), nanofiltration, reverse osmosis, and liquid membrane permeation. Most of these methods have been used extensively for the treatment of heavy metal wastewater (Sylwia et al., 2014). For instance, wastewater that contains Cu2+, Cd2+, Ni2+, and Cr2+ was treated by nanofiltration and reverse osmosis technology (Kyn et al., 1999; Qdais and Moussa, 2004).
UF as a membrane technology for wastewater treatment has received increasing attention in recent years. It has been extensively used as a separation technique in the dairy industry (Tarazaga et al., 2006). UF membrane technology in the treatment of heavy metal ions has been shown to be an effective and cheap approach, not only could it improve general water quality (Moon et al., 2009; Rojas et al., 2011), but also it could control membrane fouling (Konieczny et al., 2009; Listiarini et al., 2009; Peiris et al., 2013; Wray et al., 2014; Yu and Graham, 2015). On the other hand, membrane separation technique has shown great promise for metal ion removal due to its high efficiency, space saving, low cost, and easy operation. This could help enhance the membrane management in real practice (Li et al., 2008; Mohamed et al., 2007; Ritu et al., 2007; Tian et al., 2013).
Polyvinylidene fluoride (PVDF) membranes remain popular in many applications due to its excellent chemical resistance and good thermal and mechanical properties. 2-Mercaptobenzothiazole has adsorption capacity for chromium ions. This study used chromium adsorption material 2-mercaptobenzothiazole to modify the PVDF membrane; this was done to prepare a type of modified PVDF membrane for hexavalent chromium adsorption and purification. The mechanism of this method was that 2-mercaptobenzothiazole contains some functional groups, such as =C–H and H–C–H could generate clathrate with chromium ions. Those functional groups could improve the affinity between modified UF membrane and chromium ions. The method in this paper that blending 2-Mercapto benzothiazol during membrane preparation which could improved the adsorbability has never been reported. The tests were performed under several operating conditions: the main parameters investigated were the pure water flux and the adsorption capacity of chromium ion.
Experimental procedure
Materials
PVDF was purchased from Shanghai San Aifu New Chemical Materials Co.,
Preparation of PVDF/2-mercaptobenzothiazole blend membrane
Before the experiment, PVDF should dry for 24 h in the oven with 70℃. A certain amount of solvent (DMAc) and 2-mercaptobenzothiazole were put into a flask, then PEG400 (11 wt%) and Twain80 (1 wt%) were added in as additives.The reaction was conducted at 70℃ in the water bath for 6 h and was stand defoaming for 12 h at room temperature. Then membrane casting was conducted into a clean glass plate to prepare UF membrane and was put into the deionized water for 24 h.The deionized water was changed every 3 h and the membrane was dried for later experimental.
Characterization of PVDF/2-mercaptobenzothiazole blend membrane
All the experiments temperature and barometric was 25℃ and 0.1 MPa, respectively.
The determination of pure water flux
The pure water flux of the blend membrane can be calculated according to the following equation

The determination of rejection
Bovine serum albumin (BSA) was used frequently as a standard substance to evaluate the protein rejection of the prepared membrane. In this experiment, maintain a stable laboratory state as the same as the condition for determination of pure water flux. The initial concentration of BSA was 1.0 g/l. The absorbance was determined at the distance of 280 nm by UV–visible spectrophotometer. The determination of rejection was calculated by the following formula

Here,
The determination of water contact angles
The water contact angles of the membranes were measured on a contact angle goniometer (OCA40, Dataphysics, Germany) at room temperature. To minimize the experimental errors, the contact angle was measured at least three times for each sample and then the average was reported.
The determination of the Fourier transform infrared spectroscopy (FTIR)
To confirm the chemical modification, the FTIR spectra of the UF membranes were recorded by using a Bruker instrument (VERTEX-70). Each spectrum was captured by averaging 64 scans at a resolution of 4 cm−1. The structural morphology of the surface and a cross-section of the membrane were studied by SEM (JEOL Model S-4800, Japan). Samples of the membranes were frozen in liquid nitrogen and then fractured. All results were measured at least three times.
Adsorption of chromium ion by modified membrane
The amount of chromium ion adsorbed on membranes was one of the most important indexes in evaluating the removal ability of heavy metals of membranes. In this process, the initial concentration of chromium ion solution was 100 mg/l. The pH was adjusted to 7 and the temperature was controlled at 25℃. “Photometry of diphenylcarbazide” was used to measure the concentration of chromium ion. Adsorption experiments for chromium ion were carried out by varying the adsorption time (2, 4, 6, 8 h). The form of adsorbed chromium is described as follows

Desorption progresses were carried out in hydrochloric acid solution (0.5 mol/l), and the chromium ion concentration of the stripping liquid was measured at certain period of time until the concentration of chromium ion was approximately steady. The blend membranes were taken adsorption experiment after rinsed clean with deionized water. The rinsing process was repeated three times.
Results and discussion
Effect of 2-mercaptobenzothiazole dosage on membrane performance
As could be seen from Figure 1, the pure water flux increased considerably with the addition of the 2-mercaptobenzothiazole dosage into the blend membrane. This was because higher dosage of 2-mercaptobenzothiazole resulted in the increase of the porosity of the membrane. However, the higher 2-mercaptobenzothiazole dosage did not have significant effect on increasing the retention rate. This was due to the increase in the membrane pore size which was unconducive to the entrapment of chromium ion molecules. Figure 2 represents the effect of 2-mercaptobenzothiazole dosage on adsorption amount of Cr6+, and the results illustrate that chromium ion adsorption increased at first, and then decreased with the rising of 2-mercaptobenzothiazole. This was due to the agglomeration and decreased effective surfaces of the active groups in the high blending ratio, which lead to reduction of the membrane surface functional groups. Overall consideration, 0.2 wt% of 2-mercaptobenzothiazole was chosen as the best dosage, the adsorption quantity was 241 µg/cm2, the pure water flux was 260 l/m2 h, and the rejection rate was 85%.
Effect of 2-mercaptobenzothiazole dosage on water flux and rejection rate of Cr6+. Effect of 2-mercaptobenzothiazole dosage on adsorption amount of Cr6+.

Effect of blending time on membrane performance
As shown in Figures 3 and 4, chromium ion adsorption and the pure water flux increased prominently in the first few minutes of operation and then decreased with the later time. The rejection rate drops distinctly, with about 19% during the whole process. This was due to the 2-mercaptobenzothiazole distributed more uniformly in the membrane casting solution as blending time prolonged, it was beneficial to the chromium ion adsorption. However, after arriving at a certain time the continuous heating would cause 2-mercaptobenzothiazole generate a mass of micellar aggregates, which would lead to the loss of characteristic functional of the blend membrane. So chosen 6 h as the optimal blending time, and the adsorption quantity was 0.248 mg/cm2, the pure water flux and rejection rate were 260 l/m2 h and 87%, respectively.
Effect of blending time on water flux and rejection rate of Cr6+. Effect of blending time on adsorption amount of Cr6+.
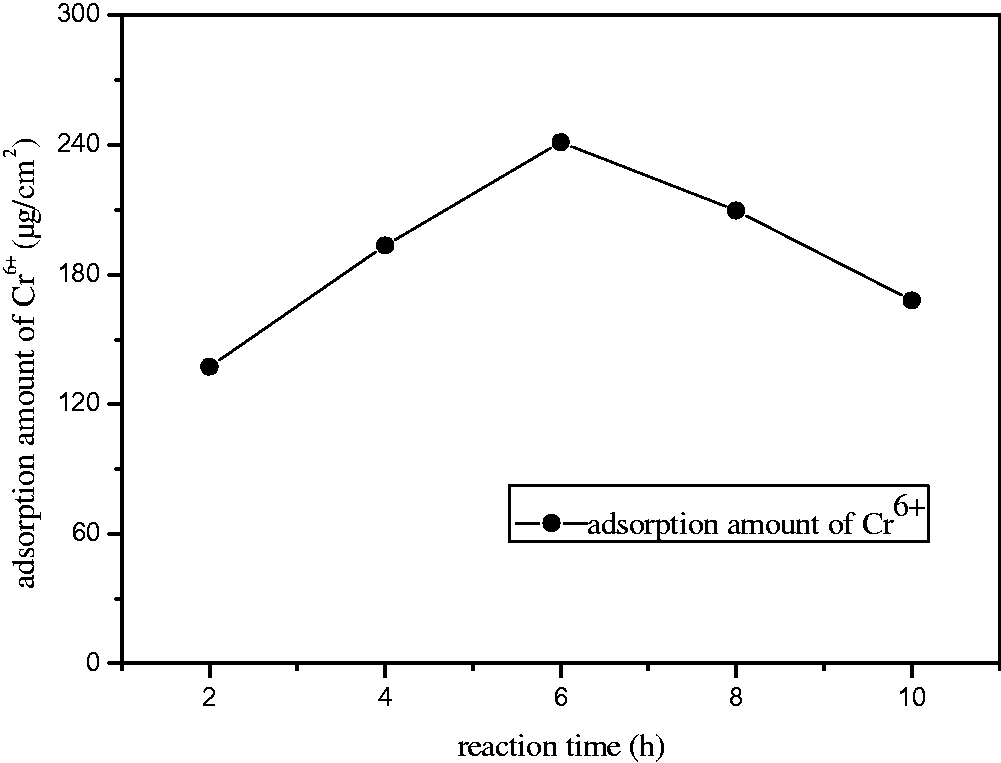
Effect of blending temperature on membrane performance
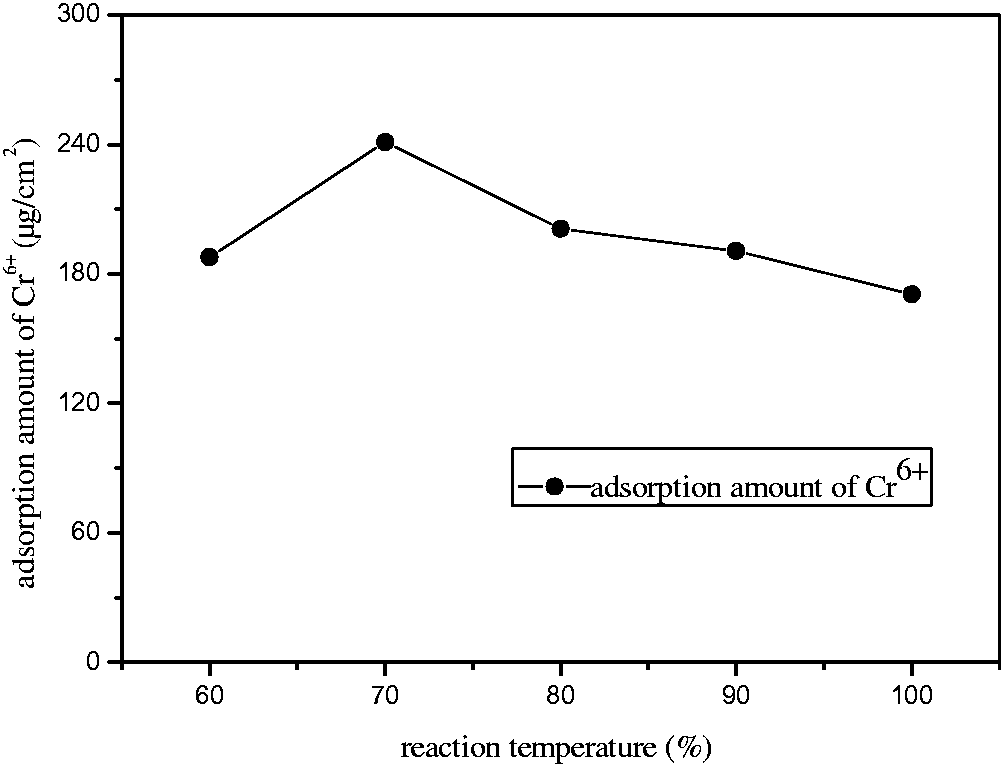
As can be seen from Figures 5 and 6, the chromium ion adsorption showed a similar consequence. They all reached the ideal state when the temperature was adjusted to 70℃. This was because high temperatures could lead to the structural change of the blend membrane and the reduction of functional groups, thus decreasing their adsorption and rejection capacities. The pure water flux reached its optimal state at 80℃. While with increasing of temperature, the pure water flux reduced. This may be because with the temperature increasing, the porosity has a decreasing trend, thus the pure water flux descends. The further research of the mechanism should be carried out in the future. Taking into account all these factors, we confirmed that 70℃ was the optimal blending temperature. The adsorption quantity was 0.240 mg/cm2, the pure water flux was 250 l/m2 h, and the rejection rate was 87% at the moment.
Effect of blending temperature on water flux and rejection rate of Cr6+. Effect of blending temperature on adsorption amount of Cr6+.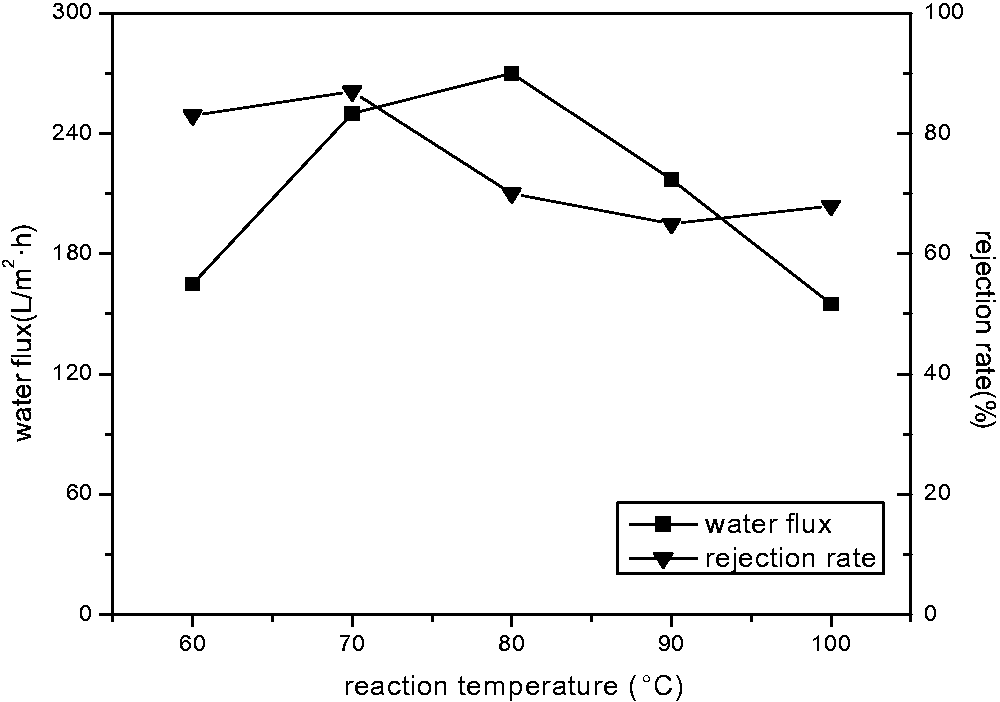
After study the effect of 2-mercaptobenzothiazole dosage, blending time, and blending temperature on membrane performance, the optimal membrane preparation conditions were obtained. In this case, pure water flux was 255.57 l/m2 h and the BSA rejection rate was 87.85+% of the optimal membrane. Then the water contact angle, FTIR, and adsorption capacity for chromium ion of the modified UF membrane were tested in the following work.
Water contact angle analysis
Contact angle analysis.

A stands for traditional PVDF ultrafiltration membrane; B stands for modified PVDF ultrafiltration membrane.
FTIR
As shown in Figure 7, the adsorptions between 3000 and 3100 cm−1 which stand for vibration characteristic adsorption frequency of unsaturated hydrocarbon key (=C–H) were stronger than the unmodified membranes. Modified membranes’ characteristic peaking in the range 800–1000 cm−1 shows the presence of C–N key. Compared with the unmodified membrane the 2-mercaptobenzothiazole-modified membrane showed the stronger adsorption at 1450 cm−1 which could be attributed to the H–C–H that 2-mercaptobenzothiazole peculiarly owned (Wang et al., 2014). This result proved that the 2-mercaptobenzothiazole had been blended in the membranes. These data indicated that the 2-mercaptobenzothiazole has been successfully blended into the PVDF.
The FTIR image of membrane. A stands for unmodified PVDF UF membrane; B stands for modified PVDF UF membrane. FTIR: Fourier transform infrared spectroscopy; PVDF: polyvinylidene fluoride; UF: ultrafiltration.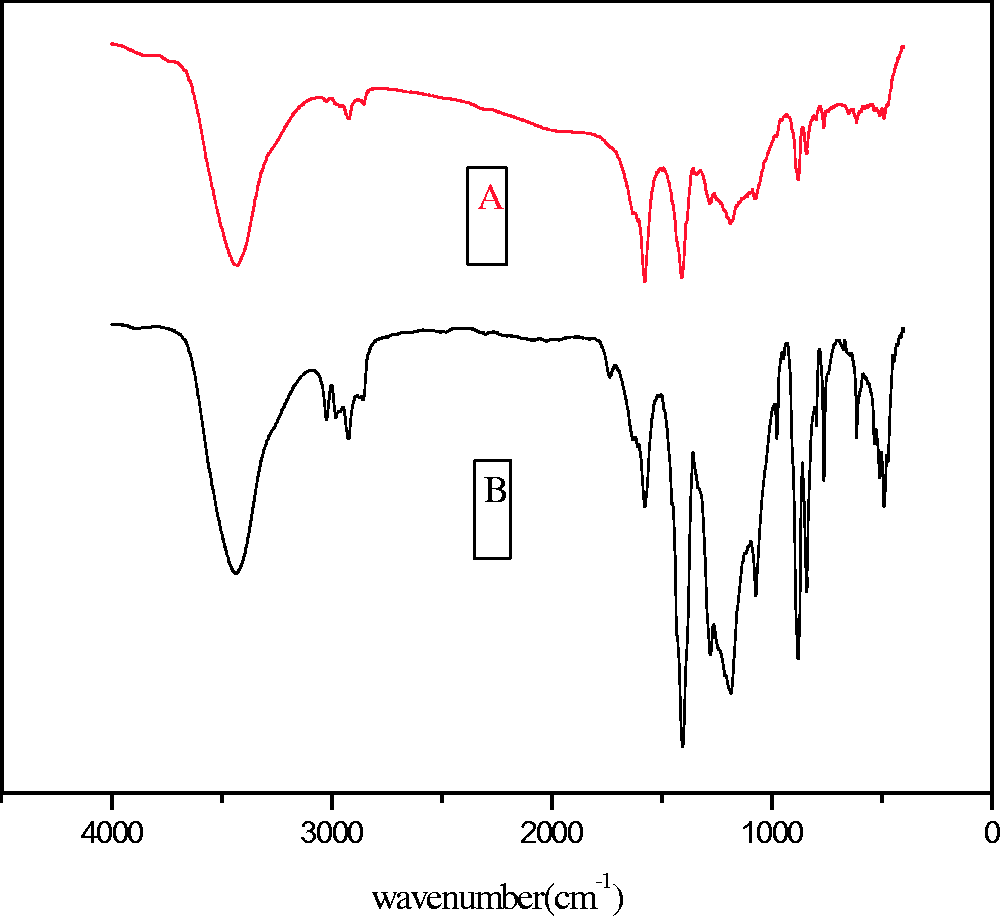
Scanning electron microscope (SEM) analysis
In order to assess the combination characteristic of PVDF/2-mercaptobenzothiazole-modified membrane and traditional membrane, the SEM images of the surface and the cross-section were shown in Figure 8(a) to (d). The SEM images provided qualitative information which showed that the bore diameter of the surface image and the number were smaller than traditional membranes. This was because 2-mercaptobenzothiazole could increase the pore channels. And the cross-section image of PVDF/2-mercaptobenzothiazole-modified membrane showed that the cortical were denser. On the other side, the modified membranes’ pores were smaller than traditional membranes. In short, the water flux of 2-mercaptobenzothiazole/PVDF-modified membrane was lower than traditional membrane, but the BSA retention rate was improved.
The SEM image of membrane. (a) Surface of 2-mercaptobenzothiazole-modified membrane, (b) surface of traditional PVDF membranes, (c) cross-section of 2-mercaptobenzothiazole/PVDF-modified membrane, and (d) cross-section of traditional PVDF membrane. PVDF: polyvinylidene fluoride.
Performance analysis of PVDF-modified membrane adsorption for chromium ion
The adsorption/desorption of PVDF-modified membrane and pure water flux.

PVDF: polyvinylidene fluoride.
Conclusion
In this paper, the influence of various factors on the PVDF-modified membranes was studied and the optimal conditions for the preparation of it were determined. Based on the experiments conducted here, the following conclusions can be drawn:
The results obtained show that chromium can be successfully removed from the waste water by blend membrane due to its high adsorption capacities. The results showed that the optimal UF membrane was obtained when 2-mercaptobenzothiazole content was 0.2 wt%, and the blending time and temperature were 6 h and 70℃, respectively. In this case, its pure water flux was 255.57 l/m2 h and the BSA rejection rate was 87.85%. The adsorption capacity of chromium ion was 241 µg/cm2. By studying the change of the contact angle, we can confirm that the contact angle of PVDF/2-mercaptobenzothiazole blend membrane was higher, but the hydrophilicity of modified UF membrane was lower than the traditional membrane.
Although this paper comes to the conclusion that 2-mercaptobenzothiazole blend with PVDF was feasible, the pure water flux was not high enough, it was even lower than the traditional separation membrane. Therefore, we need to resolve this problem and find a way to enhance the pure water flux of the modified membrane in further research.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (21006100), Shandong Science and Technology Development Plan (2012SGGX10220), Shandong Province Natural Science Foundation (ZR2011BL009), Science and Technology Development Plan for University of Shandong Province (J11LB51), Special funds of innovative Projects for Shandong Province postdoctoral (201103006), Open fund of Key Laboratory of Marine Chemistry Theory and Technology of Ministry of Education, Jinan Science and Technology Development Plan (201303066).
