Abstract
The present study reports the synthesis of molecularly imprinted chitosan-based adsorbents using 4-hydroxy benzoic acid (4HBA) as a dummy template for the selective extraction and analysis of salicylic acid (SA) using suspension polymerization method. Dummy template strategy is useful when templates like salicylic acid exhibit intramolecular hydrogen bonding. The synthesized adsorbents were characterized by Fourier transform infrared spectroscopy (FTIR) and scanning electron micrograph (SEM). The adsorption capacity of the adsorbents was investigated as a function of time, concentration of analyte and mass of adsorbent employed and was found to be 500.96 µmol g−1 for the chitosan-based 4-hydroxy benzoic acid molecularly imprinted polymer (4HBACTSMIP), 221.2 µmol g−1 for the salicylic acid MIP (SACTSMIP), 220 µmol g−1
Keywords
Introduction
Contamination of aquatic media by organic pollutant is a serious environmental problem. The main causes for the contamination are the discharge of industrial waste, excretion of unmetabolised medication into wastewater collection systems, excretion of veterinary drugs by farm animals into fields run off and surface water, improper commercial disposal methods and the release of the personal care products (PPCPs) like cosmetics, perfumes, and sunscreens into water while bathing. Amongst various techniques, adsorption has been reported as the most efficient method for removal of residual pollutants from wastewater (Dutta et al., 2004; Guibal, 2004; Varma et al., 2004). A wide range of adsorbents have been used for removing organic pollutant from contaminated water such as cellulosic wastes and their carbonisation products, bituminous coal and commercial activated carbons. However, the high cost of the activation process limits their use in wastewater treatment (Beltran et al., 2010; Mourya and Inamdar, 2008).
Chitosan (CTS) is a cheap bio-sorbent and is the N-deacetylated product of chitin which is the second most abundant natural biopolymer after cellulose. Both chitin and CTS have unique combination of properties like biodegradability, biocompatibility and bioactivity. These biodegradable polymers with good physical, chemical and mechanical properties have gained more attention in the application of adsorption techniques. Combined with the advantage of abundant availability and low price, their use has been prevalent in the pollution control operations. Presence of variety of the functional groups like primary amino, acetamido, and hydroxyl groups on CTS is responsible for its high chemical reactivity and therefore supports its use for removal of pollutants. CTS can be modified by chemical or physical processes in order to improve the mechanical and chemical properties (Dutta et al., 2004; Mourya and Inamdar, 2008). Numerous CTS derivatives have been reported with good mechanical stability by crosslinking CTS with various crosslinking agents like with epichlorohydrin, glutaraldehyde or ethylene glycol dimethacrylate. Grafting new functional groups on the CTS backbone can lead to increase in the adsorption selectivity. One of the novel methods of improving the adsorption capacity of CTS-based adsorbents is by molecular imprinting technique which creates specific recognition sites for higher adsorption. Recently, stress has been placed on the synthesis of molecularly imprinted polymers (MIP) using CTS as functional monomer for enhancing the adsorption efficiency of CTS-based adsorbents. CTS because of its versatility can be easily modified into different forms which increase its adsorption efficiency. Molecularly imprinted membrane has been synthesised using CTS as functional monomer for selective recognition of flavonoids in aqueous media. A novel molecularly imprinted nanofibre was synthesised by using β-cyclodextrin as monomer for the selective recognition of flavonoids using a pore forming agent to enhance the binding capacity of the polymers. MIPs have been effectively used as sensor material for sensing trace quantity of emerging persistent organic pollutants (Ma et al., 2011, 2013, 2016).
MIP has selective recognition sites for the template molecule which are generated within the polymer by allowing the template molecule to react with the functional monomer in the presence of porogen to form a template–monomer complex (Pardeshi et al., 2012). The type of interaction involved between the template and the monomer may be either covalent or non-covalent which should result in the formation of highly stable prepolymerisation complex (template–monomer complex). On polymerisation, the crosslinking agent freezes the prepolymerisation complex within the polymeric matrix and extracting the template molecule using a suitable solvent from the prepolymerisation complex results in the vacant binding sites with shape and size complementary to the structure of template molecule. The selectivity of the MIP is directly proportional to the number of such selective binding site within the polymeric matrix. However, the template with strong intramolecular hydrogen bonding generally leads to very weak non-covalent interaction between the template and the monomer. In such cases ‘dummy template strategy’ can lead to the higher selectivity in MIPs. Molecularly imprinted CTS-based adsorbents are found to be efficient at adsorbing residual pollutants from wastewater (Bai et al., 2008; Jiang et al.,2013; Kyzas et al., 2008, 2013; Shinde et al., 2013; Yu et al., 2008).
One large class of chemicals that have received comparatively little attention is Pharmaceuticals and active ingredients in the PPCPs, which are extensively used throughout the world. PPCPs and their metabolites are ubiquitous and display persistence into the aquatic environment. SA is a prominent metabolite of aspirin (acetylsalicylic acid, ASA), which is widely used as an analgesic for the prevention of heart attack, in keratolytic and dermatitis treatment and also as a preservative in the food items. SA has been identified as one of the PPCPs (Huang et al., 2003; Meng et al., 2013; Shi et al., 2014). In the United States alone, approximately 20,000 million tablets of Aspirin are consumed annually. ASA has been reported as the world’s first synthetic drug possessing anti-inflammatory activity against rheumatism. At present, ASA has achieved clinical prominence as a globally relied therapeutic agent not only because of its traditional anti-inflammatory paradigm, but also for its more advanced life-saving perspective related to cardiovascular events such as heart attacks and stroke (Sreenivasan, 2007). Generally, aspirin can be easily hydrolysed to SA during storage. Therefore, excess concentration of SA is often detected in aspirin, which can cause harmful effect to stomach and intestine, may induce the mucosa bleeding and even cause death in serious case. Therefore, monitoring the concentration of SA is important for clinical and environmental applications (You et al., 2014; Zhang et al., 2001, 2002).
As per the definition of MIP, it could be possible to synthesise a corresponding MIP for every compound. However, literature survey reveals that only limited number of compounds have been used as templates for the synthesis of MIP. The stability of MIP depends upon the stability of prepolymerisation complex formed between template and functional monomer. Compounds like SA with strong intramolecular hydrogen bonding interaction generally exhibit weak intermolecular hydrogen bonding interaction with the functional monomer resulting in poor imprinting effect. This can lead to corresponding MIP with very low selectivity. However this can be overcome by using ‘dummy templates strategy’ for the synthesis of MIP. Dummy templates are the structural analogues of target analytes. For the present study, 4-hydroxy benzoic acid (4HBA) was used as dummy template for the selective enrichment and analysis of SA (Huang et al., 2003; Meng et al., 2013; Shi et al., 2014; Sreenivasan 2007; You et al., 2014).
Computational approaches, such as molecular modeling, have been used in the molecularly imprinted polymer area to guide synthesis and performance by the design of virtual libraries that screen the best possible functional monomers for the preparation of an MIP for a specific template and also to study the nature of interaction between template–monomer complex or prepolymerisation complex (Pardeshi et al., 2012).
Re-binding capacities of different adsorbents for SA prepared using dummy template.

Materials and methods
Chemicals
CTS with >75% degree of deacetylation, epichlorohydrin, 4HBA, SA, 2,5 dihydroxy benzoic acid (2,5DBA), gallic acid (GA), granular activated carbon (GAC), sorbitan monooleate (Span 80), and formaldehyde were supplied by sigma Aldrich. Liquid paraffin and sucrose were obtained from Fisher scientific. Sodium hydroxide, acetic acid, Acetonitrile, and high performance liquid chromatography (HPLC) grade water were provided by Merck chemicals. All other chemicals were of analytical grade and supplied by Rankem.
Spectrophotometric analysis
Spectrophotometric analyses were performed using a Shimadzu (Japan) UV-visible spectrophotometer with matched 1 cm quartz cells. Calibration curves at 298 nm for salicylic acid adsorption experiments were prepared using standard solutions of salicylic acid in methanol and water (10:90 v/v).
Morphological characterization
4HBACTSMIP, SACTSMIP, NIP, CTS, and GAC were analysed using a scanning electron microscope (SEM, JEOL JSM-6360LV). The samples were coated with a thin gold film before observation in SEM.
The FTIR spectra of compounds were obtained using a Fourier transform infrared spectrometer (IR-Affinity-1, Shimadzu). The samples were ground with anhydrous KBr and the spectra were recorded between 4000 and 400 cm−1, by averaging 45 scans for each spectrum.
Preparation of 4HBACTSMIP and SACTSMIP
CTS (0.6 g) was dissolved in 30 mL of 2% acetic acid solution (v/v) and mixed with 15 mL of methanol containing 193 mg of dummy template 4HBA. This mixture was stirred for 30 minutes; 50 mL of liquid paraffin containing 0.3 mL of span 80 and 2 g of sucrose were added to this mixture and stirred at 50℃ for 10 minutes. Further, 1 mL of formaldehyde solution (37%, w/w) as amino protective solute was added to this mixture and stirred for 30 minutes. After adjusting the pH to 9.0 with 2 M sodium hydroxide solution, 1 mL epichlorohydrin was added drop wise into the mixture for crosslinking. The crosslinking reaction was performed by agitating the above mixture at 70℃ for 3 hours with the pH maintained at 9.0 throughout the process. The prepared 4HBACTSMIP was washed with acetic acid: methanol (20:80 v/v) to remove the unreacted precursors. The dummy template was extracted using acetic acid: methanol (20:80 v/v) in a Soxhlet apparatus for 24 hours followed by methanol and finally digested with 0.1% hydrochloric acid to remove the amino protective solute. 4HBACTSMIP was thoroughly washed with deionized water and dried in a vacuum drier before storing in vacuum desiccator. SACTSMIP was prepared using the same method with SA as the template. The control polymers non-imprinted polymer (NIP) were also prepared using same procedure in the absence of template molecule 4HBA or SA (Wang et al., 2014).
Binding experiments
The binding experiments were carried out batch-wise in triplicate to study the recognition performance of 4HBACTSMIP, SACTSMIP, NIP, GAC, CTS in the binding solvent methanol and water (10:90 v/v) using the method given below.
Dose optimisation
The dose of each adsorbent was varied in the range 10–100 mg and equilibrated in 10 ml solutions of 4 mmol L−1 SA using a water bath shaker for 2 hours and analysed for SA.
Time optimization
The contact time was optimised by equilibrating the optimised dose of each adsorbent for the fixed time period ranging from 10 minutes to 3 hours with 4 mmol L−1 of SA solution. SA was analysed after each time interval.
Selectivity study
4HBA, GA, 2,5DBA and SA were used as structural analogues to verify the selectivity of MIPs for the template over other structurally similar compounds.
SEM images of (a) 4HBACTSMIP, (b) NIP, (c) SACTSMIP, (d) CTS and (e) GAC.
4HBACTSMIP, SACTSMIP, and NIP were equilibrated with a mixture containing 5 mmol L−1 solutions of SA and 4HBA, at optimized condition for the optimum time period. After equilibration, the equilibrium concentrations of SA and 4HBA were analysed using HPLC.
4HBACTSMIP, SACTSMIP, GAC, CTS, and NIP were equilibrated with a mixture containing 5 mmol L−1 solution of SA, 4HBA, 2,5DBA, and GA at optimized condition for the optimum time period. The residual concentration of SA and 4HBA, 2,5DBA, and GA in the solution was analysed after equilibration, using HPLC.
HPLC and Chromatographic conditions
Waters HPLC equipped with a quaternary pump (2545 waters HPLC pump and photodiode array (PDA) detector (2998 Waters)), ODS-SP C18 Column (5 µm, 4.6 × 250 mm; Merck) was used for the selectivity studies. Chromatographic conditions were as follows: water/acetonitrile/acetic acid (70:30:0.2 v/v) as mobile phase, flow rate of 1 mL min−1, injection volume of 10 µL, and UV detection at 281 nm. The concentrations were determined using peak area.
Binding isotherm
Re-binding experiments were carried out to study the binding capacity of the MIPs. A series of standard SA solutions ranging from 0.5 to 5 mmol L−1 were prepared; 10 mL aliquots of each standard solution were mixed with the optimised dose of each adsorbent and shaken for the determined optimum time period. The solutions were then analysed for SA. The binding capacity, B (µmol g−1) of each adsorbent was calculated using the following equation

Kinetic study
For kinetic experiments, 50 mg of each adsorbent was suspended in 10 mL of 4 mmol L−1 SA solution and continuously shaken at room temperature. The concentrations of SA at the time intervals of five minutes (5–40 minutes) were analysed by UV-visible spectrophotometer at 298 nm and then the adsorption capacity Qt (µmol g−1) at different contact times t was calculated.
Molecular modeling
Molecular modeling studies can prove to be useful for the selection of suitable grafting agent for the functionalisation of CTS and also for the selection of suitable monomer for MIP synthesis (Farrington et al., 2006). Molecular modeling studies include several steps, as follows (Pardeshi et al., 2012).
Building of models: The first step is building of molecular models using Chem 3D Ultra software. The 3D structures are drawn and Cartesian coordinates of stable conformers are generated to prepare input file for running the Gaussian simulations.
Geometry optimization: The B3LYP (Becke-Style 3-parameter density functional theory (DFT) using the Lee–Yang–Parr correlation function) with 6-31 G (d) basis set is used for geometry optimisations to obtain structures with minimum energy.
Frequency calculations: The harmonic vibrational frequencies confirmed the structures as minima with no imaginary frequencies and enabled the calculation of Gibbs free energies.
Results and discussion
SEM result
Scanning electron micrographs of 4HBACTSMIP, NIP, SACTSMIP, GAC, CTS are presented in Figure 1. It can be seen that the NIP presents a relatively smooth surface compared to the rough porous surface of 4HBACTSMIP. This must have resulted in the higher binding capacity of 4HBACTSMIP as compared to NIP. SEM images also indicated that the beads formed were different in size (polydispersed). Amongst GAC and CTS, GAC depicted relatively smooth surface.
FTIR spectra of CTS, 4HBACTSMIPBE (before Extraction), 4 HBACTSMIPAE (after extraction) and NIP.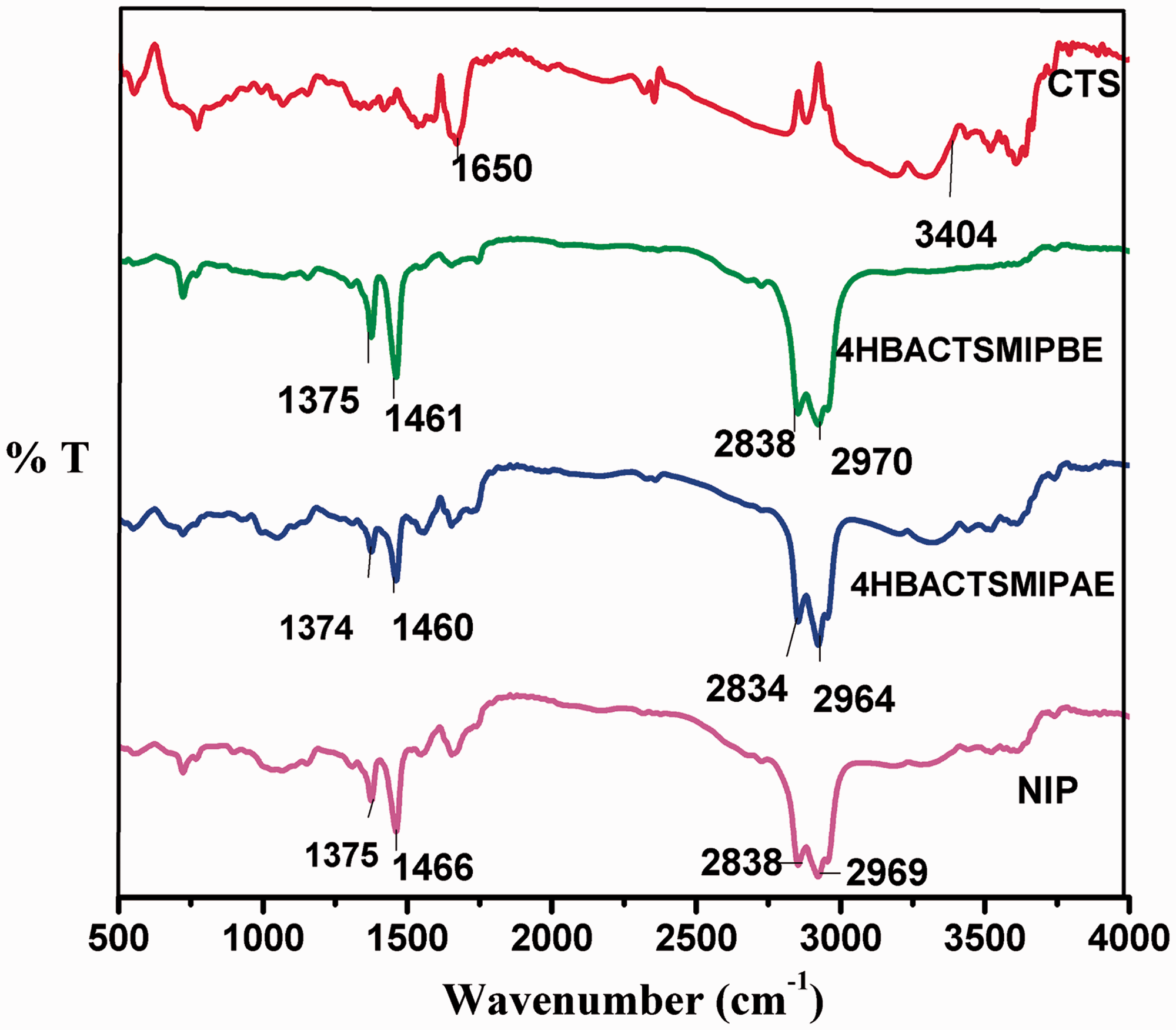
FTIR result
The FTIR spectra of CTS, 4HBACTSMIPAE (after extraction of 4HBA), 4HBACTSMIPBE (before extraction of 4HBA), and NIP are presented in Figure 2. The spectrum of CTS exhibits a broad peak around 2400 to 3400 cm−1 due to the presence of intramolecular H bonding between –NH2 and –OH groups present in CTS. The peak around 1650 cm−1 can be attributed to the amide group in CTS. (Brugnerotto et al., 2001). The spectra of 4HBACTSMIPBE (before extraction of template 4HBA) show decrease in the intensity around 2400 to 3400 cm−1 which can be attributed to the formation of intermolecular hydrogen bond between –OH group of CTS and carbonyl group of 4HBA, thus increasing the O–H bond length in CTS. These results are in accordance with the molecular modeling studies. The spectra of NIP and 4HBACTSMIPAE (after extraction of 4HBA) were seen to be almost similar. FTIR results depict that the dummy template 4HBA has been successfully imprinted in CTS via hydrogen bonding between carbonyl group of 4 HBA and –OH group in the functional monomer CTS.
Effect of time on adsorption of salicylic acid onto 4HBACTSMIP, SACTSMIP, NIP, CTS and GAC.
Suspension polymerisation method
Generally in suspension polymerisation method, water is used as a continuous phase to suspend a droplet of prepolymerisation complex (template–monomer complex). MIPs prepared by suspension polymerisation method are polydispersed but shows enhanced affinity for template molecule in comparison to NIP. The presence of water as a continuous phase during polymerisation is expected to negatively affect the efficiency of imprinting process by disrupting the weak hydrogen bonding or dipolar interactions involved in imprinting process. To overcome this limitation, alternative solvent has been used as dispersing medium which preserves non-covalent interactions in the template–monomer complex (4HBA-CTS or SA-CTS). In the present method, mineral oil was employed as continuous phase instead of water. The added advantage of using mineral oil is that no stabiliser is needed during synthesis (Biffins et al., 2012).
Batch re-binding studies
All experiments were performed in triplicate for reasons of accuracy and precision.
Optimization of time
The adsorption experiment was carried using 20 mg of 4HBACTSMIP, in neutral pH at room temperature using 10 mL of 4 mmol SA solution, for contact time ranging from 5 minutes to 180 minutes. The results are shown in Figure 3. It was observed that the adsorption capacity of 4HBACTSMIP for SA increased with the increase in the contact time and reached adsorption equilibrium within 100 minutes. Similar trend was observed for SACTSMIP, NIP, CTS, and GAC. Therefore, all subsequent adsorption experiments were performed at optimum equilibrium time of 100 minutes.
Effect of adsorbent dose on Binding capacity of 4HBACTSMIP, SACTSMIP, NIP, CTS and GAC.
Optimization of dose
It was observed that for the given SA concentration, increase in adsorbent dosage of 4HBACTSMIP, CTS, GAC, SACTSMIP, and NIP in each case leads to the increase in adsorption capacity as shown in Figure 4. The maximum binding was observed at the dose of 50 mg of adsorbents in most of the adsorbents and was used for the further experiments. The probable reason is that the increase in adsorbent dosage provides more functional group and active sites, thus leading to the increase in adsorption capacity (Liu et al., 2010).
Effect of concentration on binding capacity of 4HBACTSMIP, SACTSMIP, NIP, GAC and CTS.
Adsorption isotherms and binding site characterization
It was observed that the binding capacity of 4HBACTSMIP increased with the increase in concentration of SA and reached adsorption equilibrium at higher concentration as shown in Figure 5. With increase in the SA concentration, more active sites of 4HBACTSMIP were involved in the adsorption process. Higher concentration enhances the mass transfer driving force, thus increasing the adsorption capacity at equilibrium. In addition, with increasing concentration, the number of collision between template molecule and adsorbent also increases which in turn enhances adsorption process (Jin et al., 2008; Umpleby et al., 2001). Similar trend was observed for SACTSMIP, NIP CTS, and GAC.
Adsorption isotherm of 4HBACTSMIP, SACTSMIP, NIP, GAC, and CTS using Freundlich (F) adsorption isotherm model.
The purpose of the adsorption isotherms is to relate the adsorbate concentration in the bulk with the adsorbed amount at the interface. The concentration of SA bound to the polymers, i.e. the binding capacity (B), was plotted against the free concentration of SA in the solution .The isotherm results were analysed using the Langmuir and Freundlich isotherms. The experimental data were found to fit better into the Freundlich isotherm equation as compared to the Langmuir isotherm equation (Umpleby et al., 2001).
The Freundlich isotherm is an empirical equation employed to describe heterogeneous systems. The Freundlich equation is expressed as
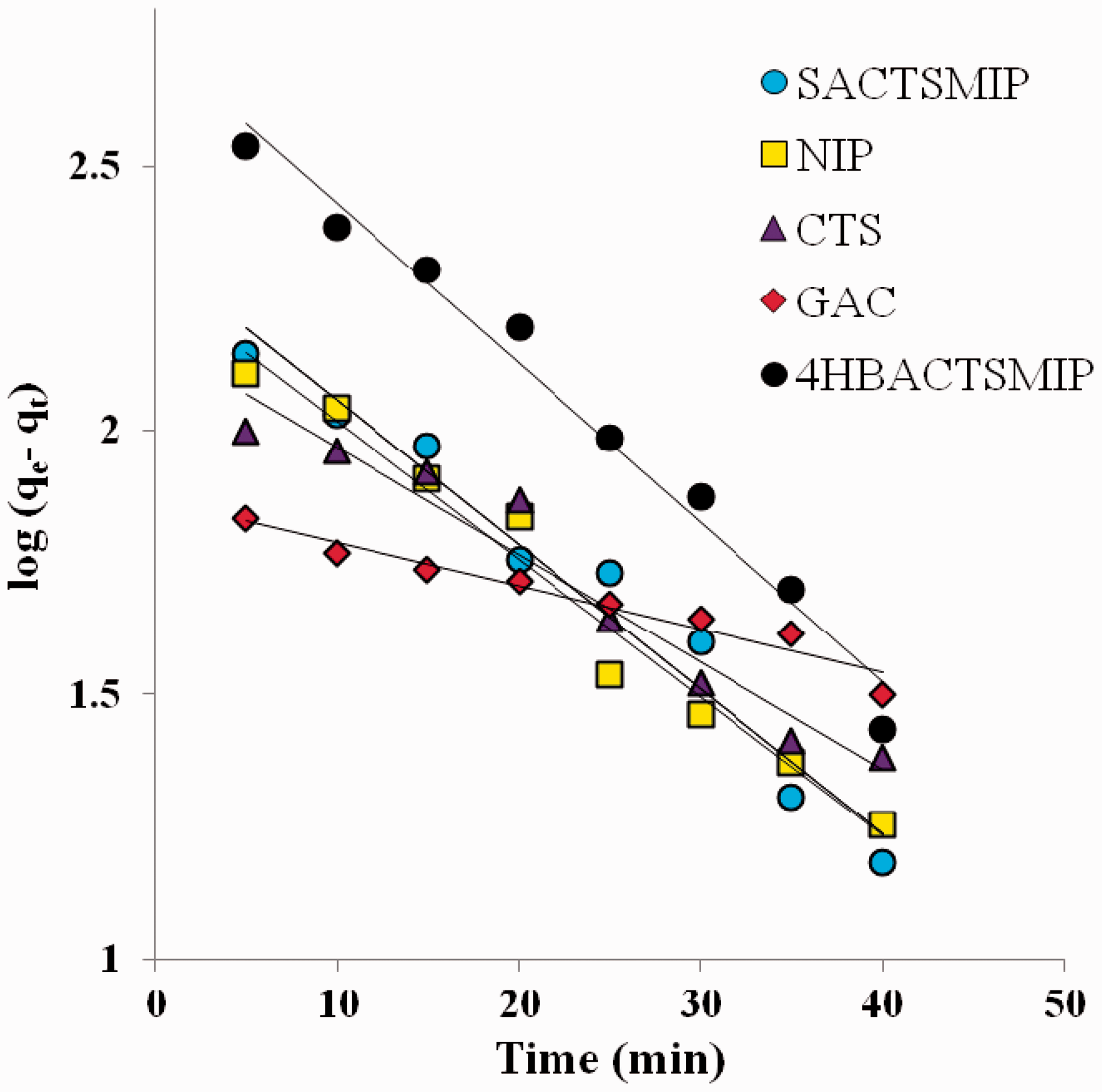
Pseudo first-order kinetic plot for 4HBACTSMIP, SACTSMIP, NIP, GAC, CTS.

Fitting parameters of the Freundlich model for different adsorbents.

Kinetic Study
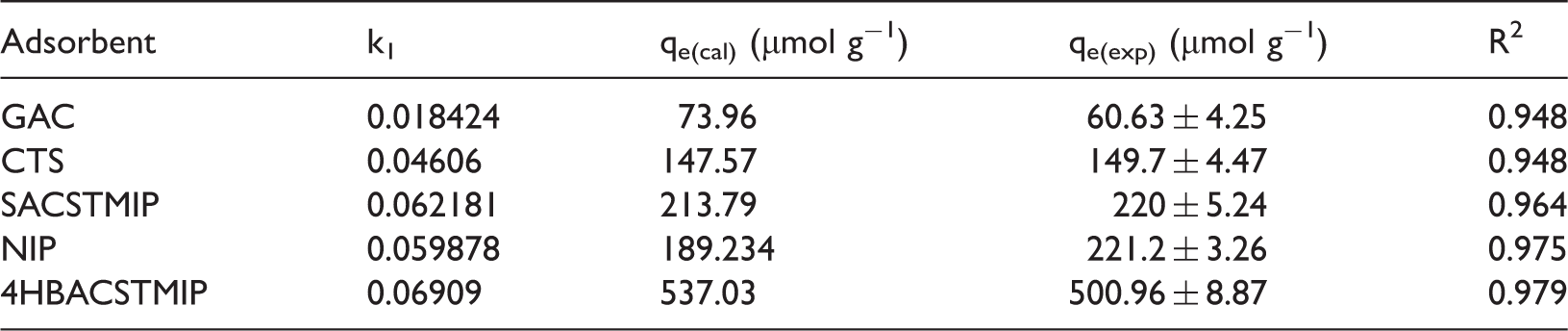
Kinetic studies were performed for different adsorbents and the time dependent adsorption profiles are shown in Figure 3. SA adsorption on 4HBACTSMIP was quite rapid in the first 80 minutes, accounting for 84% of SA removal (414 µmol g−1). Adsorption on SACTSMIP and NIP on the other hand, proceeded much more slowly, with 40% removal of SA achieved after 90 minutes contact time. Adsorbent saturation was reached at 90 minutes for 4HBACTSMIP, whereas NIP and SACTSMIP attained saturation in 45 minutes. The fast adsorption kinetics and high capacity of 4HBACTSMIP resulted from improved imprinting effect and porous structure. To better understand the adsorption behaviour of SA onto the 4HBACTSMIP, two conventional kinetic models were applied to analyse the kinetic data. The pseudo first-order and pseudo second-order kinetic equations are shown in equations (4) and (5), respectively (An et al., 2011; Hameed et al., 2008).
Structures of 4-hydroxy benzoic acid (4HBA), Gallic acid(GA), 2,5 Dihydroxy Benzoic Acid (2,5DBA) and Salicylic acid (SA) and the intramolecular hydrogen bonding in 2,5DBA and SA.


Adsorption kinetic data of different adsorbents for pseudo first-order model.
Selectivity study
Binding capacities of SACTSMIP, 4HBACTSMIP, NIP, GAC, and CTS for SA and its structural analogues.
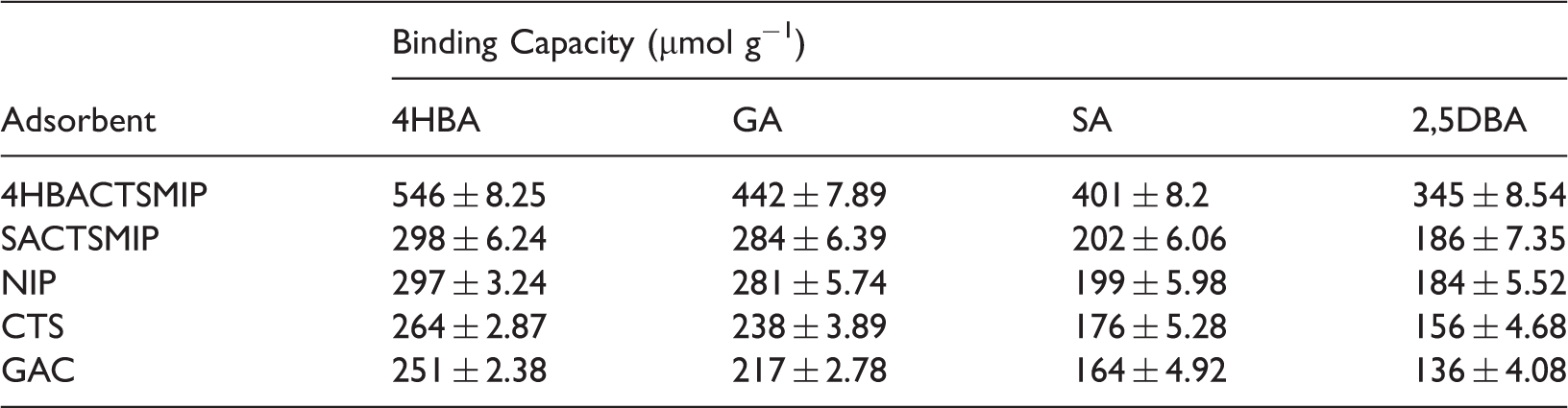
It is also observed that the binding capacity of SA is less than that of GA although SA has two hydroxyl group less than GA. Least binding capacity was observed for 2,5DBA which has one hydroxyl group lesser than GA. Low binding capacity for 2,5DBA and SA may have resulted due to weak interaction between 2,5DBA or SA with the binding cavities in the 4 HBACTSMIP because of the intramolecular hydrogen bonding between the carboxylic group and the adjacent hydroxyl group in 2,5DBA and SA molecule. This clearly indicates that the intramolecular hydrogen bonding within the template molecule can adversely affect the imprinting efficiency. Amongst 2,5DBA and SA, SA had greater binding capacity due to the molecular volume being nearly same as 4HBA than 2,5DBA (61.56 Å3). Thus, it can be concluded that the 4HBACTSMIP was found to be much selective for 4HBA over its structural analogues.
SACTSMIP was synthesised to study the influence of intramolecular hydrogen bonding of templates on imprinting using SA as template which exhibits intramolecular hydrogen bond, and its molecular recognition ability was tested. Table 4 clearly indicates that the binding capacity of SA and other structural analogues (GA, 2,5DBA, and 4HBA) onto SACTSMIP is almost similar to that of NIP. Thus, it can be concluded that SACTSMIP had no molecular imprinting effect.
Chromatogram for 4HBA and SA before and after equilibration with NIP, SACTSMIP, and 4HBACTSMIP is presented in Figure 9. The chromatogram shows a decrease in peak intensity after equilibration. The decrease in peak intensity was found to be greater for 4HBACTSMIP and was nearly same for SACTSMIP and NIP for SA.
Chromatogram for 4HBA and SA after equilibration with NIP, SACTSMIP, and 4HBACTSMIP.
Thus, it can be concluded that 4HBACTSMIP was found to be more selective for 4HBA over its structural analogues because of molecular imprinting effect. However, in case of SACTSMIP, the binding capacity of SA and all other structural analogues was found to be almost similar as that of NIP because of no molecular imprinting effect. The binding capacity of SA on 4HBACTSMIP was found to be greater than on SACTSMIP. Thus, the use of the dummy template 4HBA, the structural analogue of SA for the selective enrichment, and analysis of SA can be justified for the synthesis of MIP.
Amongst GAC and CTS, it was observed that CTS had greater binding capacity than GAC for all structural analogues which may be due to the presence of more active sites on CTS.
Molecular modeling to study the influence of intramolecular H-bond of 4HBA and SA on molecular recognition of molecularly imprinted polymer
Compounds like SA with strong intramolecular hydrogen bond exhibit weak intermolecular hydrogen bonding interactions with functional monomers in the imprinting process. Consequently, the corresponding MIPs exhibit no specific adsorption ability. In SA, a strong intramolecular hydrogen bond is formed between the adjacent –OH and –COOH groups on benzene ring resulting into the formation of six membered ring (Figure 8), as a result the intermolecular H-bonding interaction between SA and CTS is weakened, resulting into poor imprinting effect. But in 4HBA molecule the –COOH and –OH groups are at para position (Figure 8) so they are unable to form intramolecular H-bonding although it exhibits intermolecular H-bonding, not as strong as the intramolecular H-bond in solution (Meng et al., 2013; Shi et al., 2014; Sreenivasan 2007; You et al., 2014; Zhang et al., 2001, 2002). The strength of intermolecular H-bonding is related to the concentration of components. In the present study the concentration of 4HBA is in low concentration of 1 mmol. Therefore, the intermolecular H-bonding would not be very strong amongst the 4HBA molecules in the solution. When suitable excess amount of the functional monomer CTS is added to the polymerisation mixture, the intermolecular H-bonding among 4HBA will be weakened by large amount of CTS because the chance of interaction between CTS and 4HBA is increased. Thus, the stable 4HBA-CTS complex is formed through intermolecular H-bonding between CTS and 4HBA consequently leading to the formation of specific binding sites for 4HBA in 4HBACTSMIP with high imprinting effect. When the binding capacity of 4HBACTSMIP and SACTSMIP for SA was compared, it was observed that 4HBACTSMIP had greater binding capacity than SACTSMIP as SA had poor imprinting effect (Table 4). These results are in line with the molecular modeling studies.
Molecular modeling
The type of interaction between templates (4HBA, SA) and CTS was also confirmed with the help of DFT by calculating Gibbs free energy (ΔG) during the complex formation. The Gibbs free energy gains of the complexes were calculated using equation (6)

Figure 10(a) to (e) (see online data supplement for supporting information) shows the optimised geometries of the template (SA and 4HBA), monomer (CTS), and template–monomer complexes. Hydrogen bonds can be formed between proton donor such as amide, amine or hydroxyl group and proton acceptor such as carbonyl group. Similar interactions were observed between proton donor (CTS) and proton acceptor (SA, 4HBA) to form template–monomer complex. Figure 10(d) and (e) shows that there is a strong interaction between CTS and 4HBA (1.858 Å) as compared with CTS and SA (1.897 Å). After H-bond formation with template molecule, an increase in the O–H bond length of CTS was observed in the case of CTS-4HBA complex (0.970 Å to 0.978 Å Figure 10(c) and (d)), while in case of CTS-SA complex, the O–H bond length of CTS changed from 0.970 to 0.977 (Figure 10(c) and (e)). Increase in the O-H bond length of CTS was observed to be greater for CTS-4HBA complex due to strong interaction between CTS and 4HBA (1.858 Å) as compared to CTS and SA complex (1.897 Å).
(a) Optimized structure of SA with Intramolecular H-bonding. (b) Optimized structure of 4HBA. (c) Optimized structure of chitosan monomer. (d) Optimized structure of chitosan −4 HBA complex (template–monomer complex). (e) Optimized structure of chitosan-salicylic acid complex (template–monomer complex).
Gibbs free energy of template–monomer complex.

CTS formed stable complex with 4HBA than with SA. A possible explanation for the stability of the CTS-4HBA complex is that SA forms strong intramolecular hydrogen bond; as a result, the intermolecular H-bonding interaction between SA and CTS gets weakened. But in 4HBA molecule, the –COOH and –OH groups are at para position and are therefore unable to form intramolecular H-bonding. This leads to the formation of stable CTS-4HBA complex through intermolecular H-bonding between CTS and 4HBA.
Comparative study
Comparative study for binding of salicylic acid using different adsorbents.

Conclusion
Present work demonstrates the preparation of an efficient 4HBACTSMIP for the uptake of SA from the aqueous solution with specific binding capacity of 38.8 mg g−1. The use of CTS as the functional monomer complies with greener approach for the polymer preparation. Experimental and modeling results show that the molecular recognition ability of the MIP decreases if the template forms intramolecular hydrogen bonding, when hydrogen bonding is responsible for the molecular recognition of the imprinted polymer as in case of SACTSMIP. The equilibrium data fitted well to Freundlich equation and the adsorption process could be described by pseudo first-order model. The synthesized adsorbent can prove useful for the removal as well as recovery of SA from aqueous matrix.
Footnotes
Acknowledgements
Sincere thanks to Kavita Pande and Dr. Rita Dhodapkar for their continuous help and support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
The online supporting information for Figure 10(a) to (e) is available at ![]() .
.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
