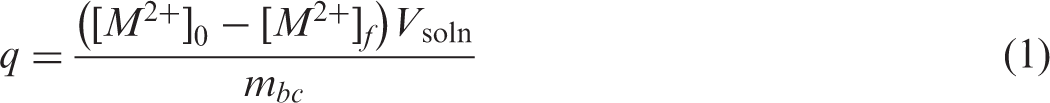Abstract
This paper evaluates the pyrolysis conditions applied during the synthesis of bovine bone char and the effect of these parameters in its adsorption properties for heavy metal removal from aqueous solution at batch reactors. The synthesis route has been analyzed in detail and the surface interactions involved in the adsorption process has been studied and discussed using different characterization techniques, given a special emphasis on X-ray photoelectron spectroscopy (XPS) analysis. A proper selection of the pyrolysis conditions improved the metal uptake of bone chars by 143% where the adsorption capacities ranged from 68.3 up to 119.4 mg/g. The removal performance followed the trend Cd2+ > Zn2+ > Ni2+. However, the multicomponent removal of Zn2+, Cd2+, and Ni2+ ions in both binary and ternary mixtures was a strong antagonistic adsorption process. XPS analysis confirmed that the ion exchange between the calcium, from the hydroxyapatite structure of bone char, and the heavy metals in solution played an important role in the adsorption process. These findings are useful to enhance the efficacy of heavy metal removal from aqueous solution using bone char.
Introduction
Tailoring the physicochemical properties of adsorbents for water treatment and purification is relevant from an economical and technical point of view (Dastgheib et al., 2004). Process efficacy and operational costs of the adsorption process highly depend on the characteristics of the adsorbent, especially its porous structure, and surface chemistry. The synthesis conditions have a significant impact on the physicochemical properties of the adsorbent and a key issue is to identify the best route for the desired application. However, this is not an easy task because several variables are involved in the synthesis (Rivera-Utrilla et al., 2011). To date, several routes have been reported for the preparation of tailored adsorbents for the removal of both inorganic and organic pollutants, but these studies have mainly focused on activated carbons obtained from lignocellullosic precursors (Bhatnagar et al., 2013; Hokkanen et al., 2016; Rivera-Utrilla et al., 2011). The assessment of alternative materials obtained from other sources and the analysis of their surface characteristics seem mandatory to identify promising adsorbents.
Bone char (BC) is playing an important role as adsorbent in water pollution control because of its outstanding physicochemical properties (Flores-Cano et al., 2016). This material is characterized by a mesoporous structure with a specific surface area from 80 to 120 m2/g and can be obtained via the thermal treatment of different bone wastes (Leyva-Ramos et al., 2010; Moreno-Piraján et al., 2010; Patel et al., 2015). It has been widely used in water defluoridation processes (Medellin-Castillo et al., 2014; Rojas-Mayorga et al., 2013) and, during last decades, new applications have emerged for this adsorbent (Ko et al., 2004; Mendoza-Castillo et al., 2015; Pan et al., 2009; Reynel-Avila et al., 2015, 2016). Several studies have reported the removal of a variety of organic and inorganic toxic compounds from aqueous solutions with BC including, for example, pharmaceuticals (Reynel-Avila et al., 2015), dyes (Reynel-Avila et al., 2016) and heavy metal ions (Ko et al., 2004; Mendoza-Castillo et al., 2015; Pan et al., 2009).
The best synthesis route of BC for water treatment is a matter of debate and different experimental conditions have been reported in the literature, e.g. Moreno-Piraján et al. (2010) and Mwaniki (1992). Despite these discrepancies, it is widely accepted that the temperature and the atmosphere used during the thermal treatment are the most important operating parameters that can affect the final adsorbent properties (Moreno-Piraján et al., 2010; Mwaniki, 1992; Rojas-Mayorga et al., 2013, 2015). It is clear that differences in the synthesis conditions of BC may impact its pollutant uptakes. For instance, commercial BCs show fluoride uptakes from 1 to 4 mg/g (Tovar-Gómez et al., 2013). However, recent studies from our research group have concluded that the fluoride adsorption properties of BC can be improved if an optimum pyrolysis process is used where the final fluoride uptake can be as high as ∼7.0 mg/g (Rojas-Mayorga et al., 2013). Similar trends can be expected to enhance the adsorbent performance for other organic and inorganic water pollutants.
Herein, it is convenient to remark that a standardized and optimized synthesis route for BC with tailored characteristics for the adsorption of heavy metal ions has not been identified, despite the relevance of these pollutants in the context of water treatment. Some authors have shown that the metal uptakes of BC may be higher than those obtained for activated carbons and other adsorbents (Choy and McKay, 2005; Ko et al., 2004; Mendoza-Castillo et al., 2015). However, the discrepancies in the synthesis routes cause a high variability on its final physicochemical properties. The identification of appropriate preparation conditions for this adsorbent is relevant to improve its removal performance and to reduce the production costs.
With this in mind, the aim of this study is to identify the best pyrolysis conditions to synthetize bovine BCs with tailored adsorption properties for heavy metal removal from aqueous solutions. This synthesis route has been analyzed in detail to improve the adsorption of Cd2+, Ni2, and Zn2+ ions at batch reactors. Surface interactions involved in the removal of these metallic cations with BC have been studied using different characterization techniques, given an emphasis in the discussion of XPS analysis.
Methodology
Synthesis of BC and tailoring of its adsorption properties for heavy metal removal
Metal uptakes and yields of bone char obtained at different pyrolysis conditions.

The best adsorbent was utilized in additional removal experiments for quantifying adsorption kinetics and isotherms of Cd2+, Ni2, and Zn2+. Kinetics studies were performed with an initial concentration of the heavy metals of 500 mg/L and an operating time up to 38 h at pH 5 and 30℃. Adsorption and intraparticle diffusion rates were calculated using the pseudo-second order kinetic model and the Weber-Morris approach, respectively. These models are defined as




Results of kinetic and isotherm data modeling for the adsorption of heavy metals on bone char.

Multicomponent adsorption of heavy metal ions on bone char using binary mixtures Cd2+–Ni2+, Zn2+–Cd2+, and Zn2+–Ni2+ at pH 5 and 30℃.

Multicomponent adsorption of heavy metal ions on bone char using ternary mixtures Cd2+–Ni2+–Zn2+ at pH 5 and 30℃.
Preparation of metallic solutions and its quantification
All removal experiments were done using metal solutions prepared with reactive grade nitrate salts (JT Baker) and deionized water. Concentrations of heavy metals in aqueous solution were quantified via atomic absorption spectroscopy (ICE 3000 Thermo Scientific) with linear calibration curves.
Study of surface chemistry of BC
Surface chemistry of BC was studied using different characterization techniques. Transmission FTIR spectra were utilized for identifying the surface functional groups with a Bruker IFS 66/S spectrophotometer. All samples were analyzed with spectroscopic grade KBr using a scan time of 200 scans and a resolution of 4 cm−1. The analysis of adsorbent crystalline structure was performed using X-ray diffraction (XRD) measurements in the 10 ≤ 2θ ≤ 80 range, which were obtained with a X-ray diffractometer Bruker D8-Advance equipped with Göebel mirror that has a tube with copper anode RX and radiation Cu Kα (λ = 1.5406 Å). Database from ICDD (International Center for Diffraction Data) was used in this analysis. X-ray photoelectron spectroscopy (XPS) analysis of BC samples was carried out with a K-ALPHA Thermo Scientific spectrometer using Al K-alpha radiation (1486.6 eV), which was monochromatized by a twin crystal monochromator yielding a focused X-ray spot with a diameter of 400 µm at 3 mA × 12 kV. Details of the experimental protocol of the XPS measurements have been reported in a previous study (Rojas-Mayorga et al., 2015). XPS results were analyzed considering a data fitting based on a combination of Lorentzian (30%) and Gaussian (70%) lines. Binding energies were obtained with an accuracy of ±0.2 eV and they were referenced to the C 1s line (i.e., 284.6 eV). The elemental composition was quantified by X-ray fluorescence (XRF) spectroscopy using a sequential X-ray spectrometer (Philips Magix Pro), which was equipped with a rhodium tube and beryllium window. The textural parameters of BC were determined via N2 adsorption–desorption isotherms at −196℃ with a home-made fully automated equipment designed and constructed by the Advanced Materials group (LMA) of University of Alicante (Spain). Morphology and composition of the BC were obtained by scanning electron microscope/energy-dispersive X-ray spectroscopy (SEM/EDX) with a FE-SEM system (Quanta FEG 650, FEI). Removal mechanism and surface interactions between heavy metal ions and BC were discussed using the results from these characterization techniques.
Results and discussion
Best conditions for the synthesis of BC for heavy metal removal from water and its adsorption properties
Table 1 shows the metal uptake for the twenty samples of BC obtained via pyrolysis. Adsorbent yields ranged from 70.5% to 75.4% and it is clear that the impact of the thermal treatment on this parameter was insignificant. Concerning the adsorption capacity, the metal uptake of tested samples ranged from 50.2 to 66.7 mg/g for Zn2+, 54.5 to 78.5 mg/g for Ni2+ and 66.3 to 81.9 mg/g for Cd2+ ions. Pyrolysis conditions have a significant impact on the metal adsorption properties. The adsorption capacities increased with the pyrolysis temperature from 650℃ to 700℃ and decreased at synthesis temperatures >700℃. Note that this trend prevails for the three heavy metal ions. The highest metal uptakes were obtained at 700℃ being 81.9 ± 0.79 for Cd2+, 78.5 ± 0.85 for Ni2+ and 66.7 ± 0.71 for Zn2+. Results showed that the best pyrolysis conditions correspond to 700℃ during 2 h and a heating rate of 10℃/min. It is interesting to remark that this synthesis route agreed with that reported for BC synthesis using fluoride ion as target pollutant (Rojas-Mayorga et al., 2013). The dehydroxylation process of hydroxyapatite and the decomposition of carbonates of BC occurred at pyrolysis temperatures higher than 700℃ (Rojas-Mayorga et al., 2013). This thermal effect on adsorbent structure may impact its ion exchange properties for heavy metal removal.
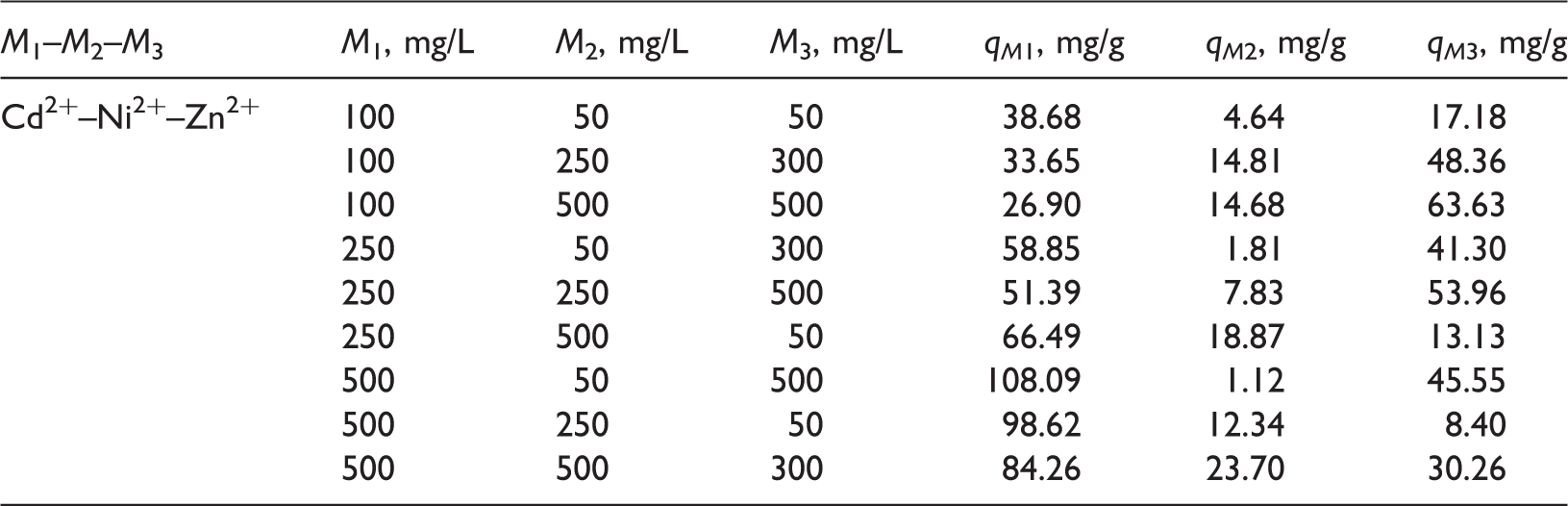
Results for kinetic and equilibrium adsorption studies in mono-metallic solutions are reported in Figures 1–3. Metal adsorption kinetics fitted the pseudo-second order model and the initial adsorption rates ranged from 17.7 to 20.5 mg/g h, while the kinetic constants k varied from 0.00216 to 0.00372 g/mg h. Note that a larger k means a lower adsorption rate (see Table 2). Weber-Morris kinetic analysis showed that the intra-particle diffusion is not the rate limiting stage (Figure 2), which is consistent with the results reported in other studies for heavy metal removal using BC (Mendoza-Castillo et al., 2015). Intraparticle diffusion rates for heavy metal adsorption are reported in Table 2 and they ranged from 0.9 to 11.3 mg/g h0.5. These results suggested that the surface diffusion has a relevant role as the rate-limiting step in the adsorption of metallic ions on BC.
Kinetics for the adsorption of Cd2+, Ni2+, and Zn2+ ions on bone char at 30℃ and pH 5. Weber-Morris analysis for the kinetics of the adsorption of (a) Cd2+, (b) Ni2+, and (c) Zn2+ ions on bone char at 30℃ and pH 5. Isotherms for the adsorption of Cd2+, Ni2+, and Zn2+ ions on bone char at 30℃ and pH 5.

Adsorption isotherms for tested metal ions are given in Figure 3. They can be classed as Langmuir-type isotherm (L2) of Giles classification (Giles et al., 1974), which corresponds to a favorable adsorption process. Results of isotherm data fitting with Langmuir and Sips models are reported in Figure 3 and Table 2. Overall, Sips equation satisfactorily fitted these experimental data but with an overestimation of the monolayer adsorption capacity, see Table 2. The saturation experimental uptakes obtained for this adsorbent ranged from 68.3 to 119.4 mg/g where the removal behavior was: Cd2+ > Zn2+ > Ni2+. These adsorption capacities are higher than those reported for other carbon-based adsorbents used in heavy metal removal (Reynel-Avila et al., 2011). As stated above, previous studies have reported the removal of heavy metal ions with BC, e.g. Ko et al. (2004) and Moreno et al. (2010). Ko et al. (2004) analyzed the adsorption of Cd2+ and Zn2+ ions on the commercial BC Brimac 216 where the monolayer adsorption capacities were 67.8 and 33.7 mg/g, respectively. On the other hand, Moreno et al. (2010) reported the Ni2+ adsorption using a BC obtained from pyrolysis at 800℃ during 5 h where the metal uptake was ∼40 mg/g. These results confirm that the proper selection of the pyrolysis conditions is paramount for obtaining the best adsorbent. In this study, the metal adsorption properties of BC have been improved up to 76%, 71%, and 143% for Cd2+, Ni2+, and Zn2+ ions, respectively. It is also relevant to highlight the outstanding performance of BC for Zn2+ adsorption, which is a difficult water pollutant to be removed.
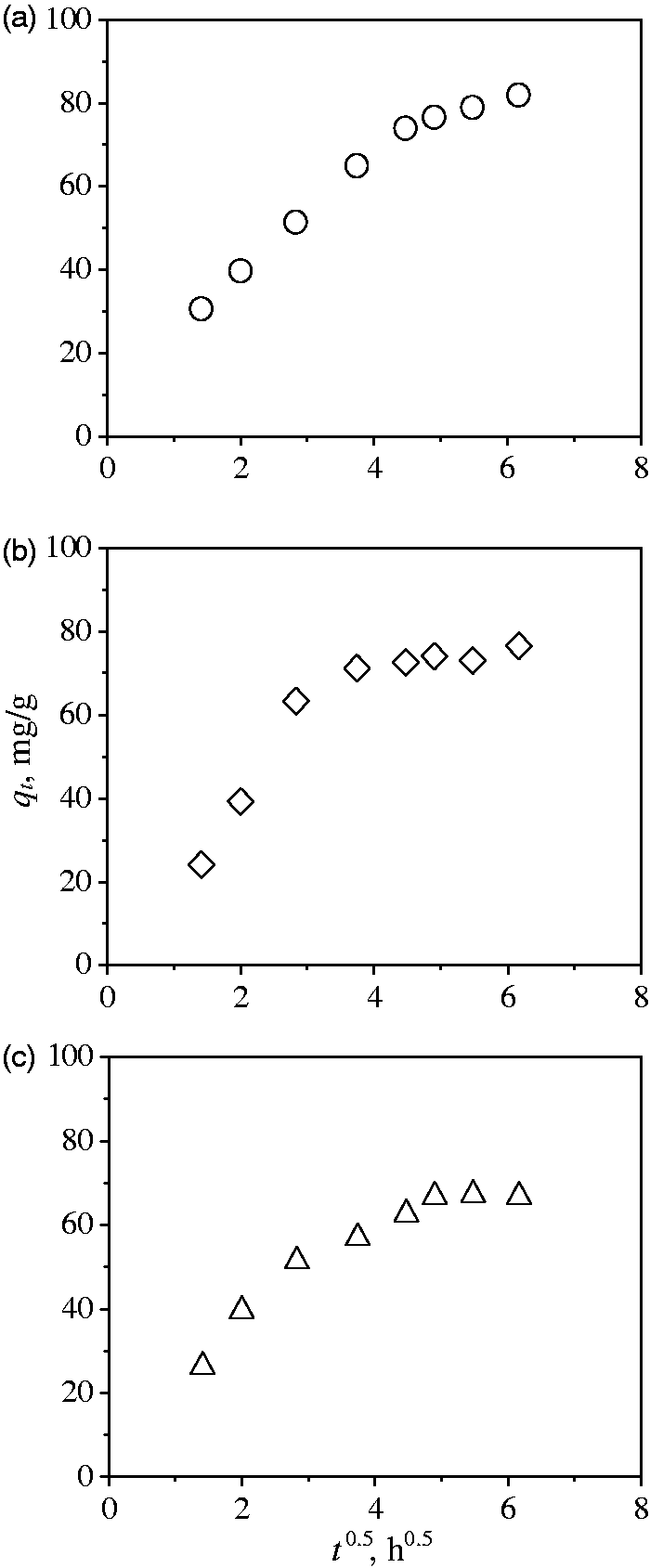
Tables 3 and 4 show the results of multicomponent adsorption of heavy metal ions in both binary and ternary systems using the best BC. These results confirmed the presence of a strong competitive (i.e., antagonistic) adsorption process where the presence of co-ions decreased the metal uptakes. The multicomponent adsorption capacity of BC in binary mixtures for Cd2+ ions varied from 24.7 to 108.4 mg/g, while the uptakes of Ni2+ and Zn2+ ions were 0.4–52.3 mg/g and 7.8–62.4 mg/g, respectively. The impact of co-ion concentration on multicomponent adsorption tests can be determined via the ratio of adsorption capacities Rqmetal = qmetal,mix / qmetal where qmetal,mix is the uptake of BC for metal i in the multicomponent solution and qmetal is the uptake of the same metal using a mono-metallic solution at the same operating conditions (i.e., initial metal concentration, pH and temperature). For binary mixtures, Rq of tested metal ions ranged from 0.03 to 0.94, with Ni2+ showing the lowest values. Note that these ratios decreased with increments of the co-ion concentration due to the competition of metallic ions by the binding sites of the adsorbent surface. Cd2+ and Ni2+ ions have the highest values of electronegativity and the lowest value of hydrated ionic radii, which are cation properties that could favor the selectivity of the ion exchange process involved in the multi-metallic adsorption on BC (Choy et al., 2005). The adsorption capacities in ternary mixtures were 26.9–108.1 mg/g for Cd2+, 1.1–23.7 mg/g for Ni2+, and 8.4–63.6 mg/g for Zn2+ ions, see Table 4. Ni2+ uptake was highly impacted by the presence of both Cd2+ and Zn2+ ions. Values of Rq ranged from 0.09 to 0.95 for the heavy metal ions in the ternary mixture at tested experimental conditions.
Surface chemistry of BC and adsorption mechanism
Figure 4 shows a SEM image and EDX results for the BC sample obtained at 700℃. SEM micrograph displays a rough morphology, non-porous surface without defined geometry, see Figure 4(a). EDX analysis is reported in Figure 4(b) and indicates that the adsorbent is mainly composed of calcium (Ca) and phosphorus (P) where there are also some minor components such as oxygen (O) and carbon (C). Textural parameters of the BC obtained at the best pyrolysis conditions are reported in Figure 5(a). This adsorbent has a Brunauer–Emmett–Teller (BET) surface area of 85 m2/g, a total pore volume of 0.24 cm3/g with a mesopore volume of 0.20 cm3/g, respectively. On the other hand, results of XRD analysis are given in Figure 5(b). XRD patterns showed the characteristic peaks of the known phases of pure hydroxyapatite. The main (h k l) indices reported for the pure hydroxyapatite are (002), (211), (112), (300), (202), (310), (222), (213), and (004), which were identified in the pattern of the BC sample. Note that this result is also consistent with the phases listed in the ICDD database. XRD analysis showed that there were no major changes in the peak position resulting from the metal ion adsorption on BC (i.e., samples labeled as BC-Cd, BC-Ni and BC-Zn in Figure 5b). However, the characteristic reflections of the crystalline phase of the hydroxyapatite partially lost intensity. This result indicated that the hydroxyapatite lost its crystallinity due to the deposition of heavy metal ions on the adsorbent surface. These metallic ions are adsorbed on the hydroxyapatite of BC surface via an ion exchange mechanism with Ca2+ ions leading to the formation of a new structure [Ca10-xMx(PO4)6(OH)2], where x is the adsorbed heavy metal (Choy and McKay, 2005; Mendoza-Castillo et al., 2015).
(a) SEM image and (b) EDX results of bone char sample. Results of (a) textural parameters, (b) XRD, (c) XRF, (d) FTIR, and (e) XPS of the best bone char obtained via pyrolysis. Label BC is used for raw bone char, while BC–Cd, BC–Ni, and BC–Zn correspond to the adsorbent loaded with Cd2+, Ni2+, and Zn2+ ions, respectively.

The elemental composition (wt %) of the best adsorbent is reported in Figure 5(c). XRF analysis confirmed the chemical composition of hydroxyapatite in BC, which is mainly constituted of calcium and phosphorus, i.e., 39 and 19%, respectively. This analysis also showed the presence of Cd2+, Ni2+, and Zn2+ in the samples BC-Cd, BC-Ni, and BC-Zn where the Ca content decreased in these samples. Previous studies have concluded that divalent metal cations have a strong selectivity towards the Ca2+ in the hydroxyapatite lattice via an ion exchange mechanism (Cheung et al., 2000; Rosskopfová et al., 2011; Stötzel et al., 2009).
Figure 5(d) shows the FTIR characteristic absorption bands of the hydroxyapatite and carbonaceous phase of BC at 3430, 2930, 1630, 1460, 1420, 1030, 960, 875, 605, and 565 cm−1 (Rojas-Mayorga et al., 2013, 2015). The stretching vibration of the OH− groups corresponds to the signal at ∼3430 cm−1 (Mondal et al., 2012; Rojas-Mayorga et al., 2013). The asymmetric stretching vibration mode of
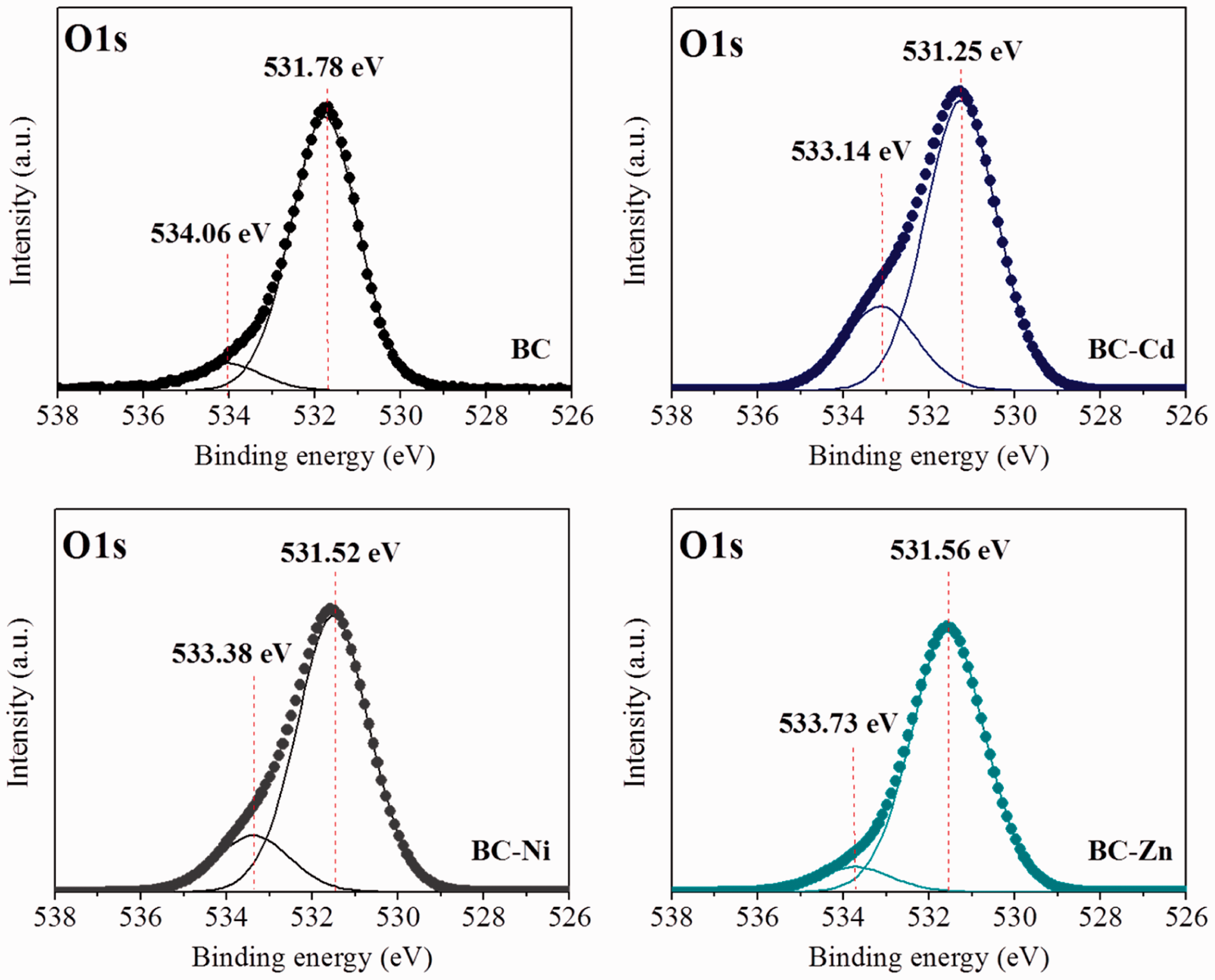
Figure 5(e) shows the XPS survey spectra of BC samples that contain the photoelectron peaks belonging to oxygen (O), calcium (Ca), phosphorus (P), and carbon (C). These peaks correspond to the hydroxyapatite and the amorphous carbonaceous phase from the adsorbent. Note that the peaks of Cd, Ni, and Zn are identified for samples BC-Cd, BC-Ni, and BC-Zn. A detailed analysis of these XPS results was performed. The Ca 2p peak for raw BC has been deconvoluted into two peaks consisting of Ca 2p1/2 peak at 351.4 eV and Ca 2p3/2 peak at 347.9 eV (see Figure 6). These signals indicate the interaction Ca–O. However, the Ca 2p1/2 peak at 351.4 eV corresponds to the presence of Ca-OH and to the calcium linked to carbonate group (Wang et al., 2014). The main Ca 2p3/2 peak located at 347.9 eV can be associated to calcium atoms bonded with a phosphate group (Dupraz et al., 1999). The P 2p spectrum has been deconvoluted into two peaks P 2p1/2 at 134.7 eV and P 2p3/2 at 133.5 eV (see Figure 7). These energies can be associated with P=O and P–O bonds from phosphate group. The oxygen spectra (O 1s) for BC sample showed two peaks at 534.1 and 531.8 eV, see Figure 8. The O 1s signal at 534.1 eV corresponds to –OH groups, while the peak at 531.8 eV could be attributed to Deconvolution of XPS Ca 2p spectra of raw bone char (BC) and metal-loaded bone chars (BC-Cd, BC-Ni and BC-Zn). Deconvolution of XPS P 2p spectra of raw bone char (BC) and metal-loaded bone chars (BC-Cd, BC-Ni and BC-Zn). Deconvolution of XPS O 1s spectra of raw bone char (BC) and metal-loaded bone chars (BC-Cd, BC-Ni and BC-Zn).

After heavy metal adsorption, several changes in XPS spectra of BC were identified in the region of Ca 2p, P 2p, and O 1s, while new contributions were observed for Cd 3d, Ni 2p, and Zn 2p XPS spectra, see Figure 9. The intensity of binding energies of Ca 2p XPS spectra decreased and a chemical shift to lower energy region was observed for samples BC-Ni, BC-Zn, and BC-Cd. This effect is due to the loss of Ca–O bonds and the change in electron density of remaining Ca atoms in the BC structure after the adsorption process. As stated, this phenomenon is caused by the ion exchange of the calcium from the hydroxyapatite structure of BC by the heavy metals in solution. However, the intensity of this ion exchange process depends on the nature of the heavy metal used in the adsorption experiment. It is convenient to remark that the Ca 2p spectrum of sample BC-Cd showed the major change indicating that cadmium removal caused the highest calcium exchange. These findings are consistent with the BC adsorption isotherms, which showed the highest uptakes for cadmium. The new Ca 2p binding energy identified for samples BC-Cd, BC-Ni, and BC-Zn corresponds to Ca–O interactions from hydroxyapatite. These results also confirmed that the affinity of BC was different for the tested metal ions. For illustration, the elemental composition (at %) of different BC samples is reported in Table 5.
Deconvolution of XPS cadmium, nickel, and zinc spectra of metal-loaded bone chars. Atomic ratios (%) obtained from XPS analysis for bone char samples before and after heavy metal adsorption.

XPS spectra of samples BC-Ni and BC-Cd showed the largest change in the P 2p region. These spectra get wider after the removal process, thus revealing the presence of additional contributions to the phosphate group. These new binding energies correspond to P–O and P=O bonds of the phosphate group from hydroxyapatite. These contributions may be associated with the interaction of Cd and Ni with the phosphate group, due to the aforementioned displacement of the calcium atoms from BC structure by these ions. The O 1s energy level for the initial contribution of the BC at 534.1 eV shifted to lower energy values of 533.1, 533.4, and 533.7 eV in the samples BC-Cd, BC-Ni, and BC-Zn, respectively. This is due to the displacement of calcium atoms from hydroxyapatite by heavy metal atoms during adsorption on BC leading to the formation of new metal–OH interactions (Su et al., 2013). The peak at 531.8 eV in sample BC also shifted to lower energy values: 531.2, 531.5, and 531.6 eV for samples BC-Cd, BC-Ni, and BC-Zn, respectively. This change could be caused by the interaction of the phosphate groups with the heavy metal ions.
Figure 9(a) shows the Cd 3d energy level that has been deconvoluted into two Cd 3d5/2 peaks at 407.0 eV and 405.4 eV. Both binding energies are associated with the interactions Cd–O (Haemers et al., 1984) and, according to O 1s and P 2p energy levels, these contributions could be the interaction of Cd ions with hydroxyl and phosphate groups. Ni 2p spectrum has been deconvoluted into four peaks at 865.45, 862.05, 858.29, and 856.29 eV (see Figure 9b). These signals can be associated with the Ni–O interactions such Ni–phosphate, Ni-OH and nickel oxides with different electronic density (Nesbitt et al., 2000). The Zn 2p XPS spectra showed a single signal at 1022.6 eV that corresponds to the Zn–O bond, which is formed between the oxygen from hydroxyapatite surface and the adsorbed Zn2+ ions (see Figure 9c). This interaction can correspond to Zn-OH (Haemers et al., 1984).
These characterization results confirmed that the adsorption of heavy metals on BC occurred via an ion exchange process between the structural calcium from hydroxyapatite and the heavy metal ions in solution (Mendoza-Castillo et al., 2015)

Conclusions
A tailored BC for heavy metal adsorption from aqueous solution has been obtained via an optimized pyrolysis process. Results showed that a pyrolysis temperature >700℃ has a significant impact on metal uptakes of this adsorbent due to the dehydroxylation of hydroxyapatite and the decomposition of carbonates from BC. The best synthesis route improved the heavy metal adsorption properties of BC up to 143% with respect to the results reported in literature for other commercial adsorbents. BC showed an outstanding performance for the adsorption of Zn2+ ions, which is a relevant and persistent water pollutant. However, the multicomponent removal of Zn2+, Cd2+, and Ni2+ ions in both binary and ternary mixtures is a strong antagonistic adsorption process. XPS characterization analysis confirmed that the main mechanism involved in heavy metal adsorption with BC is an ion exchange process where metal–oxygen interactions from hydroxyapatite could play a relevant role. In summary, this study reports new insights to improve the surface chemistry of BC for water treatment and purification.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.