Abstract
The ability of the cleaning agents such as cyclohexane, acetone, isopropanol and hydrogen peroxide to remove oil from technical steel and titanium samples was studied in systematic experiments. The composition of the organic residues and the layer thickness on the differently cleaned surfaces were determined via X-ray photoelectron spectroscopy analysis, and the corresponding water contact angles were measured. The ability of the cleaning agents to remove oil was found to increase in the following order: acetone, isopropanol, cyclohexane and hydrogen peroxide. The relationship between the chemical composition of the residues and the observed contact angles is investigated, and a quantitative correlation is developed.
Keywords
Introduction
The wetting and the cleanliness of steel and titanium surfaces play an important role in various applications. For example, the cleanliness is crucial for medical implant surfaces (Keller et al., 1994; Zhao et al., 2008), and the wetting of surfaces has a strong influence on heat and mass transfer (Antonio Rocha et al., 1996; Lyklema, 1995; Wen and Wang, 2002).
Many applications require a “clean” surface. The demands on cleanliness, however, vary for different applications. A distinction is drawn between atomically clean surfaces and technically clean surfaces (Mittal, 1979). Atomically clean metal surfaces exist under special conditions only, e.g. after plasma cleaning and storage in ultra-high vacuum. The focus of the present work is on technical surfaces.
Different techniques for cleaning surfaces and monitoring the cleanliness have been examined. Surface analytical techniques yield detailed information on properties of surfaces. Since the advent of surface analysis techniques such as Auger electron spectroscopy or X-ray photoelectron spectroscopy (XPS) (Briggs and Grant, 2003) in the late 1960s, much insight has been gained into the nature and sources of surface contamination (Castle, 2008; Olivas et al., 1999; Tlili et al., 2012). Studies of carefully prepared metal surfaces yielded, e.g. information on adsorbed water and contamination by polar and non-polar organic matter and salts (Castle, 2008; Eirich, 1968). Such studies improved the general understanding of the chemistry and physics of surfaces.
A comparatively simple method for monitoring the cleanliness makes use of the wettability of a surface (So et al., 1999; Williams and O’Bryon, 2013), i.e. the contact angle is measured. Often, water is used as the test fluid (Schrader, 1979). The intrinsic contact angle θY establishes in the three-phase equilibrium on an ideal, i.e. plane and homogeneous surface. It is related to the interfacial free energies by the Young (1805) equation
The factor r is the ratio of the actual and the projected surface area. It characterizes the roughness of the substrate. From equation (2), the measured contact angle θ can be related to the intrinsic contact angle θY and to the interfacial tensions.
The interfacial tensions can be related to the intermolecular interactions. A simple approach is based on the concept of separating a body of medium i in vacuum into two parts (Israelachvili, 2011). For the separation to infinite distance, the intermolecular forces have to be overcome. The work per unit surface area spent on the separation is called the work of cohesion.
Fox and Zisman (1950) distinguish between solids with high- and low-energy surfaces according to the corresponding work of cohesion. The high-energy surfaces are found for solids with very low vapor pressures and strong molecular bonds, like metals. They have interfacial free energies of several thousand mJ/m2. On the other hand, the low-energy surfaces are found for solids with higher vapor pressures that are bound by, e.g. van der Waals forces, like many polymers or organic compounds (De Gennes, 1985; Fox and Zisman, 1950). These solids have usually interfacial free energies of up to a few hundred mJ/m2.
Using this classification, solids with a high-surface free energy like metals or metal oxides tend to be completely wetted by water whereas samples with a low-surface free energy tend to be non-wetting (De Gennes, 1985; Fox and Zisman, 1950). For example, Harkins and Feldman (1922) as well as Fox et al. (1955) state that most pure liquids spread on high-energy surfaces; Bernett and Zisman (1970) as well as Schrader (1970) show that water spreads spontaneously on a pure, clean gold surface. On the other hand, there is experimental evidence of liquids not spreading on metal surfaces (Erb, 1965). The reason for this is that the surface free energy of metals depends strongly on the sample pretreatment, as it has been shown for stainless steel (Boulange-Petermann, 1993; Mantel and Wightman, 1994). Even slight contamination can convert a high-energy surface into a low-energy surface (Bernett and Zisman, 1968; Schrader, 1968) so that the liquid does not spread spontaneously anymore. Gledhill et al. (1977) confirmed the decrease of surface energy of steel as a function of the relative humidity of the surrounding air.
The contamination can result from different effects. Adsorption is always present and can range from weak physisorption to strong chemisorption. Metal surfaces may be covered by oxygen in form of a monolayer of chemisorbed molecules, but thick surface oxide layers are also observed. Furthermore, water adsorption is ubiquitous. Also organic contaminants play an important role. Traces of organic contaminants can prevent water from spreading on a metal surface (Erb, 1965; Fox et al., 1955).
Transition metal oxides are very sensitive to organic contamination and show individual contamination kinetics (White, 1970). Matsunaga (1979) studied the contamination rate of six metal oxides and found a relation between the contamination rate and the solid–water interaction energy. TiO2 had the highest contamination rate of all examined metal oxides.
As the wetting behavior of surfaces is closely related to its cleanliness, the aim of the present study is twofold. First, we are interested in studying the cleaning of technical steel and titanium surfaces from oil with common solvents. A better understanding of the nature and the amount of contaminations remaining at the surface after typical cleaning procedures is gained. XPS analysis is used for this purpose. Furthermore, the relationship between the water contact angle and the amount and nature of the contaminations is of interest. The question is addressed whether it is possible to measure the amount of the remaining contaminations after typical cleaning procedures simply by water contact angle measurements instead of the more extensive XPS analysis.
Experimental
Materials
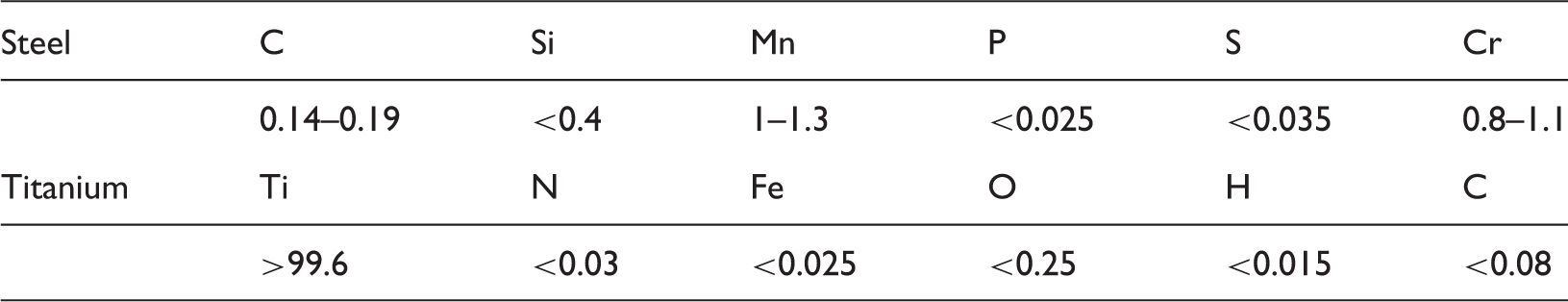
Chemical composition of the steel (material number 1.7131) and titanium (material number 3.7035) bulk materials in atom Ma%.
The oil used in this study was a fully synthetic poly-alpha-olefin from Evonik Industries according to SAE J300 Class 0 W 20. No friction and wear modifying additives were blended. The four cleaning agents are cyclohexane (purity better than 99.5%, obtained from Sigma Aldrich), acetone (purity better than 99.8%, obtained from Sigma Aldrich), isopropanol (purity better than 99.5%, obtained from J. T. Baker) and hydrogen peroxide (purity better than 30%, stabilized in water, obtained from Th. Geyer GmbH).
The cleaning agents can be divided into two groups according to their cleaning mechanism: cyclohexane, acetone and isopropanol are pure non-reactive solvents that remove the oil via dilution in the solvent. The cleaning mechanism of hydrogen peroxide is presumably more complex. Hydrogen peroxide is catalytically decomposed at the steel and titanium surfaces and forms hydroxyl radicals (Lin and Gurol, 1998; Tengvall et al., 1989). Due to the excess of hydrogen peroxide in the aqueous solution, the free radicals are able to initiate a chain reaction in form of a hydroxylation of the saturated hydrocarbons of the oil (Clerici, 1991; Greiner, 1970). The polar hydroxyl groups increase the solubility of the hydrocarbons in water. They are presumed to be subsequently dissolved in the aqueous medium.
Sample pretreatment
The treatment for both material types, i.e. steel and titanium was identical. The samples were divided into two groups. The samples of one group were put into an oil bath for 5 min. Gross oil residues were removed with lint-free laboratory tissues. These samples are called “oil samples,” in the following. The second group of samples was not exposed to oil, called “no-oil samples,” in the following.
Both the oil and the no-oil samples were identically cleaned in three steps: Each sample was put in a clean Erlenmeyer flask filled with a cleaning agent and put in an ultrasonic bath at room temperature. After 10 min, the samples were taken out of the flask and rinsed with the corresponding fresh cleaning agent. Then, the metal disks were vertically put on a draining board for drying. When employing hydrogen peroxide as the cleaning agent, the samples were rinsed with fresh hydrogen peroxide and subsequently with highly pure, double-distilled water. Then, they were put on a heating plate. This was done because the drying of hydrogen peroxide was too slow. After no longer than 2 min, the samples looked dry to the naked eye. The entire procedure was carried out three times. In the following, the samples are designated according to their surface treatment: AAA, CCC, III and PPP correspond to the cleaning in three steps with acetone, cyclohexane, isopropanol and hydrogen peroxide, respectively.
The samples’ surfaces were analyzed immediately after the cleaning procedures to avoid the growing of new adsorption films. The contact angle measurement was carried out typically a few hours after the XPS analysis. For storage and transport, the samples were wrapped in aluminum foil.
Surface analysis
X-ray photoelectron spectroscopy
The chemical analysis was done by XPS (Al Kα1486.6 eV) under ultra-high vacuum conditions with an Axis Nova (Kratos Analytical). The working pressure was below 10−8 mbar in the analyzing chamber. Therefore, adsorption films of volatile materials like water from environmental humidity are not present.
The analysis was carried out for the elements carbon (C), oxygen (O), iron (Fe) and titanium (Ti) in their different binding states. Trace contaminations of sodium, calcium, potassium and nitrogen were found at less than 1–2 at% and are not further considered here.
Hydrogen is not directly detectable by XPS. Adsorbed hydrocarbon films are identified via the XPS signal of the photoelectrons from the carbon 1 s (C1s) orbital. Different binding states of carbon atoms are identified via the chemical shift of the C1s photoelectron line. Thus, carbon bound in non-polar aliphatic or aromatic hydrocarbons (in the following called “non-polar carbon”) at an electron binding energy of EB = 285 eV is distinguished from carbon bound in polar groups (in the following called “polar carbon”), like hydroxyl, carbonyl and carboxyl groups with one (EB = 286.4–286.6 eV) or two oxygen atoms (EB = 286.6–289.2 eV) as binding partners (Mantel and Wightman, 1994), respectively (cf. Figure 1, left).
XPS detail spectra of the C1s (left) and O1s (right) photoelectron signals from a steel sample after oil exposition and III cleaning. Different binding states of the C and O atoms can be distinguished. Oxygen is found in an organic and an oxidic binding environment. The three peaks on the left in the C1s spectrum are lumped together for the quantification and are designated as polar carbon. The peak on the right in the C1s spectrum is designated as non-polar carbon.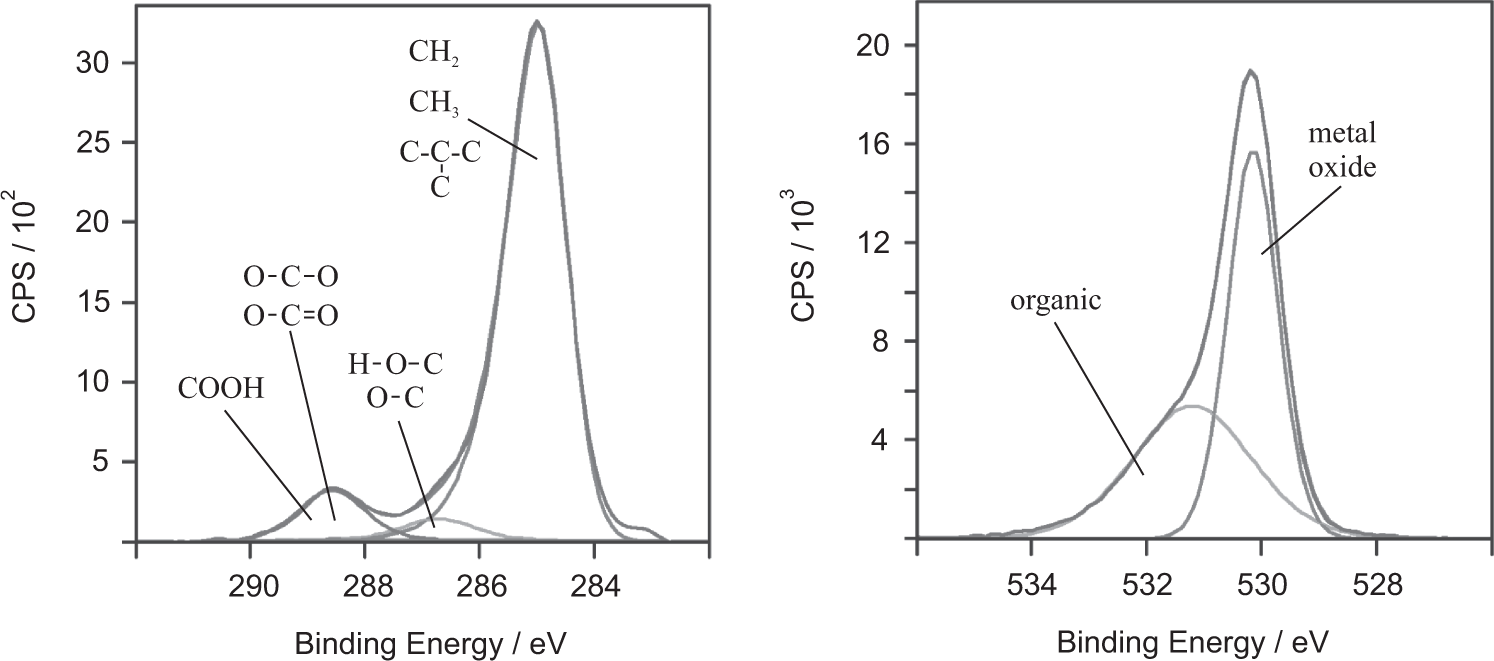
Oxygen is detected from the XPS signal of the photoelectrons from the 1 s (O1s) orbital. By the chemical shift of the O1s photoelectron line, oxygen bound in metal oxide (EB = 529.9–530.1 eV) is distinguished from oxygen bound in organic molecules (EB = 532.9–533.9 eV) (cf. Figure 1, right). The photoelectron intensities were determined as peak area of the photoelectron peaks using a Shirley background.
The information which is obtained by the XPS measurements reveals a two-layer structure of the chemical compounds on the surface. There is a metal-oxide layer and an organic layer that is located on top of the oxide. Both layers were analyzed.
Analysis method
For brevity, the atom number fractions obtained by the XPS analysis are called concentrations in the following. The apparent XPS concentrations obtained from the measured intensities (the set of atomic sensitivity factors supplied by the instruments manufacturer was used, see Table S.1 in the Supporting Information) have to be converted into the concentrations in the two surface layers, i.e. the metal-oxide and the organic layer. The ratio of Fe:O in the metal-oxide layer on the steel samples was found to be 1:1.4, and the ratio of Ti:O in the metal-oxide layer on the titanium samples was found to be 1:1.9. The spectra of the metal reveal predominantly oxidic metal and the pure metallic contributions are negligible in any case.
For the organic layer, the concentrations of organic oxygen
Contact angle measurement
Water contact angles were measured with a drop shape analysis system (Krüss DSA100). It consists of a light source and a camera that records the photographs of the sessile drop. The environmental chamber was equipped with two heat exchangers for temperature control. The chamber was filled with nitrogen that was presaturated with water at 20℃. After a steady state was reached, the drop was disposed. The test fluid for the contact angle measurement was double-distilled water. The measurements were carried out at 20℃. The images were processed with the instrument’s software. A tangent fitting method was used to determine the contact angle of the sessile drop. In preliminary work, the reproducibility was tested by measurements of the water contact angle on three samples that were prepared identically. This was done for different cleaning methods on no-oil as well as on oil samples. The reproducibility of the contact angle measurements was found to be of the order of ±5° which is taken as the error in the contact angle measurements in the present study.
Surface roughness
As the contact angle on a rough surface is related to the intrinsic contact angle via the roughness parameter according to equation (2), surface topography maps for the steel and titanium samples were determined. The surface topography maps obtained by white light interferometry are shown in Figure 2. From the results, different measures of the roughness were determined. The root mean squared roughness of the samples was Surface topography map of the flat finished sample surfaces (left: steel, right: titanium) obtained by white light interferometry.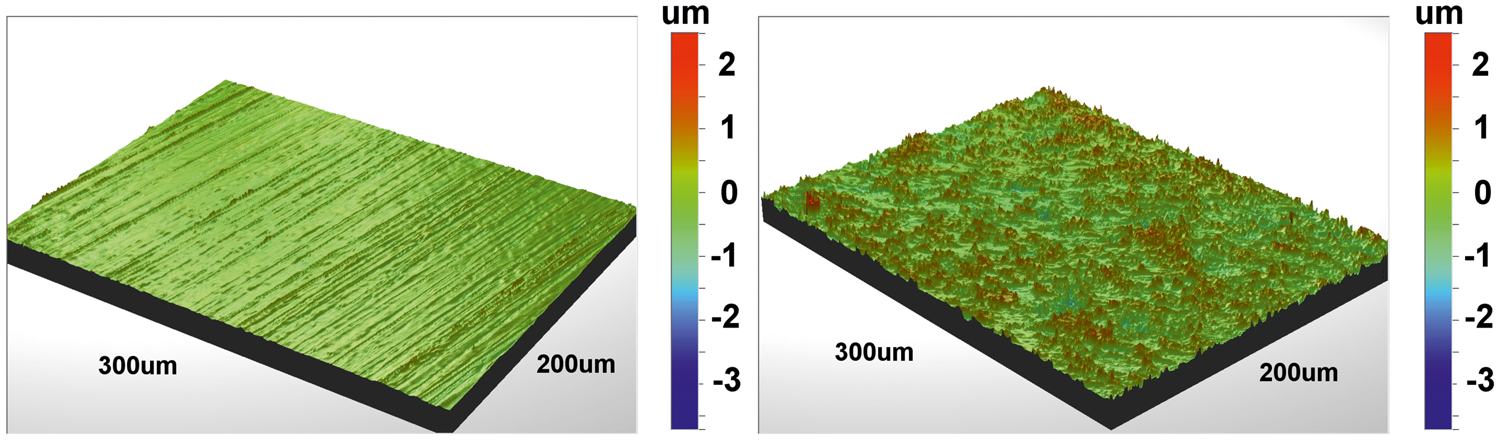
Experimental results
Thickness and chemical composition of the organic layer
Water contact angle, layer thickness and layer concentrations of oxygen and carbon, respectively, in its different binding states, measured on the differently prepared metal samples.

Oxygen located in the organic layer is distinguished from oxygen bound as metal oxide, and carbon bound in non-polar binding states is distinguished from carbon in polar binding states.
AAA: acetone; CCC: cyclohexane; III: isopropanole; PPP: hydrogen peroxide.
As expected, the results reveal that, both for the steel and the titanium samples, none of the cleaning treatments was able to provide an atomically clean oxide surface, the thickness of the organic layer is between 1.4 nm and 14.3 nm.
No-oil samples
For the no-oil steel samples, the organic layer thickness δ increases in the order PPP, III, AAA and CCC. The organic layer concentration of oxygen is related to the portion of oxygen atoms per molecule of the cleaning agent: PPP produces the highest oxygen concentration. While the concentration of oxygen in the organic layer is the lowest for CCC, no significant difference is found between III and AAA. For the latter two cases, the values of the oxygen concentration are between the ones of PPP and CCC. As the composition of the organic layer correlates strongly with the cleaning agent used, it can be concluded that in case of the steel samples a significant portion of the organic layer consists of the cleaning agent itself.
In contrast, no such correlation is found for the no-oil titanium samples. The reason for this finding could be that a chemical reaction between the titanium oxide surface and the solvents takes place which influences the concentrations of oxygen in the organic layer. The thinnest layer is again found for the PPP treatment. After that treatment, the layer is significantly thinner than on the no-oil steel samples. The other agents yield a similar thickness of the organic layer. The numbers are in the same range as observed for the steel samples.
Oil samples
For both steel and titanium, the thickness of the organic layer δ and the concentration of non-polar carbon found on the oil samples are significantly higher as compared to the no-oil samples. This is ascribed to residues of oil remaining on the samples. The values of δ increase in the order PPP, CCC, III and AAA for both steel and titanium. Correspondingly, an increase of the non-polar carbon concentration is observed. These findings are attributed to the chemical nature of the cleaning agents. Cyclohexane is non-polar which makes it most appropriate for the cleaning of oil, out of the three solvents. Due to the different cleaning mechanism of hydrogen peroxide, it is most efficient in removing the oil from the surface even though it is a polar substance.
Overview
Overall, Table 2 shows that the oil samples have distinctly larger concentrations of non-polar carbon than the no-oil samples, as expected. The samples with the highest concentration of non-polar carbon tend also to have the highest thickness of the organic layer. The steel samples tend to have slightly lower concentrations of polar carbon than the titanium samples.
Water contact angle
The measured water contact angles are also included in Table 2. Depending on the pretreatment, they vary from 38° to 103° for the titanium samples and from 46° to 95° for the steel samples. On average, the values observed for the steel and the titanium samples contaminated by oil are 35% above those of the no-oil samples both for the steel and the titanium samples.
Discussion
From the water contact angles θ that were measured in the present study, the intrinsic contact angles θY are determined via equation (2). The experimental data (cf. Table 2) show that there is a correlation between the composition of the sample surfaces as measured by XPS and the water contact angle. A simple empirical approach is used to describe this

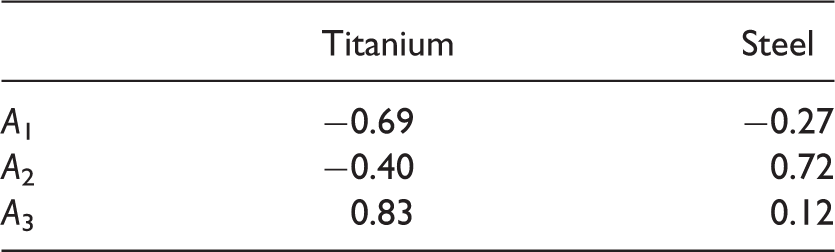
In equation (3), A1, A2 and A3 are the parameters which are fitted to the data. Using the terms, Parity plots showing the contact angles obtained by the model according to equations (2) and (3) with the model parameters according to Table 3 versus the measured contact angles. The open symbols represent the no-oil samples, the filled symbols oil samples. Numbers for A1, A2 and A3 in equation (3) obtained from fitting the present experimental data.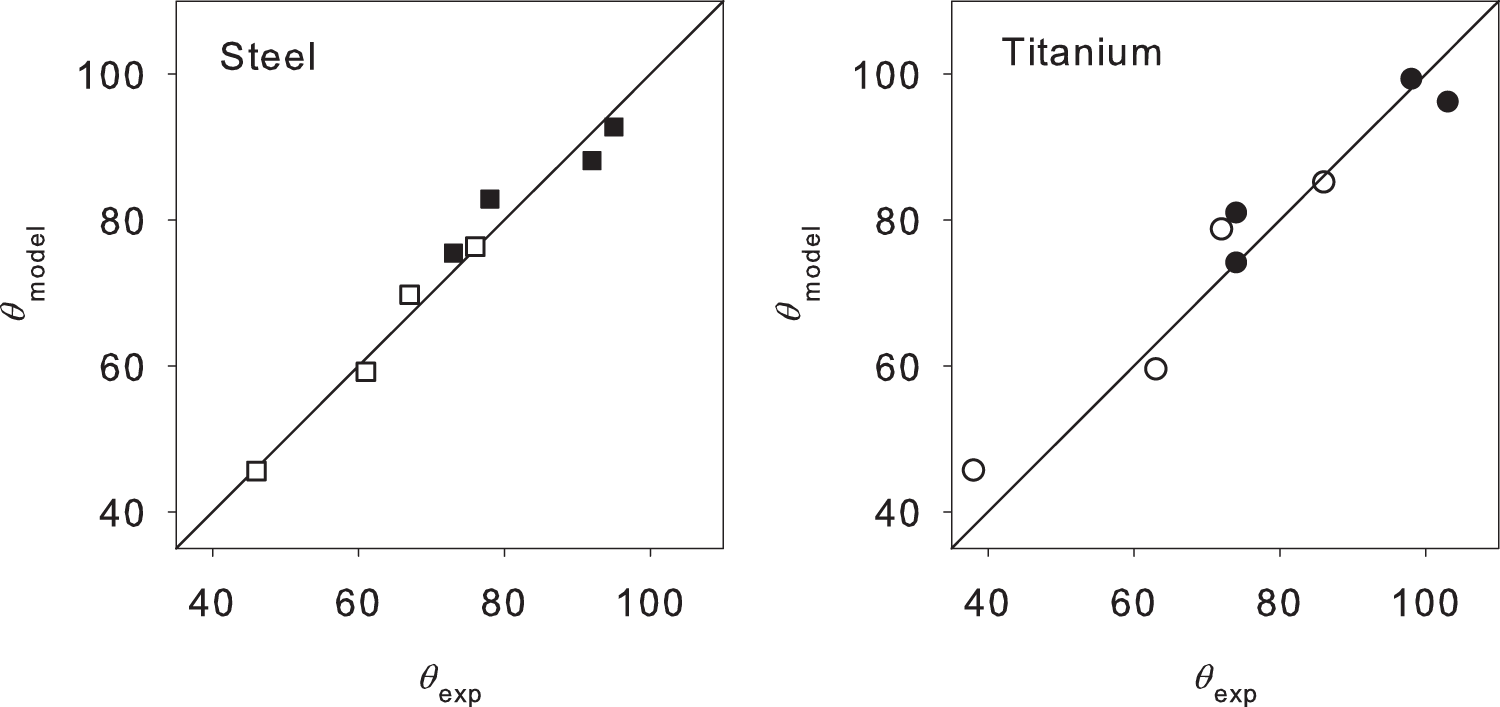
As can be seen from Table 2, the contact angle also correlates with the thickness of the organic layer. The contact angle rises as the thickness increases. For a given thickness of the organic layer, it can be observed that the contact angles on the titanium surfaces are usually higher than on the steel samples.
A correlation describing this dependence in an implicit way is presented in the Appendix. It is based on the Frumkin–Derjaguin approach (Churaev and Sobolev, 1995; Frumkin, 1938) using an effective interface potential. The four adjustable parameters of the correlation were fitted to the experimental data of the present work which results in a good description of the experimental data obtained for a broad variety of conditions, i.e. steel and titanium samples of the oil and the no-oil type, cleaned with the different solvents. As can be seen from Figure 4, the correlations of the water contact angle for the steel and the titanium samples increase with increasing thickness of the organic layer and converge to a value of slightly below 100° which is the same both for the steel and the titanium samples. The differences in the substrate mainly show up in the results obtained for low numbers of the thickness of the organic layer where the contact angles on the steel samples are below those on the titanium samples.
Water contact angles on steel and titanium samples over the thickness of the organic layer. The symbols designate experimental results: □, steel no-oil; ▪, steel oil; ^, titanium no-oil; • titanium oil. The lines represent the results of the model introduced in Appendix. The full line shows the results of the two-layer model in the case of the steel samples, the dash-dotted line in the case of the titanium samples.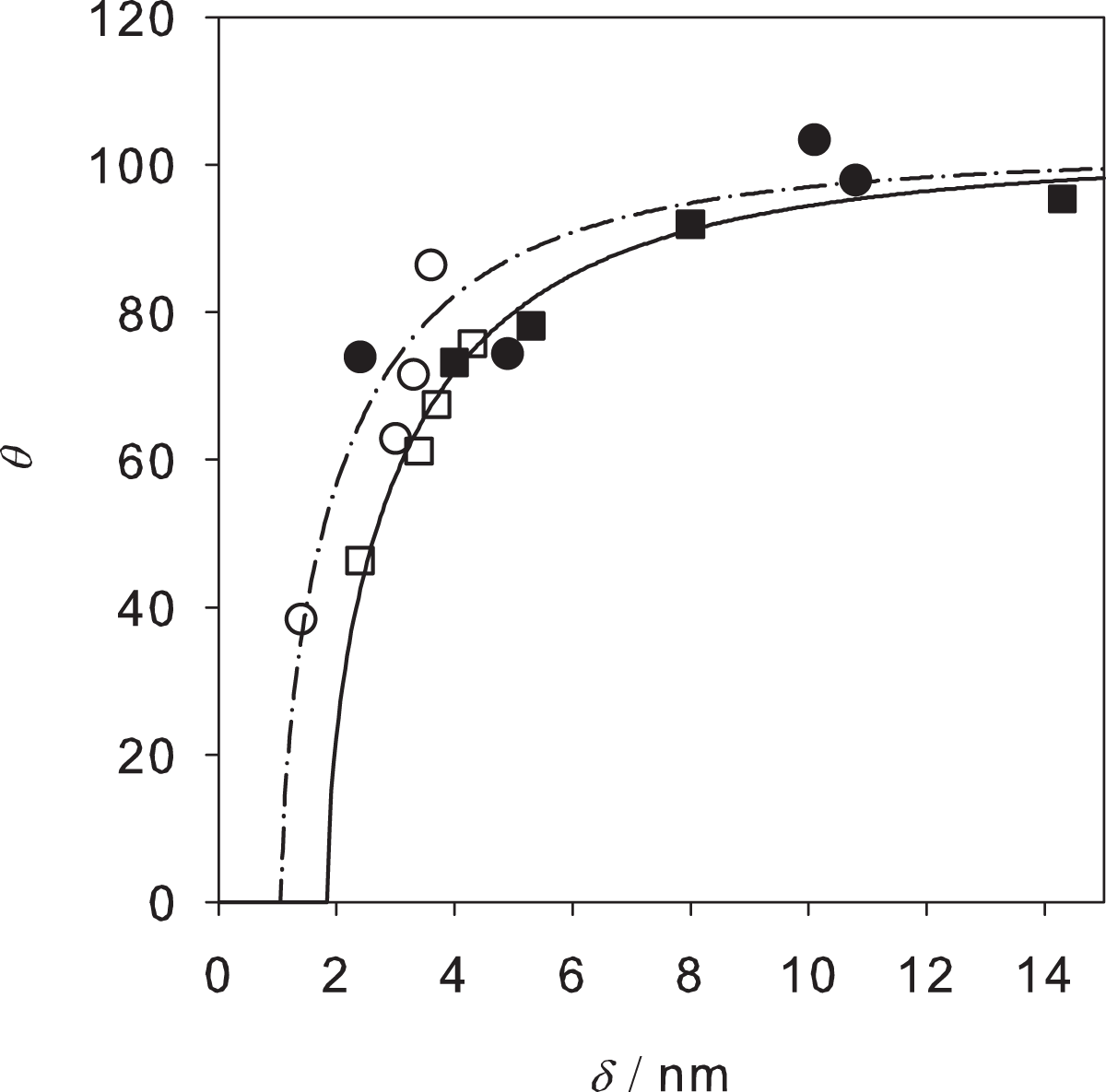
Figure 4 shows that the sensitivity of the contact angle is high for thin organic layers and decays as the thickness of the organic layer increases. In the present study, many of the samples have a thickness of the organic layer in the range between 2 and 6 nm where the sensitivity of the contact angle is high. This explains the rather high uncertainties in the contact angle measurements as a consequence of the variation in the surface properties. Even identically prepared samples may slightly vary in their surface properties, which can lead to the observed variation of the measured contact angle.
Two correlations were presented in this study. One describes the contact angle as a function of the composition of the surfaces. The other describes the dependence of the contact angle on the thickness of the organic layer. The existence of these two correlations shows that there is also a correlation between the thickness of the organic layer and its composition. That third correlation is not discussed here in detail, but it is implicitly given by the two correlations presented above.
Only the correlation layer thickness ↔ contact angle is bijective, whereas the correlation composition → contact angle is only surjective. Therefore, from the contact angle measurements estimates regarding the layer thickness are possible based on the present results, but the composition cannot be determined unambiguously based on water contact angle measurements alone.
Conclusion
The wetting behavior of technical steel and titanium surfaces is strongly influenced by their preceding treatment. Cleaning of technical surfaces was carried out here by means of four different cleaning agents (cyclohexane, isopropanol, acetone and hydrogen peroxide). Their ability to remove oil contamination was assessed. The surfaces obtained after the cleaning were characterized by XPS and contact angle measurements. Organic films adsorbed at the metal oxide surfaces are 1.4 nm to 14.3 nm thick and vary in their composition.
Some of the contamination stems from the cleaning agent itself. Exposure to oil always causes a larger thickness of the organic layer, irrespective of the cleaning treatments. The chemically reactive hydrogen peroxide performed best in removing organic substances from the surface whereas cyclohexane, isopropanol and acetone were less efficient.
0Water contact angles are often used for assessing the contamination of surfaces (Mittal, 1979; Schrader, 1979; So et al., 1999; Williams and O’Bryon, 2013). The present study investigates the background of this. It is shown that the water contact angle can be described as a function of the composition of the organic layer on the surface or, alternatively, as a function of the thickness of that layer. Both correlations work as also the thickness and the composition of the layer are related. From the water contact angle measurements, information on the layer thickness can be determined for the systems studied here. However, the XPS compositions cannot be retrieved from contact angle data alone. Our results show that in-depth analysis of the result of cleaning of technical surfaces is possible and yields results which give insight in the cleaning process and the properties of the resulting surfaces. The study is a step on the long way to establish physical insight in the cleaning of technical surfaces.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge financial support of the present study by Deutsche Forschungsgemeinschaft DFG, Collaborative Research Center CRC 926 “Morphology of component surfaces (MICOS)” and thank Jürgen Berje for fruitful discussions.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


