Abstract
An influence of various organic compounds on aggregation of asphaltenes separated from Tartar oil was studied by small-angle X-ray scattering and scanning tunneling microscopy methods. The compounds such as cyclohexane, methanol, formic acid, heptane, tetrabutylammonium fluoride, acetonitrile, decane, hexanol-1, furfural, and octene-1, added to the solution of asphaltenes in toluene, were shown to produce a considerable aggregation of asphaltene particles of ∼10–13 nm size and of an oblong ellipsoid shape along the longer axis. Decalin, dimethylformamide, dimethylsulfoxide, nitrobenzene, aniline, cyclohexanol, monoethanolamine, triethanolamine, 1-methylnaphtalene, and dichloroethane cause asphaltene aggregate sizes to decrease essentially. Acetone, glycerin, ethyl acetate, dioxane, benzyl alcohol, oleic acid, limonene, α-pinene, cycloheptatrien, and nitromethane were shown to influence weakly on aggregation/disaggregation processes of asphaltenes. Conceivable reasons of the observed effects are discussed from general physico-chemical properties of the compounds used.
Introduction
Heavy oils and oil fractions are characterized by a high content of asphaltenes, high-molecular compounds soluble in aromatic hydrocarbons (toluene) and insoluble in n-alkanes (heptane). Asphaltenes represent complex mixtures of polycyclic molecules whose carbon skeleton includes naphtene, aromatic, and heteroaromatic cycles with aliphatic substituents along with sulfur, nitrogen, and transition metals, such as Ni, V, etc. (Chilingarian and Yen, 1994; Mullins, 2007). Asphaltene molecules tend to the aggregation to form large aggregates resulting in depositions. This makes mining, transporting, and processing of heavy oils difficult.
Studies of asphaltene aggregation become more topical by virtue of increasing heavy oil mining. A typical concentration of asphaltenes in heavy oils of various origins generally lies between 1% and 15% (Chilingarian and Yen, 1994; Mullins, 2007; Simanzhenkov et al., 2003). The aggregation of asphaltene molecules is observed at these concentrations and oil can be thought of as a colloidal solution of asphaltene nanoaggregates (from units to tens of nanometers) in lighter hydrocarbon fractions (Espinat et al., 2004; Lesueur, 2009). The stability of the solution depends not only on the temperature and the pressure (Fahim et al., 2001; Verdier et al., 2006) but on oil chemical composition as well.
A determination of sizes and shapes of asphaltene-containing aggregates is complicated by a great variety of their chemical composition and polydispersity, resulting in ambiguous interpretation of experimental data. Typical sizes of asphaltene aggregates and their shape were determined by small-angle X-ray scattering (SAXS) and small angle neutron scattering (SANS) methods in (Barre et al., 2008; Eyssautier et al., 2011, 2012; Fenistein and Barre, 2001; Gawrys and Kilpatrick, 2005; Gawrys et al., 2006; Herzog et al., 1988; Mullins, 2007; Spiecker et al., 2003; Tanaka et al., 2004; Tuzikov et al., 2011; Xu et al., 1995). Asphaltene-containing particles of 2–50 nm in sizes were observed depending on the type of the samples studied, asphaltene aggregates being rod (Gawrys and Kilpatrick, 2005; Gawrys et al., 2006) or disc (Barre et al., 2008; Eyssautier et al., 2011; Xu et al., 1995) shaped.
Sizes, shape, and aggregative stability of asphaltenes may considerably vary with the raw oil composition. These processes may take place in the simple mixing of various oils as well as in addition of a variety of compounds required to dissolve asphaltene deposits during the oil production. The addition of aliphatic hydrocarbons into an asphaltene-containing solution gives rise to a further aggregation of asphaltenes since those are insoluble in aliphatic hydrocarbons (ASTM, 2012; Chilingarian and Yen, 1994; Gabrienko et al., 2014a, 2014b; Mullins, 2007; Simanzhenkov et al., 2003). Aromatic hydrocarbons capable of solving asphaltenes may create disaggregation of large asphaltene aggregates (Chilingarian and Yen, 1994; Mullins, 2007; Simanzhenkov et al., 2003). The role of other types of organic compounds giving rise to aggregation or disaggregation of asphaltenes is unfortunately one-sided studied. There are literature data on the feasibility of certain compounds primarily. An example is the information on the effective usage of organic chlorine and sulfur compounds to dissolve asphaltene sediments (Mullins, 2007; Mullins and Sheu, 1999; Sharp, 1983; Varadaraj and Brons, 2009). Nevertheless, the compounds have found no wide use in the oil production because of their high toxicity and incompatibility with further oil processing. There are also data on the application of alcohols, esters, amines, olefins, and other compounds as a part of solvents to dissolve asphaltene deposits in pipelines (Al-Yazdi, 1992; Aymes et al., 2012; Becker and Wolf, 1996; Castellano, 2008; Elochukwu et al., 2014; Li et al., 2010). Almost each oil deposit has the own empirical set of solvents. However, little is known about the changes in the solution after dissolving asphaltene deposits. For the most part, there are data on variations in viscosity, solidification temperature, and other macroscopic parameters.
An aim of this work is data acquisition on the structure and dimensional features of asphaltene particles in solutions containing an essential number of various organic compounds to influence on the aggregation. We report the study of the asphaltene aggregation by 30 organic compounds of various classes but commonly available.
Experimental section
Asphaltenes extracted from a sample of heavy oil produced in Tartar Republic (Trukhan et al., 2014) by a standard procedure (ASTM, 2012) were studied. The real density of the separated asphaltenes measured by a helium pycnometer was 1.178 g/cm3. The asphaltenes were solved in toluene and then various organic compounds were added to the solution. The maximum solubility of these asphaltenes in toluene is about 20 weight %. In all solutions, the asphaltene and the additive concentrations were 5.0 and 20.0 weight %, respectively. All experiments described below were performed at 22℃.
SAXS was registered by a S3-MICRO diffractometer (Hecus, Austria) with a Cu anode (λCuKα = 1.541 Å) and the point collimation system. SAXS patterns were registered over the 0.01 < h < 0.6 Å−1 range where h is the wave vector determined as h = 4 π* sin(θ)/λ, 2θ is the scattering angle, and λ is the wave length of the applied radiation with using 1.5 mm quartz capillaries. SAXS patterns of initial solvents were subtracted from the corresponding SAXS patterns of asphaltene solutions taking into account X-ray absorption coefficients. The radius of gyration of the particles (RG) was determined by the slope of the small angle curve in coordinates (ln(I(h); h2), and the radius of gyration of the cross section of the particles (RG(cross)) was determined by the slope of the small angle curve in coordinates (ln(I(h)*h); h2) in ranges h*RG < 1.3 (Feigin and Svergun, 1987). The ATSAS program package was used for data processing (Konarev et al., 2006; Svergun, 1992). The fitting of the SAXS data was performed via ellipsoid model using the following mathematical expression:
Scanning tunneling microscopy (STM) experiments were performed on an UHV variable temperature STM VT-7000 (RHK Technology, USA). The cut Pt/Ir tips were used for STM measurements. The quality of the tips was tested periodically by the atomic resolution calibration experiments on HOPG and on Si(111)-(7 × 7). Scanning step better than 0.08 nm was used. To prepare the samples for STM study, the solution of asphaltenes in toluene (<0.1–0.2 weight %) was dropped on a copper foil, pre-annealed and treated with acid etching, with a micropipette. After evaporation of toluene resulted in a thin film on the foil the sample surface was suitable for the STM study. The usage of the higher asphaltene concentrations in the solution gave no way of preparing the sufficiently thin film and deterring unambiguously sizes and the shape of individual asphaltene aggregates. In the course of the STM research, the topography and current images were recorded simultaneously (Nartova et al., 2005). The STM images were analyzed with WSxM 5.0 Nanotech (Horcas et al., 2007) and XPM Pro V2.0.1.6 (RHK Technology) software's. The determination of size characteristics of the particles has been done manually using reconstruction of the profile section for each particle with the following set of statistically significant number of measurements. In case of any ordering of the particles observed on the surface, the results were confirmed using Fourier transformation analysis.
Results and discussion
At the first stage of the SAXS research, the shape and sizes of asphaltene-containing particles dissolved in toluene were determined to get reliable reference for the further analysis of evolution peculiarities of morphology and dimensional features of asphaltenes and their aggregates under various conditions. The data were analyzed in the approximation of two weakly interacting systems with a separated asphaltene fraction as a disperse phase and toluene as dispersion medium. Small-angle scattering curves for 0.2 and 5.0 weight % solutions of asphaltenes in toluene are shown in Figure 1. The scattering intensity is seen to decrease by ∼25 times in passing to a smaller concentration, however, with no essential change in the curve shapes. Such a behavior testifies that the dilution of asphaltene solution gives no rise to smaller particles or non associated asphaltene molecules in the solution. For the large h values SAXS curve is well described by the power law I(h)–h−2.6, suggesting that asphaltene particles lack both compact and sphere-like shapes (Feigin and Svergun, 1987).
Small-angle scattering curves for 0.2% and 5.0% solutions of asphaltenes in toluene.
An additional information on the distinctive particle features may be obtained by analyzing dependencies of the ln(I(h)·hx) function on h2 (Feigin and Svergun, 1987). Some structure parameters, among them inertia tensor components of a three-dimensional body, may be determined in this case. Figure 2 shows small-angle scattering curves in various coordinates for asphaltene solution (5.0 weight %) in toluene. The analysis of the SAXS data (Figure 2) allowed an inherent form factor of asphaltene-containing particles dissolved in toluene to be determined. The particles have the extended form and may be characterized by the radii gyration, RG = 3.0 ± 0.1 nm and radii gyration of cross-section RG (cross) = 1.0 ± 0.1 nm, along the axial and cross axes, respectively (Table 1, Figure 3), the average particle length being ∼9.8 nm (Table 1). A model of the triaxial ellipsoid provided the best fit of experimental data at the 1:5:15 axis ratio. Figure 3 gives such a description by means of the form-factor matching. The resultant distribution mainly agrees with the values obtained. The shape of asphaltene particles may differ greatly depending on the origin of crude oil. The similar rod-like asphaltene particles for instance was found in the sources of crude oils included Hondo (HO), Arab Heavy (AH), Gulf Coast (GC), and Canadon Seco (CS) (Gawrys and Kilpatrick, 2005; Gawrys et al., 2006).
The small-angle scattering curve for 5.0% solution in various coordinates to determine the average form-factor of asphaltene particles. Average values of radii gyration (RG), radii gyration of cross-section (RG (cross)), and average length (L) of asphaltene particles in toluene before and after introduction of organic additives into the initial solution. <L>2 = 12(RG2 − RG(cross)2). The estimation of the average length for the extended particle (Feigin and Svergun, 1987). TBAF is tetrabutylammonium fluoride (7% additive is limited by its solubility). The small-angle scattering curve for 5% solution and its approximation with using the ellipsoid form-factor. The obtained distribution of ellipsoids by lengths is given in the inset.

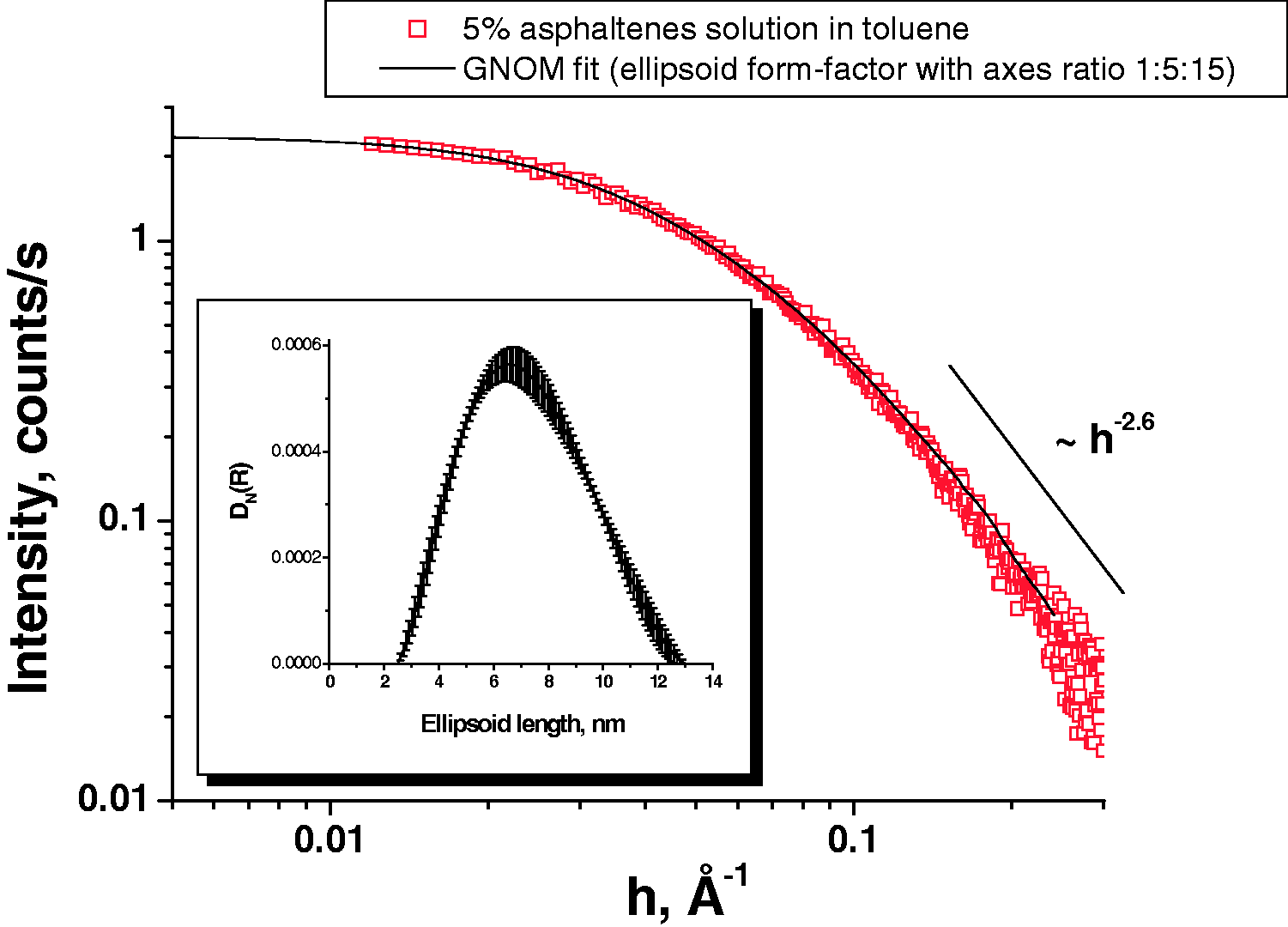
The ellipsoid model for fitting the SAXS data a priory cannot be considered as the only possible and unambiguous one. For example the power law I(h) ∼ h−2.6 in principle can imply the fractal structure of asphaltene particles also. To elucidate the situation and choose the correct model that can be used for the SAXS data fitting we used STM as an independent method to check our approximation. Asphaltene aggregates were deposited on a copper foil from the toluene solution by the procedure above. According to STM data the asphaltene film coats a considerable part of the foil surface (Figure 4), uncoated surface regions being observed. A comparison of topographic and current STM images confirmed the difference between electronic properties of the copper foil and asphaltene film to use for a qualitative differentiation of the registered topographic features. The asphaltene film thickness was about 2.5 nm; the measured “height” of the particles forming particular agglomerate (the height difference within the agglomerate) was ∼1.7 ± 0.2 nm.
The STM image of asphaltene film on the copper foil surface: scanning range (93.2 nm × 51.7 nm × 12.5 nm), tunneling voltage (Ut = −1.5 V), tunnel current (It = 0.5 nА).
From STM data, the asphaltene film is formed from agglomerate particles of the extended ellipsoid shape with the average sizes of 13.0 ± 1.8 nm and 5.7 ± 0.8 nm along the longest and shortest axes and with the average particle length/least size ratio of 2.3 ± 1.9. The particles of one such agglomerate are oriented in one direction, a certain disorientation of the neighboring agglomerates relative to each other being observed.
Data obtained by SAXS and STM methods agree well and testify that asphaltene aggregates have an extended shape. Particular good agreement is observed for the average greatest size (the length) of those. The average particle length obtained by two methods was 9.8 and 13.0 nm, respectively. The form-factor values somewhat differ, 1:5:15 for SAXS and 1.7:5.7:13.0 for STM. Figure 5 shows a comparison of the particle distribution in lengths. According to STM data, the distribution has a tightened “tail” on the side of great sizes. Such a discrepancy in data is probably associated with the additional asphaltene aggregation on the copper support. Despite the dilute asphaltene solution, the particles may be aggregated during the solvent evaporation resulting in changing of the dimensional features of asphaltenes on the copper foil surface as compared to those in solution in toluene. It may be suggested that the observed large particles of the lognormal distribution “tail” (Figure 5) are actually agglomerates of smaller asphaltenes, resulting in invalid determination of their average size.
The comparison of distributions by sizes for asphaltene particles in the solution obtained by the SAXS method and after depositing on the support obtained by the STM method.
At the next stage, an influence of various organic compounds on aggregative stability of asphaltenes was studied. 20% additives (7% for tetrabutylammonium fluoride (TBAF)) were added to asphaltene solution in toluene to provide the 5% end concentration of asphaltenes in the solution. Table 1 gives a full list of various organic compounds used in this work and typical radii gyration of asphaltene aggregates (RG), radii gyration of cross-sections (RG(cross)), and average particle lengths (L) before and after insertion of 20% organic additives into the mother toluene solution.
Nonpolar compounds would be expected to give rise to the additional asphaltene aggregation while polar compounds to promote the disaggregation of asphaltene particles in the solution. Nonetheless, the experiments revealed a rather ambiguous picture. From the data obtained (Table 1), all additives used may be divided into three groups. The first group includes the additives giving rise to the aggregation, namely cyclohexane, methanol, formic acid, heptane, acetonitrile, decane, hexanol, octene-1, furfural, and t TBAF having the strongest influence. To the second group may be assigned the additives promoting the disaggregation: decalin, dimethylformamide (DMF), dimethylsulfoxide (DMSO), nitrobenzene, aniline, cyclohexanol, monoethanolamine, triethanolamine, 1-methylnaphtalene, and dichloroethane. The third group includes inert or very weakly affecting additives: acetone, glycerin, ethyl acetate, dioxane, benzyl alcohol, oleic acid, limonene, α-pinene, cycloheptatrien, and nitromethane.
Note that RG and RG(cross) values change differently upon the insertion of the additives (Table 1). The absence of symbate changes during aggregation or disaggregation processes may suggest different influence mechanisms of various organic compounds. From SAXS data, the insertion of additive molecules between the asphaltene layers or the formation of a certain layer around asphaltene particles may give rise to such changes in the structure parameter values. Figure 6 gives SAXS data for asphaltene solution in toluene with some additives of the first and second groups. In principle the intensity of X-ray scattering from the asphaltenes depends on the density of additives—the increase of the additive’s density results in the decrease of the scattering intensity and vice versa. It was checked that the variation of the solvents density in our study from 0.83 to 0.95 g/cm3 (toluene density is 0.87 g/cm3) does not have noticeable influence on the scattering curves due to the contrast variation. The changes of the scattering pattern for the asphaltene solutions can be registered only if the density of the solution is higher than 1.03 g/cm3 (the density of asphaltene is 1.178 g/cm3). Figure 7 shows the appropriate distribution of asphaltene particles by the lengths in the approximation of the ellipsoid particle shape with the 1:5:15 ratios of the ellipsoid axes. One can see that the distribution of the asphaltene particles is shifted into the range of smaller sizes for disaggregation additives while the obtained distributions are shifted to the larger size range upon the insertion of aggregation compounds (Figure 7).
Small-angle scattering curves for asphaltenes in toluene with some additives to give rise to disaggregation (left) and aggregation of asphaltenes (right). The distribution of the particles by the lengths after the addition of disaggregation additives (left) and aggregated additives (right).

The data indicate that compounds of different polarity, polar and nonpolar, may give rise to the aggregation or disaggregation. As the example, cyclohexane and decalin posses the opposite aggregated properties despite their belonging to the same naphthenes. So, the aggregation occurs with the insertion of cyclohexane into the asphaltene solution, whereas decalin promotes their disaggregation. The insertion of hexanol-1 and cyclohexanol also result in the opposite aggregation affects. The first gives rise to the aggregation and the second to the disaggregation. It is interesting to note the different influence of nitromethane and nitrobenzene on the aggregation. The essential disaggregation effect is observed for the latter while nitromethane leaves the aggregation virtually unaffected. This effect may testify the absence of the direct interaction between nitro group and polar asphaltene groups.
The observed variations may perhaps be explained by different steric effects occurring between the appropriate molecules and asphaltene particles. It is likely that the planar molecules fit into the supramolecular asphaltene structure with a partial removal of the asphaltene molecules with large aliphatic substituents, resulting in a decrease of particle sizes. The aliphatic hydrocarbon additives, such as heptane, octene-1, and decane, give rise to an increase of the particles. Note that the presence of the double bonds in the additives has no essential influence on the aggregation processes though terpenes were reported as potential compounds for solving asphaltene deposits (Aymes et al., 2012; Castellano, 2008; Elochukwu et al., 2014). Limonene, α-pinene, and cycloheptatrien have no any effect on aggregation/disaggregation processes of asphaltenes.
Small polar molecules may also influence differently on the aggregation. So, methanol, acetonitrile, and formic acid produce the aggregation of the particles while ethanolamines, dichloroethane, DMF, and DMSO give rise to the disaggregation. Some polar substances are capable of influencing on the aggregation very strongly. As the example, the addition of furfural or TBAF gives rise to a considerable agglomeration of the asphaltene aggregates. One can suggest that there occurs a partial condensation of furfural with the asphaltene molecules resulting in larger aggregates, as for TBAF, the ionic compound, conceivably there occurs a loss of the aggregated stability of asphaltene colloids in the solution. Other polar substances of the third group (acetone, glycerin, ethyl acetate, dioxane, benzyl alcohol) virtually had no effect on aggregated processes. Note that oleic acid also did the same. Nevertheless, some works reported that the application of fatty acids and their derivatives (oleic acid among them) prevents the formation of aggregates in oils (Breen, 2000; Miller et al., 2001; Mohamadshahi and Nazar, 2012).
We can conclude that the additives of the third group, efficient upon solving asphaltene sediments, have no in fact an immediate effect on disaggregation of asphaltenes in solutions. Efficiency of the additives in solving is perhaps governed by larger stability of the colloid oil solution formed.
Chemical compounds also may act differently to influence on aggregation/disaggregation processes of asphaltenes. For nonpolar compounds, such as decalin, methylnaphthalene, steric effects perhaps are the decisive factor to permit the molecules to introduce into the structure of asphaltene-containing aggregates and to destroy it partially with the formation of the smaller stable aggregates of asphaltene molecules. One can suggest that similar planar molecules also would display disaggregation effects for asphaltene aggregates by such a way.
For polar compounds, such as nitrobenzene, triethanolamine, DMSO, of great importance are the local specific interactions between the additive and asphaltene molecules rather than the polarity degree of the compound. One can suggest that polar compounds possessing strongly disaggregating properties form thermodynamically favorable complexes with asphaltenes resulting in the disruption of the initial asphaltene aggregate and in the decrease of its sizes.
To recognize the origin of the effects observed we analyzed the interrelation between physical properties of the compounds used and their influence on the asphaltene aggregation. It turned out that the characteristics such as the dipole moment value, dielectric constant, partition coefficient in water/octanol mixture, and molar volume are associated with the aggregation/disaggregation ability in no way. Some researchers (Acevedo et al., 2010; Oh et al., 2004; Sato et al., 2014), however, performed studies on asphaltene solubility in various solvents with using the so-called Hansen solubility parameter (Hansen, 2007). The parameter is a measure of the intermolecular energy value. We compared the Hansen solubility parameters (δd) with a change in the radii gyration of the asphaltene particles after the addition of the appropriate compound (Figure 8). The δd parameter means the energy originated from the dispersion coupling between the molecules. The detail description of Hansen solubility parameters can be found elsewhere (Hansen, 2007).
The relationship between the Hansen solubility parameter (δd) of the compounds and their influence on aggregation/disaggregation processes.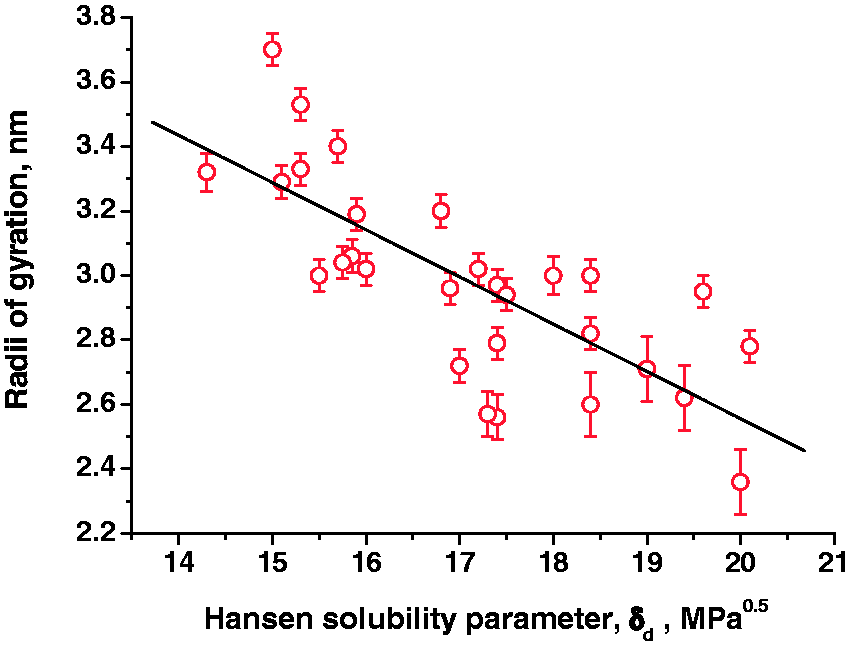
As seen from Figure 8, the solubility parameter of the compound is observed to be related to its aggregated ability, the correlation coefficient being 80% (except furfural and TBAF). Strong deviation in the cases of furfural and TBAF could be explained as possible reactions between such reagents with asphaltene molecules. Note that the correlation is observed only with the parameter δd taking into account the disperse interaction. The similar interrelation is missed for the solubility parameters which take into consideration the polarity (δp) and the presence of the hydrogen bonds in the compound (δh).
As seen from the obtained correlation (Figure 8), the polarity has little value by itself for aggregation/disaggregation processes while the affinity of compounds and asphaltenes described by the solubility parameter has greater importance. At the same time a sufficient number of compounds having no influence on aggregation/disaggregation processes (at the constant inert radius ∼3.0 nm) have the scatter in the solubility parameter values (15.5–18.5 MPa0.5). For other substances, a certain scatter is observed that deviates from the description by standard disperse interactions between asphaltene molecules and other organic compounds. This may testify that in some cases specific interactions prevail to result in the ordered complexes.
Conclusion
Asphaltene aggregates separated from heavy oil were studied by SAXS and STM methods. A good agreement is observed between the particle sizes determined by different procedures. Asphaltene aggregates were shown to have an extended shape of 10 nm length. A wide particle distribution by sizes is typical of those.
The influence of various organic additives on asphaltene aggregation processes was studied. It was revealed that cyclohexane, methanol, formic acid, heptane, TBAF, acetonitrile, decane, hexanol-1, furfural, and octene-1 when being added into the toluene solution of asphaltenes give rise to the aggregation of asphaltene particles. The compounds such as acetone, glycerin, ethyl acetate, dioxane, benzyl alcohol, oleic acid, limonene, α-pinene, cycloheptatrien, and nitromethane weakly influence on aggregation/disaggregation processes. On the contrary, DMF, DMSO, nitrobenzene, aniline, cyclohexanol, monoethanolamine, triethanolamine, 1-methyl-naphtalene, and dichloroethane give rise to the essential decrease in asphaltene aggregate sizes. A certain correlation is observed between those and the solubility parameters of the added compounds.
It was found that the correlation coefficient between RG value and Hansen solubility parameter is about 80% only. It implies that in some cases the complexes between asphaltene molecules and additives can be formed. So, for understanding the origin of the intermolecular interactions responsible for the observed phenomena the further quantum chemistry calculations are needed as well as the experimental identifications of complexes formed from the asphaltenes and specific organic molecules.
Footnotes
Acknowledgments
The authors are grateful to KV Obida and IV Kozhevnikov for assistance in the investigations of the samples. This research was performed within the project №15-19-00119 of Russian Science Foundation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Russian Science Foundation (project 15-19-00119).

