Abstract
The article reports an experimental study of the metal uptake properties of poly(1-methylpyrrol-2-ylsquaraine) particles, in the context of 51 elemental metal complexes. Vagaries of the experimental results meant that the final analysis took place for 43 of these metals. After an initial descriptive analysis, an Analysis of Variance model was fitted. This revealed that several factors, some intrinsic to the metals themselves, others relating to the experimental conditions, have an important influence on the adsorption characteristics of the poly(1-methylpyrrol-2-ylsquaraine) particles. Of these, the most important is the ratio of the number of moles of the metal to the number of moles of the adsorbent particles. X-ray mapping of the poly(1-methylpyrrol-2-ylsquaraine) particles treated with contrast agent revealed that, although adsorption was all throughout the particles, the majority of the agent was adsorbed within 10–15% (diameter distance) of the surface of the particles.
Introduction
Squaraine dyes and polymers since their first reported synthesis in 1965 have been studied for a range of interesting chemical, physical and photophysical properties (Ajayaghosh, 2005; Beverina and Salice, 2010; Hu et al., 2013; Lynch, 2015; Patsenker et al., 2010). The three main areas of squaraine chemistry (in order of decreasing numbers of publications produced) are those based on derivatives of aniline, indolenine and pyrrole, although quite a number of other squaraine derivatives have been reported as well. Of the polymeric squaraine compounds reported in the literature, those made from derivatives of pyrrole have been studied the most. In fact, the first few polysquaraine compounds reported were the products of polycondensation reactions between squaric acid and 1H-pyrrole (Triebs and Jacob, 1965), 1-methylpyrrole (Triebs and Jacob, 1965, 1966) (Figure 1) or 3-ethyl-4-methyl-1H-pyrrole (Triebs and Jacob, 1966). In each of these, pyrrole molecules both α-positions are free, promoting ease of polymerization. Mention of the polysquaraine made from 1H-pyrrole (or its derivatives substituted in the 3- or 4-position) in the literature (Binda et al., 2009; Chenthamarakshan and Ajayaghosh, 1998a; Chenthamarakshan et al., 1999a; Hasegawa et al., 2009; Ko et al., 2011; Lu et al., 2010a,b, 2013; Triebs and Jacob, 1965, 1966) is about equal (in terms of numbers of articles) to mention of the polysquaraine made from 1-methylpyrrole (Begum et al., 2010; Courgneau et al., 2013; Lynch et al., 2001, 2005, Lynch, 2011; Sant’Ana et al., 2006; Spicer et al., 2006; Triebs and Jacob, 1965, 1966; Yu et al., 1990), with both limited by the fact that (without the aid of 3-position substituted long alkyl chains on the pyrrole groups to aid solubility) they can best be described as ‘black intractable powders’. Irrespective of this, poly(1-methylpyrrol-2-ylsquaraine) (PMPS) particles have been studied for third-order nonlinear optical properties (Yu et al., 1990), both electrical conduction (Sant’Ana et al., 2006) and electrical insulation properties (Lynch et al., 2001); utilized for their size and shape to produce hollow silica particles (Lynch et al., 2005) and core-shell particles (Begum et al., 2010), used as support particles for the addition of ammonium phosphate in polymer fibres (Spicer et al., 2006), and used as an odour adsorbent agent (Courgneau et al., 2013). In the article reporting that PMPS particles could be used to produce core-shell Fe2O3@SiO2 particles (Begum et al., 2010), where the Fe2O3 resided inside the silica shells as concentric shells or, as it was referred to in that article as hollow Russian doll microspheres, it was inferred that PMPS particles were porous. Although not extensively discussed (in that article), the production of these Russian doll microspheres was only possible because the PMPS particles adsorbed the iron salt dissolved in HCl solution. Lithium ion interactions in solution with polysquaraines made from N-[[(methoxyethoxy)ethoxy]ethyl]pyrrole (Chenthamarakshan and Ajayaghosh, 1998b; Chenthamarakshan et al., 1999b) and N-[(methoxyethoxy)ethyl]pyrrole (Chenthamarakshan et al., 1999b) have also been previously reported, but, in these studies, the association with lithium was attributed to the presence of the polyethoxy groups. The ability of PMPS particles (that do not contain any specific chemical groups known for metal ion complexation) to uptake both inorganic and organic molecular species makes them a potentially useful support for active ingredients across a whole range of applications. But before individual applications can be determined, the general molecular transport capabilities of PMPS particles need to be studied. Thus, here we report the PMPS particle uptake of 51 elemental metal compounds with the emphasis being on the study of the metal cation as opposed to the non-metallic anion, although in two cases, the metal element under investigation was incorporated in the cation.
General synthesis of PMPS and an electron microscope image of the particles.
Experimental
Chemicals and materials
All chemicals were purchased from Sigma-Aldrich and were used as-received. Forty-three of the metal complexes purchased were chloride salts with emphasis on hydrated species. Eight metal species were purchased in other forms either because the chloride salt was insoluble, hazardous or was otherwise unobtainable. These eight include Ti2(SO4)3 45% wt solution in dil. H2SO4, (NH4)10W12O41.5H2O, AgNO3 and AgSO4, Ga(NO3)3.xH2O, Tl2SO4, GeO2, As2O3, Bi2O3 and SeO2. Reverse osmosis (r/o) water was prepared in-house. PMPS particles were prepared according to the literature procedure (Lynch et al., 2001) by refluxing equimolar amounts of squaric acid and 1-methylpyrrole in butanol for 18 hours, filtering, and then washing (using a Soxhlet extractor) with hot ethyl acetate for 4 hours before drying in a heated cabinet (60℃). All glassware, including all volumetric flasks for analysis, was cleaned by soaking in a 5% H2SO4 solution for 24 hours, followed by rinsing in r/o water and drying in a heated cabinet (60℃), prior to use.
Instrumentation
Electron microscope images were recorded on either a FEI Quanta 200F environmental SEM, a Jeol 1200 EX transmission electron microscope (TEM) fitted with a LaB6 filament or a Jeol 7000F SEM in STEM mode using an Oxford Instruments EDS detector with INCA software (for X-ray mapping). Ultrathin slices of PMPS particles in resin were prepared according to the literature (Begum et al., 2010). The staining solution used was 30% uranyl acetate in methanol; stained for 7 minutes and then 7 minutes in Reynolds lead citrate. Atomic force microscope images were recorded on a Veeco Dimension 3100 AFM. Hydrogen storage experiments were run on a Hiden IGA constant pressure thermogravimetric balance. A pressure composition isotherm was collected as the sample was first degassed and then run up to 18 bar at 77 K (adsorption and desorption). Brunauer–Emmett–Teller (BET) nitrogen adsorption measurements were recorded on a Micrometrics Tristar 3000 V6.08A.
Uptake of metal complexes
Unless otherwise indicated, 1.0 g of each metal complex (either salt or oxide) was dissolved in 30 mL of either r/o water (H2O), 0.1 M hydrochloric acid or conc. hydrochloric acid with stirring (up to 16 hours in some cases), and then maintained at temperatures of either 4℃, room temperature (25℃ ± 10%) or 50℃. One gram of PMPS particles were added to each of the nine solutions (of each metal complex) with stirring for 30 minutes, filtered under vacuum and dried in a heated cabinet (60℃).
To overcome difficulties with insolubility 10 M HCl was used instead of conc. HCl for NaCl, 6 M HCl was used instead of conc. HCl for KCl, conc. H2SO4 was used instead of conc. HCl for both SrCl2.6H2O and BaCl2.2H2O, and AgNO3 was used for dissolution in H2O and 0.1 M HNO3 while Ag2SO4 was used for dissolution in conc. H2SO4. TaCl5 was only soluble at 50℃ in conc. HCl, (NH4)10W12O41.5H2O was only soluble in H2O and 0.1 M HCl, and H2SO4 was used for Tl2SO4 instead of HCl. Low solubility was experienced for ReCl5, NbCl3, As2O3 and SbCl3 in both H2O and 0.1 M HCl, for IrCl3.H2O, MoCl3 and GeO2 in all solutions, and for RhCl3 in conc. HCl. Few solutions where some re-precipitation of a metal salt occurred were filtered immediately before addition of the PMPS particles.
Titanium samples were prepared using Ti2(SO4)3 45% wt solution in dil. H2SO4. The solution was used as purchased (as the conc. sample) and diluted 25:75 with H2O (to provide a dilute sample). PMPS particles treated with AgSO4 in conc. H2SO4 could not be appropriately dried (in the heated cabinet) so were further washed with 0.1 M H2SO4 before re-filtering and drying. No sample was analysed for thallium in H2O at 4℃ due to insolubility. SnCl2.2H2O was only soluble in conc. HCl. Antimony samples at all three temperatures were prepared in H2O and conc. HCl. Bi2O3 was only soluble in conc. HCl.
Acid–peroxide digestion of samples
Approximately 100 mg of each sample (accurately weighed) was dissolved in a 1:1 mixture of 100 vol. H2O2 and conc. H2SO4 (5 mL each) added sequentially in a 100 mL volumetric flask. Upon dissolution of the particles, the flask volume was made up using r/o water. Blank samples were prepared using untreated PMPS particles. The metal ion concentration in each solution was analysed using a Perkin-Elmer 5300DV ICP.
Vanadium samples required cooling in ice with very slow addition of the sulphuric acid and waiting for the reaction to subside before continuing the digestion. Ruthenium samples reacted violently when hydrogen peroxide was added, resulting in cracking of the volumetric flask. To digest these samples, it was necessary to cool the samples in ice before the addition of conc. H2SO4 first with very slow addition of the H2O2 with gentle swirling, waiting for the reaction to subside before adding more H2O2. Palladium samples were similarly volatile during digestion. These samples had to be cooled in ice and the conc. H2SO4 added very slowly allowing the reaction to subside before continuing.
Barium samples were expectedly found to precipitate insoluble BaSO4 upon addition of conc. H2SO4. Instead, microwave digestion was undertaken for barium using a Milestone 1200 Mega HPR 600/10 and using 30/70 v/v of 100 volume hydrogen peroxide and conc. HNO3 in place of H2SO4–peroxide digestion. HNO3 was also added to the standards to matrix match with the samples. The three tin samples were analysed by direct injection of suspended solutions of PMPS particles in H2O into the ICP.
Calibration standards – Quality control
For each metal analysis, four calibration standards and one blank standard were prepared with all standard solutions being of the same anionic species of the sample to ensure matrix matching. Spiked samples (fortified blanks) of a known concentration (50 mg/L) were subsequently analysed with each set of samples as a quality control procedure. The purpose of preparing spiked and blank samples was to reveal errors due to interfering contaminants from the reagents and vessels used during the analysis. A complete set of new standard solutions, and spiked samples and blanks were prepared for any set that fell outside the error limit of ±5%. To ensure consistency of both analysis methodology and synthesis of PMPS particles, five elements (Na, Mg, Fe, Se and Ag) had each of their samples replicated 3 times over the course of the study.
Supplementary material
Adsorption data for each metal analysed, in this study, boxplots for temperature and solvent concentration, and hydrogen storage pressure composition isotherm for PMPS particles have all been deposited as supplementary material.
Results and discussion
The purpose of this study, in the first instance, is to examine general trends (if any) in the uptake of metal compounds into PMPS particles such that any overall model developed could be applied in further studies targeting the PMPS uptake of specific metal complexes (which have not necessarily been studied here). For each of the 51 metals examined, nine separate results were possible with uptake into PMPS particles being undertaken in three different solutions (water, dil. acid, conc. acid), each at three different temperatures (4℃, room temp., 50℃), although considering the difficulties encountered with insolubilities, not all metals returned this number of results. For this reason, the data set is by no means complete and it is recognized that not all the metals were examined in an equivalent form, but fortuitously the only element (not counted in the 51 metals) not to return any results in any category was Germanium. It is also acknowledged that not only are there no comparative anion studies so as to normalise the use of differing anions (other than chloride) for some elements, and there are no differing oxidation state studies to normalize the effect of oxidation state, but there is also no way of normalising the results for either tungsten or gold, where the metal resides in the anion ((NH4)10W12O41.5H2O and HAuCl4.3H2O, respectively). It was considered that such anion results for tungsten may more accurately reflect the uptake of the ammonium cation (in the PMPS particles), although comparative experiments with both ammonium sulphate and mono-ammonium phosphate suggested that this was not the case and the results obtained for tungsten were unique to that complex. However, for the purposes of this particular study, all data were included and initially considered of equal relevance.
Statistical analysis
As previously stated, not all 51 metals provided a full set of measurements,
1
or were anomalous for other reasons, which complicated the ability of standard statistical methods to provide a complete analysis. Metal concentrations were measured in mg/g but were converted to mmol/g indicating the number of moles of each metal element taken into 1 gram of PMPS particles. For this reason, the amount of PMPS particles made available for each adsorption process was held constant, as was the amount of metal complex added to each solution so as to give variation in the solution concentration of the adsorbed species. It was considered that the metal concentration in each solution was likely to have an important effect on the results but, by means of the statistical analysis proposed, it was expected that this effect could be quantified and removed from consideration before investigating other effects. To facilitate this analysis, the ratio:
On examining the results numerically, it was immediately obvious that there was indeed considerable variation between the different groups. Figure 2 shows this by means of a boxplot. The boxes here represent the variation between the upper and lower quartiles, whereas the bold line represents the median value. It is immediately clear that the ‘surplus’ group (r ≥ 1.07) has a much greater average adsorption than the others; for instance, not only is the median much greater but also the lower quartile is actually greater than the upper quartiles of both the other groups. Even though these two latter groups have different medians, there is also considerable overlap. In many respects, these results are not surprising; ceteris paribus, the more metal that was present, the greater the amount that we would expect to be adsorbed, but it is also clear that this cannot fully explain the data. The ‘surplus’ group (r ≥ 1.07) has greater variation for the bulk of its range, whereas the others are more compact. Nonetheless, there are a substantial number of outliers in these groups (circles in the diagram), and the picture as a whole suggests that other sources of variation remain.
Adsorption characteristics of metal groups (def = r < 0.7, par = 0.7 ≤ r < 1.07, sur = r ≥ 1.07).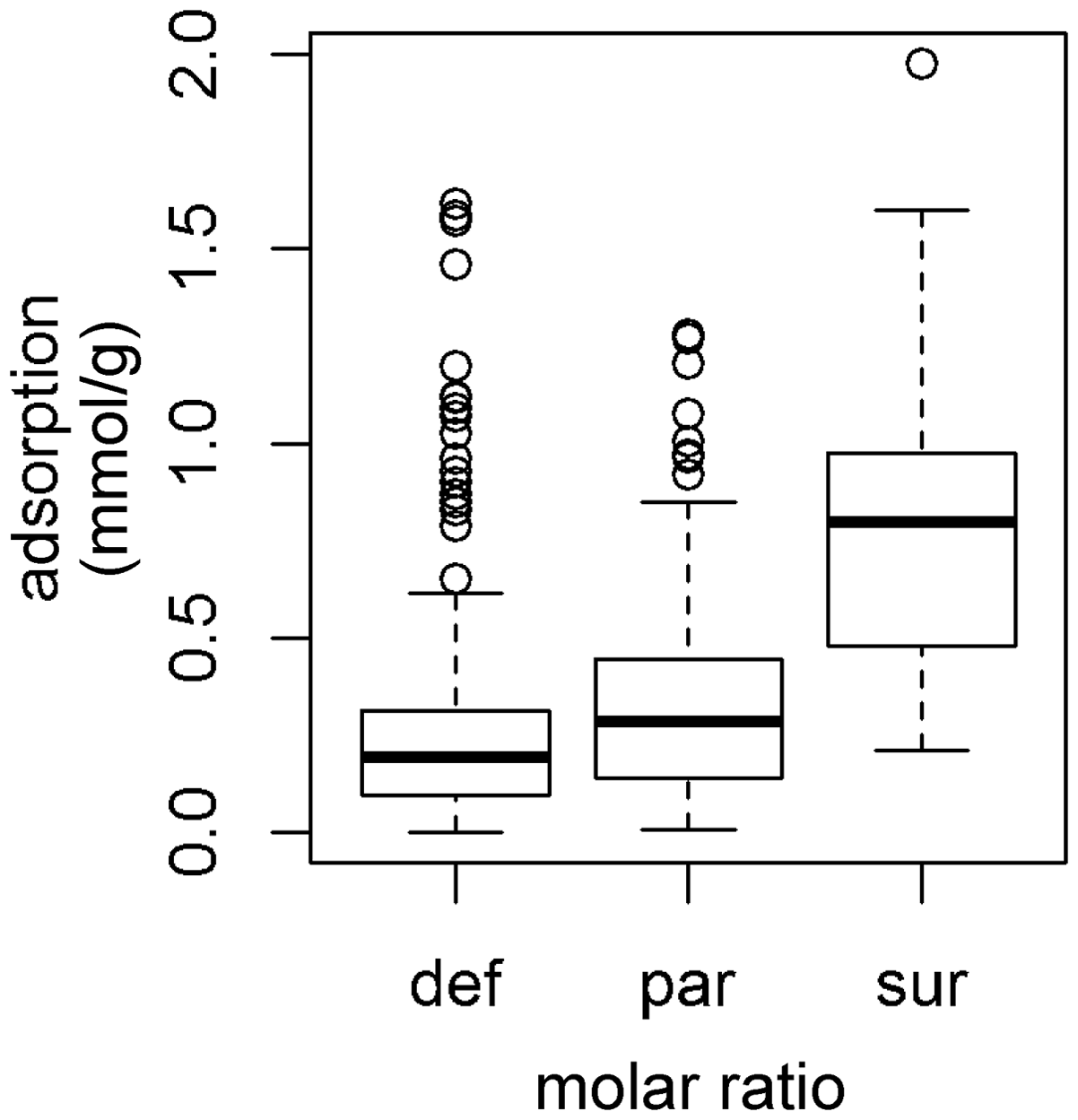
One such potential source of data variation that was considered was the Lewis acidity classification (of Hard, Borderline, Soft)
2
and a boxplot showing how adsorption was classified with respect to this factor is shown in Figure 3. In this case, although the Hard acid class has the least adsorption on average, the considerable variation within all three classes is the main feature. Boxplots for temperature and solvent concentration were similarly plotted (included in the Supplementary material). There is some evidence that uptake tends to increase with temperature; otherwise, in both cases, large variation was again the most obvious characteristic. However, it should be noted that the existence of large variability relative to any factor on its own does not necessarily preclude its relevance within the context of a more comprehensive model. This is where the statistical technique of Analysis of Variance (ANOVA) becomes important in uncovering potential explanatory factors for the observed variation. In effect, different sources of variation can be isolated and the relative importance of each factor assessed. A standard, and very comprehensive, explanation of ANOVA modelling has been presented by Montgomery (2012).
Adsorption characteristics of Lewis acidity classes (H = hard, B = borderline, S = soft).
ANOVA
ANOVA for initial model: Adsorption measured in mmol/g.

ANOVA: analysis of variance; df: degrees of freedom; SS: total sum of squared deviations.
The use of the model stated above gave a general picture that was encouraging in its analysis, in that, a large SS (total sum of squared deviations) had been reduced substantially. All of the F-ratios implied very small P-values, i.e. the probability of wrongly rejecting a null hypothesis that the effect was due to chance. As expected from the boxplots, MOLE-RATIO was very highly significant, accounting for more than 20% of the total variation with only 2 df (degrees of freedom). There was also a difference between the characteristics of METALs in different ACIDITY classes, while within each class, there was considerable variation between the METALs in that class. The different concentrations of SOLVENT were significant within each ACIDITY class, and TEMP had a strong effect across all classes. A model in which SOLVENT was included as a main effect across all classes was also fitted, but it failed to provide any very useful additional explanation of the data; i.e. the P-value was not significant at a 1% level. SOLVENT does, however, have a strong interaction with METALs; in other words, some metal complexes preferred more concentrated solvent than others. There was still some variability that was unexplained by these factors, but no other interactions could be found. Attempts to fit a more comprehensive model did not find any other significant effects beyond the marginal cross-class effect of SOLVENT referred to above (as metal:solventjk).
Other variables
As well as the variables representing experimental conditions, it was also possible to calculate other variables relating to the experimental units themselves, in particular, the CHARGE (valency) and IONIC RADIUS (crystal ionic radii) of each metal element examined. Possible dependence on these was also investigated, but no evidence was found that either was important in accounting for any residual variation after the model discussed above had been fitted.
Further analysis
These results were in a sense merely preliminary; finding out exactly how the interactions take place requires further analysis. However, caution suggests a consideration of the units in which uptake was measured, in that there did not appear to be a ‘well-behaved’ distribution for the residual errors. It is possible that some of the extremely significant effects observed are to some extent an artefact of a highly skewed distribution, whereas the P-values were all calculated on the assumption of an underlying ‘normal’ distribution. But, given the very small P-values found, these would have to change by many orders of magnitude before the conclusions that have been drawn from the results were affected. Thus, although a Box–Cox transformation or similar could be applied (see Chapter 15 of Montgomery (2012)), on balance it is unlikely to make a material difference.
Tables of means
Mean adsorption for main factors (mmol/g).

Mean adsorption (mmol/g) for interaction between

Mean adsorption (mmol/g) for all

Physical properties of PMPS particles
Apart from synthesis (Triebs and Jacob, 1965,1966; Yu et al., 1990), thermal decomposition (Lynch et al., 2005), size, shape, colour and crush strength (Begum et al., 2010) and possibly molecular structure (Sant’Ana et al., 2006), very little else is known about the physical characteristics of PMPS particles. To date, no density values have been reported for these particles, and neither has any surface area nor porosimetry data been disclosed. Previous electron microscopy studies have determined that the particles are spherical, with diameters ranging from 1.3 to 4.0 µm (average diameter ∼1.9 µm) (Begum et al., 2010; Lynch et al., 2005), and studies utilizing PMPS particles have also implied that they must be porous (Courgneau et al., 2013; Lynch et al., 2005; Spicer et al., 2006) although there has been no examination thus far on these supposed pores. The metal element uptake data presented in this article, with respect to the amount of material carried by the particles and the differences between metal element uptake, not only suggests that they are porous, which could be attributed to just physical adsorbance, but also suggests that there is other chemistry involved. Chemisorbance can be ruled out on the grounds of the results from a previous article that use PMPS particles as a support for ammonium phosphate (Spicer et al., 2006). If either component of ammonium phosphate was chemisorbed onto the surface of the PMPS particles, then the particles, either before or after compounding into polymer fibres, would not consistently leach phosphate over numerous washing events. However, there is evidence, in two previously reported X-ray crystal structures, that squarate oxygen atoms are capable of co-ordination with metals (Hsueh et al., 2007a,b). Thus, direct metal co-ordination with the squarate oxygens in the PMPS pores must also be taken into account, which is suspected, in corporation with the specific pore sizes, to give rise to the differences in metal element uptake.
Electron microscopy for pore determination
To visualize any porosity within the PMPS particles, ultrathin slices of the particles were treated with known metal element contrast agents and examined under a TEM such that any pores would show up as dark patches or lines. It is common in these types of experiments that both uranyl acetate and lead acetate are consecutively used to stain the ultrathin sections in what is known as ‘double contrasting’ for examination under an electron microscope. Figure 4 shows two TEM images of an ultrathin section of a stained PMPS particle at two different magnifications. Figure 4(a) shows an entire particle (magnification × 50 k) and reveals that there are cavities within the particles where the stain has been concentrated, thus they are resolved as dark patches. Upon closer examination, Figure 4(b) (magnification × 100 k; arranged to avoid the large dark patches) reveals that the particles contain an elaborate network of smaller pores, in addition to the larger cavities. However, although these images highlight internal pores, because the ultrathin particle sections were treated with the stain solutions, the images do not reveal either an accurate size distribution of the internal pores or whether or not the internal pores are accessible from the surface of the particles. Therefore, particles were soaked in a solution of 1% uranyl acetate in water, filtered and dried, before being secured in resin and sliced. TEM images (similar to Figure 4) revealed very little, so X-ray mapping images were taken. Figure 5 is a composite image showing (a) two joined particles identified for X-ray mapping, (b) the X-ray map measuring the Kα1line for chlorine and (c) the X-ray map measuring the Mα1line for uranium. It should be noted that neither the stain nor the particles should have any chlorine anion present, so it must be assumed that some was present either in the stain or on the glassware as a contaminant. Fortuitously, this image better demonstrates the concentration of the stain than does the uranium image. Both images indicate a concentration of the stain in the surface regions, approximately 10–15% of the diameter of each particle, as well as lesser distribution throughout the particles. Figure 6 is a cross-sectional profile of the strength of the signal from both elements across the two particles, which again indicate that the majority of the stain resides in the surface regions with lesser stain throughout the middle of the particles. Although the highest concentration of stain appears to be in the surface regions, these images indicate that uptake of the stain still occurs throughout the entirety of the particles. The observance of higher concentration of adsorbed material nearer the surface of the particles explains why these can act as sacrificial templates for hollow silica shells if they are treated with a silica precursor and then thermally disintegrated, and it also explains the release profile observed for ammonium phosphate, where significant amounts of the adsorbent leached within the first few washes but measurable amounts continued to be released over numerous washing events (Spicer et al., 2006).
TEM images of an ultrathin section of a stained PMPS particle at two different magnifications: (a) ×50 k and (b) ×100 k. Composite image showing (a) TEM image of an ultrathin slice of pre-stained PMPS particles including an outline of the area X-ray mapped, (b) X-ray map of Cl Kα1 and (c) X-ray map of U Mα1. Composite image showing (a) TEM image of an ultrathin slice of pre-stained PMPS particles including the line of measurement of cross-sectional X-ray strength and (b) X-ray profile for both Cl Kα1 and U Mα1.


Surface analysis
Previous SEM images (Begum et al., 2010; Lynch et al., 2005), plus the image in Figure 1, indicate that the surface of PMPS particles is relatively featureless. This is also evident in Figure 4 where little can be gained on the size and shape of the pore openings. Thus far, SEM images of the particles have been recorded with the use of a conductive surface coating on the particles, which could obscure any fine surface detail. Figure 7 shows two images of a single PMPS particle, without any surface coating, taken using an environmental SEM image. Unfortunately, at these magnifications, the resolution in both images is low and no surface features can be verified. As an alternative, Figure 8 is an atomic force microscope image of an exposed portion of a single PMPS particle, pressed into wax (to keep the particle stable), which gives better resolution than the environmental SEM but still indicates that PMPS particles have a featureless surface. A featureless surface additionally indicates that the metal element uptake in PMPS particles is due to internal pores and not just outer surface adhesion.
Environmental SEM images of an uncoated PMPS particle at magnifications (a) ×43.5 k and (b) ×95 k. AFM image of the surface of a PMPS particle pressed into wax (total image width = 1.1 µm).
BET nitrogen adsorption measurements to determine the surface area of the particles returned a value of 2.013 ± 0.034 m2/g, which is a reasonable measurement for solid particles but not for porous ones. The discrepancy between the measured surface area and a value that better represents the porosity of the particles may be explained if the pore sizes are below the limit of detection for BET nitrogen adsorption measurements, i.e. pore width <2 nm (Wang et al., 2013). A more appropriate approximation of surface area may come from hydrogen storage measurements, which were also recorded on the PMPS particles. A number of independent hydrogen adsorption results have shown that there is a correlation between weight percentage uptake and surface area (Thomas, 2009) and the general value range for the conditions under which this experiment was run is 300–500 m2/g surface area per weight percent hydrogen adsorption, thus indicating a surface area of no more than ∼450 m2/g for PMPS particles. It has also been reported that, in activated carbons, the relationship between storage capacity and pore size shows that pores 0.6–0.7 nm provide the largest hydrogen uptake per unit surface area at elevated pressures and liquid nitrogen temperatures (Wang et al., 2013). Thus, further investigation of the pore sizes in PMPS particles is warranted; although the introduction of dopants to enhance the hydrogen adsorption capabilities may be required. The results presented in this article may be useful in the determination of such dopants.
Conclusion
Analysis of the data set revealed that there was clear evidence that the ratio of the amount of metal to the amount of adsorbent (both measured in moles) was highly significant in determining the uptake of each metal into the PMPS particles. This was the most important factor, albeit an expected one. Nevertheless, even when this effect has been removed from consideration by ANOVA, other factors were found to be relevant as well. Metals classed as Hard Lewis acids were on average substantially less efficiently adsorbed than those that were Borderline or Soft. However, the most efficiently adsorbed metal (on average) was found to be Na+, which is classified as a Hard Lewis acid, with other metals in the same periodic (table) group (Li+ and K+) also featuring prominently, although other (Hard) metals were very poorly adsorbed. Within the Borderline class, Hg2+, Zn2+ and Fe3+ were highly placed, as were Au3+, Ag+ and Cu2+ in the Soft category. Incidentally, in each category, the three top metals were found to have considerably higher mean values than the fourth (or lower) ranked, and this order possibly indicates a relationship to periodic table group (as might be implied by Na+, Li+ and K+ for the Hard Lewis acids; Au3+, Ag+ and Cu2+ for the Soft Lewis acids). As for the experimentally controlled variables, it appeared that metal uptake was substantially improved by raising the solvent temperature. The effect of solvent acidity depended on the Lewis acidity class of the metal concerned. Solvent acidity made little difference for the Hard Lewis acids, but in the Borderline cases, a high acid (solvent) concentration was preferred, whereas Soft metals were better adsorbed when the acid (solvent) concentration was lowered. Ultrathin sections of PMPS particles that had been stained with a contrast agent revealed that the particles possess an elaborate network of pores. X-ray mapping of the PMPS particles treated with contrast agent revealed that, although adsorption was all throughout the particles, the majority of the agent was adsorbed within 10–15% (diameter distance) of the surface of the particles.
Footnotes
Acknowledgments
The authors thank Teresa Morris (Birmingham University) for the ultrathin slice images, Simon Henley and Cristina Giusca (University of Surrey) for environmental SEM and AFM work, respectively, and Allan Walton (Birmingham University) for hydrogen storage experiments.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors wish to thank the Faculty of Health and Life Sciences, Coventry University, for financial assistance. All electron microscopy work on ultrathin slices was funded by the West Midlands Manufacturing Advisory Service (TOR No. 09/188). The environmental SEM and AFM work was funded by the BlueSkyNano program. Hydrogen storage experiments were funded by the Advantage West Midlands Science City Hydrogen Energy Project (BG/IN7008).


