Abstract
Silicone rubbers are high-performance polymers which, in the case of high consistency silicone rubber (HCR), are processed in the extrusion process, for example. Due to the relatively high material price, the material can be foamed to reduce costs and to adjust certain properties. This study therefore investigated the influence of nucleating agents on extruded high consistency silicone foams where water was used as blowing agent. The types of crosslinker investigated showed clear differences in their resulting foam structure and mechanical properties with the talcum and calcium carbonate agents used.
Introduction
Silicone rubber is a synthetic rubber which, unlike organic rubbers, has no carbon compounds in its main chain, but alternating silicon and oxygen atoms.1–3 This results in high temperature stability and good chemical resistance.4–6 These properties make silicones suitable for use in the automotive and pharmaceutical industries, for example.7,8 As with other plastics, the desired component can be foamed with blowing agents to make it lighter and cheaper. 9
In the field of silicone rubbers, chemicals such as AIBN (azobisisobutyronitrile) or azodicarbonamide are usually used as chemical blowing agents according to the current state of the art, but these are usually harmful to health and by-products and degradation products from the reaction remain in the foam. The decomposition reaction of the blowing agent must be coordinated with the cross-linking reaction of the silicone rubber, as both are thermally activated, which makes the process control much more complex. 10
In the case of physical blowing agents, gases such as carbon dioxide and nitrogen or liquids such as water can be injected into the rubber during the extrusion process. In the case of water, the blowing agent can also be incorporated into the rubber compound using a carrier substance such as silica. When water is used, the foaming process is achieved by the evaporation of the water, whereas when gases are used, foaming takes place as soon as the rubber leaves the die due to the pressure drop that occurs. 11
In the field of silicone rubbers, the focus is mainly on the use of blowing agents such as CO2 12 to foam the silicone rubber during processing. In the experimental environment, tests were also carried out with alcohols, 13 water,14,15 and thermoplastic microspheres.9,16
The foaming of silicone rubber is achieved with water by heating the silicone during the processing stage. This simultaneously initiates the vulcanization process and the evaporation of the water, which leads to the formation of the foam structure. 17
The foam structure is influenced by various factors, including rheological properties, 18 crosslink density, 19 temperature, 20 and nucleating agents. 21
Nucleating agents are generally used to control cell nucleation in the production of high-quality foam products. Effective nucleating agents include calcium carbonate, magnesium silicate or talcum. The criteria for an ideal nucleating agent include the fact that nucleation must be energetic compared to homogeneous nucleation and must have a uniform surface and dispersibility. 21
In the case of rubbers, it was also found that finer particle sizes of nucleating agents calcium carbonate, talcum or carbon black lead to a finer pore distribution. 22
The cellular structure of foamed components significantly influences their properties. The introduction of foamed structures reduces mechanical properties compared to compact profiles; however, this reduction is less pronounced in fine-celled structures with a high cell density and small cell size. In addition to the mechanical properties, this also affects the density. The reduction in density resulting from the cellular structures leads, for example, to a decrease in tensile strength. 10
The relationship between mechanical properties and density with the foam structure therefore illustrates why controlling pore density and pore size is crucial for the properties of foamed components.
In this study, we investigate the influence of nucleating agents on the resulting foam structures in two different crosslinking systems, at a constant temperature and viscosity and with materials that have undergone a constant crosslinking process. This has not yet been done for silicone rubbers in the field of water-based blowing agents in previous studies. Cell morphology can be adjusted by altering foam parameters, such as the type of nucleating agent or crosslinking system used. This is followed by a presentation of the relationship between cell morphology and the mechanical properties of silicone rubber foams.
Experimental
The high-consistency silicone rubber (HCR) used in this study was Elastosil R 401-40 S and Elastosil R plus 4305/40 from Wacker Chemie AG. For the peroxide crosslinking 401-40, the peroxide Peroxan PMB (di-(4-methylbenzoyl)peroxide) from Pergan Hilfsstoffe für industrielle Prozesse GmbH was used. Elastosil AUX BATCH PT1 from Wacker Chemie AG was used as a catalyst for the addition-curing rubber 4305/40.
The Elastosil R plus 4305/40 is a two-component addition curing silicone rubber exhibiting good mechanical properties and excellent transparency. The Elastosil R 401-40 S has good flexibility and mechanical properties and is used for gaskets, sheets or tubings. The 401-40 is a variant that is preferably crosslinked with peroxide. A water/silica mixture in a ratio of 2:1 served as physical blowing agents. Therefore, a fumed hydrophile silica Aerosil 200 from Evonik Industries AG (Essen, Germany) with a specific surface area of 200 m2/g was used.
Talcum (Mg3Si4O10(OH)2) and calcium carbonate (CaCO3) from Carl Roth GmbH + Co. KG were used as nucleating agents. The calcium carbonate used here is precipitated calcium carbonate with a purity of ≥99%. The density of the CaCO3 was 2.93 g/cm3 with a grain size of approx. 150 µm. The talc used has a density of 2.7 g/cm3 and a grain size of approx. 44.
For the compounding of the HCR, a twin-screw internal mixer (CTM-25) from COLMEC SPA was used, which can mix 1.5 kg per batch. Each batch is made up of the four components silicone rubber, crosslinker or catalyst, blowing agent and the dispersing or nucleating agent, as shown schematically in Figure 1. Composition of the compound.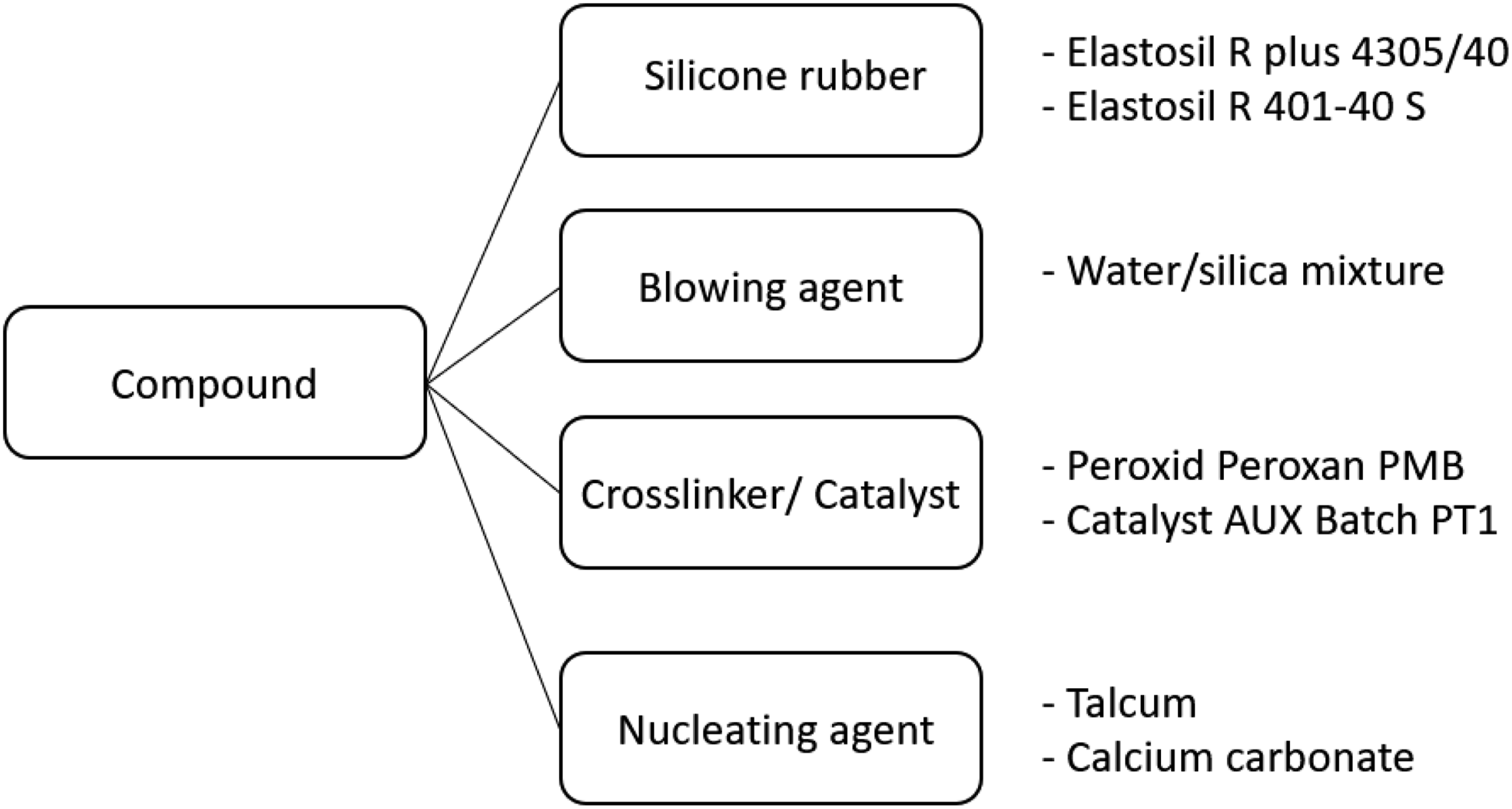
Mixture of the testet compounds.

After the mixing process, the finished compounds were discharged in strip form for further processing on a silicone extruder from Rubicon Gummitechnik und Maschinenbau GmbH. Shaping was carried out using a tool measuring 2 mm × 40 mm with subsequent vulcanization in an infrared tunnel. The control was set so that the extrudate had a temperature of 180 ± 5°C after leaving the tunnel. The temperature was determined using an infrared pyrometer.
Characterization of compounds and extrudates
After the compounds were produced, they were first examined using a Rubber Process Analyzer (RPA). The aim was to analyze interactions between the blowing agent and the crosslinking and to identify the further possible influence of the nucleating agents. For this purpose, an RPA from Montech was used with the vulcanization settings at 180°C for 3 min with an angle of 1° and a frequency of 1.67 Hz. The density of the foamed elastomer was examined on the profiles produced using a YDK04 density measuring set from Sartorius with demineralized water. Furthermore, the Shore A hardness was examined using an automatic testing machine from Bareiss Prüfgeräte GmbH. The foam structure was determined using a CamScan MV 3200 scanning electron microscope from Electron Optic Services Inc.
Results
The individual results of the material and profile analysis are explained below. The mixture containing all of the proportions described above, but without the nucleating agents, serves as a reference. The cellular structure of foamed components significantly influences their properties. The introduction of foamed structures reduces mechanical properties compared to compact profiles; however, this reduction is less pronounced in fine-celled structures with a high cell density and small cell size.
Rubber process analyser
The evaluation of the RPA curves showed that the incorporation of the blowing agent led to a decrease in the elastic torque, regardless of the type of crosslinking system used. The incorparation of the nucleating agent also led to an increase in the torque of the peroxides with increasing nucleating agent content. In the case of addition crosslinking, 0.5 phr talc and calcium also lead to an increase in the torque, whereby a further increase leads to a drop in the elastic torque (Figure 2). Influence of nucleating agents on vulcanization during peroxide crosslinking (left) and addition crosslinking (right).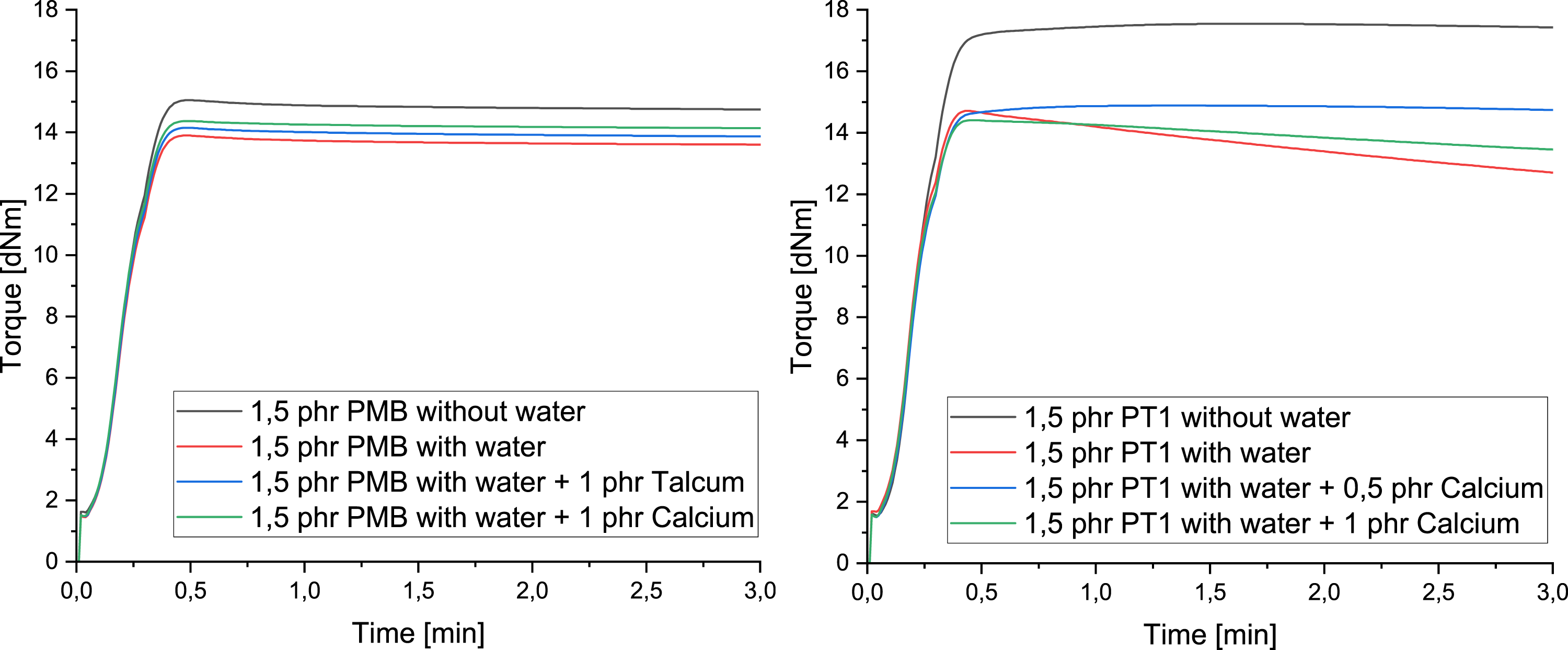
Hardness
When evaluating the hardness parameters, the samples foamed with peroxide initially show a very slight drop in hardness values, regardless of the type of nucleating agent used. However, as the nucleating agent content increases, it approaches the reference value again. In the samples with addition crosslinkers, the addition of talc and calcium leads to varying increases in hardness due to the nucleating agents. However, the increase in hardness from 8 to 37 Shore is most pronounced with the addition of 1 and 1.5 phr calcium carbonate, as shown in Figure 3. Influence of nucleating agents on the hardness of peroxide crosslinking (left) and addition crosslinking (right).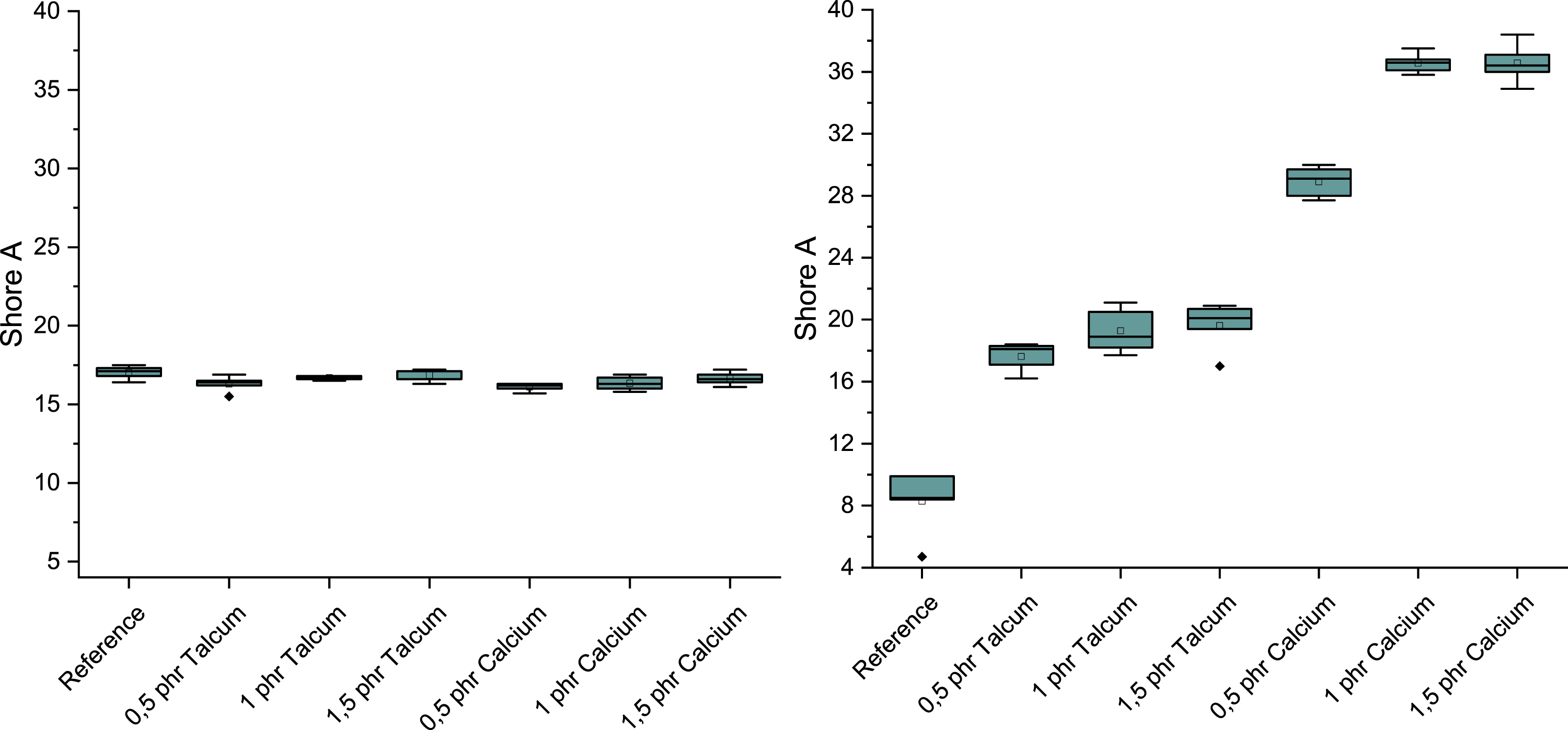
Density
When analyzing the density values, clear differences between the two crosslinking systems and the influence of the nucleating agents became apparent (Figure 4). In the case of peroxide crosslinking, there was no significant influence on the measured density, regardless of the type and quantity of additive used. With addition crosslinking, on the other hand, there was a sharp increase in density with talc at 0.5 phr and a further increase with an increase to 1 phr talc. This increase was even more pronounced with calcium carbonate than with talc, where the density initially rose to approx. 0.8 g/cm3 at 0.5 phr calcium and then to over 1 g/cm3 at 1 phr. Influence of nucleating agents on the density of peroxide crosslinking (left) and addition crosslinking (right).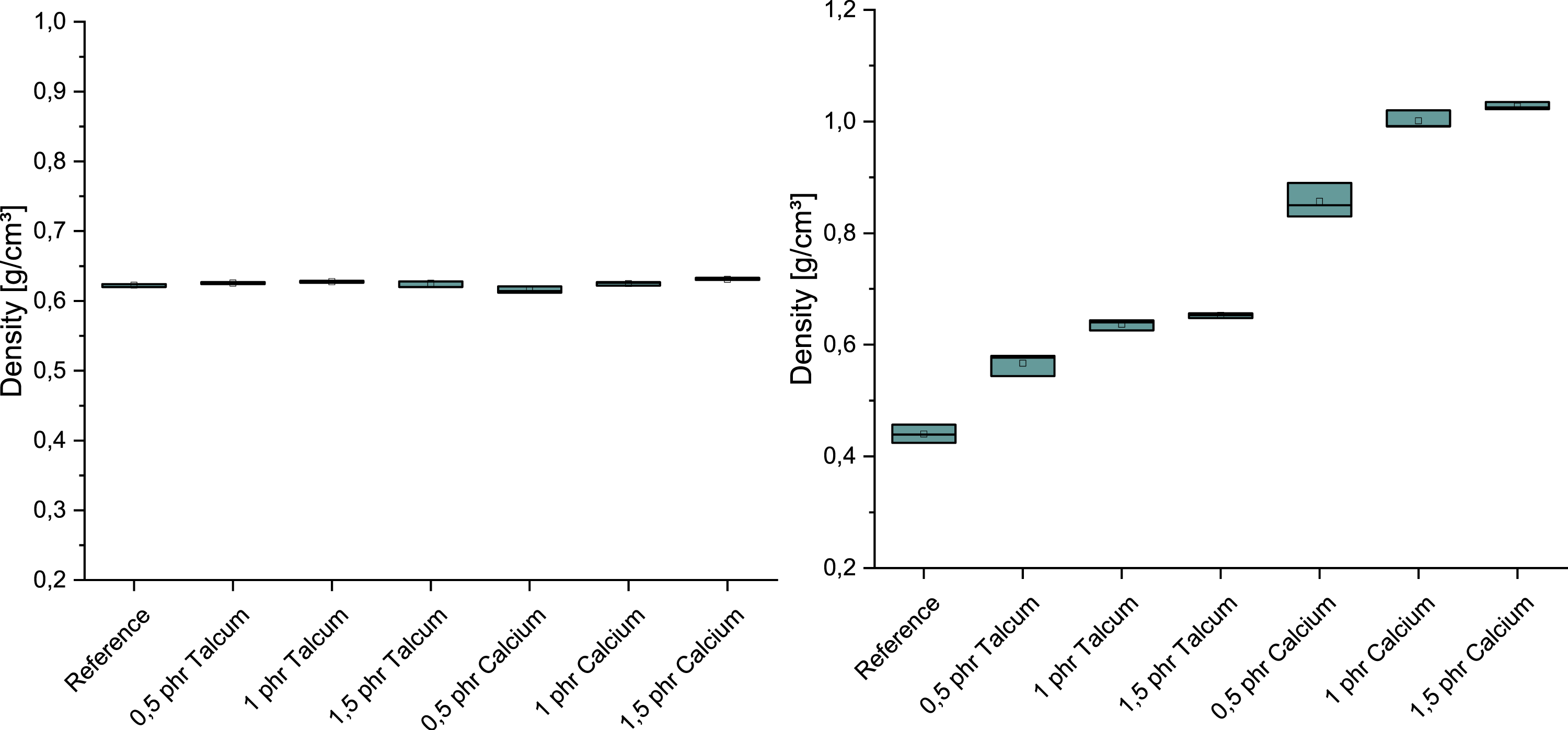
SEM
Figure 5 shows the change in the silicone structure caused by the formation of foam. As can be seen, the material in its unfoamed and uncrosslinked state initially has no pores, but these develop during the extrusion process in the vulcanisation tunnel, simultaneously with crosslinking. Unfoamed rubber (left) and foamed elastomer after vulcanization (right) at a magnification of 60X.
The evaluation of the foam structure (Figure 6) showed that both nucleating agents lead to a reduction in the average pore size during peroxide crosslinking, whereby the minimum is reached for both types with an amount of 1.5 phr. With regard to pore density, it is clear that both agents lead to an increase in pore density as the proportion increases. With addition crosslinking, the influence of the nucleating agents is different compared to peroxide crosslinking. With calcium carbonate, there is a reduction in pore formation with increasing proportion, which is close to 0 at 1.5 phr. Both the pore size and the pore density decrease. In contrast, this trend is not discernible for talc, where the pore diameter also decreases, but at the same time there is an increase in pore density. Influence of nucleating agents on pore size and pore density in peroxide crosslinking (left) and addition crosslinking (right).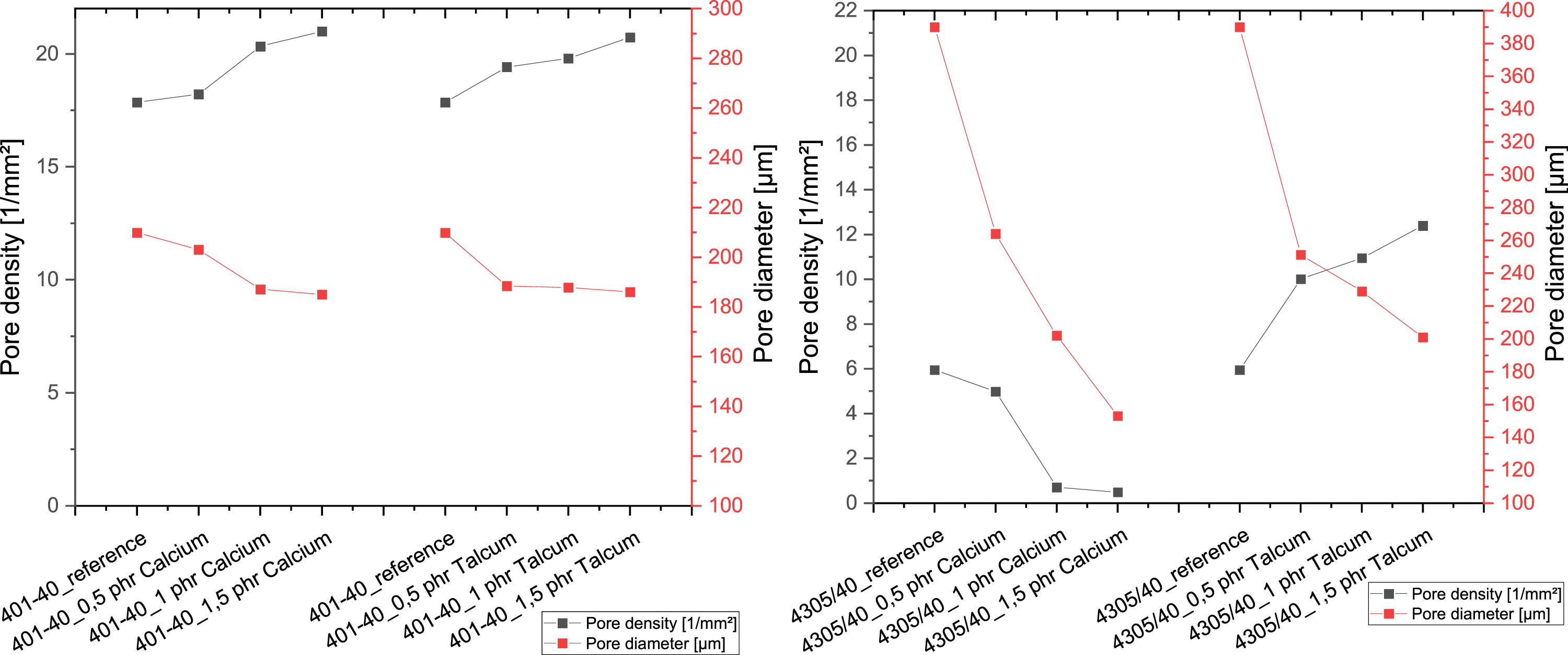
Discussion
As can be seen in the RPA measurements presented, the blowing agent has an influence on the measured vulcanization behaviour or torque, regardless of the type of crosslinking system. This can be explained by the formation of microcavities in the test specimen during the measurement, which was already recognized in studies by Pechurai 24 in the field of rubber. The addition of the nucleating agent leads to an increase in the torque of the peroxide, which is presumably caused by a strengthening effect of the matrix due to the talc and calcium carbonate. However, this effect is not recognizable when compared with the density and hardness values determined, as there is no significant change compared to the reference value, regardless of the type and amount of nucleating agent. The addition crosslinking, on the other hand, provides a different picture.
With addition crosslinking, however, there is initially also an increase in torque until there is no further change from 1 phr. Compared with the hardness and density parameters, parallels become clear here, as the values of 1 and 1.5 phr also differ only slightly. However, here the increasing hardness and density values contradict the decreasing values of the RPA, as these should actually be increasing. It is therefore clear that no further findings about the properties can be derived on the basis of the RPA measurements.
When comparing the foam structure with the mechanical properties of the foam with peroxide, it becomes clear that although the talc and calcium carbonate generally lead to a finer pore structure, this does not significantly change the mechanical properties of density and hardness. However, their influence is different for the samples that were crosslinked with addition crosslinkers, as shown in Figure 7. In the case of calcium carbonate, an increase in hardness and density can be observed due to the simultaneous decrease in pore size and pore density, which can be attributed to the reduction in the total foamed area. One explanation for this could be that the calcium carbonate reacts with the water and the components of the additional crosslinker before it is converted into steam or gas. This would give it a defoaming effect in the process, which would explain the significant reduction in the number of pores as the nucleating agent content increases. In contrast, the influence of the talc on the foam structure and mechanical properties is evident. Although the finer foam structure with increasing talc content also leads to a reduction in the foamed surface area, the decrease is significantly smaller compared to calcium carbonate, which is reflected in the hardness and density. Comparison of the characteristic values determined during addition crosslinking.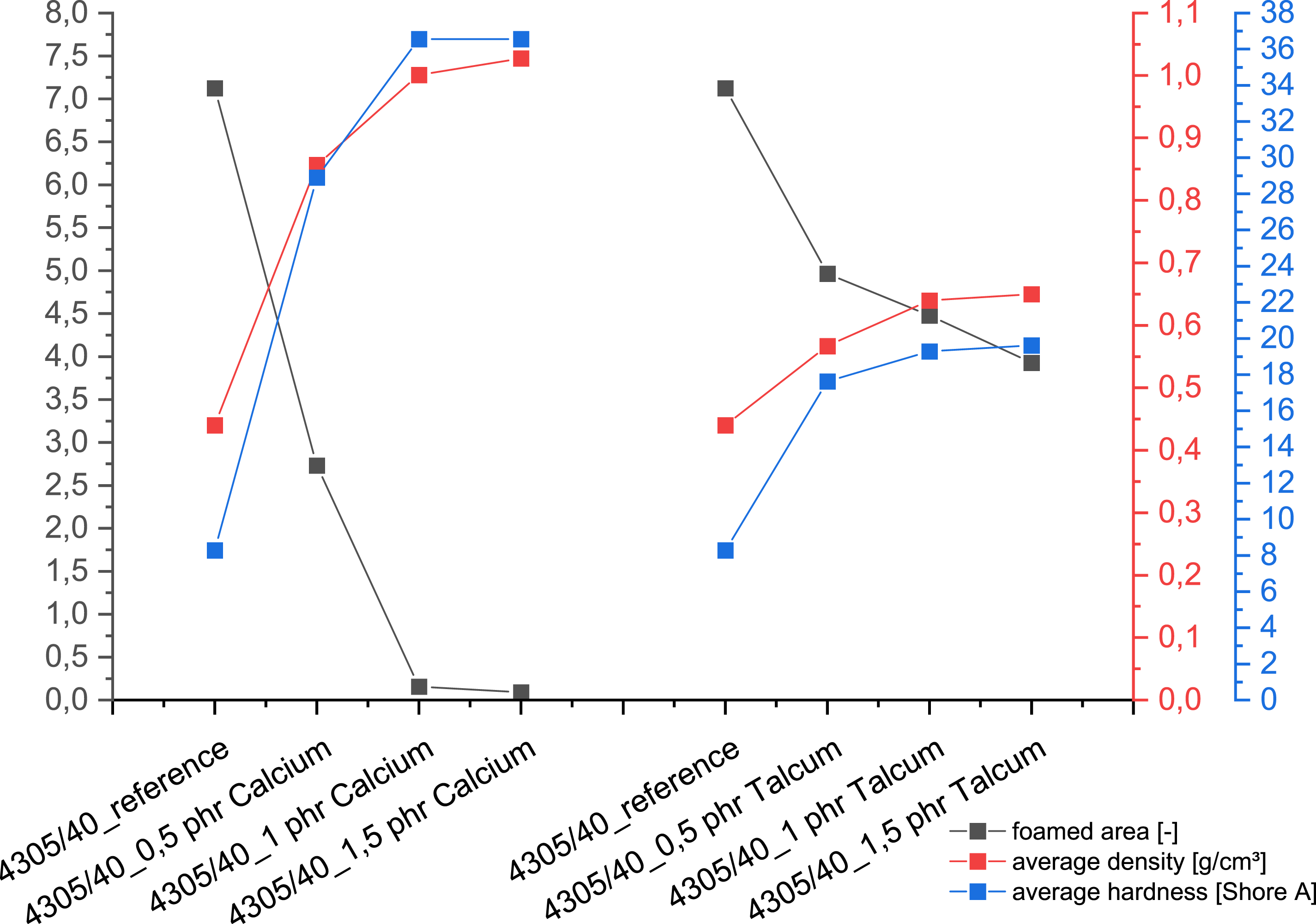
A comparison of the two cross-linking methods also shows that the type of cross-linking has a major influence on the resulting foam structure. The reason for this could lie in the different behavior with regard to the cross-linking mechanism. In the case of peroxide, the cross-linking agent forms radicals, which then attach themselves to other polymer chains by splitting off a hydrogen atom. 25 In addition crosslinking, on the other hand, crosslinking occurs through hydrosilylation, whereby the Si-H groups of the crosslinking agent react with the vinyl groups of the polyorganosiloxanes. 26 However, this can lead to catalyzed dehydrocondensation between the water and the polymer, resulting in the formation of hydrogen. 13 Depending on the crosslinking system, two different blowing gases would be formed: H2O and H2. This would explain the different foaming behaviour.
Conclusion
As part of this work, the influence of the nucleating agents calcium carbonate and talc on the foaming process of solid silicone rubber with different crosslinking systems was investigated. A comparison of the two crosslinking systems showed that the pore density and size can be specifically adjusted when foaming with a water/silica mixture, whereby the influence is greatest with addition crosslinking. Overall, the findings can be summarized as follows: • Talc leads to smaller pore sizes with a simultaneous increase in pore density independent of the crosslinking system • Calcium carbonate achieves a similar effect to talc during peroxide crosslinking, but during addition crosslinking there is a decrease in pore size and pore density • Due to the strong influence of the calcium carbonate on the foam structure, the hardness and density can be adjusted over a wide range during addition crosslinking
Further investigations should analyze the process-related influences on the structure for both crosslinking systems. Of particular interest here are the pressure 22 and temperature27,28 during the processing are particularly relevant here, as previous investigations of similar materials have shown.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
