Abstract
In animal-based toxicology studies, oral gavage is the predominant technique for administering compounds directly into the stomach. Recognised as a stressful procedure for animals, gavage is associated with accidents and ‘gavage-related reflux’ (GRR), which may lead to respiratory distress and/or death. This paper estimates the prevalence of incidents (deaths and/or non-fatal clinical signs) attributed to the gavage procedure in a representative sample of repeated dose toxicity test reports from the last 20 years, sourced from the ECHA CHEM database of REACH-registered chemicals. In 21% of the 300 reviewed studies, at least one death or clinical sign in rats was attributed to the gavage procedure in the study report. An additional 16% of these studies reported clinical signs associated with GRR in the animals, with no other adverse effects, but without attribution to the gavage procedure. Among those studies with incidents attributed to the gavage procedure, incidents were three times more common in 90-day studies than they were in 28-day studies. Incidents were also more likely to have occurred with the test substance rather than with the control, with highly viscous substances, and in more recently conducted studies. There was no association between the number of studies with a gavage-attributed incident and the strain, size or age of the rats, the dose volume or the testing of irritant substances. The current study suggests that incidents attributed to gavage are more prevalent than previously thought, and that a review of the gavage procedure is urgently needed.
Introduction
Gavage is one of the most frequently used methods for the oral dosing of substances in studies involving animals. 1 The technique involves the manual restraint of a conscious animal and the insertion of a rigid metal hollow needle or flexible plastic tube through the oesophagus to deliver a bolus of substance directly into the stomach. Although it is technically demanding, one of the advantages to the technique, in contrast to dosing in food, is that it more readily provides confidence that each animal has received the intended dose. 2 However, it is considered to be the most stressful of the oral administration routes for the animal. 3 The majority of studies that have examined the impact of the procedure have focused on rodents, as these are the most commonly used animals in biomedical research and testing. These studies have reported that the procedure can lead to increased blood pressure and heart rate in rodents for approximately 30 minutes,4,5 as well as elevated body temperature 4 for up to one hour post-procedure. Other studies have found that the technique elevates blood corticosterone levels for some time after the procedure,6,7 with effects lasting up to four hours. 8 Studies have also reported a reduction in weight gain over time 9 or, in some cases, weight loss, 10 along with effects on the liver. 11 Reviews of studies such as these have concluded that the procedure causes stress12,13 and should be avoided, particularly in longer-term studies.1,6
Repeated gavage has also been implicated in lesions in the oral cavity,14,15 in the trachea 16 and in the lungs.17,18 In a rat carcinogenicity study, a mortality rate of 53% was attributed to oesophageal impaction of food and bedding. 16 The authors concluded that chronic oesophageal irritation from repeated gavage led the rats to overeat in an attempt to alleviate the symptoms. 16
The technique also carries the risk of technical errors, such as accidental insertion of the gavage tube into the lungs (leading to suffocation), or puncturing of the oesophagus, trachea or stomach with the gavage needle.1,19 These incidents typically result in rapid death, 20 and are often attributed to errors made by the technician performing the procedure.19–22 However, otherwise unexpected respiratory signs or death in studies using gavage in rodents have also been reported.2,20 A three-week study by Damsch et al. 20 that used the gavage procedure, resulted in the death of 13% of the rats. This prompted the authors to coin the term ‘gavage-related reflux’ (GRR). 20 The deaths were preceded by recurrent dyspnoea (laboured breathing), rales (noisy breathing) and/or excessive salivation. The authors proposed that the deaths were not due to gavage accidents, but to breathing difficulties caused by inadvertent aspiration of the test substance. They identified two possible mechanisms for this: 1) spontaneous reflux of the test substance into the nasal cavity due to an overly full stomach; or 2) mechanical reflux, where small droplets from the tip of the gavage needle entered the nasal cavity during needle withdrawal. Upon subsequent inhalation, some of the substance may enter the lungs. Rats are obligate nose breathers and are virtually anatomically incapable of emesis. 20 This unique physiology has led to rats evolving a highly sensitive sense of taste and smell to prevent them from ingesting harmful foods. In cases where rats do ingest something harmful, they may engage in pica (eating non-nutritive substances), which might explain the observed eating of bedding in the study mentioned above. The authors of the GRR theory suggest that because of this physiological characteristic, if the conditions are met, rats may experience reflux rather than emesis following gavage. 20
Damsch et al. 20 pointed out that it is difficult to differentiate between compound-related respiratory effects, technical gavage errors and GRR. They suggested that a gavage accident is characterised by the sudden onset of audible respiratory signs prior to death, with histopathological evidence of erosions in the larynx, trachea or bronchi, as well as inflammation, oedema or bleeding in the lungs. In contrast, GRR is characterised by recurrent respiratory signs following dosing, nasal cavity erosions and changes in the lungs that are indicative of low-level irritation. The authors also noted that compound-related respiratory effects are unlikely to cause audible breathing problems.
It has been suggested that certain properties of a substance may increase the likelihood of a gavage incident, whether by accident or GRR, although no statistical analysis has been published. The testing of irritant substances has been implicated,16,20,23,24 potentially explaining signs such as oesophageal irritation and salivation. The use of highly viscous substances8,20,24 or higher dose volumes4,8,20,25 has also been implicated, which would support the spontaneous reflux mechanism hypothesis. It has also been reported that studies of longer duration are more likely to be associated with gavage incidents, which could be a result of continued irritation or the increased statistical likelihood of accidents with repeated gavage procedures.9,18 The Fischer 344 strain of rat 16 and mice 6 have been suggested to be at higher risk of gavage incidents, and we hypothesise that this could be due to their relatively smaller size compared to Sprague Dawley rats, making them more difficult to handle during dosing.
There have been attempts to refine the procedure with the use of rubber tubes instead of steel needles, 23 but it is not known whether this has had an impact on the rate of incidents. Suggestions have been made to sugar-coat the gavage needle 26 or even anaesthetise the animal,10,27 the latter of which was reported to reduce the risk of gavage incidents.10,27 Voluntary dosing by hiding the substance in attractive food items has been shown to be a successful and less stressful method of delivery in several studies.6,14,28–30 Rodents have also been trained to accept being fed by syringe with substances in a sweetened solution.7,26,31,32 However, it has not yet been demonstrated that such techniques could be routinely used in regulatory toxicology studies where, for example, the animals may be required to consume a significant portion of a potentially noxious substance daily for up to 90 days (up to 1000 mg/kg body weight, equating to around 200 mg per day). It should also be noted that daily anaesthesia may carry an increased welfare burden, 33 and so may not be an effective mitigating option. Currently, therefore, the procedure remains the preferred technique for oral administration in regulatory toxicology studies. 1
There are several reports in the literature of high rates of respiratory signs and/or sudden deaths in academic studies that have used the gavage procedure in rodents.10,16,19,23,24,34 It has been suggested that the rates of gavage accidents should be lower in toxicology studies compared to academic studies, as technicians in these settings perform the procedure routinely and may therefore be more competent.21,22 However, the actual prevalence of gavage accidents or clinical signs of GRR is not known. Some studies have experimentally investigated the risk and reported no significant negative effects on the animals following repeated gavage, but these studies primarily used behaviour and weight gain as indicators rather than signs of GRR.19,35,36 To date, no systematic review has been conducted to determine the prevalence of gavage incidents across the regulatory sector. The current study was conducted to provide this information.
A fair and robust approach necessitates the evaluation of a representative sample of studies conducted in a consistent manner. To this end, the robust study summaries of repeated dose toxicity tests using rats that had been submitted under the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) regulation 37 were obtained from the European Chemicals Agency’s (ECHA) publicly available database, ECHA CHEM (https://www.echa.europa.eu/web/guest/information-on-chemicals). These tests are performed in a similar manner according to Organisation for Economic Co-operation and Development (OECD) Test Guidelines (TGs) 407 38 and 408. 39 The robust study summaries available in the ECHA CHEM database are not full study reports — however, they must contain the results and a discussion of them (see https://echa.europa.eu/support/registration/creating-your-registration-dossier/what-is-iuclid-). The public availability of these summaries provides the opportunity to assess, for the first time, the prevalence of gavage incidents and to test the hypotheses that substance properties, dose volumes, dosing frequency and rat strain may affect the likelihood of a gavage incident.
Methods
The objective of this study was to determine the prevalence of reported gavage incidents over the last 20 years in regulatory repeated dose toxicity tests involving rats. It took the form of a systematic review, examining reports from a single source, and was conducted in accordance with the reporting principles for systematic reviews. 40 The presentation of the search results follows the PRISMA statement, as applied by Yao et al. 41 The raw data can be supplied upon request.
Search strategy and database
Search strategy.

Screening and selection process
Record inclusion and exclusion criteria.
Selection of studies for review
Number of records at each selection stage.
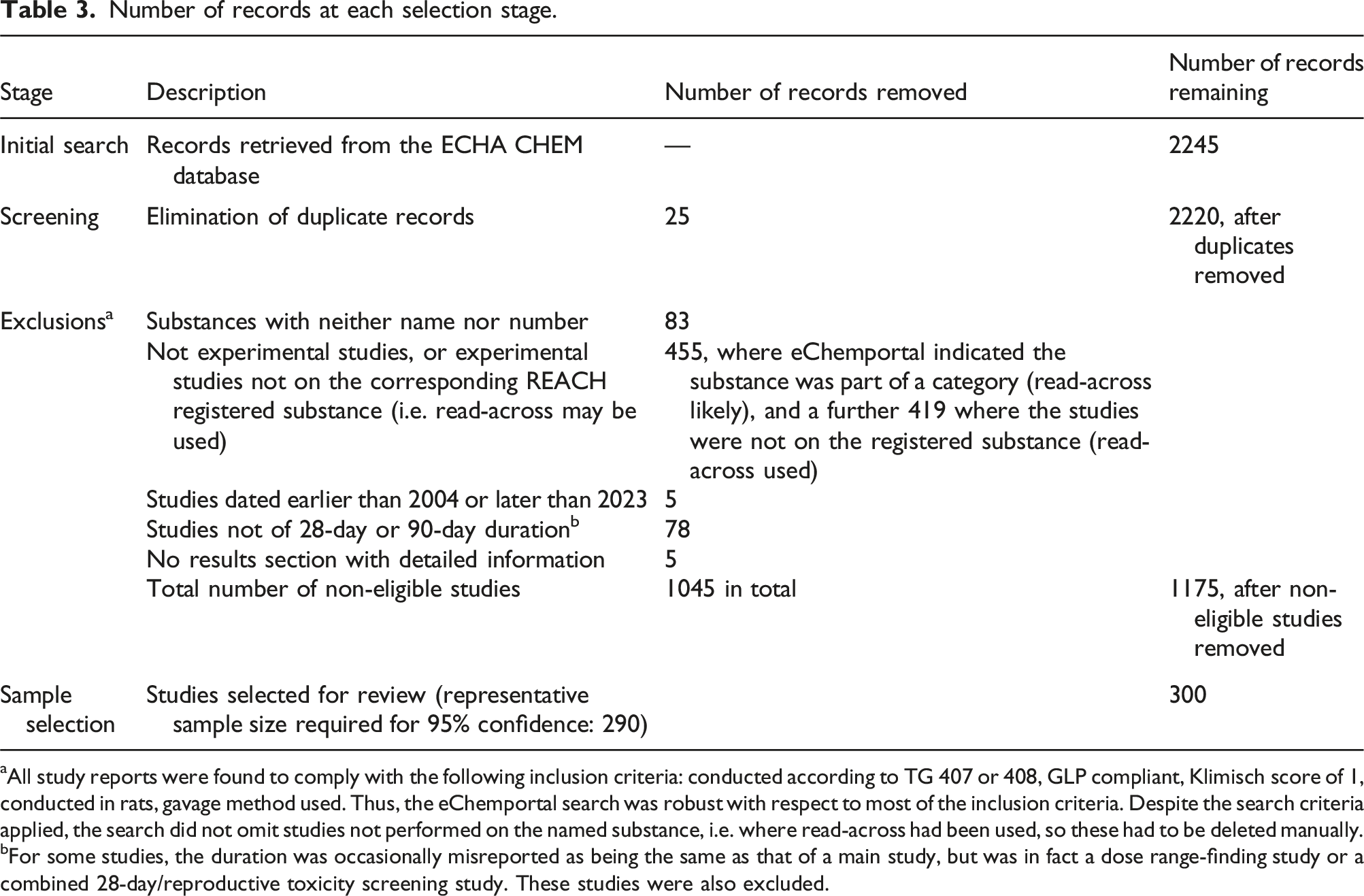
aAll study reports were found to comply with the following inclusion criteria: conducted according to TG 407 or 408, GLP compliant, Klimisch score of 1, conducted in rats, gavage method used. Thus, the eChemportal search was robust with respect to most of the inclusion criteria. Despite the search criteria applied, the search did not omit studies not performed on the named substance, i.e. where read-across had been used, so these had to be deleted manually.
bFor some studies, the duration was occasionally misreported as being the same as that of a main study, but was in fact a dose range-finding study or a combined 28-day/reproductive toxicity screening study. These studies were also excluded.
Some substances had more than one repeated dose study associated with them. If a study was selected that featured a substance that had been the subject of an already-selected study, then the next random number was chosen. This ensured that all of the studies selected for review were unique with regard to the substances tested. This was done to ensure complete independence within the data set, which was important when testing the hypotheses relating to the properties of the substances. The study reports were then reviewed by the lead author alone and the data items listed below were recorded.
Data items
A study was considered to have had a gavage incident if the study report attributed sudden deaths or observed clinical signs to the gavage procedure itself, rather than the test substance, in at least one animal. We noted whether the report identified the cause as a gavage accident (i.e. technical administration error) or reflux (i.e. aspiration), and the basis for that conclusion, such as histopathology. We recorded the number of studies that reported the presence of the following key clinical signs associated with GRR2,20 in animals that died prematurely, or in those exhibiting non-fatal clinical signs following dosing during the study period. This was done both for studies where gavage was attributed as the cause and those where it was not. The key clinical signs were: — laboured breathing, gasping, shortness of breath (dyspnea) or rales (audible respiration); — excessive salivation (ptyalism); — nasal discharge (rhinorrhea); or — eating or rooting in bedding or chin rubbing. — The percentage of studies with one or more deaths attributed to the gavage procedure in the study report, the average number of animals affected per study and the overall prevalence of deaths. A t-test was used to assess whether deaths attributed to the gavage procedure were significantly more likely in the treatment groups. — The percentage of studies reporting one or more animals with non-fatal clinical signs attributed to the gavage procedure in the study report. — The total percentage of studies affected by a gavage incident (deaths and/or clinical signs attributed to the gavage procedure, either an accident, GRR or both).
The following outcomes were determined:
Variables
The variables recorded.
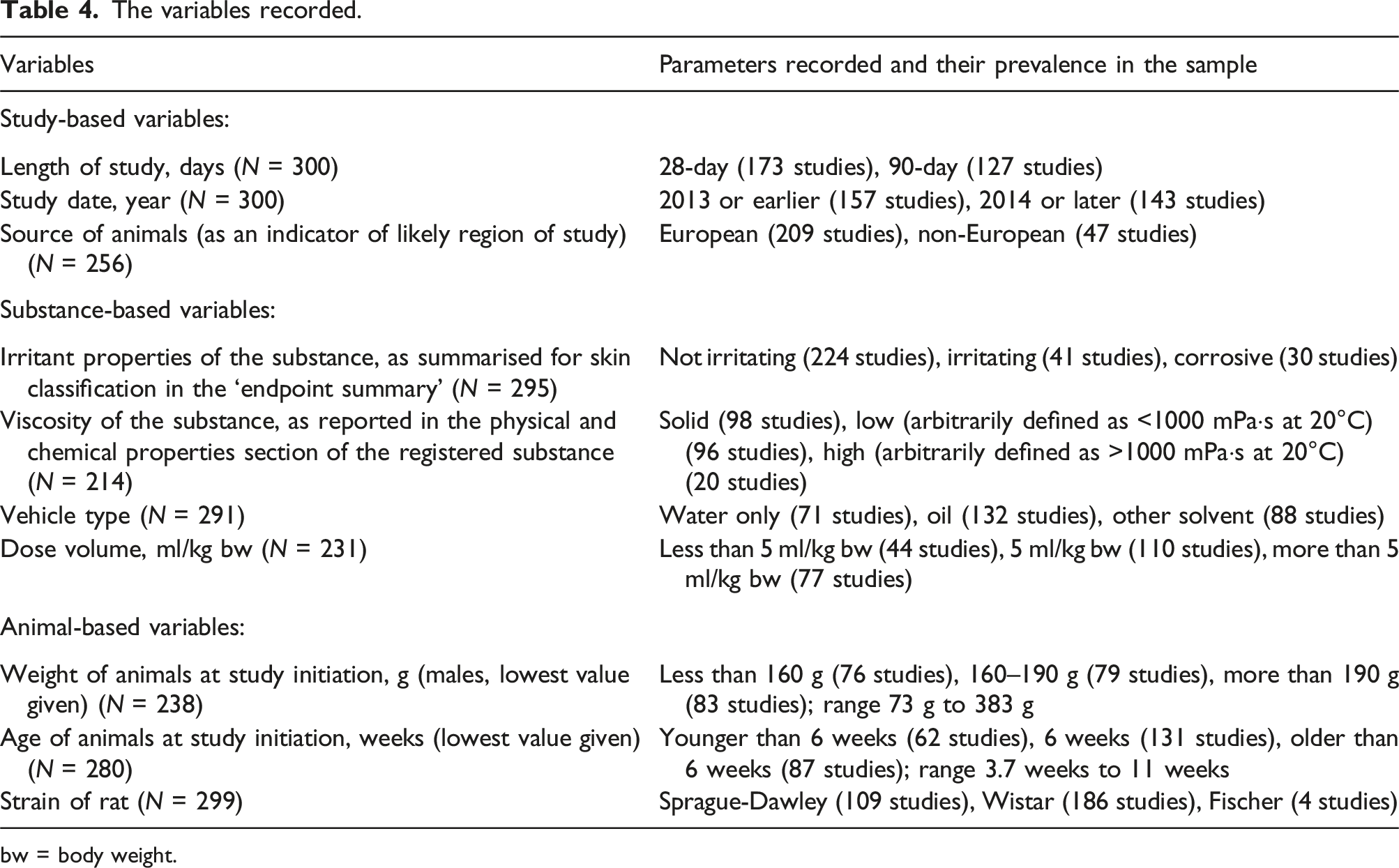
bw = body weight.
It should be noted that all studies included an equal number of males and females as standard. The type of gavage needle was only occasionally specified in the study reports, so it was noted when the information was available. The competence of personnel was not reported and is assumed. In both 28-day and 90-day studies, dosing occurred once daily, resulting in 28 doses for 28-day studies, and 90 doses for 90-day studies. The location of test facilities is not provided in the ECHA CHEM database; therefore, the source of the animals (European or non-European) was used as the proxy, based on the assumption that, in regulatory testing using standard-bred rodents, European laboratories are unlikely to source animals from non-European laboratories, and vice versa.
Chi-square analysis (2 × 2 table) was used to determine whether studies with incidents attributed to the gavage procedure in the study report were more likely to be of longer duration, less recent in terms of study year, from non-European regions (study-based variables), and whether they involved irritant substances or viscous substances. The analysis also considered the type of vehicle used, the use of higher dose volumes (substance-based variables), and whether the study involved smaller, younger animals or a particular strain of rat (animal-based variables).
Results
Prevalence of reported gavage incidents
Out of the 300 reviewed studies, 41 reported one or more deaths attributed to the gavage procedure, accounting for 14% (or one in seven) of the studies. The percentage of affected animals ranged from 1 to 12.5% of the animals used, with a weighted mean of 2.2%. A total of 78 deaths were reportedly attributed to the gavage procedure out of a total of 20,180 animals used in 300 studies. Therefore, the probability of any one animal reported dying as a result of the gavage procedure alone was 0.4% (or 1 in 250).
Studies with a gavage incident.
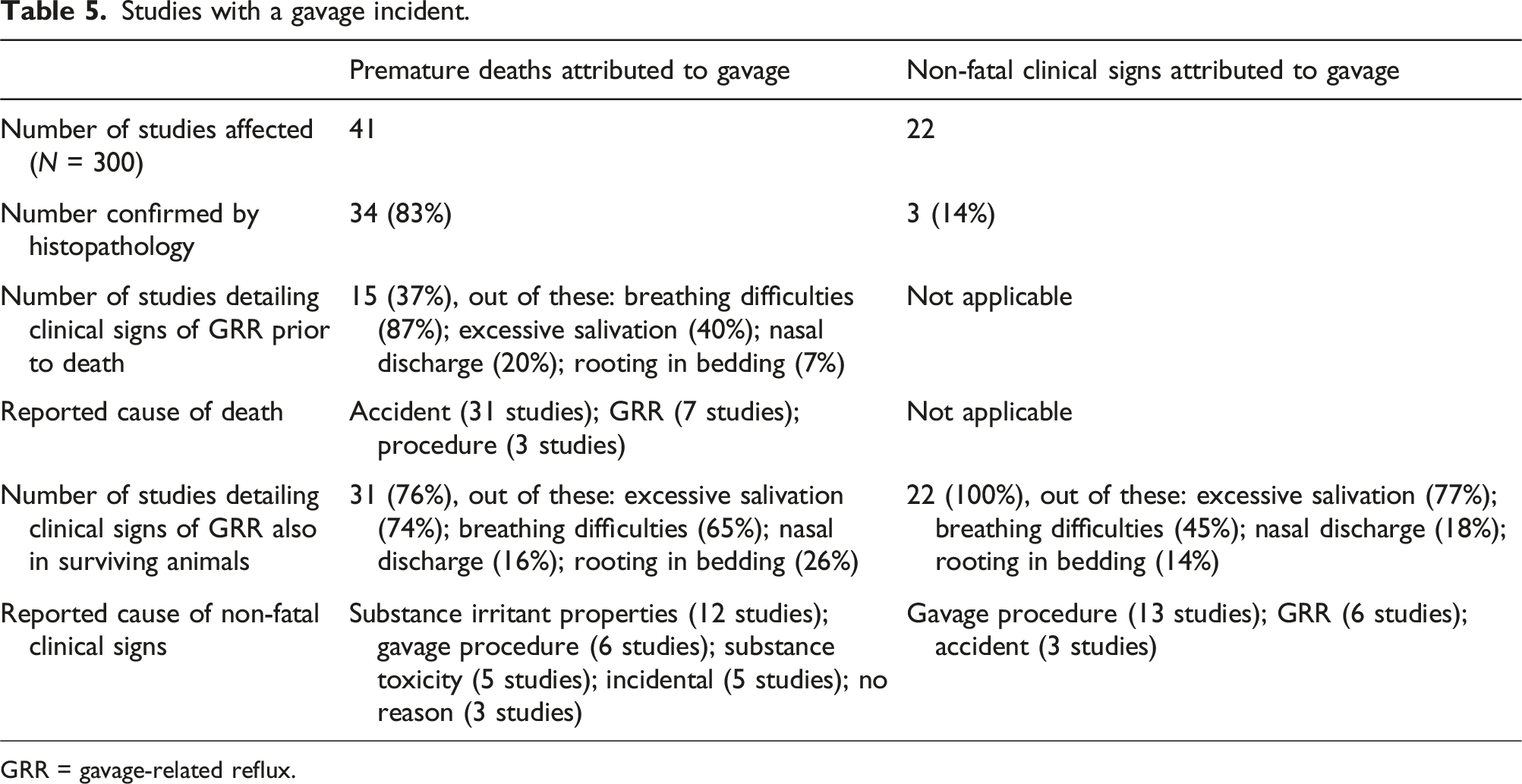
GRR = gavage-related reflux.
The probability of a repeated dose toxicity study, conducted in the last 20 years and recorded in the ECHA CHEM database, having an incident (i.e. death and/or clinical sign) attributed to the gavage procedure in the study report was therefore 21% (one in five), as shown in Table 5
Presentation of gavage-attributed incidents
Only 37% of the studies reporting animals that died prematurely due to a gavage incident also detailed the clinical signs associated with the gavage incident prior to the animal’s death (see Table 5). Among these, breathing difficulty was the primary sign (87% of those with signs), while excessive salivation was also noted in 40% of the reported cases. The majority (83%) of the gavage-attributed deaths were confirmed by histopathology. Fluid or blood in the lungs was reported in 15 cases, fluid or blood in the thoracic cavity in 12 cases, and perforation of the trachea or oesophagus in seven cases (all reported to be gavage accidents). Signs of damage or irritation were also observed in the trachea (4 cases), stomach (3 cases), lungs (3 cases) and nasal cavity (1 case), with some reports indicating more than one histopathological finding.
The cause of death was attributed to a gavage accident in 31 studies (76%), to GRR in 7 studies (17%), and to the gavage procedure in 3 studies (7%), without further specifying whether it was due to an accident or GRR. While statistical analysis was not possible, sudden death — sometimes preceded by breathing difficulties and accompanied by histopathological findings consistent with perforation of the trachea, or oesophagus, and/or blood or fluid in the thoracic cavity — was generally reported as a gavage accident. Other histopathological signs, such as contents in the lungs, were equally attributed to either a gavage accident or GRR.
Of the studies with gavage-attributed deaths, 31 (76%) also reported clinical signs in surviving animals that are consistent with GRR in the literature.2,20 Breathing difficulties were reported in 65% of these 31 studies, excessive salivation in 74%, nasal discharge in 16% and rooting in or eating bedding in 26%. However, 81% of these 31 studies did not attribute the signs to the gavage procedure. Instead, they attributed the signs to the irritant properties of the substance (12 reports) or its toxicity (5 reports); in 8 reports, no explanations for the clinical signs were given.
An additional 22 studies reported non-fatal clinical signs associated with GRR that were attributed to the gavage procedure. Breathing difficulties were reported in 45% of these studies, excessive salivation in 77%, nasal discharge in 18% and eating or rooting in bedding in 18%. The signs were mostly attributed to the gavage procedure in general, with accidents specifically implicated in 14% of cases and GRR in 27% of cases (Table 5).
We also identified 48 studies (16% of the total) where one or more of the four signs associated with GRR were reported prior to unexplained sudden deaths or in surviving animals, with no other adverse effects reported apart from incidental reports of alopecia and scabs. Of these 48 studies, only 6% reported sudden deaths. Out of the remainder, 67% reported excessive salivation only, while 33% reported more than one clinical sign. The reasons provided for these signs included the unpleasant taste or irritant properties of the substance, or occasionally its toxicity.
Risk factors
Study-based variables
Risk factors for incidents attributed to gavage in reports.
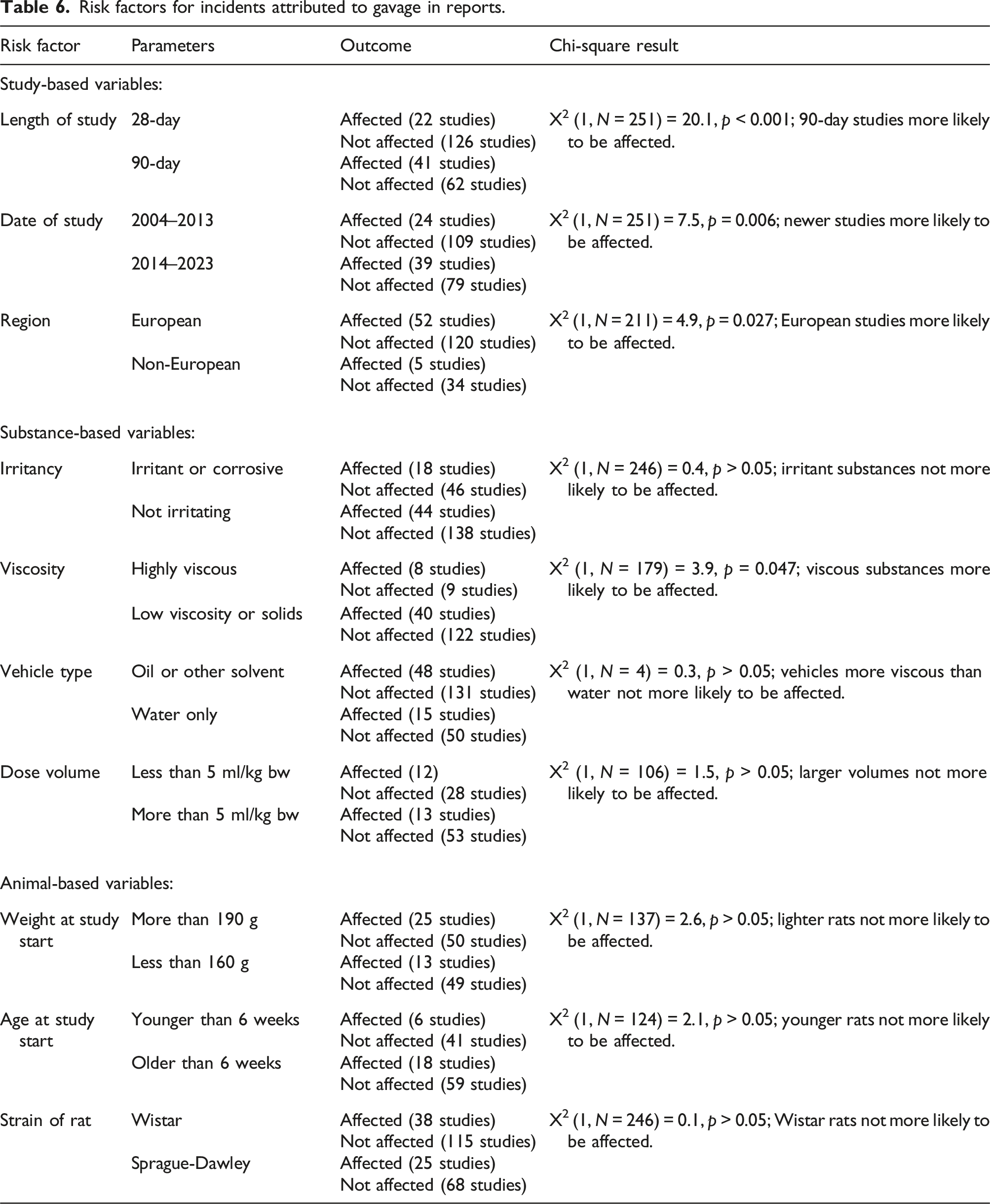
Substance-based variables
Out of those studies where the death was attributed to the gavage procedure in the study report, deaths were more likely to occur in the treatment groups than they were in the control groups (t-test, mean number of deaths in control groups 0.23, mean number of deaths in treatment groups 1.64, t = −6.00, d.f. = 38, p < 0.001).
There was no association between the testing of irritant substances or the use of larger dose volumes and the likelihood of a study having an incident attributed to the gavage procedure in the study report (Table 6). There was a marginally significant effect of viscosity, with highly viscous substances more likely to have an incident attributed to the gavage procedure in the study report. However, there was no effect of vehicle type (water only, compared with other solvents of varying but higher viscosity than water).
Animal-based variables
There was no association between the weight, age or strain of the rats used and the likelihood of a study having an incident attributed to the gavage procedure in the study report (Table 6). Fischer rats were rarely used, so only Sprague Dawley and Wistar strains could be compared.
Discussion
Prevalence of reported gavage incidents
This is the first survey to assess the prevalence of reported gavage incidents across a specific area of animal use. To ensure consistency in the type of studies reviewed, and because these reports were publicly available, we focused on repeated dose testing of chemicals submitted under the EU REACH regulation.
Of the 300 reviewed studies, 21% were affected by deaths and/or clinical signs attributed to the gavage procedure. Among these, 65% reported one or more deaths attributed to the gavage procedure, accounting for 14% of the total sample. Overall, the risk of an individual animal reported as dying as consequence of a gavage incident was 1 in 250. These proportions suggest that gavage incidents may be more prevalent than previously thought, even in regulatory toxicology studies, where it has been suggested that such accidents should be rare due to the technicians’ experience in performing the procedure.21,22
Overall, 52% of the incidents were attributed to a gavage accident, 21% to GRR, and the remainder did not specify which aspect of the gavage procedure caused the deaths or non-fatal clinical signs. Deaths were most commonly attributed to gavage based on histopathological evidence of consequential trauma to the lungs, trachea, oesophagus or stomach. Most deaths occurred suddenly without preceding signs. However, 76% of the studies with deaths attributed to gavage also reported clinical signs that have been associated with GRR2,20 in the surviving animals, namely: breathing difficulties, excessive salivation, nasal discharge and/or eating or rooting in the bedding. Despite reporting that one or more of their animals had died as a consequence of the gavage procedure, the authors of only 19% of these studies also attributed the observed non-fatal clinical signs to the gavage procedure. This suggests that non-fatal clinical signs are more likely than deaths to be overlooked and not attributed to the gavage procedure, despite their potential association as proposed by the authors of the GRR theory.2,20,34 Additionally, 16% of studies in the sample reported one or more of these same clinical signs in the animals used, with no other adverse effects. However, they did not attribute these signs to the gavage procedure, but were instead considered incidental, due to factors such as the unpleasant taste or irritant properties of the substance, or occasionally the toxicity of the substance.
Interestingly, there was no decrease in the proportion of studies affected by gavage incidents in the last 10 years (2014 to 2023). In fact, the proportion has nearly doubled compared to the previous decade (2004–2013). This may be attributable to the improved quality of reporting in toxicology studies in recent years, as a consequence of the REACH regulation implemented in 2007 37 that required the submission of full data packages that could be subject to compliance checks by ECHA. 42 Our analysis also revealed that studies that were assumed to have been conducted in Europe based on the source of the animals, were more likely to have reports of gavage incidents. This may be due to a greater familiarity with REACH reporting requirements among test facilities located in the EU.
Risk factors
The higher incidence of gavage-attributed deaths in the treatment groups compared to the control groups (that also undergo gavage but with an inert vehicle), suggests that the properties of the substance may influence the likelihood of a gavage incident. Previous studies have similarly reported a higher incidence of deaths in the treatment group(s).2,16,23,24 Additionally, associations have been made between increased risk and the administration of irritant or highly viscous substances by gavage.8,16,20,23,24 Interestingly, those studies that reported few or no adverse effects from the gavage procedure only had water administered.35,36 Whilst no clear association between gavage incidents and the testing of irritant substances was observed in our survey, we did find that highly viscous substances were slightly more likely to be associated with an incident attributed to gavage in the study report. This observation may support the hypothesis that GRR is more related to the movement of the substance into the nasal cavity, 20 rather than a direct physiological reaction to irritation. Excessive salivation, which is the most commonly reported clinical sign of GRR, warrants further consideration, as it may result from the substance being expelled from the animal’s nasal and oral cavities rather than a physiological reaction to irritation. Notably, if some of the substance is being expelled through the nose or mouth, the animal may not be ingesting the full dose that was administered, which could have implications for the study’s validity. Jones et al. 27 observed that awake mice undergoing gavage had a 20-fold higher incidence of incomplete retention of the administered saline compared to anaesthetised mice. To our knowledge, no further studies have explored the scientific implications of this observation.
We also did not observe an effect of dose volume, which is surprising if the spontaneous mechanism of GRR is the correct hypothesis. This hypothesis suggests that GRR may result from the animal being administered too large a volume of material, forcing it up the oesophagus and into the nasal cavity.4,8,20,25 Consequently, several studies have recommended using dose volumes of less than 10 ml/kg body weight (bw).2,20,43 The majority of studies in our survey used lower volumes — 5 ml/kg bw was the most common dose volume, compared to the maximum 10 ml/kg bw recommended in the relevant OECD test guidelines TG 407 38 and 408. 39 Despite this, we found a prevalence of reported gavage incidents in 21% of the reviewed studies (i.e. at least one death or clinical sign was reported), suggesting that gavage incidents can still occur at low dose volumes. Given that a high proportion of incidents were attributed to technical accidents, dose volume may not be a major contributing factor here. Some studies have reported deaths with doses as low as 1 ml/kg bw, 23 further suggesting that dose volume may not be a determining factor. However, if at the time of dosing the rat’s stomach is full of food, 20 even low volumes could be sufficient to induce spontaneous reflux. 8 In repeated dose studies, animals are not fasted overnight, 8 as depriving them of food during their peak feeding period over long periods would negatively affect their weight and welfare.44–46 Some studies have suggested that shorter periods of fasting (six hours or less) can result in a sufficiently empty stomach and should be used if fasting is necessary.45,46 Fasting prior to gavage dosing has been suggested as a way to reduce the risk of GRR.2,20 It might be possible that careful consideration of the timing of the gavage procedure — for example, at the end of the day if food is withdrawn during the day when the animals are less likely to eat — could reduce the risk of GRR, whilst minimising any further impact on the animals’ welfare.
We did observe an effect of study length, as noted by other authors,9,18 with 90-day studies being more prone to gavage-attributed incidents than 28-day studies. In fact, 90-day studies were nearly three times more likely to be affected by a gavage incident. This may reflect the approximately three-fold increase in the number of gavage procedures, i.e. 90 versus 28. It is not possible to conclude from our study whether the increase in gavage incidents is due to cumulative damage and irritation from the greater number of procedures performed, or simply reflects the increased statistical probability of an accident or reflux occurring when more procedures are performed.
One study reported that the Fischer 344 strain of rats were more likely to experience gavage incidents and hypothesised that they were more susceptible to irritation. 16 Since Fischer 344 rats are smaller than Sprague-Dawley rats, 47 we hypothesised that the effects observed in that study might be related to the smaller size of the animals, which could make them more difficult to handle and gavage successfully. However, we found no difference in the proportion of incidents between the smaller Wistar and larger Sprague-Dawley rats (Fischer 344 rats were actually rarely used), nor an effect of weight or age, which casts doubt on that hypothesis.
Information on the competency of the technician was not provided in the study reports, so we could not investigate any association there. However, it should be assumed that the personnel conducting the procedures are competent. In the EU, individuals conducting procedures on animals must be competent in accordance with EU Directive 2010/63/EU on the protection of animals used for scientific purposes. 48 It could be expected that there would be fewer incidents in Europe-based studies compared to studies from other regions, as the requirements for competence are higher in Europe than elsewhere in the world, 49 but our findings did not support this. It did appear that some studies were notably affected by gavage incidents, including multiple deaths and clinical signs in surviving animals. While this might suggest that the study had employed a poorly trained technician, it could also indicate that GRR contributed to both the deaths and clinical signs, with the properties of the substance also playing a role.
Study limitations
This study relied on the judgement of the authors of the study report to determine whether a gavage incident had occurred, based on the clinical signs and the histopathology of the animal tissue. The study report author is likely to be a senior toxicologist within the test facility, motivated to explain any unexpected signs or deaths in their animals to ensure accurate toxicity assessment of the substance. In most cases, the deaths were confirmed by histopathological findings consistent with the literature on gavage accidents, such as signs of trauma to the respiratory or digestive system resulting from the accidental insertion of the gavage needle into the lungs or surrounding tissues. 34 Furthermore, all studies reporting non-fatal clinical signs attributed to the gavage procedure included one or more of the four key clinical signs or histopathological signs linked with gavage accidents or GRR in the literature.2,20 For these reasons, we are confident that the majority of these reports correctly attributed the deaths and/or clinical signs to the gavage procedure. The prevalence observed could, in fact, be an underestimate, as an additional 16% of studies reported no other adverse effects aside from one or more of the clinical signs associated with GRR.
Damsch et al. 20 did note that differentiating between compound-related respiratory effects, technical gavage errors and GRR can be challenging. They suggested that a gavage accident is typically characterised by the sudden onset of audible respiratory signs prior to death, and histopathological findings that include erosions in the larynx, trachea or bronchi, as well as inflammation, oedema or bleeding in the lungs. In contrast, they described GRR as being characterised by recurrent respiratory signs following dosing, nasal cavity erosions and changes in the lungs indicative of low-level irritation. In our study, we also found that sudden deaths were more likely to be attributed to gavage accidents rather than to clinical signs alone, though we were not able to test this statistically. However, a significant proportion of studies with deaths, most of which were attributed to gavage accidents, also reported non-fatal clinical signs in surviving animals that were also consistent with GRR. As a result, we were not confident in separating the reports into distinct categories of ‘accidents’ and ‘GRR’ for separate risk factor analyses. Consequently, the risk factors identified cannot be definitively linked to accidents, GRR or both. This is particularly challenging since we found that substance properties do appear to play a role — deaths were more common in treatment groups and with viscous substances, but the exact impact of these factors on the likelihood of accidents or GRR remains unclear. Previous studies on this issue have also faced this challenge, often failing to specify whether they are addressing accidents, GRR or both.
The study summaries rarely included information on the material of the gavage needle, so we could not assess its potential impact on the prevalence of gavage incidents. Of those that did report the material, 11 indicated the use of steel needles, while 12 indicated plastic needles. However, the sample size was too small to establish any association with gavage incidents. Since the late 1980s, 23 plastic tubes have been suggested to be less likely to trigger a gavage accident and subsequent studies have indicated that the use of plastic needles can reduce stress4,50 and mortality,16,23 as compared to metal needles. However, plastic needles do not appear to be used exclusively in practice.
Animal welfare implications
According to our survey, the risk of an individual animal used in a 28-day or 90-day study experiencing a death attributed to the gavage procedure, as reported by the authors of the reviewed study reports, is 1 in 250. This corresponds to the ‘uncommon’ rating, according to the system for reporting side effects in medicines. However, the risk of a 28-day or 90-day study being affected by at least one death attributed to gavage by the study report author is one in seven, which corresponds to the ‘very common’ rating. It is important to consider prevalence at the level of the study when considering the requirements of the EU Directive 2010/63 on the protection of animals used for scientific purposes. 48 According to this legislation, the prospective severity of an animal procedure informs the cost–benefit analysis of a project, the outcome of which influences whether it should be authorised in the EU. The severity categories are: mild, moderate, severe and non-recovery (where the animal remains under anaesthesia for the entire procedure). Annex VIII of the Directive provides examples of the prospective severity of some procedures; gavage is included in those considered to cause ‘mild’ pain, suffering or distress, the lowest category of suffering. However, prospective severity is intended to “be based on the most severe effects likely to be experienced by an individual animal [in a study] after applying all appropriate refinement techniques.” 48
If there is a one in seven chance that a death will occur as a direct consequence of the gavage procedure within each regulated study, the classification of ‘mild’ may not be an appropriate severity category for this procedure. Closer attention to technician training and competence in the gavage procedure could potentially reduce the associated risk.
Scientific implications
We found evidence suggesting that the study author did not always associate the clinical signs of GRR, such as respiratory problems and excessive salivation, with the gavage procedure — even in cases where they may have been related. The implication of this is that the substance itself might be identified as the cause of these effects, potentially leading to an overestimate of its potential toxicity. This could impact its use, which would be problematic if the substance otherwise offers economic or other benefits to society and the environment. In fact, we found that the clinical signs of GRR were often dismissed as incidental or attributed to the irritancy of the substance, regardless of whether it was classified as an irritant or not. In contrast, for substances with other reported adverse effects, we noted that the clinical signs that could be attributed to GRR were frequently included in the overall toxicity conclusion, potentially exaggerating the perceived toxicity of the tested substance.
If GRR is being overlooked, the additional stress on affected animals may not be fully recognised. Stress can lead to physiological, metabolic, endocrine and behavioural changes that may interfere with study outcomes. 5 Several authors have identified an association between the gavage procedure and stress parameters.4–8 Roberts et al. 11 suggested that the stress of gavage could cause liver damage, which should be considered when designing toxicology studies. De Meijer et al. 9 reported that gavage-induced stress affected metabolism, indicating that the procedure itself should be taken into account in obesity research. Scarborough et al. 7 proposed that the stress response from gavage could potentially affect neurobehavioural readouts, such as locomotor activity and anxiety-like behaviour. Vandenburgh et al. 51 suggested that gavage-induced stress could alter endocrine responses, and that it should therefore be avoided for the toxicity testing of endocrine disruptors.
There are recent studies suggesting that the gavage procedure should be avoided due to fundamental issues related to absorption. One review of the use of gavage in pharmacological studies highlighted evidence that the gavage route disregards the possibility of absorption in the oral cavity, as it by-passes this area. 1 For studies where absorption and immune responses in the oral mucosa could be relevant, such as in the case of therapeutic drugs, the use of gavage is therefore not appropriate. 1 Similarly, Vandenburgh et al. 51 argued that this administration route is not suitable for toxicology studies because it underestimates effects in the oral mucosa and does not replicate human dietary exposures. Two studies also reported that the gavage procedure caused damage to the oral mucosa and interfered with oral uptake relevant to their research.14,15 Furthermore, one study found that the gavage procedure delayed gastric emptying by up to three hours and interfered with intestinal absorption of the drug. 52 Other studies have similarly observed delayed gastric emptying in gavage-based studies, 34 suggesting that it may contribute to reflux. Stress has been linked to delayed gastric emptying in rats. 53 If the gavage procedure itself causes delayed gastric emptying, it could not only cause reflux in the animals but also interfere with the absorption of the test substance, thus impacting the study results. This could have significant scientific implications, which are not mitigated by the use of control animals, as control animals also undergo gavage.
Conclusions
This is the first survey of any kind to examine the prevalence of reported gavage incidents across a specific sector where the use of gavage is common. We focused on 28-day and 90-day repeated dose toxicity studies located within the EU’s REACH ECHA CHEM database. Our findings indicate that, over the last 20 years, the risk of any one study reporting deaths and/or clinical signs in one or more of the animals as a consequence of the gavage procedure is approximately one in five — and that this prevalence is not decreasing. Furthermore, our study suggests that non-fatal clinical signs of GRR are not consistently recognised as such in a significant proportion of studies. The test substance appears to play a role, as incidents were more common in the treatment groups. We found that the viscosity of the test substance was associated with a higher risk, but not its irritant properties, despite this frequently being cited as a cause in the study reports. While it is not possible to determine whether the risk of gavage incidents would increase with higher dose volumes, this survey indicates that there is a significant risk of gavage incidents in studies using dose volumes of 5 ml/kg bw or less. In our view, a review of toxicology studies that use gavage is urgently needed, on both animal welfare and scientific grounds.
