Abstract
Background
Dietary quality is a critical determinant of reproductive health, yet little is known about how distinct dietary patterns and food processing affect fertility outcomes in the general population.
Aim
To examine the associations between ultra-processed food (UPF) intake and Mediterranean diet (MD) adherence and self-reported fertility among American women of reproductive age.
Methods
We analyzed data from 2582 women aged 20–45 years participating in the 2013–2018 National Health and Nutrition Examination Survey. Fertility status was defined based on self-reported attempts to conceive for ≥12 months. Dietary data collected by two 24-h dietary recalls was used to classify UPFs according to the Nova classification and UPF intake was defined as % grams per day. MD adherence was assessed using a modified 8-point score. Multivariable logistic regression models were used to examine associations, adjusting for sociodemographic and health covariates, including obesity.
Summary
The mean UPF intake was 27.15% of daily intake (g/day) and the mean MD score was 3.35 out of 8. Women reporting infertility had significantly higher UPF intake and lower MD scores compared to fertile women. Higher UPF intake was significantly associated with lower odds of fertility in the fully adjusted model (OR = 0.32; 95% CI: 0.14, 0.75). MD adherence was initially associated with higher odds of fertility (OR = 1.16; 95% CI: 1.00, 1.33), though this association was no longer significant when adjusted for obesity. These findings underscore the need for dietary guidance targeting women of reproductive age.
Keywords
Introduction
Infertility, defined as the inability to conceive after 12 months of regular unprotected intercourse, is recognized as a global health burden affecting approximately 15% of American couples (Thoma et al., 2013). Beyond its clinical implications, infertility is associated with considerable psychological distress, social stigma, economic hardship, and strain on intimate relationships (Sun et al., 2019a). While assisted reproductive technologies have become more widely used, their high cost and limited accessibility have increased interest in identifying modifiable risk factors for infertility (Gaskins and Chavarro, 2018).
Diet quality, in particular, has emerged as a potentially important determinant of fertility, with evidence suggesting that dietary patterns emphasizing nutrient-dense, anti-inflammatory foods may support hormonal balance, ovulatory function, and overall reproductive capacity (Berrino et al., 2001; Chavarro et al., 2008, 2009, Gates et al., 1996; n.d.; Gaskins and Chavarro, 2018; Wise et al., 2018). Among these, adherence to the Mediterranean diet (MD), a pattern characterized by high intake of fruits, vegetables, whole grains, legumes, nuts, and healthy fats, has been associated with improved fertility outcomes in clinical and observational settings (Karayiannis et al., 2018; Sun et al., 2019b; Toledo et al., 2011). However, much of this work has been conducted in non-U.S. populations or fertility clinic cohorts, limiting its generalizability to the broader population.
Concurrently, the global food supply has shifted toward increased availability and consumption of ultra-processed foods (UPFs), which are industrial formulations high in added sugars, fats, sodium, and synthetic additives (Monteiro et al., 2018). According to the most recent U.S. data from the National Health and Nutrition Examination Survey (NHANES), UPFs now account for approximately 52% of Americans adults’ daily energy intake (Juul et al., 2022). Emerging evidence links high UPF consumption to adverse cardiometabolic (Chen et al., 2020; Dos Santos et al., 2020; Mambrini et al., 2023), psychological (Contreras-Rodriguez et al., 2023; Gomes Gonçalves et al., 2023; Lopes Cortes et al., 2021), and endocrine outcomes (Zhang et al., 2022b; Zhao et al., 2024), yet little is known about its potential impact on reproductive health. UPFs may influence fertility both directly—via suboptimal intakes of fertility-supportive nutrients—(Moubarac et al., 2017) and indirectly, through their role in metabolic dysfunction (Mambrini et al., 2023), endocrine disruption (Buckley et al., 2019), and inflammation (dos Santos Martins et al., 2022). These pathways may be particularly relevant during reproductive years, when hormonal regulation and metabolic health are closely intertwined.
Despite the biological plausibility of diet–fertility relationships and growing public health interest in UPFs and dietary patterns, no study to our knowledge has examined the associations between UPF intake and MD adherence with fertility-related outcomes in a nationally representative U.S. population. The NHANES provides a unique opportunity to explore these relationships using rigorously collected dietary, sociodemographic, and biomarker data. By analyzing fertility status based on self-reported attempts to become pregnant within the past year, this study provides novel insights into how contrasting dietary patterns may influence reproductive health at the population level.
Methods
Study design and participants
This study used data from three consecutive cycles of the NHANES spanning 2013–2018. NHANES is a nationally-representative, cross-sectional survey of the non-institutionalized, U.S. civilian population (National Health and Nutrition Examination Survey, 2025). A four-stage, stratified, and clustered sampling approach was undertaken to ensure national representation, with some subgroups being oversampled to increase precision for subgroup estimates. Sociodemographic and health data are collected through in-home interviews, followed by physical examinations and biological sample collection at a mobile examination center (MEC). Data collection for NHANES was conducted with approval from the National Center for Health Statistics (NCHS) Ethics Review Board (ERB), and all participants provided written informed consent (CDC, 2024).
Female participants aged 20–45 yrs. who participated in the reproductive questionnaire were included in our study. Participants were excluded if they had history of hysterectomy or oophorectomy, and if they had unreliable or incomplete dietary recall status for one or both recall days (Supplementary Figure 1). Missing data for covariates were handled using complete-case analysis, as is standard in SAS; thus, sample sizes varied slightly across multivariable models depending on covariate availability. Participants were not excluded upfront based on missing covariates.
Dietary data
Dietary intake was assessed using two non-consecutive 24-h dietary recalls administered using the USDA Automated Multiple Pass Method (Moshfegh et al., 2008). Trained interviewers administered the first recall in-person at the MEC and the second by telephone 3–10 days later (Moshfegh et al., 2008). Nutrient and food group intakes were averaged across both days.
Ultra-processed food intake
All reported food and beverage items were classified using the Nova system, which categorizes items based on the extent and purpose of industrial processing into four groups: 1) unprocessed or minimally processed foods; 2) processed culinary ingredients; 3) processed foods; and 4) ultra-processed foods (Monteiro et al., 2018). The Nova classification has been outlined in greater detail elsewhere briefly however, Group 4, includes industrially formulated foods that typically contain cosmetic additives and/or substances of rare culinary use such as sugar-sweetened beverages, packaged snacks, frozen meals, and reconstituted meat products (Martinez-Steele et al., 2023; Monteiro et al., 2018). Foods and beverages consumed by participants were reported in grams and labelled with 8-digit food codes (FNDDS : USDA ARS, n.d.). “Main Food Description”, “Additional Food Description”, and “Ingredient Code Description” were used to classify foods according to their level of processing. Potential homemade dishes were disaggregated and classified at the ingredient level.(Steele et al., 2023) UPF intake was expressed as the proportion of total food intake (e.g., 0.25 = 25%) composed of Nova Group 4 items. Grams rather than calories were used to account for low- or zero-calorie UPFs. UPF intake was considered as a continuous variable in all analyses, and estimated odds ratios reflect the effect per 10 percentage point increase in UPF intake, consistent with prior epidemiological studies (Nardocci et al., 2021; Srour et al., 2019; Suksatan et al., 2021).
Mediterranean diet (MD) score
Adherence to the Mediterranean Diet was measured using a modified version of the alternate MD score developed by Trichopoulou et al.(Gross et al., 2025; Trichopoulou et al., 2003) Dietary data were mapped to the USDA Food Patterns Equivalents Database (FPED) (Bowman et al., n.d.a, n.d.b, n.d.c), and scores were computed based on eight components: fruits, vegetables, whole grains, legumes, nuts and seeds, fish, red and processed meat, and the ratio of monounsaturated to saturated fat. Individuals who had intake above the median for beneficial components of the MD (all components but red and processed meat) scored 1 point and those who had intake below the median scored 0 points, for each category respectively. Participants scored 1 point if their intake was below the median intake for red and processed meats, and 0 points if the intake was above the median intake. Thus, the total MD score ranged from 0 (lowest adherence to MD) to 8 (highest adherence to MD). Traditional MD scoring includes alcohol intake, with moderate intake contributing positively (Gross et al., 2025; Sofi et al., 2014; Trichopoulou et al., 2003). However given evidence suggesting that even moderate alcohol intake may negatively impact fertility (Eggert et al., 2004; Fan et al., 2017; Jensen et al., 1998), and due to the potential for underreporting in NHANES, alcohol was excluded from the MD score. Furthermore, preliminary analyses indicated that alcohol intake was not significantly associated with fertility status or either dietary exposure. As such, alcohol was also excluded from covariate adjustment. This approach aligns with previous studies on reproductive health and reflects both the biological rationale and concerns about data reliability (Chavarro et al., 2007; Karayiannis et al., 2017).
To account for the potential effect of total energy intake on the relationship between diet quality and fertility, average daily calories (from both dietary recall days) were calculated and included as a covariate in multivariable models.
Outcome variable
Fertility status was defined based on responses to the question, “Have you ever attempted to become pregnant over a period of at least a year without becoming pregnant?” Participants who answered “yes”, were categorized as infertile; those who answered “no” were considered fertile.
Covariates
Covariates were selected based on prior literature and included factors associated with fertility and dietary behaviors. Sociodemographic covariates were: age (years), race/ethnicity (Mexican American, other Hispanic, non-Hispanic White, non-Hispanic Black, Asian, other/multiracial), education level (less than ninth grade, ninth to eleventh grade with no diploma, high school graduate, some college with no diploma, college graduate or above), poverty-to-income ratio (PIR), and marital status (ever married vs. never married). Health and lifestyle covariates included birth control use (yes/no), other hormone use (yes/no), history of pelvic infection (yes/no), age at menarche, smoking status (never, former, current), obesity status (body mass index ≥30 measured at MEC; yes/no) and self-reported physical activity (PA) (meeting ≥150 min/week of moderate-to-vigorous activity; yes/no) and total daily caloric intake (Current Guidelines | odphp.health.gov, n.d.). Alcohol intake was not included as a covariate for the reasons outlined above.
Statistical analyses
Descriptive statistics were computed for all study variables. Means and standard errors (SE) were reported for continuous variables, and proportions with SE for categorical variables. Group comparisons by fertility status were assessed using t-tests for continuous variables and chi-square tests for categorical variables.
Separate logistic regression models were constructed to examine the associations between MD score and fertility status, and between UPF intake and fertility status. These exposures were not included in the same model to avoid mutual adjustment, which may obscure independent associations due to overlapping dietary behaviors and shared variance across dietary patterns.
Each dietary exposure was modeled using crude and multivariable logistic regressions: Model 1: Adjusted for sociodemographic variables (age, race/ethnicity, PIR, education, marital status); Model 2: Model 1+ health and lifestyle covariates (hormonal birth control use, hormone use, pelvic infection, age at menarche, smoking status, physical activity, caloric intake); Model 3: Model 2+ obesity status. Interaction terms for age, PIR, education, and obesity status and the main exposure (MD score or UPF intake) were tested in fully adjusted models. As a sensitivity analysis, we excluded participants in the 5th and 95th percentiles of caloric intake (<602.5 kcal or >3792.0 kcal/day) to assess potential misreporting and found no difference in estimates or statistical significance.
All analyses were conducted using SAS 9.4. NHANES dietary sample weights and Taylor series linearization was applied to account for the complex survey design and to produce nationally representative estimates. Statistical significance was set at
Results
Sample characteristics
The analytic sample included 2582 women aged 20–45 years, of whom 87.83% (SE: 0.84) were classified as fertile and 12.16% (SE: 0.84) as having experienced infertility. The mean UPF intake was 27.15% (SE: 0.01) of total daily grams and the mean MD score was 3.35 (SE: 0.07). Compared to fertile women, women who experienced infertility had significantly higher UPF intake (30.64% vs. 26.67%) and lower MD scores (3.03 vs 3.40). Women with reported infertility were also older, and were more likely to be obese, married, and to report a history of pelvic infection (Table 1). No significant differences were observed in race, PIR, educational attainment, birth control use, hormone use, physical activity levels, smoking status, age at menarche, or total daily energy intake between groups (Table 1).
Sociodemographic, lifestyle, and dietary characteristics of US women aged 20–45 years, by fertility status (NHANES 2013–2018).
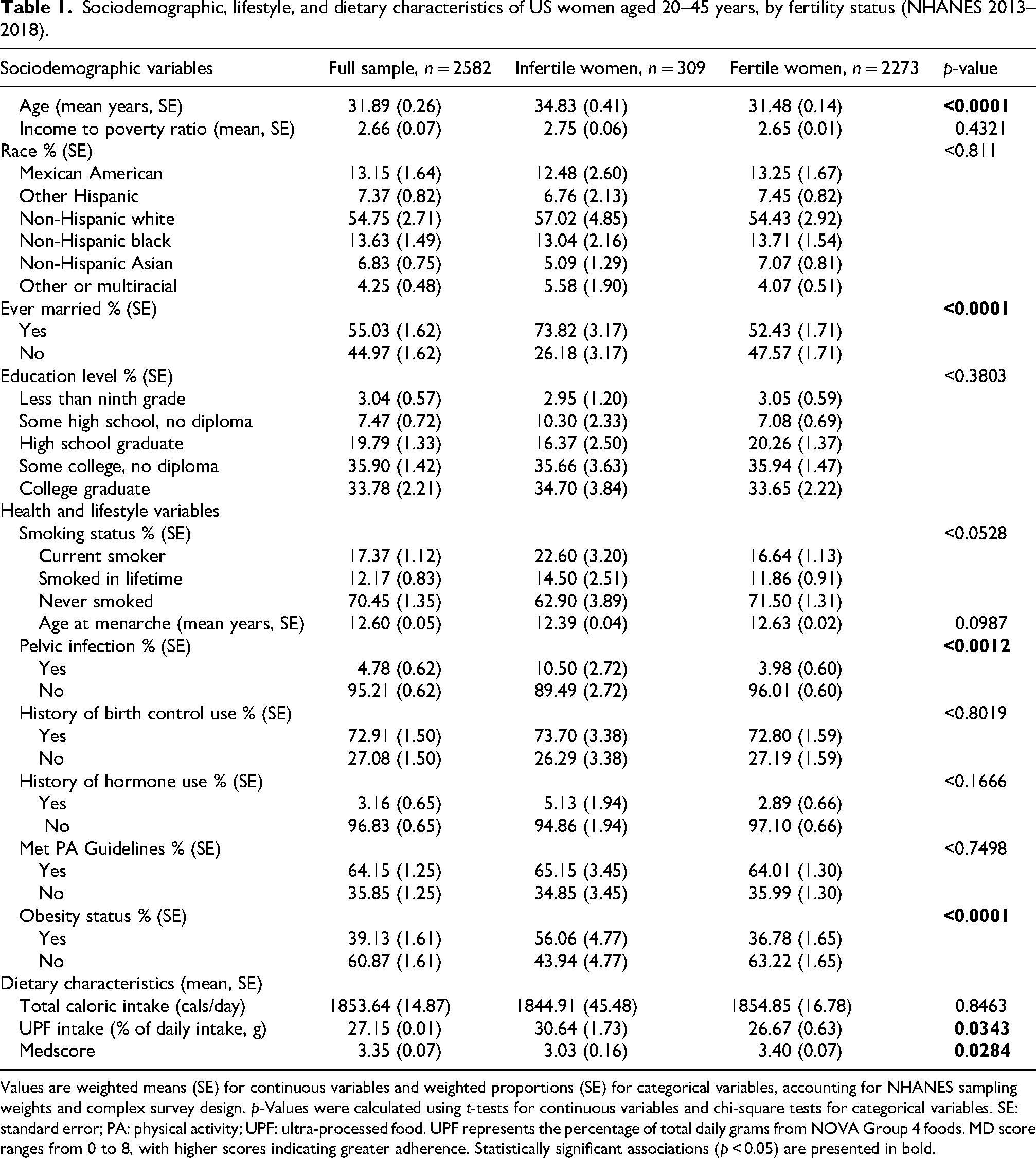
Values are weighted means (SE) for continuous variables and weighted proportions (SE) for categorical variables, accounting for NHANES sampling weights and complex survey design.
Association between UPF intake and fertility status
Across all models, higher UPF intake was significantly associated with lower odds of being fertile (Table 2). In the unadjusted model, higher UPF consumption was significantly associated with lower odds of being fertile (Odds Ratio (OR) = 0.52; 95% Confidence Interval (CI): 0.29–0.95). This inverse association remained statistically significant after adjusting for sociodemographic factors (Model 1: OR = 0.32; 95% CI: 0.13–0.75), health and lifestyle factors (Model 2: OR = 0.32; 95% CI: 0.14–0.75), and obesity status (Model 3: OR = 0.39; 95% CI: 0.17–0.91).
Odds ratios for fertility status by UPF intake in US women of reproductive agea.
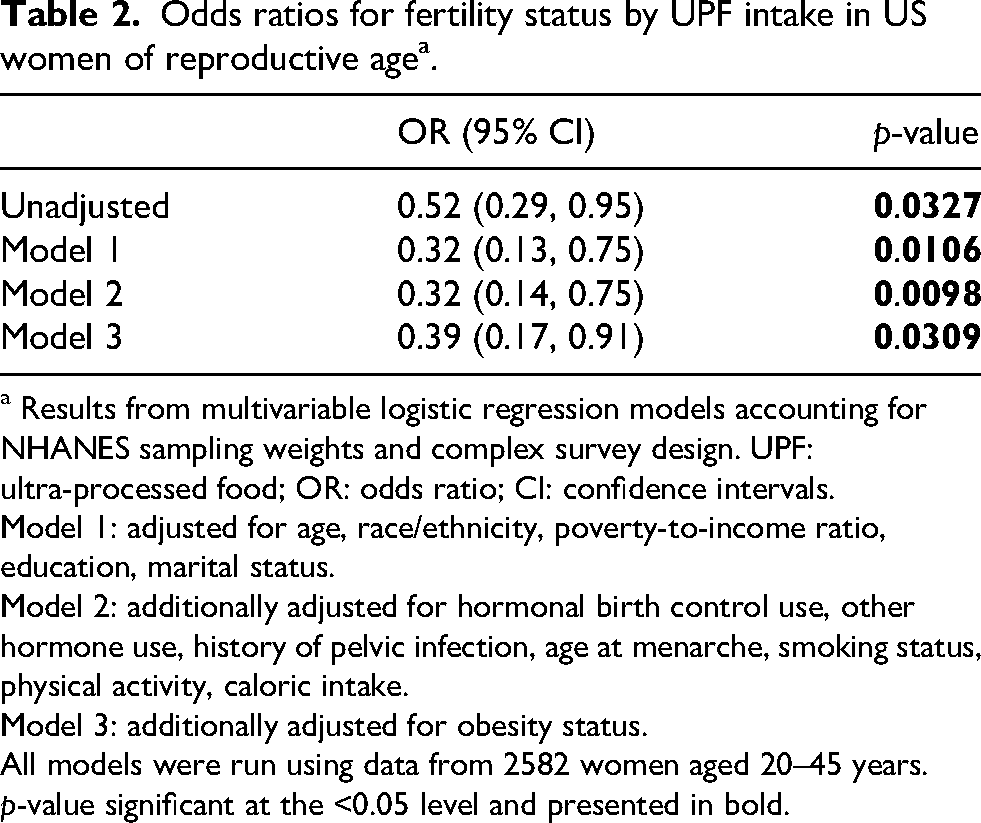
a Results from multivariable logistic regression models accounting for NHANES sampling weights and complex survey design. UPF: ultra-processed food; OR: odds ratio; CI: confidence intervals.
Model 1: adjusted for age, race/ethnicity, poverty-to-income ratio, education, marital status.
Model 2: additionally adjusted for hormonal birth control use, other hormone use, history of pelvic infection, age at menarche, smoking status, physical activity, caloric intake.
Model 3: additionally adjusted for obesity status.
All models were run using data from 2582 women aged 20–45 years.
Association between MD score and fertility status
Higher MD scores were associated with significantly greater odds of being fertile in the unadjusted model (OR = 1.10; 95% CI: 1.02–1.20) (Table 3). This association remained significant in models adjusting for sociodemographic (Model 1: OR = 1.16; 95% CI: 1.02–1.33) and additional health and lifestyle factors (Model 2: OR = 1.16; 95% CI: 1.00–1.33). However, in the fully adjusted model including obesity status (Model 3), the association was attenuated and no longer significant (OR = 1.12; 95% CI: 0.93–1.27).
Odds ratios for fertility status by Mediterranean Diet (MD) adherence score in U.S. women of reproductive agea.

a Results from multivariable logistic regression models accounting for NHANES sampling weights and complex survey design. MD: Mediterranean diet; OR: odds ratio; CI: confidence intervals.
Model 1: adjusted for age, race/ethnicity, poverty-to-income ratio, education, marital status.
Model 2: additionally adjusted for hormonal birth control use, other hormone use, history of pelvic infection, age at menarche, smoking status, physical activity, caloric intake.
Model 3: additionally adjusted for obesity status.
All models were run using data from 2582 women aged 20–45 years.
Interaction terms for age, PIR, education, and obesity status with the main exposures (MD score or UPF intake) were tested in fully adjusted models and were not statistically significant (all
Discussion
To the best of our knowledge, this study is the first to examine the independent relationships between UPF intake, MD adherence and self-reported infertility in a nationally representative sample of U.S. women of reproductive age. Higher UPF intake was significantly associated with lower odds of fertility, even after accounting for sociodemographic factors, reproductive health indicators, and obesity. In contrast, greater MD score adherence was initially associated with higher odds of fertility, but this relationship was attenuated after adjusting for obesity, suggesting that part of the association may be mediated through body weight, particularly given the MD's known effects on adiposity and metabolic regulation.
Infertile women in our sample were more likely to be older, married, and have obesity, which are well-established correlates of fertility challenges due to age-related declines in ovarian reserve (Amanvermez and Tosun, 2016), increased likelihood of attempting conception within marriage (Lichter et al., 2014), and adiposity-mediated hormonal dysregulation (Androulakis et al., 2014; Bloom et al., 2021; Chen et al., 2025; Kim et al., 2017; Kuang et al., 2024; Mongraw-Chaffin et al., 2015; Yeung et al., 2013). Interestingly, adherence to physical activity guidelines did not differ significantly between fertile and infertile women. This may reflect limitations of self-reported estimates or variability in physical activity type and intensity, given that both excessive and insufficient levels have been linked to reduced fertility (Foucaut et al., 2019; Mussawar et al., 2023).
Our findings are consistent with, and extend, previous research linking suboptimal diets and female fertility. Diets characterized by high intakes of minimally-processed plant-based foods, healthy fats and high in micronutrients have been associated with improved fertility outcomes (Chavarro et al., n.d.; Moludi et al., 2023; Toledo et al., 2011; Wang et al., 2024).
The relationships between both dietary patterns and fertility were attenuated after adjustment for obesity, but only the association with UPF intake remained statistically significant. This pattern suggests that obesity may potentially mediate part of the observed associations, particularly between MD adherence and fertility, while other factors may more strongly contribute to the relationship between UPF and fertility. The attenuation of the relationship between MD and fertility after controlling for obesity aligns with evidence linking the MD to lower risk of obesity (Buckland et al., 2008), type 2 diabetes (Esposito et al., 2015), and other cardiometabolic morbidities known to impair fertility (Chen et al., 2025; Galbete et al., 2018; Hunter et al., 2021; Kuang et al., 2024; Liu et al., 2021; Mattsson et al., 2021). Given that our models controlled for total energy intake, these associations are unlikely to reflect caloric intake alone, and may instead relate to the MD's overall nutrient density and anti-inflammatory properties (Castro-Quezada et al., 2014).
In contrast, the persistent association between UPF intake and lower fertility, even after adjustment for obesity, suggests that UPFs may in influence reproductive health through additional, weight-independent pathways such as poor nutrient quality, endocrine disruption, inflammation, or other metabolic effects unrelated to adiposity.
While Western-style diets high in processed meats, refined carbohydrates and added sugars have inconsistent associations with fertility (Sugawa et al., 2018; Toledo et al., 2011), our study is the first to evaluate UPF intake using the Nova classification in relation to self-reported infertility. This provides a more precise examination of how industrial food processing may independently affect reproductive function. Importantly, the persistent association between UPF intake and fertility, even after adjustment for energy intake and obesity, suggests that additional mechanisms beyond adiposity may be involved. Prior studies from the U.S., Canada, Brazil, and Europe have established that high UPF intake is associated with poorer diet quality (da Costa Louzada et al., 2018; Lauria et al., 2021; Liu et al., 2022; Martínez Steele et al., 2017; Moubarac et al., 2017) and lower intake of nutrients critical for reproductive health, including fibre, folate, iron, and antioxidant vitamins (Ji et al., 2023). Although UPF consumption is also linked to higher total caloric intake (Hunter et al., 2021), our findings suggest that the relationship with fertility extends beyond energy excess, implicating additional metabolic and hormonal mechanisms.
Emerging evidence indicates that UPFs may be detrimental to health independent of their nutrient composition. Industrial processing methods can alter the food matrix and nutrient bioavailability, impairing satiety signaling and increase postprandial glycemic response (Dicken and Batterham, 2021; Fardet, 2016; Forde et al., 2020; Hall et al., 2019). Higher UPF consumption has also been shown to associate with increased urinary concentrations of endocrine-disrupting chemicals such as phthalates and bisphenols, compounds used in food packaging and processing (Baker et al., 2024; Buckley et al., 2019; Huang et al., 2021; Steele et al., 2020), as well as acrylamides, compounds formed during high-temperature processing (Martínez Steele et al., 2023). These compounds have been linked to hormone dysregulation (Dubey et al., 2022; Golestanzadeh et al., 2020), metabolic disturbances (Duan et al., 2019; Golestanzadeh et al., 2019; Ribeiro et al., 2019; Zhang et al., 2022a; Zhu et al., 2021), as well as infertility in both human and animal models (Amjad et al., 2021; Duan et al., 2015; Louis et al., 2014; Tian et al., 2024; Trnka et al., 2021).
Additionally, UPFs have been associated with systemic inflammation through both nutrient-related (e.g., high in sodium, added sugars, low fibre) and non-nutrient-related (e.g., additives and chemicals) pathways. Higher intake has been correlated with elevated circulating levels of inflammatory markers including pro-inflammatory cytokines (Dos Santos et al., 2023; dos Santos Martins et al., 2022; Millar et al., 2025), C-reactive protein (Baric et al., 2025; Hall et al., 2019; Lane et al., 2022; Lopes et al., 2019), and white blood cells (Baric et al., 2025; Contreras-Rodriguez et al., 2023; Mete et al., 2024). Chronic low-grade inflammation may impair fertility (He et al., 2020; Li et al., 2023) by disrupting oocyte and embryo development or altering endometrial receptivity (Bai et al., 2021; Topkara Sucu et al., 2024). The gut microbiota also plays a critical role in modulating inflammatory and hormone pathways relevant to reproduction (Beni et al., 2024; Hakansson and Molin, 2011). Diets high in UPFs have been associated with unfavorable shifts in microbiota composition, including reduced microbial diversity, fewer beneficial bacteria, and a greater abundance of pro-inflammatory species (Cuevas-Sierra et al., 2021; Fernandes et al., 2023; Rondinella et al., 2025). These changes may contribute to systemic inflammation and hormonal dysregulation, potentially affecting fertility through disrupted ovarian function, implantation, or broader endocrine effects (Hakansson and Molin, 2011).
These findings suggest that public health guidance addressing food processing, not just nutrient profiles, may be warranted in dietary recommendations targeting women of reproductive age. We did not observe significant interactions between obesity and either dietary exposure (MD or UPF intake), indicating that the associations between diet quality and fertility were consistent among women with or without obesity. This suggests that dietary quality may influence reproductive health irrespective of weight status, supporting broader public health messaging that promotes healthy dietary patterns for women of reproductive age.
While this study provides important insights, certain limitations should be considered in the interpretation of our results. Firstly, the cross-sectional design of the NHANES limits the ability for causal inference, and it is conceivable that fertility challenges influence dietary choices rather than the reverse. The large sample size provided by the NHANES, however, enabled adjustment for a range of sociodemographic and reproductive factors to mitigate this concern. Additionally, dietary intake was assessed through two 24-h dietary recalls, which may introduce recall bias and limit the ability to capture habitual intake. Nonetheless, this method is widely considered the gold standard in nutritional epidemiology (Zhang et al., 2015). Similarly, while 24-h recalls are widely used and validated, they were not specifically designed to assess the degree of food processing. Details such as brand names or preparation methods are not always reported, which may introduce some misclassification of UPF intake. However, any such error is likely to be non-differential and would bias associations toward the null. Even if reporting differed by obesity status, a factor linked to both UPF intake and infertility, this would likely attenuate rather than exaggerate the observed associations, suggesting our findings are likely to be conservative. Finally, it is also important to note that our fertility measure was based on self-reported attempts to conceive over a 12-month period. While this aligns with standard epidemiologic definitions (Vander Borght and Wyns, 2018), it may not capture unrecognized subfertility or those not attempting to become pregnant. Despite these limitations, the consistency of the observed association across multiple adjusted models supports a potentially meaningful association that warrants further investigation through longitudinal and mechanistic research.
Our findings, drawing on a nationally representative sample of U.S. women of reproductive age, show that higher intake of UPFs was associated with significantly lower odds of fertility, independent of sociodemographic factors, total energy intake, and obesity. In contrast, greater adherence to a MD pattern was positively associated with fertility status, although this relationship was attenuated after adjustment for obesity, suggesting a potential mediating role of adiposity. These findings contribute new evidence to the growing literature on dietary patterns and reproductive health and highlight the importance of considering both nutrient quality and the degree of food processing in relation to fertility outcomes.
Given the rising global consumption of UPFs and the continued public health burden of infertility, our findings underscore the need for public health guidance that extends beyond single-nutrient recommendations. Longitudinal and mechanistic studies are warranted to further elucidate the pathways linking diet quality to reproductive health and to support the development of dietary recommendations that promote fertility and broader metabolic well-being in women of reproductive age.
Supplemental Material
sj-jpg-1-nah-10.1177_02601060261433154 - Supplemental material for Ultra-processed food intake and Mediterranean diet adherence in relation to fertility status in U.S. women: Findings from NHANES 2013–2018
Supplemental material, sj-jpg-1-nah-10.1177_02601060261433154 for Ultra-processed food intake and Mediterranean diet adherence in relation to fertility status in U.S. women: Findings from NHANES 2013–2018 by Angelina Baric, Lauryn Flannagan, Euridice Martinez-Steele, Annika Bauer and Anthea Christoforou in Nutrition and Health
Footnotes
List of abbreviations
Acknowledgments
The linkage of the Nova Classification System for Industrial Food Procession with ASA24 (version) and NHANES Dietary Data (years/cycles) is supported in part by the National Cancer Institute, Bethesda, MD.
Ethical considerations
The ethical approval was granted by the U.S. National Center for Health Statistics Ethics Review Board (Protocols # 2011-17 and #2018-01).
Consent to participate
The National Center for Health Statistics Research Ethics Review Board approved the NHANES study protocol, and participants provided written informed consent at enrollment.
Consent for publication
Not applicable.
Authors' contributions
AB (Baric) and AC designed research; EMS provided essential materials; AB (Baric) and AB (Bauer) contributed to material preparation. AB (Baric) and LF performed statistical analyses; AB (Baric), AC, and LF wrote the paper. AC had primary responsibility for final content. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Data described in the manuscript can be made available upon request.
Statements and declarations
Not applicable.
Supplemental material
Supplemental material for this article is available online.
