Abstract
Introduction
Ageing can be defined as a time-dependent decline in cellular and systemic physiological functions. Environmental and endogenous stressors contribute to ageing by accumulating macromolecular damage and impairing repair mechanisms, thus increasing the risk of suffering cardiovascular, metabolic, neurodegenerative diseases and cancer, eventually leading to mortality (Guo et al., 2022; Richardson and Schadt, 2014). Up to 12 molecular, cellular, and systemic hallmarks of ageing have been currently proposed, with telomere attrition being considered one of the major mechanisms underlying cellular ageing (López-Otín et al., 2023a, 2023b, 2013).
Telomeres are complex nucleoprotein structures found at the end of linear chromosomes that play a crucial role in maintaining chromosome stability and integrity, preventing chromosome end fusion (Saretzki, 2018). In mammals, telomeric DNA consists of tandem repeats of the hexanucleotide sequence TTAGGG ranging from 5 kbp in humans to 100 kbp in mice (Zhu et al., 2019). Telomeric DNA is associated with the shelterin complex, a group of six proteins involved in capping and protecting telomeres that also facilitates the access to telomeric DNA to the telomerase reverse transcriptase enzyme (TERT), which recognizes the 3’-hydroxyl groups at the end of chromosomes to extend telomeres (Wu et al., 2017). In humans, TERT activity seems to be mainly restricted to early development, and in that way, stem cells have been found to exhibit robust activity (Hiyama and Hiyama, 2007), whereas it is downregulated in most somatic cells (Cong et al., 2002).
Currently, telomeres are recognized as important biomarkers of cellular ageing as they continuously shorten with cell division due to the “end-replication problem”, which leads to telomere shortening after each cell division or replication event (Bernardes de Jesus and Blasco, 2012; Boccardi and Marano, 2024; Saretzki, 2018). Once a critical telomere length is reached, it leads to telomere uncapping and activation of the DNA damage response (DDR). Then, cells stop dividing and enter a state of replicative senescence, leading to cell cycle arrest, or even apoptosis, and loss of regenerative capacity of tissues. In this scenario, and in preclinical models, interventions aimed at maintaining telomere length have led to preservation of cell survival, lifespan extension, and delayed ageing (Bernardes de Jesus et al., 2012; Jaskelioff et al., 2011; Shim et al., 2021; Tomás-Loba et al., 2008).
It has been reported that every cell division telomeres can shorten by 30 to 200 bp, although actual contribution of the end-replication problem to telomere shortening is thought to be no longer than 15–20 bp per cell division in human cultured cells (Von Zglinicki et al., 2001). Oxidative stress has been considered the main factor responsible for the remaining loss in cultured cells (Von Zglinicki, 2002). Free radicals can modify nucleotides or induce single- and double-strand DNA breaks (Saretzki, 2018), and accumulation of single-strand DNA breaks at telomeric regions has been reported as the most important cause of telomere shortening in cultured cells due to the low efficiency of DNA repair mechanisms at telomeric regions (Von Zglinicki et al., 2000). In addition, there is also evidence that oxidative stress shortens telomeres in living organisms, and thus dietary antioxidant supplementation has shown to diminish telomere shortening and/or increase telomere length, whereas antioxidant depletion has been related to a reduction in telomere length (Armstrong and Boonekamp, 2023; Reichert and Stier, 2017).
A freeze-dried ingredient derived from the marine green eukaryotic microalga Tetraselmis chui (phylum: Chlorophyta, family: Chlorodendraceae) with a significantly high superoxide dismutase (SOD) activity (from now on, TSOD) has been shown to behave as a promoter of cellular health (Cocksedge et al., 2025b). In the European Union, TSOD was authorized as a novel food for human consumption in 2014 according to Regulation (EC) No 258/97, and more recently, as a food supplement in 2017 (Hurtado et al., 2017). TSOD has been demonstrated to possess high antioxidant and ROS scavenging activities in a variety of in vitro tests (Cokdinleyen et al., 2024; Mantecón et al., 2019). In addition to SOD, a range of compounds present in TSOD (polyunsaturated fatty acids, vitamins, carotenoids, polyphenols, or phytosterols) likely contribute to its high antioxidant capacity (Cokdinleyen et al., 2024; Mantecón et al., 2019). Of note, this ingredient has been demonstrated to behave as an indirect antioxidant, stimulating the own cellular antioxidant mechanisms both in vitro and in vivo (Gil-Cardoso et al., 2022; Ramírez et al., 2020; Sharp et al., 2020), which might be related to its cytoprotective effects against oxidative insults in human neuronal cells (Cokdinleyen et al., 2025). The aim of present study was to evaluate the effect of TSOD on telomere length in human neonatal primary fibroblast cells. Cultures of human primary fibroblasts have previously been employed to study the process of telomere shortening under both standard and mild oxidative conditions (Levy et al., 2024; Mehkri et al., 2019; Quiles et al., 2022; Samuel et al., 2022). These cells exhibit normal ageing with a lifespan of 2–3 months, during which telomeres shorten and cells enter senescence, making them an excellent in vitro model for telomere research (Levy et al., 2024; Samuel et al., 2022). Given the relationship between oxidative stress and telomere shortening, telomere length was evaluated both under standard culture conditions and in the presence of an oxidative insult.
Materials and methods
TSOD extract
TSOD freeze-dried microalgae biomass was provided by Fitoplancton Marino, S.L. (El Puerto de Santa María, Cádiz, Spain). A 10% (w/v) biomass suspension in cold phosphate-buffered saline (PBS) 1X (Biowest) was rehydrated on ice for 45 min, and then lysed at 1100–1200 bar in a high-pressure homogenizer (PandaPLUS 1000, GEA Niro Soavi). After centrifugation at 30,000 g, 4°C, for 15 min, the supernatant was aliquoted and immediately kept at −80°C. Three aliquots of the supernatant were used for SOD activity determination using a kinetic method with the SOD Assay Kit—WST (Dojindo) according to the manufacturer's instructions, yielding an average value of 3,545 U/mL.
Cell culture
Neonatal human fibroblasts (ATCC PCS-201–010TM) were cultured in six-well plates at a seeding density of 5 × 103 cells/cm2 in fibroblast medium kit (Innoprot). The culture medium, buffered with HEPES and bicarbonate to maintain pH 7.4, was refreshed every 2–3 days. Cells were subcultured upon reaching 70–80% confluence. Cell counts at each passage were obtained using Automated Cell Counter EVETM (NanoEntek). Population doubling (PD) was calculated using the following formula: 3.322 × (log Cf – log Ci) + x, where Cf is the final cell count, Ci is the initial cell count, and x represents the PD value from the previous passage. One PD corresponds to a single complete cell division.
MTT assay
Primary neonatal human fibroblasts (≤ 3 passages) were cultured under standard conditions and seeded in 96-well plates at 5000 cells/well (three days) or 3500 cells/well (one week) using the Fibroblast Medium Kit (Innoprot). Twenty-four hours following the seeding process, cells were treated with the respective compounds in cell culture media. Each condition was tested using three biological replicates with and without 10 µM H2O2 (for mild oxidative stress and standard culture conditions, respectively). For positive and negative controls, 8 mM methyl methane sulfonate and PBS at 3% were used, respectively. Following compound addition, the plates were incubated for three days and one week, while the medium with the compound was changed every two days. After the treatment period, cells were washed twice with PBS and incubated with 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), 0.5 mg/mL in phenol red-free DMEM. The plates were gently shaken and incubated for 4 h. After incubation, the medium was removed and replaced with DMSO (Sigma), and the plates were gently shaken again to solubilize the formazan crystals. Absorbance was then measured at 570 nm using an Envision multiplate reader. To assess compound effects, five serial dilutions of TSOD extract (1:40, 1:80, 1:160, 1:320, 1:640) were prepared and evaluated under both standard and mild oxidative stress conditions.
Treatments
Aliquots of TSOD extracts were thawed and centrifuged at 4°C for 15 min at 15,000 r/min prior to treating cells. Considering the results of the MTT assay, cells were exposed to TSOD extract at dilutions of 1:160 (TSOD_A), 1:320 (TSOD_B), and 1:640 (TSOD_C) on a weekly basis with each media change. Treatments were conducted both under standard conditions and under mild oxidative stress conditions induced with 10 µM H2O2, with each condition assessed separately. All samples were plated in three biological replicates. Control cells on day 0 received no TSOD nor H2O2. At appropriate time points (4 and 8 weeks), cells were detached using TrypLETM (Gibco) and frozen in 90% fetal bovine serum (FBS) (Cultek), 10% DMSO (Sigma).
Sample preparation and high-throughput quantitative fluorescence in situ hybridization
Frozen cell samples and control cell lines were thawed at 37°C, and both cell count and viability were evaluated. Only cells with viability exceeding 60% were selected for further processing. These cells were seeded into 384-well black-walled, clear-bottom plates at a density of 12,000 (test samples) and 35,000 (control cell lines) cells per well. Test samples and control cell lines were plated in five technical replicates per biological replicate. Cells were then fixed with a methanol/acetic acid solution (3:1, v/v). Pepsin digestion was used to remove the cytoplasm, preparing the nuclei for hybridization of a specific peptide nucleic acid probe against telomere repeated sequence (Alexa 488-OO-CCCTAACCCTAACCCTAA, from Panagene). Following a series of washes, cells were stained with DAPI to label DNA. Finally, wells were filled with PBS before proceeding with imaging.
HT microscopy
Quantitative imaging was performed using the High Content Screening Opera Phenix System (Revvity) A 40x/0.95 NA water immersion objective was used for image acquisition. UV and 488 nm excitation wavelengths were applied to detect cell nuclei (DAPI) and telomeric spots (AF488 signals), respectively. For each well, 15 images were acquired at distinct positions under fixed exposure and Z-stack settings. Images were analyzed with SIMA software Version 1.2 (Revvity).
Telomere analysis technology
Telomere length was assessed using Life Length's proprietary high-throughput quantitative fluorescence in situ hybridization method. The intensity of the fluorescent signal, directly proportional to telomere length, was converted to absolute values in base pairs using a calibration standard curve generated from reference cell lines with known telomere lengths. Life Length's proprietary algorithms were used to calculate the telomere length variables. This approach provides a complete telomere length distribution for each sample. From this, median telomere length (MTL), the 20th percentile telomere length, and the proportion of critically short telomeres (< 3 kbp) were determined. The telomere analysis technology platform maintains an interassay coefficient of variation below 5%.
Statistical analysis
All statistical analyses were conducted using Prism 9.2.0 (283) for Mac OS X (GraphPad Software, LLC). Data were expressed as mean ± standard deviation (SD) in all charts. The Shapiro-Wilk test for normal distribution was applied, and then differences between groups were determined by one-way ANOVA followed by Tukey's multiple comparisons test. Differences were considered significant for a p-value < 0.05.
Results
MTT assay
MTT assay results showed that, under standard conditions, 1:40 dilution induced > 90% cell death at both three days and one week, far exceeding the 20% cytotoxicity threshold (Samuel et al., 2022). At 1:80 dilution, a toxic effect was observed only at one week. More diluted concentrations of TSOD (1:160, 1:320, and 1:640, corresponding to approximately 22, 11, and 5.5 U/mL of SOD activity) did not surpass the cytotoxicity threshold at either three days or one week (Table 1), indicating no significant toxic effects. Morphological analysis of cells through optical microscopy confirmed the absence of any deleterious effects at these three diluted concentrations.
Results of the MTT assay under standard culture conditions.
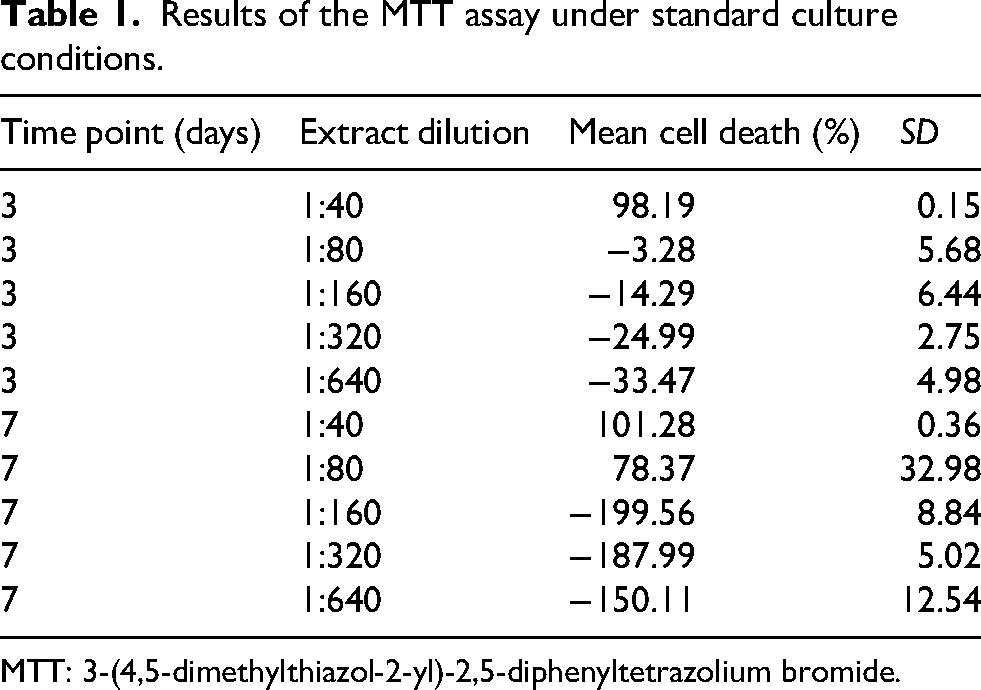
MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide.
Under oxidative stress, the cytotoxicity profile was consistent with the results observed under standard conditions. At three days, 1:40 dilution induced marked cell death (> 90%), clearly exceeding the 20% toxicity threshold. On the other hand, 1:80 dilution remained toxic after one week, whereas 1:160, 1:320, and 1:640 dilutions exhibited nonsignificant toxic effects after three days and one week of exposure (Table 2). Optical microscopy confirmed the absence of morphological alterations at these three dilutions also under oxidative conditions.
Results of the MTT assay under oxidative stress culture conditions.
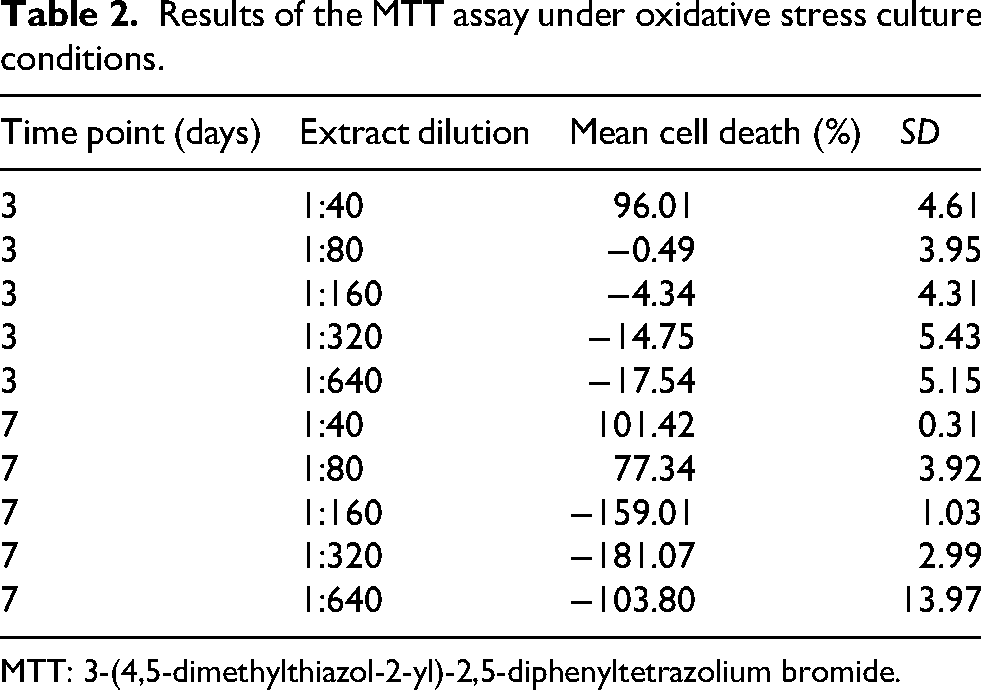
MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide.
Since 1:160, 1:320, and 1:640 dilutions did not show cytotoxicity under either standard or oxidative stress conditions at three days or one week, these concentrations, referred to as TSOD_A, TSOD_B, and TSOD_C, respectively, were selected for subsequent assays.
Median telomere length
The MTL (50th percentile) is a more representative indicator than the mean, since telomere length is not normally distributed within the sample. Representative images of telomere analysis by confocal microscopy to determine MTL under standard culture conditions are shown in Figure 1(a) (control cells) and Figure 1(b) (TSOD_A treated cells). MTL slightly increased after 4 weeks of treatment with TSOD_A and TSOD_B, without reaching statistically significant differences compared to the other two groups. After 8 weeks of exposure, a slight increase in MTL was also observed in the three TSOD-treated groups, but without reaching statistically significant support (Figure 1(c); Supplemental Material S1).
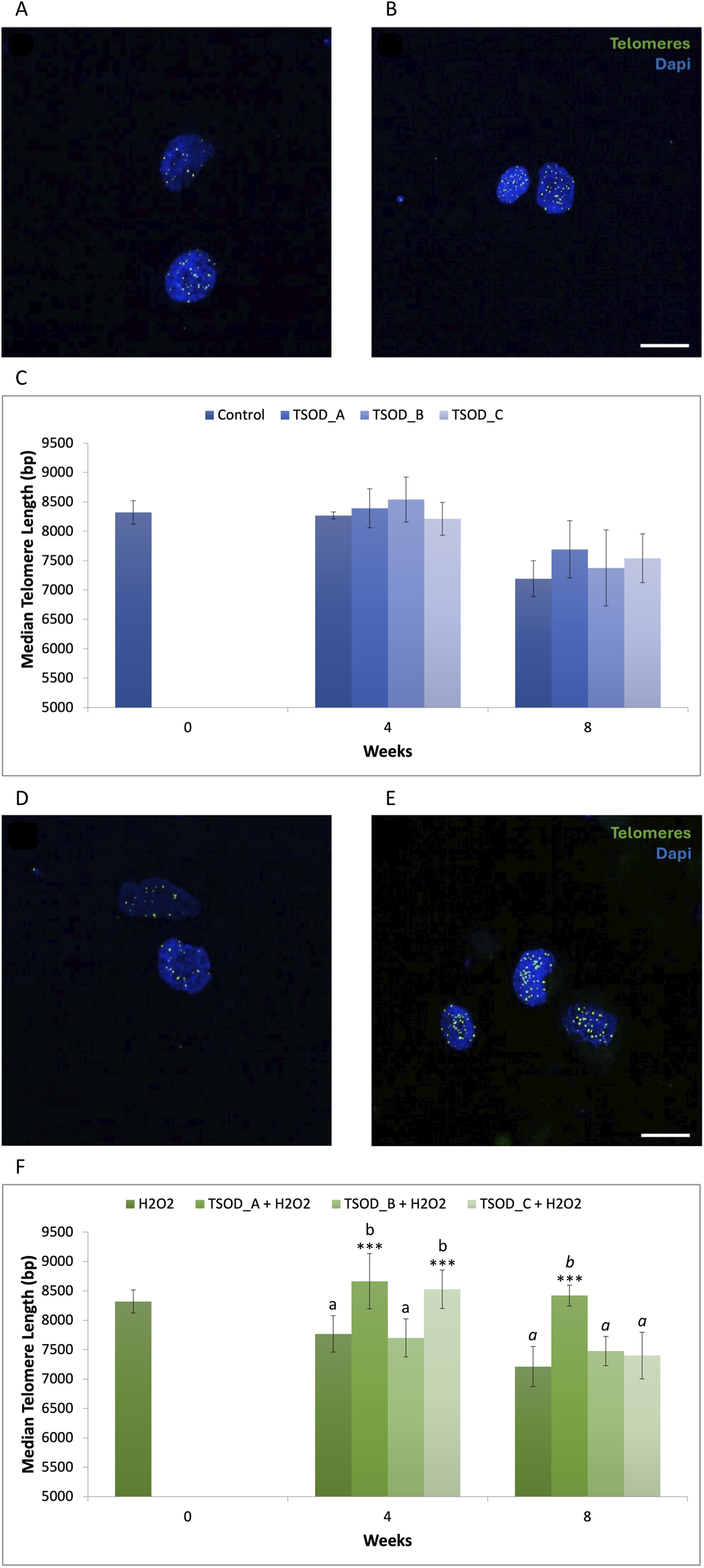
Representative image of telomere analysis by confocal microscopy: untreated cells (a) and TSOD_A treated cells (b) at week 8 under standard conditions, and untreated cells (d) and TSOD_A treated cells (e) at week 8 under oxidative conditions. Scale bar 20 µm. Bar charts of MTL at 4 and 8 weeks for the different TSOD extract concentrations under standard culture conditions (c) and under oxidative stress conditions (f). In both instances, statistically significant differences were evaluated using one-way ANOVA followed by Tukey's multiple comparisons test and are indicated by asterisks (***p < 0.001). Different letters denote statistically significant differences between experimental groups, with plain text indicating comparisons at 4 weeks and italics indicating comparisons at 8 weeks.
Representative images for telomere analysis by confocal microscopy under oxidative stress conditions are shown in Figure 1(d) (control cells) and Figure 1(e) (TSOD_A treated cells). At 4 weeks, TSOD_A and TSOD_C significantly lengthened MTL compared to the control group (H2O2) at the same time point (1.12- and 1.10-fold, respectively; ***p < 0.001), and even increased compared to the control at baseline. After 8 weeks, the antiageing effect of TSOD was maintained, with TSOD_A showing a significant increase in MTL (1.17-fold; ***p < 0.001) compared to H2O2 control at the same time point (Figure 1(f); Supplemental Material S1). Collectively, these findings indicate that TSOD lengthens telomeres under the assessed experimental conditions.
20th percentile telomere length
The 20th percentile is a telomere length metric derived from the telomere length distribution. It represents the length below which the shortest 20% of telomeres fall, providing insight into the shortest telomeric fraction within the sample. Under standard conditions, the 20th percentile telomere length slightly increased after 4 weeks of treatment with TSOD_A and TSOD_B concentrations, without reaching statistically significant differences compared to the control at the same time point. However, a slight decrease in 20th percentile was detected with TSOD_C in relation to the other groups, which was statistically significant when compared to TSOD_B (0.91-fold; *p < 0.05). After 8 weeks of exposure, TSOD-treated groups showed an increase in 20th percentile telomere length compared to the control, but it was statistically significant only with TSOD_A (1.13-fold; *p < 0.05) (Figure 2(a); Supplemental Material S1). Under oxidative stress conditions and at 4 weeks, TSOD_A and TSOD_C significantly increased 20th percentile telomere length compared to the H2O2 control at the same time point (1.23- and 1.20-fold, respectively; ***p < 0.001). This effect persisted at 8 weeks for TSOD_A, which again showed a highly significant difference in 20th percentile telomere compared to the H2O2 control at the same time point (1.35 fold; ***p < 0.001), being comparable to the control at baseline (Figure 2(b); Supplemental Material S1). Consistent with the MTL results, these findings indicate that TSOD has a protective effect on the shortest telomeres both under standard and oxidative cell culture conditions.
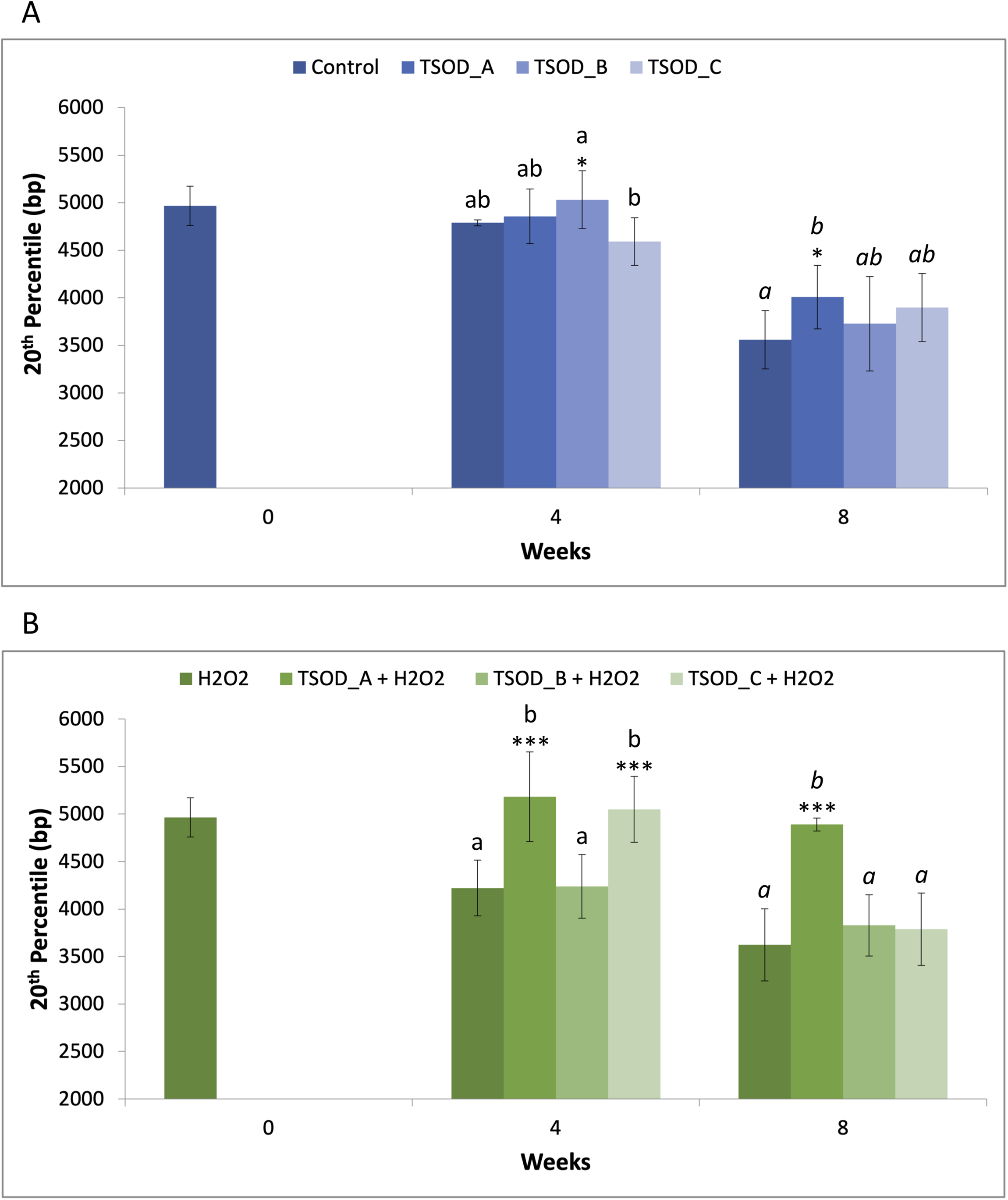
20th percentile telomere length (in base pairs) after 4 and 8 weeks of treatment with different TSOD concentrations under standard (a) and oxidative stress (b) conditions. Bars represent median ± standard deviation. Statistical differences were evaluated using one-way ANOVA followed by Tukey's multiple comparisons test and are indicated by asterisks (*p < 0.05; ***p < 0.001). Different letters denote statistically significant differences between experimental groups, with plain text indicating comparisons at 4 weeks and italics indicating comparisons at 8 weeks.
Percentage of telomeres < 3 kbp
The percentage of telomeres < 3 kbp is another indicator of short telomeres, which are associated with genomic instability and cellular senescence. Under standard conditions, TSOD treatment showed a decrease in the percentage of these critically short telomeres at both 4 and 8 weeks relative to the respective control, but with no statistical support. Strikingly, a statistically significant increase was observed at 4 weeks with TSOD_C when compared to TSOD_A (1.16-fold; *p < 0.05) and TSOD_B (1.25-fold; **p < 0.01), but it was not maintained at 8 weeks (Figure 3(a); Supplemental Material S1). Under oxidative stress (10 µM H2O2), TSOD treatment significantly reduced the percentage of the shortest telomeres. At 4 weeks, TSOD_A and TSOD_C showed a statistically significant reduction compared to the H2O2 control (0.66- and 0.71-fold, respectively; ***p < 0.001) at the same time point, and also relative to TSOD_B. At 8 weeks, the highest concentration (TSOD_A) maintained this significant reduction (0.60-fold; ***p < 0.001) compared to the H2O2 control at the same time point, while the other concentrations also showed a slight reduction in the percentage of short telomeres although without reaching statistically significant differences (Figure 3(b); Supplemental Material S1). Notably, cells under TSOD_A treatment even showed a similar percentage of the shortest telomeres to that exhibited by the H2O2 control at baseline. Taken together, these findings indicate that TSOD lengthens the shortest telomeres.
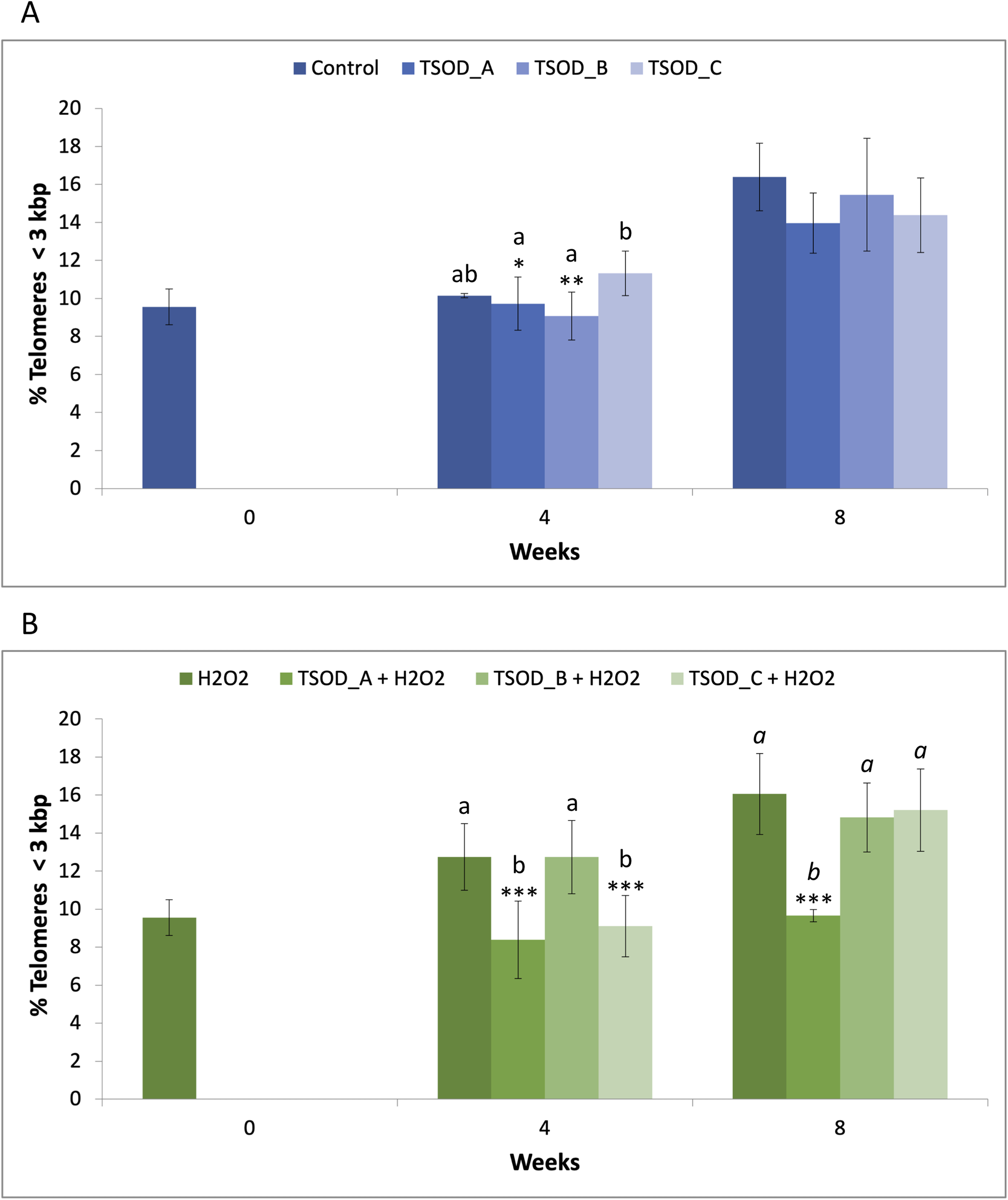
Percentage of critically short telomeres (< 3 kbp) after 4 and 8 weeks of treatment with different TSOD concentrations under standard (a) and oxidative stress (b) conditions. Bars represent mean ± standard deviation. Statistical differences were evaluated using one-way ANOVA followed by Tukey's multiple comparisons test and are indicated by asterisks (*p < 0.05; **p < 0.01; ***p < 0.001). Different letters denote statistically significant differences between experimental groups, with plain text indicating comparisons at 4 weeks and italics indicating comparisons at 8 weeks.
Telomere shortening rate
Since cell replication is one of the main contributors to telomere shortening, telomere length measurements were normalized by the PD levels in each condition and at each time point. Under standard cell culture conditions, cells treated with TSOD_A and TSOD_B exhibited enhanced proliferative capacity, starting from the fifth and second week of expansion, respectively (Supplemental Material S2). Under oxidative cell culture conditions, cells treated with TSOD_C showed reduced proliferation from the first week of expansion, while those treated with TSOD_A displayed decreased proliferative capacity until the fifth week of expansion (Supplemental Material S3).
Under standard conditions, the telomere shortening rate showed variable values across the different TSOD concentrations at both 4 and 8 weeks, but without reaching statistically significant differences (Figure 4(a); Supplemental Material S1). Under oxidative stress conditions (10 µM H2O2), a statistically significant reduction in telomere shortening rate was observed relative to the control with TSOD_A (−0.63-fold; ***p < 0.001) and TSOD_C (−0.35-fold; *p < 0.05) at 4 weeks, and the same occurred when compared to TSOD_B. After 8 weeks, TSOD_A showed a significantly reduced shortening rate (−0.09-fold; ***p < 0.001) compared to the H2O2 control, and also relative to TSOD_B and TSOD_C (Figure 4(b); Supplemental Material S1). These results indicate that over time, TSOD treatment might help maintain telomere length by mitigating the cumulative effect of cell replication.
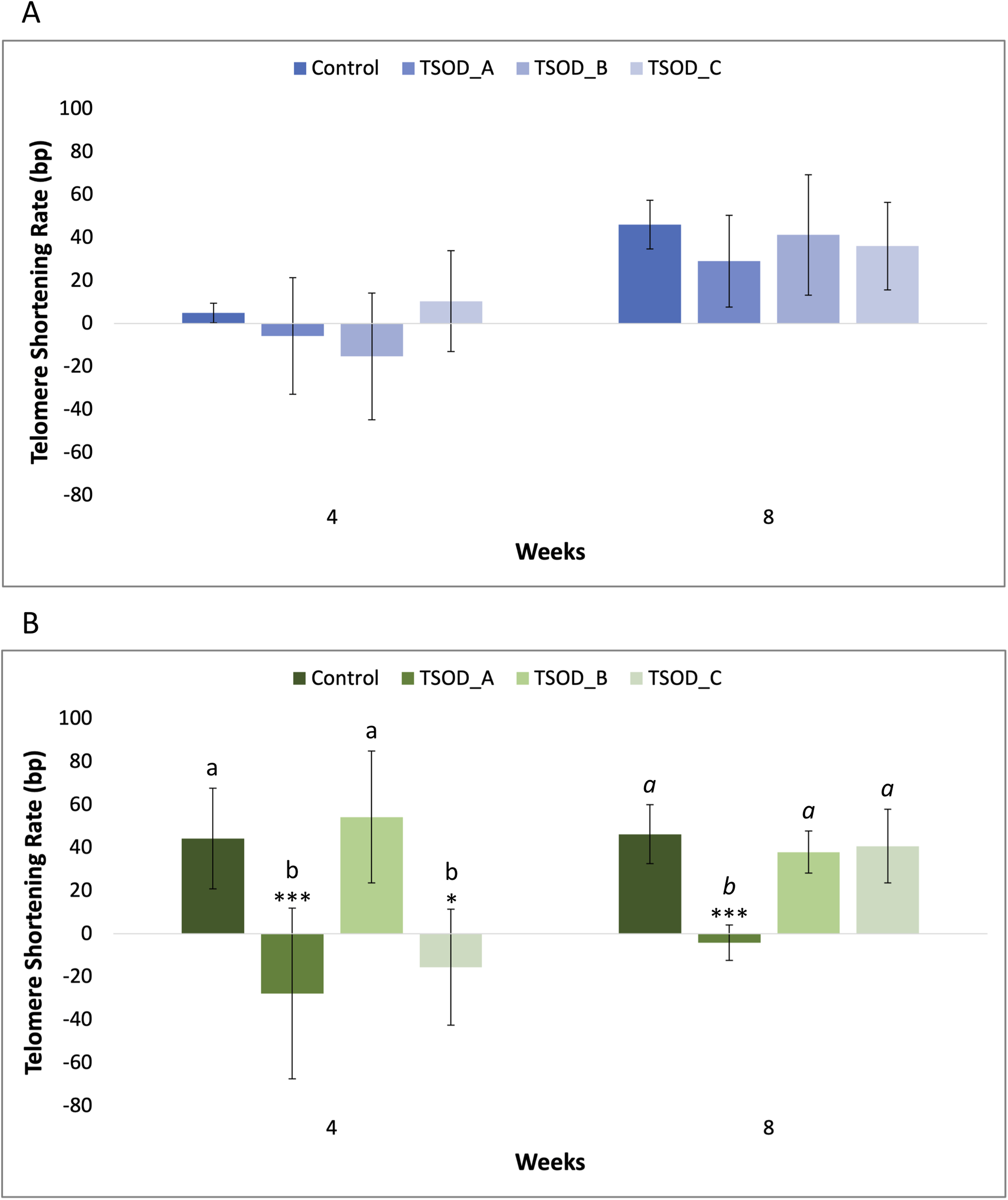
Telomere shortening rate (in base pairs per PD) at 4 and 8 weeks under standard (a) and oxidative stress (b) conditions for the different TSOD concentrations. Data were normalized to PD levels for each condition. Bars represent mean ± standard deviation. Statistical differences were evaluated using one-way ANOVA followed by Tukey's multiple comparisons test and are indicated by asterisks (*p < 0.05; ***p < 0.001). Different letters denote statistically significant differences between experimental groups, with plain text indicating comparisons at 4 weeks and italics indicating comparisons at 8 weeks.
Discussion
Telomeres are recognized as critical biomarkers of cellular ageing, and hence of disease susceptibility. Telomere shortening is significantly associated with a variety of pathological conditions such as cardiovascular disease, diabetes mellitus, neurological disorders, pulmonary fibrosis, and cancer (Blackburn et al., 2015; López-Otín et al., 2023a, 2023b), making strategies to slow this process crucial for health and longevity. Compared to the respective control, TSOD elicited a significant increase in MTL and 20th percentile telomere length, as well as a significant reduction in the percentage of critically short telomeres (< 3 kbp), under oxidative culture conditions at both 4 and 8 weeks of treatment. Interestingly, less remarkable results were observed under standard culture conditions. Moreover, TSOD was found to significantly reduce telomere shortening rate at both 4 and 8 weeks of exposure, indicating that reduction in telomere shortening was due to a direct effect of TSOD rather than differences in the number of cell divisions. Collectively, these findings demonstrate a protective effect of TSOD on telomere length, significantly pronounced under oxidative stress conditions. This seems to be in agreement with the observed homeostatic response induced by TSOD in human tissues, acting as a modulator of cellular responses that need enhancement (Cocksedge et al., 2025a, 2025b). Thus, mild oxidative stress might have triggered a stronger protective response leading to higher telomere protection.
The reduction in the percentage of critically short telomeres (<3 kbp) observed in this study in response to TSOD treatment deserves special interest. Actually, this reduction might be particularly relevant to understanding cellular senescence pathways, as these dysfunctional telomeres represent key triggers of the DDR and replicative senescence in somatic cells (Huang et al., 2025; Pizzul et al., 2023). Critically shortened telomeres become uncapped and are recognized as DNA double-strand breaks, activating DNA damage checkpoint kinases, ultimately enforcing permanent cell cycle arrest and cellular senescence (Ajoolabady et al., 2025). By mitigating the accumulation of critically short telomeres through reduction in telomere shortening rate, TSOD may help preserve cellular proliferative potential and delay the transition to replicative senescence, thereby supporting the maintenance of tissue regeneration capacity and promoting healthy ageing.
Oxidative damage has been considered one of the most important elements contributing to telomere shortening, and hence the antioxidant properties of TSOD might underlie its protective effects on telomere length. Consistently, diverse antioxidants have previously been shown to protect telomeres in in vitro studies (Alt et al., 2022; Levy et al., 2024; Mehkri et al., 2019; Quiles et al., 2022; Samuel et al., 2022), and an association between diets rich in antioxidant nutrients and longer telomeres has been suggested based on the outcomes of clinical trials (Crous-Bou et al., 2019; Galiè et al., 2020; Rafie et al., 2017). Different indirect antioxidants mechanisms triggered by TSOD might have operated to protect telomeres from shortening. For instance, TSOD has been demonstrated to activate the main antioxidant enzymes (SOD, glutathione peroxidase, and catalase) both in vitro and in vivo (Ramírez et al., 2020; Sharp et al., 2020). In a preclinical model of metabolic syndrome, TSOD increased glutathione levels in the liver (Gil-Cardoso et al., 2022), which is known to play a crucial role in cellular antioxidant protection. Interestingly, such increase occurred with a concomitant upregulation of genes directly involved in glutathione biosynthesis and recycling, including the modifier subunit of the glutamate-cysteine ligase (GCLM), glutathione synthetase (GSH-S), and glutathione reductase (GSR) (Gil-Cardoso et al., 2022), and in agreement with this, TSOD was also able to increase the transcript levels of GCLM and GSR in human muscle tissue (Cocksedge et al., 2025a). Similarly in human muscle, TSOD upregulated an additional group of genes known to be involved in protection against oxidative damage, including peroxiredoxin 3 (PRDX3) and 6 (PRDX6), and the glutathione-transferase mu 3 gene (GSTM3) (Cocksedge et al., 2025a). Of note, and in addition to all these free radical scavenging enzymes, TSOD has been demonstrated to transcriptionally activate the nuclear factor erythroid 2-related factor 2 (NRF2) both in human myoblasts (Ramírez et al., 2020) and muscle tissue (Cocksedge et al., 2025a). NRF2 is considered the master of the cellular antioxidant response, controlling the expression of more than 200 genes, most of them with a cytoprotective function (Hayes and Dinkova-Kostova, 2014; He et al., 2020). In this regard, NRF2 is known to regulate the expression of a fundamental set of genes involved in redox metabolism, including genes of the glutathione-based system (such as GCLM, GCLC, or GSR), thioredoxin-based system (such as PRDX6), and other antioxidant systems (such as SOD) (Eleutherio et al., 2021; Hayes and Dinkova-Kostova, 2014; He et al., 2020; Singh and Bhat, 2012; Vahidinia et al., 2024). The demonstrated up-transcriptional regulation by TSOD of sirtuin 1 (SIRT1) in human tissue is also relevant when dealing with strengthening of the cellular antioxidant response (Cocksedge et al., 2025b). As a sirtuin, SIRT1 mediates the deacetylation of both histones and nonhistone proteins in an NAD+-dependent manner, and NRF2 is one of its targets (Kratz et al., 2021). Deacetylation of NRF2 has been related to an increase in stability, nuclear localization, and transcriptional activity of NRF2, thus improving cell resistance to oxidative damage (Huang et al., 2013; Kratz et al., 2021; Ma et al., 2019).
The reported upregulation of NRF2 and SIRT1 by TSOD might involve additional mechanisms acting against telomere shortening. Although not assessed in this study, potential activation of telomerase activity by TSOD has to be considered, as an increase in telomerase activity is known to be related to telomere lengthening (Quiles et al., 2022; Samuel et al., 2022). In support of this, NRF2 and SIRT1 have been shown to promote TERT expression, activity, and/or stability, contributing to telomere maintenance (Ahmad et al., 2016; Chen et al., 2020; Dong et al., 2021; Lee et al., 2024). However, further research will be necessary to demonstrate any of these effects on telomerase by TSOD.
It is known that telomeric DNA repair is crucial to maintain telomere stability and counteract telomere shortening, and thus it cannot be ruled out that the observed telomere protection induced by TSOD might be related to an activation of DNA repair mechanisms. In this regard, a significant reduction of DNA damage was found in human sperm samples after 90 days of TSOD supplementation (González et al., 2024). Thus, potential upregulation of NRF2 and SIRT1 by TSOD might also support this hypothesis, as both key cellular factors participate in a variety of DNA repair pathways (Lagunas-Rangel, 2019; Li et al., 2023). Demonstrating a direct link between activation of DNA repair mechanisms and telomere protection mediated by TSOD will require additional research.
Although the protective effects of TSOD on telomere observed in this study are promising, they should be validated through in vivo research. However, the insights here generated might open a door for the use of TSOD particularly in the context of ageing. Telomere attrition has been related to a number of diseases, but also occurs during normal ageing in humans, and a causal link between telomere loss, cellular senescence and organismal ageing has been established in genetically modified animal models (López-Otín et al., 2013, 2023a). But it is important to highlight that a decline in the activity of both NRF2 and SIRT1 is known to happen with ageing (Chen et al., 2020; Schmidlin et al., 2019; Suh et al., 2004), and hence TSOD might help counteract such a decline and combat the deleterious effects of telomere shortening, thus promoting healthy ageing and longevity.
Conclusions
TSOD has demonstrated the ability to protect telomeres from shortening in neonatal human fibroblasts, increasing MTL and 20th percentile telomere length, and decreasing both the percentage of critically short telomeres and the telomere shortening rate. However, specific limitations of this study should be noted, such as those related to in vitro model constraints, the absence of direct telomerase activity measurements, and the lack of any assessment of DNA damage markers. Nevertheless, these promising in vitro antiageing effects undoubtedly justify further evaluation in in vivo studies.
Supplemental Material
sj-docx-1-nah-10.1177_02601060261431078 - Supplemental material for An antiageing telomere-protective effect of superoxide dismutase-rich Tetraselmis chui in human cells
Supplemental material, sj-docx-1-nah-10.1177_02601060261431078 for An antiageing telomere-protective effect of superoxide dismutase-rich Tetraselmis chui in human cells by Alfonso Blázquez-Moreno, Lorena Orgaz, Estefanía Rodríguez-Moreno, Adrián Asensio, Marta Marsal, Lalia Mantecón, Carlos Infante, Enrique Samper and Emmanouil Flemetakis in Nutrition and Health
Footnotes
Acknowledgments
This work was conducted in the scenario of a collaborative research program between Fitoplancton Marino, S.L. and Agricultural University of Athens.
Ethical considerations
Not applicable for this manuscript.
Consent for publication
The manuscript has been read and approved by all the authors, who also approved the order in which they are listed.
Authors’ contributions
ABM and LO contributed to conceptualization, data curation, formal analysis, writing—original draft, and writing—review and editing; ERM and AA contributed to investigation; LM and CI contributed to writing—review and editing; MM and ES contributed with project administration, writing—original draft, and writing—review and editing; EF contributed with writing—original draft and writing—review and editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Fitoplancton Marino, S.L.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Lalia Mantecón and Carlos Infante are employees of Fitoplancton Marino, S.L. who funded the current work.
Data availability statement
The data that support the findings of this study are available from the corresponding author, EF, upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
