Abstract
Background:
Choline is an essential nutrient involved in human health and development across the lifespan. The evidence on choline and its health effects has been growing; yet the findings are inconsistent.
Aim:
We aimed to map the current evidence and identify gaps in knowledge.
Methods:
We conducted a scoping review of the studies that examined relationships between choline exposure and any health outcomes. MEDLINE, CINAHL, and the Cochrane Central Register of Controlled Trials were searched for research involving humans and published between January 2000 and May 2025. Screening and data abstraction were performed in duplicate.
Results:
A total of 117 primary studies were included. Most of the studies were conducted in North America (54%) and Europe (22%). Cardiometabolic disease accounted for the largest proportion (34%), followed by offspring neurodevelopment (19%) and liver conditions (12%). The studies were predominantly observational (prospective 60%, retrospective 12%, cross-sectional 20%). The evidence regarding potential benefits and harms was reported to be insufficient or equivocal for many health outcomes. Based largely on data from small randomized controlled trials in women with prenatal exposure to alcohol or infants with fetal alcohol syndrome disorder, high maternal choline intake/status was associated with improved neurocognition and neurodevelopmental outcomes in the offspring.
Conclusion:
Potential benefits of high maternal choline intake/status was indicated for neurodevelopment of the offspring; however, the evidence was insufficient or equivocal for most of the outcomes reviewed. A more comprehensive synthesis incorporating preclinical evidence from animal studies will improve the current understanding of choline and its health effects.
Introduction
Choline was classified as an essential nutrient in 1998 and is thought to be involved in human health and development across the lifespan because of its wide ranging roles in human metabolism (Institute of Medicine, 1998; Zeisel and Da Costa, 2009). It is involved in phospholipid synthesis (Leermakers et al., 2015; Zeisel, 2006); synthesis of neurotransmitters (Blusztajn et al., 2017; Yang et al., 2020); and DNA and histone methylation processes (Leermakers et al., 2015; Zeisel, 2006). Choline provides the one-carbon units required for the synthesis of s-adenosylmethionine, a key methyl donor involved in DNA methylation processes, while increasing the remethylation of homocysteine into methionine (Romano et al., 2017; Zeisel and Da Costa, 2009).
In the US, a Dietary Reference Intake (DRI) for choline was established by the US Institute of Medicine (IOM) based on “adequate intake” (AI, a level assumed to ensure nutritional adequacy) in 1998 (Institute of Medicine, 1998). At that time, evidence was insufficient to establish an estimated average requirement (EAR). Similar AIs have been specified in other countries. The lack of an EAR hinders the development of targeted nutritional interventions, such as supplementation programs for vulnerable subgroups, e.g., pregnant women with an increased requirement for choline (Nguyen et al., 2025) and individuals displaced from homes due to political conflicts, food insecurity, or climate change living with an increased risk of malnutrition (Godde et al., 2021; Rother et al., 2023).
As endogenous synthesis of choline alone is not sufficient to meet the biological requirements (Institute of Medicine, 1998; Zeisel et al., 1991), dietary intake is needed. According to limited population data, average choline intake in the general population falls below established AI levels (Vennemann et al., 2015; Wiedeman et al., 2018) in North America (Lewis et al., 2014; Masih et al., 2015; Wallace and Fulgoni, 2016, 2017), Europe (Roeren et al., 2022), and globally (Bragg et al., 2022; Robb et al., 2021; Semba et al., 2016).
Epidemiological evidence on the relationship between choline status and health appears to have been growing. Low choline intake has been reported to be associated with an increased risk of type 2 diabetes (Dibaba et al., 2020; Gao et al., 2017; Virtanen et al., 2020), cardiovascular disease (Millard et al., 2018; Papandreou et al., 2021), non-alcoholic fatty liver disease (Chai et al., 2023; Yu et al., 2014), and metabolic syndrome (Gao et al., 2019). Some studies have been interpreted as suggesting that choline intake/status has beneficial effects on cognitive function among older adults (Aguree et al., 2023; Liu et al., 2021a; Smith et al., 2010; Velazquez et al., 2020; Ylilauri et al., 2019) and among infants (Irvine et al., 2022; Jiang et al., 2014; Obeid et al., 2022). However, other studies have suggested that high choline intake/status may have adverse effects on the basis of positive associations with cardiometabolic disorders in different populations (Schizttarella et al., 2017; Vallianou et al., 2024).
Several evidence syntheses have been published in the recent years reporting on the relationship between maternal choline intake during pregnancy and neurodevelopment of the offspring (Derbyshire and Obeid, 2020; Gould et al., 2025) and between choline intake/status and cardiometabolic disorders (Meyer and Shea, 2017; Yang et al., 2023a). The pooled evidence on the relationship between choline and neurodevelopment appears heterogeneous, with considerable variation in both the types of exposures assessed (e.g., dietary intake, supplementation, or biomarkers of choline status) and the outcomes measured (such as cognitive, behavioral, and structural brain indicators). This variability limits the ability to draw firm conclusions, and overall findings remain largely inconclusive. Review findings on cardiovascular disease also indicate a complex picture: studies of dietary choline intake have reported different associations than studies focusing on circulating choline concentrations, highlighting a potential discrepancy between self-reported intake and biological measures of exposure.
We therefore undertook a scoping review to scan the existing evidence landscape concerning choline and its relation to human health with the aim of mapping the evidence for different health outcomes and identifying gaps in knowledge. More specifically, we aimed to systematically search and broadly describe the evidence examining the relationship between choline intake/status and health outcomes by disease category.
Materials and methods
We conducted a scoping review in accordance with the Joanna Briggs Institute (JBI) guidelines (Peters et al., 2020) and the Preferred Reporting Items for Systematic reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) (Tricco et al., 2018).
Eligible articles were reports of studies (a) involving human participants of any age; (b) with assessment of choline intake (from diet, supplementation, or fortified foods), choline status or potential choline deficiency as exposure; (c) with comparison of different levels of exposures or comparison of exposure versus non-exposure; (d) with self-reported or clinically diagnosed health conditions as outcomes; and (e) with study designs of randomized controlled trial, observational studies or systematic reviews with or without meta-analyses (see Table 1).
Eligibility criteria for studies included in this review.
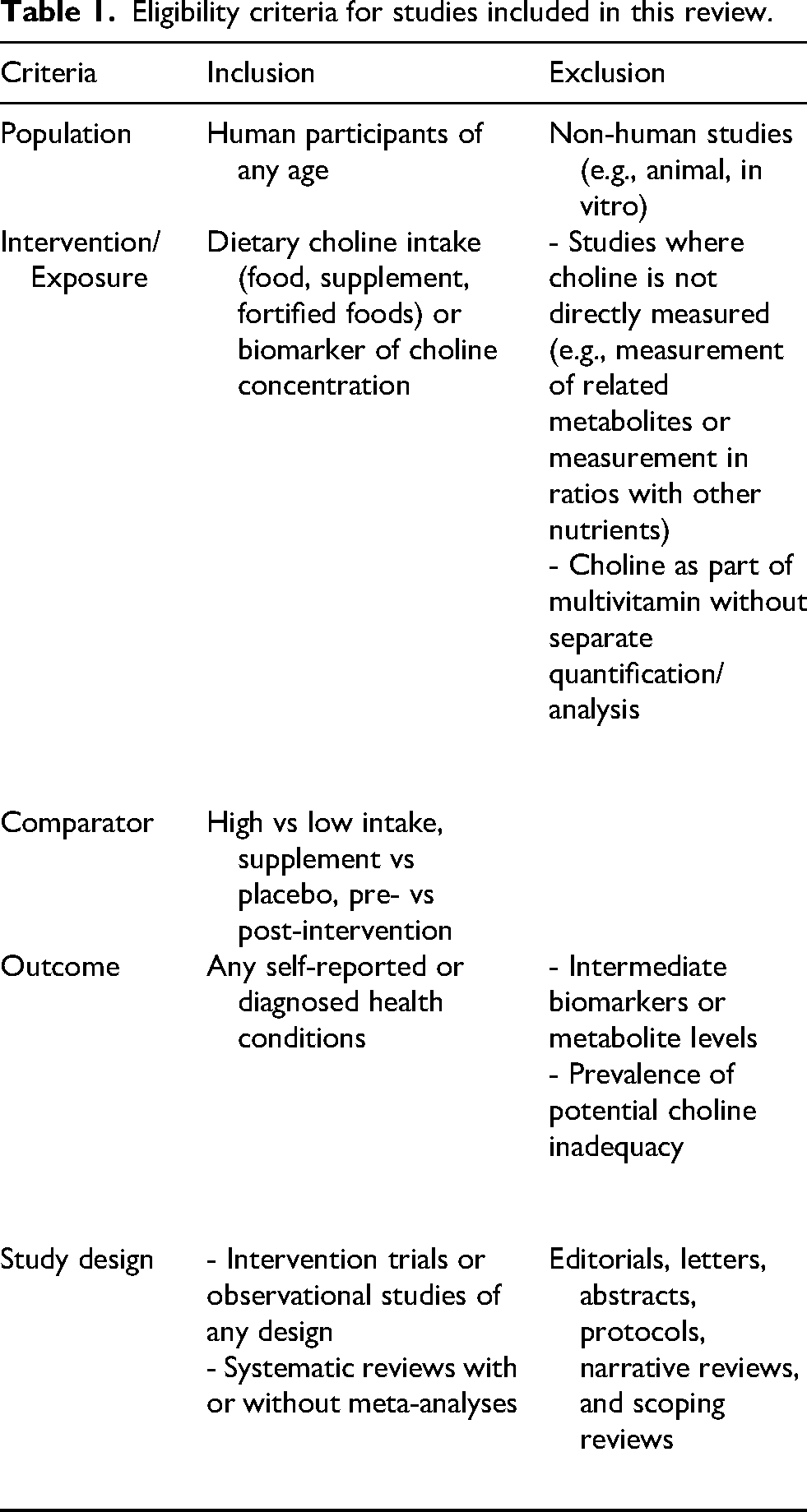
We excluded the following: non-human studies such as animal experiments or in vitro research; studies that did not measure choline directly; studies of choline as part of multivitamins without separate quantification, or studies focusing on related compounds (e.g., trimethylamine N-oxide (TMAO)) or biochemical systems (e.g., cholinesterase enzymes) without specific choline-related data; studies reporting choline only as a ratio or as part of a neuroimaging metric (e.g., N-acetyl aspartate (NAA)/total choline); studies focusing on mechanistic pathways; studies reporting on prevalence of choline intake/status above or below a threshold without corresponding health outcomes; and editorials, abstracts, narrative reviews and scoping reviews.
Data source and search strategy
We developed a search strategy using terms related to choline and health outcomes and searched three electronic databases: Medline, CINAHL, and the Cochrane Central Register of Controlled Trials (CENTRAL). Human intervention trials or observational studies published between January 2000 and May 2025 were searched. We limited the search to studies published in 2000 onward to ensure the inclusion of research that reflects more recent methodological standards, diagnostic criteria, and dietary assessment techniques. Since the release of choline DRIs in 1998, formally recognizing choline as an essential nutrient, scientific interest in choline grew rapidly with more consistent definitions of intake levels, improved study designs, and relevance to current dietary patterns. No language restrictions were applied. We further manually searched the reference lists of all included systematic reviews/meta-analyses and identified eligible primary studies not captured in the database search. The database and the manual searches were executed in May and June 2025, respectively. Full search strategies for each database are available in Supplementary Table 1.
Screening and data abstraction
All identified records were imported into Covidence. Three reviewers (SY, AM, AA) independently performed two-stage screening based on the eligibility criteria. Discrepancies were resolved through discussion or consultation with a fourth reviewer (JL). Data were abstracted using a standardized template. One reviewer (SY or AM) conducted initial abstraction, and a second reviewer (AA) independently verified the data. Discrepancies were resolved through discussion. Key information abstracted from each article include study characteristics (first author, year of publication, potential conflict of interest, source of funding), methodology (study design, follow-up duration, sample size, country), study population (age, sex, ethnicity), exposure measure (type of choline exposure, unit of measurement, dose/duration, comparator) and outcome (health condition, subgroup analyses, key findings).
Evidence synthesis
We conducted descriptive analyses summarizing the identified evidence by key parameters, i.e., year of publication, geographic region, and study design. We provided further description of the studies in each health outcome category in terms of the populations studied, types of choline exposure used, and overall findings. We also separately presented systematic reviews identified through the search as part of a broad landscape of the evidence on choline and its associated health outcomes. The volume and quality of the systematic reviews often reflect the level of clinical or policy-related interest in particular topics. The included systematic reviews were assessed for methodological quality by two independent reviewers (SY, AM) using AMSTAR-2 (A MeaSurement Tool to Assess systematic Reviews, version 2) (Shea et al., 2017).
Results
Our database search retrieved 3149 articles. After removing duplicates, 2798 titles and abstracts were screened, followed by full-text review of 237 articles. Of these, 91 articles (82 primary studies and 9 systematic reviews with or without meta-analyses) were included in the review. We identified an additional 31 non-overlapping articles from the systematic reviews and another 4 studies from other sources. A total of 117 primary studies were included for this review (Figure 1). Characteristics of all included studies are in Tables 2-(1) and 2-(2).
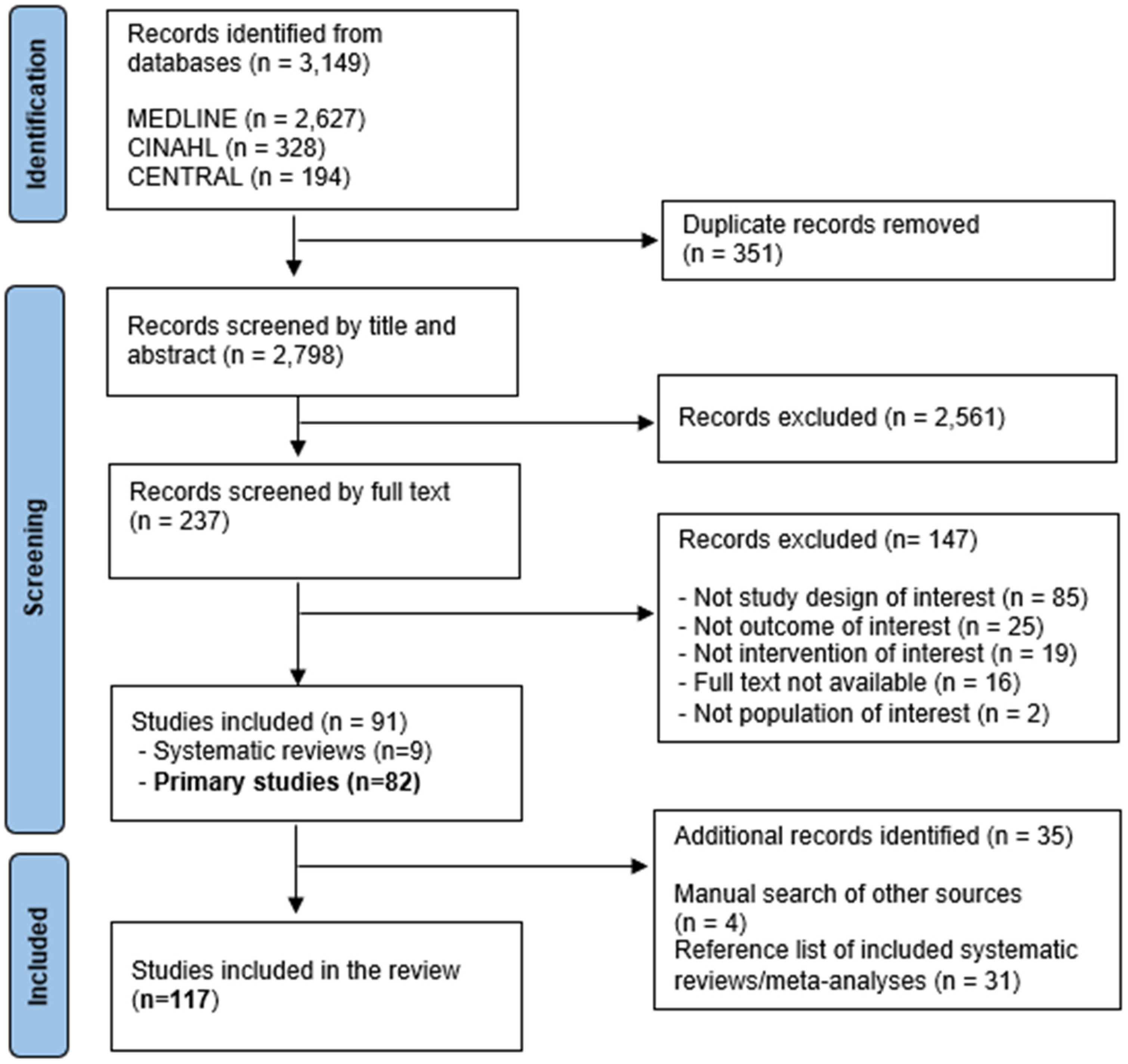
PRISMA flow diagram of systematic literature search for studies examining choline exposure and health outcomes.
Characteristics of randomized controlled trials (RCTs) examining the relationship between choline intake/status and health outcomes (in chronological order).
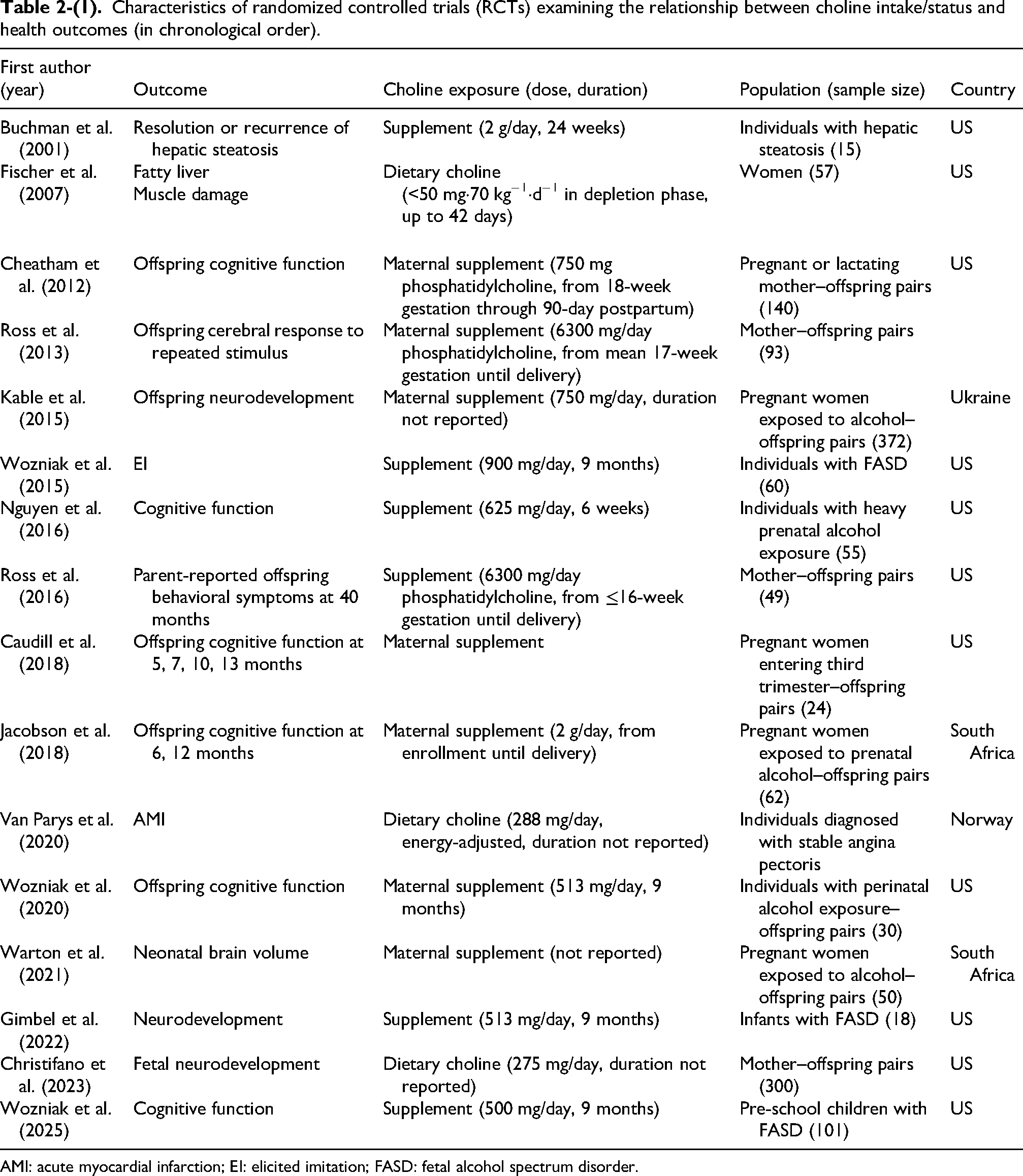
AMI: acute myocardial infarction; EI: elicited imitation; FASD: fetal alcohol spectrum disorder.
Characteristics of observational studies examining the relationship between choline intake/status and health outcomes (in chronological order).
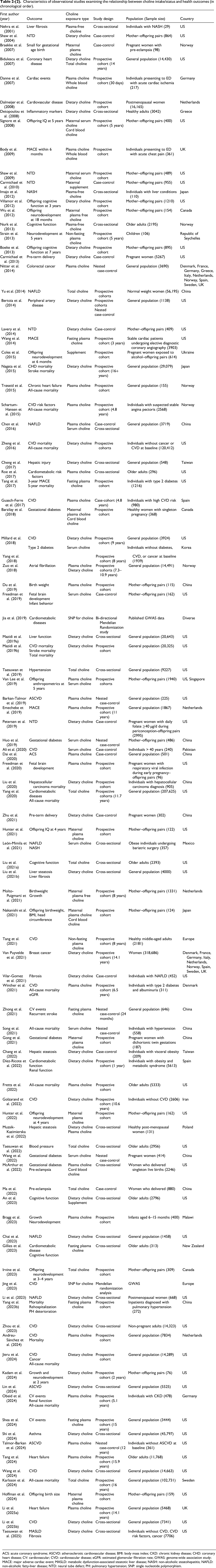
ACS: acute coronary syndrome; ASCVD: atherosclerotic cardiovascular disease; BMI: body mass index; CKD: chronic kidney disease; CHD: coronary heart disease; CV: cardiovascular; CVD: cardiovascular disease; eGFR: estimated glomerular filtration rate; GWAS: genome-wide association study; MACE: major adverse cardiac event; MASLD: metabolic dysfunction-associated steatotic liver disease; NASH: non-alcoholic steatohepatitis; NAFLD: non-alcoholic fatty-liver disease; NTD: neural tube defect; PH: pulmonary hypertension; SNP: single nucleotide polymorphism.
Distribution of the choline studies by year, region, health outcome, and study design
The number of choline studies has been slowly increasing over the years (Figure 2). By geographical region, more than half of the studies were conducted in North America, primarily the United States (54%), almost a quarter (22%) in Europe, and 17% from East Asia, mainly China. By outcome, cardiometabolic disease accounted for the largest proportion (34%) of the studies, followed by offspring neurodevelopment (19%) and liver conditions (12%). Among the studies identified, randomized controlled trials (RCTs) accounted for 14%, prospective observational studies (prospective cohorts, nested case-control, and case-cohort studies) accounted for 61%, cross-sectional studies accounted for 20% and case-control studies accounted for 12% (Table 3).
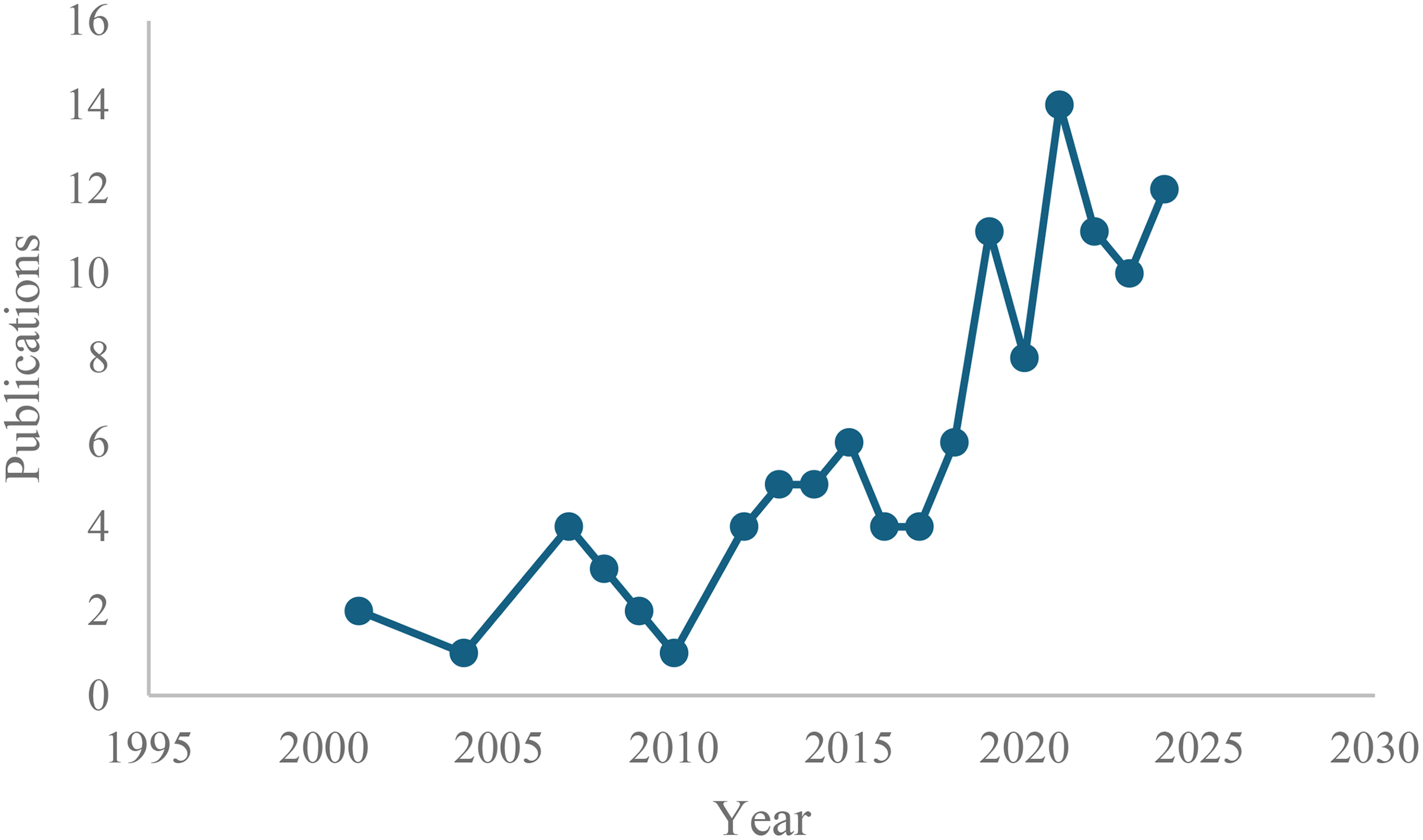
Temporal distribution of choline studies since 2000.
Classification of the included choline studies by geographical region, health outcome, and study design.
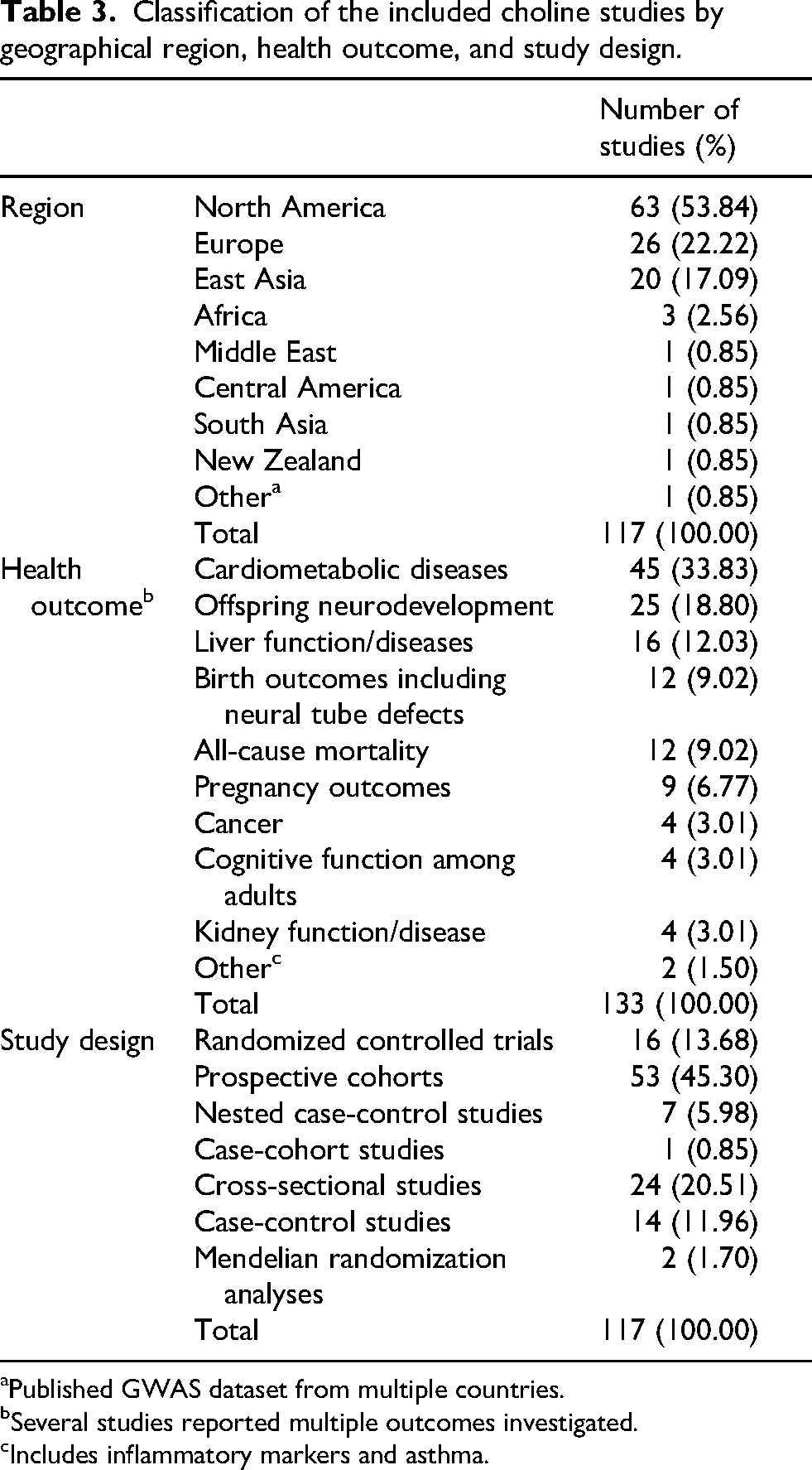
Published GWAS dataset from multiple countries.
Several studies reported multiple outcomes investigated.
Includes inflammatory markers and asthma.
Evidence on cardiometabolic outcomes
A total of 45 studies reported on cardiometabolic outcomes: 33 prospective cohort, 8 cross-sectional, 2 case-control studies, and 2 Mendelian Randomization analyses. Among the prospective cohorts (conducted primarily in Europe (n = 15) and the US (n = 12)), 13 studies examined dietary choline intake, and 20 studies examined plasma choline concentration in relationship with various cardiovascular outcomes (e.g., cardiovascular events, coronary heart disease, ischemic stroke, acute myocardial infarction, peripheral artery disease, atrial fibrillation, and cardiovascular mortality) and metabolic outcomes (e.g., type 2 diabetes).
The evidence on dietary choline was equivocal. Several studies reported an inverse association, with higher dietary choline intake linked to a reduced risk of cardiometabolic events (Díez-Ricote et al., 2022; Jieru et al., 2024; Millard et al., 2018). In contrast, other studies found positive associations, suggesting that higher intake may be related to an increased risk (Bidulescu et al., 2007; Mazidi et al., 2019a; Van Parys et al., 2020; Yang et al., 2020; Zheng et al., 2016; Zuo et al., 2018). Four cohorts did not find an association between dietary choline intake and cardiometabolic risks (Bertoia et al., 2014; Dalmeijer et al., 2008; Golzarand et al., 2022; Nagata et al., 2015). The evidence on plasma choline concentration predominantly indicated that high plasma concentration of choline was associated with an increased risk or greater severity of cardiometabolic events (Andreu-Sánchez et al., 2024; Barkan-Talmor et al., 2019; Danne et al., 2007; Emechebe et al., 2019; Guasch-Ferré et al., 2017; Li et al., 2025a; Shea et al., 2024; Talmor-Barkan et al., 2024; Tang et al., 2017, 2021, 2024; Trøseid et al., 2015; Wang et al., 2014; Yang et al., 2018; Yang et al., 2023a).
Cross-sectional studies were conducted primarily in the US (n = 7) using the National Health and Nutrition Examination Survey (NHANES) (n = 6), reporting on the relationship between dietary/total choline intake (n = 6) and plasma choline concentration (n = 2) and cardiometabolic diseases. Four studies reported an inverse association between high dietary/total intake of choline and the risk of cardiovascular or metabolic events (Li et al., 2025b; Taesuwan et al., 2019; Wang et al., 2024; Zhou et al., 2023), while two reported a positive association between high plasma choline concentration and the risk of cardiometabolic diseases (Gillies et al., 2023; Roe et al., 2017). One article reported an inverse, non-linear relationship between dietary choline and atherosclerotic cardiovascular disease, particularly in men (Lin et al., 2024). Another study found an interaction between total choline intake and body mass index in relation to hypertension risk (Taesuwan et al., 2022). Sex was reported as a potential moderator in an inverse relationship between dietary/total choline intake and hypertension risk in a different study (Taesuwan et al., 2019).
Two case-control studies (China (n = 501), Pakistan (n = 240)) reported that high serum/plasma choline concentration was associated with an increased risk of cardiovascular disease (Ali et al., 2020; Dai et al., 2020). Additionally, two Mendelian Randomization analyses suggested potential causal links between a high genetically predicted plasma choline level and the risk of type 2 diabetes (Jia et al., 2019), valvular heart disease and myocardial infarction (Jing et al., 2023).
Evidence on offspring neurodevelopment
Different measures of neurodevelopment in relation to maternal choline intake/status during pregnancy were examined in 13 RCTs (Caudill et al., 2018; Cheatham et al., 2012; Christifano et al., 2023; Gimbel et al., 2022; Jacobson et al., 2018; Kable et al., 2015; Nguyen et al., 2016; Ross et al., 2013, 2016; Warton et al., 2021; Wozniak et al., 2015, 2020, 2025), 10 prospective cohorts (Boeke et al., 2013; Coles et al., 2015; Freedman et al., 2020; Hunter et al., 2021, 2022; Irvine et al., 2023; Signore et al., 2008; Strain et al., 2013; Villamor et al., 2012; Wu et al., 2012), one cross-sectional analysis (Bragg et al., 2023), and one case-control study (Freedman et al., 2019).
Of the RCTs, eight (five of which were conducted in the US, two in South Africa, and one in Ukraine with the sample size ranging from 18 to 372) investigated the effect of choline supplementation, either on pregnant mothers exposed to alcohol (n = 2) or on the children exposed to alcohol perinatally or diagnosed with fetal alcohol spectrum disorder (FASD) (n = 6), on various neurodevelopment measures. The evidence indicated that supplementing choline on pregnant mothers exposed to alcohol (2 g/day from study enrollment until delivery) was effective in mitigating brain volume reductions in the offspring (Warton et al., 2021) and in improving visual recognition memory at 12 months compared to the placebo group(Jacobson et al., 2018). Supplementing choline on the infants/children perinatally exposed to alcohol (513–900 mg/day for 9 months) was reported to be beneficial for their cognitive function measured between 6 months and 15 years of age (Gimbel et al., 2022; Kable et al., 2015; Wozniak et al., 2020, 2025). One trial reported no difference between the choline supplementation (625 mg/day for 6 weeks on school-age children) and the placebo group in terms of cognitive performance (Nguyen et al., 2016), while another trial reported a non-significant or potentially negative effect of choline supplementation (500 mg/day for 9 months on children aged 2.5–5 years) on memory function, moderated by age (Wozniak et al., 2015).
A total of four RCTs (conducted in the US) examined the effect of choline among pregnant mothers without alcohol exposure on neurodevelopment of the offspring. Supplementing choline at 930 mg/day for 12 weeks during the third trimester resulted in cognitive benefits of the offspring compared to a lower dose of 480 mg/day (Caudill et al., 2018). Supplementing choline from the second trimester to delivery also resulted in higher cognitive functioning of the offspring measured at 12 weeks and 40 months after birth (Ross et al., 2013, 2016). A dietary choline intervention showed more favorable neurodevelopment of the fetus at 36 weeks compared to placebo (Christifano et al., 2023).
Among prospective cohort studies on choline and neurodevelopment, six examined maternal plasma concentration of choline (conducted in the US, Canada, and the Republic of Seychelles), three examined maternal dietary intake of choline (all in the US), and one assessed maternal supplementation of choline with multivitamins (Ukraine). Some evidence reported positive associations between maternal plasma concentration of choline, measured during pregnancy, and cognitive functions of the offspring, measured between 3 months and 4 years (Freedman et al., 2020; Hunter et al., 2021, 2022; Wu et al., 2012), while others reported no association measured at 5 years of age (Signore et al., 2008; Strain et al., 2013). Maternal intake of dietary choline during the first two trimesters of pregnancy was not associated with cognitive function of the offspring at 3 years of age (Villamor et al., 2012) but was positively associated at 7 years of age (Boeke et al., 2013). An interaction of dietary folate and choline was also reported, showing that high folate combined with low choline intake may negatively influence the offspring's neurodevelopment (Irvine et al., 2023). A study supplementing choline on alcohol-exposed pregnant mothers showed an inverse association between choline intake and child's neurodevelopment at 6 months (Coles et al., 2015).
A cross-sectional study reported no association between plasma concentration of choline and cognitive function among Malawi children aged 6–15 months (Bragg et al., 2023). A case-control study examining mothers with infections during pregnancy reported that higher plasma concentration of choline among mothers was associated with better cognitive development of the offspring up to 3 months of age (Freedman et al., 2019).
Evidence on birth outcomes
Neural tube defects (NTDs) were examined in five studies conducted in the US (Carmichael et al., 2010; Lavery et al., 2014; Petersen et al., 2019; Shaw et al., 2004, 2009), three in relation to maternal dietary intake of choline, one in relation to maternal intake of choline supplements, and one in relation to maternal serum concentration of choline. The studies (sample size ranging from 409 to 2995 mother–offspring pairs) found strong inverse associations between high dietary intake of choline and the risk of NTDs. This finding was supported by a strong inverse association between maternal serum concentration of choline and the risk of NTDs (n = 489 pairs). The evidence was inconclusive about the relationship between maternal choline supplementation and NTDs (n = 955 pairs).
Birthweight and birth size were investigated in four prospective cohort studies in China, Japan, the Netherlands, and the US (sample size ranging from 115 to 1331 mother–offspring pairs) (Du et al., 2019; Hoffman et al., 2024; Molto-Puigmarti et al., 2021; Nakanishi et al., 2021). Maternal plasma choline concentration and cord blood choline concentration were each inversely associated with the risk of low birthweight (defined as <10th percentile) and low body mass index. The relationship between maternal choline status and growth in childhood (up to 5 years of age) was investigated by two other studies (sample size ranging from 400 to 985 pairs) (Kadam et al., 2024; Van Lee et al., 2019). High choline status of mothers during pregnancy was positively associated with offspring growth and anthropometric measures during childhood. Another study in Malawi (Bragg et al., 2023) did not find an association between plasma concentration of choline and anthropometric measurements among infants aged 6–15 months.
Evidence on pregnancy outcomes
The relationship between choline intake/status and pregnancy outcomes were investigated in nine observational studies (gestational diabetes (GDM) n = 5, pre-eclampsia n = 2, pre-term delivery n = 2). Prospective cohort investigations (Canada, China) examining plasma choline concentrations and the risk of GDM reported no associations (Barzilay et al., 2018; Gong et al., 2021), while two nested case-control studies (China) reported an inverse relationship between serum choline status and GDM risk (Huo et al., 2019; Wang et al., 2022).
A cross-sectional study in the US (McArthur et al., n.d.) reported no association between plasma choline and the risk of GDM or pre-eclampsia, which contrasted the findings from a case-control study in Norway (Braekke et al., 2007). Another case-control study in China reported an inverse association between total choline intake and the risk of pre-eclampsia (Ma et al., 2022). Other case-control studies (n = 2; US, China) were inconclusive on the relationship between dietary choline intake and the risk of pre-term delivery (Carmichael et al., 2013; Zhu et al., 2021).
Evidence on liver diseases
A total of 16 studies involving 18 datasets (intervention trials n = 2, prospective cohort n = 2, cross-sectional n = 11, case-control n = 3) investigated the relationship between choline intake/status and various measures of liver dysfunction. One intervention trial in the US found that dietary choline deprivation resulted in development of fatty liver or muscle damage among the participants (n = 57) (Fischer et al., 2007). Another trial (US) provided choline supplements for individuals with hepatic steatosis and receiving parenteral nutrition (TPN) and reported that choline deficiency was a significant factor in the development of TPN-associated liver disease (Buchman et al., 2001). Two prospective cohorts in China reported that total choline intake was inversely associated with the risk of fatty liver and non-alcoholic fatty liver disease (NAFLD), with a potential effect modification by obesity status among women (Yu et al., 2014).
US NHANES data were used in six cross-sectional studies. Higher dietary choline intake was associated with a lower risk of non-alcoholic steatohepatitis (NASH) or NAFLD in the general population (Chai et al., 2023; Mazidi et al., 2019b) and among post-menopausal women (Li et al., 2023). Higher dietary choline was not associated with metabolic dysfunction-associated steatotic liver disease (MASLD); however, among male or obese individuals with MASLD, higher dietary choline intake was associated with higher risk of fibrosis (Taesuwan et al., 2025). One NHANES analysis reported a positive relationship between dietary choline intake and the risks of liver steatosis and fibrosis (Liu et al., 2021b).
Dietary choline intake was also associated with a reduced risk of hepatic injury among Taiwanese individuals with hepatitis (Cheng et al., 2017) and with a lower risk of fatty liver among post-menopausal women in Poland (Muzsik-Kazimierska et al., 2022). In contrast, plasma choline status was not associated with NAFLD in a community-based cross-sectional study in China (Chen et al., 2016). Other cross-sectional studies in Mexico and Japan reported elevated plasma/serum choline concentration correlating with NAFLD and NASH among obese individuals undergoing bariatric surgery (León-Mimila et al., 2021) and among patients with liver or metabolic conditions (Imajo et al., 2012). Conversely, evidence from case-control studies suggested protective associations, with higher total choline intake/serum choline concentrations linked to reduced risk of NAFLD (Chen et al., 2016), liver fibrosis in individuals with NAFLD (Vilar-Gomez et al., 2021), and liver steatosis in individuals with visceral obesity(Chang et al., 2022).
Evidence on kidney diseases
We identified three prospective cohort studies (Denmark, Germany, and Spain) and one Mendelian Randomization analysis investigating the relationship between choline intake/status and kidney functions. Among individuals with type 2 diabetes and albuminuria (n = 311), higher plasma choline concentration was associated with a higher risk of eGFR decline (Winther et al., 2021). Among individuals with pre-existing chronic kidney disease (n = 478), plasma choline concentration was not associated with adverse renal outcomes (Obeid et al., 2024). Among older adults with obesity and metabolic syndrome (n = 5613), higher intake of dietary choline was associated with improvements in renal function (Díez-Ricote et al., 2022). A Mendelian Randomization analysis did not detect a significant association between genetically elevated choline and an increased risk of chronic kidney disease (Jia et al., 2019).
Evidence on cancer outcomes
No association was found between dietary choline intake and the risk of breast cancer in a European prospective cohort (n = 318,686) (Van Puyvelde et al., 2021). A Chinese prospective cohort studied individuals with newly diagnosed hepatocellular carcinoma (HCC) (n = 905) and reported that higher total choline intake, either from water-soluble forms or lipid-soluble forms, was not associated with HCC-specific mortality (Liu et al., 2020). In colorectal cancer, a US population-based cohort (n = 14,289) found an inverse association between dietary choline intake and the risk of colon cancer (Jieru et al., 2024); while a nested case-control study conducted in European countries (n = 3690) reported an inverse association between plasma choline concentration and the risk of colorectal cancer among women (Nitter et al., 2014).
Evidence on cognitive function among adults
Cross-sectional studies from New Zealand, Norway, and the US (n = 4; sample size ranging from 313 to 2796) examined the correlation between plasma choline status or total choline intake and cognitive function among older adults. In a New Zealand study, plasma choline concentration showed no association with global cognition, as measured by the Montreal Cognitive Assessment (MoCA) (Gillies et al., 2023). By contrast, a Norwegian study reported a positive, dose-dependent association between plasma choline concentration and cognitive performance assessed with the Mini-Mental State Examination (MMSE) (Nurk et al., 2013). Evidence from the US indicated that higher total choline intake (187.06–399.50 mg/day) significantly reduced the risk of poor cognitive function, with benefits plateauing above ∼400 mg/day (Liu et al., 2021a). However, another US study found no association between dietary/total choline intake and cognitive performance (An et al., 2023).
Evidence on all-cause mortality
A total of 12 longitudinal analyses were identified in the context of choline intake/status and all-cause mortality. Findings in the general population were mixed. In a large Swedish cohort (n = 102,731), total choline intake was not associated with all-cause mortality (Karlsson et al., 2024). In contrast, higher dietary choline intake was linked to an increased risk of mortality in two US cohorts (n = 120,412 and n = 20,325) (Mazidi et al., 2019b; Zheng et al., 2016) and among black Americans (Yang et al., 2020). Conversely, in another US cohort (n = 14,289), moderate choline intake was associated with a reduced risk of total mortality (Jieru et al., 2024). Yang et al. (Yang et al., 2020) further suggested that the relationship between choline intake and mortality may be modified by alcohol consumption and pre-existing cardiometabolic conditions.
There were positive associations between plasma/serum concentration of choline and all-cause mortality among older adults (US, n = 5333) (Fretts et al., 2022) and individuals with hypertension (China, n = 558) (Song et al., 2021). Among individuals with chronic heart failure (Norway, n = 155) (Trøseid et al., 2015), type 2 diabetes (Denmark, n = 311) (Winther et al., 2021), stable angina pectoris (Norway, n = 2568) (Schartum-Hansen et al., 2015), and chronic kidney disease (Germany, n = 478), there was no association between plasma choline status and all-cause mortality. Among individuals with hepatocellular carcinoma (China, n = 905) (Liu et al., 2020), higher intake of dietary choline was associated with a reduced risk of all-cause mortality.
Systematic reviews and meta-analyses
Of the nine systematic reviews and meta-analyses that we identified, published between 2017 and 2025, four (44%) examined cardiometabolic diseases (Khan et al., 2024; Meyer and Shea, 2017; Sharifi-Zahabi et al., 2024; Yang et al., 2023) and another four (44%) examined neurodevelopmental outcomes of the offspring (Akison et al., 2018; Derbyshire and Obeid, 2020; Gould et al., 2025; Obeid et al., 2022).
A subset of meta-analyses (n = 3) found that higher choline status or intake was associated with an increased risk of cardiovascular disease (Yang et al., 2023) and major cardiovascular adverse events (Khan et al., 2024), and each 100 mg/day increase in dietary choline intake was associated with an 11% increase in cardiovascular mortality and a 6% increase in all-cause mortality (Sharifi-Zahabi et al., 2024). Conversely, one meta-analysis reported no association between choline intake and incident cardiovascular disease (Meyer and Shea, 2017).
The evidence on neurodevelopmental outcomes appears to be more nuanced. Human intervention trials demonstrated favorable effects of higher maternal choline intakes on neurocognition of the offspring; however, human observational studies were divided, particularly those using plasma choline concentration as an exposure measure (Derbyshire and Obeid, 2020; Obeid et al., 2022). Evidence from the human observational studies was appraised to be insufficient and have critical limitations (Gould et al., 2025; Obeid et al., 2022).
The methodological quality of the syntheses assessed with AMSTAR-2 was low to moderate (see Supplementary Table 2). Most of the syntheses were rated low in the comprehensiveness of the search strategy (Q4) and adequate description of the included studies (Q8). While all syntheses assessed the potential risk of bias in the primary studies (Q9), many did not consider the risk of bias in combining data or interpreting results (Q12, 13). Heterogeneity among the included studies were often not investigated or discussed (Q14).
Discussion
We mapped the broader evidence landscape on choline and human health. Research interest in choline intake/status and health outcomes has been increasing, mostly in North American and Europe. Among adults, the majority of studies have focused on cardiometabolic, liver, and kidney outcomes, whereas research on neurodevelopment has primarily examined maternal choline status and offspring health indicators.
The evidence was insufficient or equivocal for many health outcomes. Prospective and cross-sectional dietary studies tended to report inverse associations between choline intake/status and various cardiovascular events or metabolic markers, while plasma studies, both prospective and retrospective, tended to report positive relationships. A Mendelian Randomization analyses reported a causal link between high choline status and type 2 diabetes, ventricular heart disease, and myocardial infarction risks. The findings on pregnancy outcomes, cancer, and mortality were inconclusive.
Choline supplementation in pregnant women or in children prenatally exposed to alcohol appears to support neurodevelopment, with potential effect modification by the child's age. High choline status was also associated with better cognitive function among otherwise healthy mother–offspring pairs. Although evidence remains limited, there may be cognitive benefits of choline among older adults. With respect to liver health, most studies indicated an inverse association between choline status and the risk or severity of liver steatosis, NAFLD, NASH, and fibrosis.
A separate examination of the evidence syntheses showed that a contextualized understanding of the pooled evidence may be important, particularly when heterogeneity and potential risk of bias are present among the primary studies. Consideration of methodological issues specific to nutrition studies (e.g., ascertainment of exposure, baseline nutrient status) may also improve the ability to pool data and clarify relationships between micronutrients and health outcomes.
A large pool of preclinical evidence was synthesized in two reviews (Akison et al., 2018; Derbyshire and Obeid, 2020). Among infants perinatally exposed to alcohol, maternal choline supplementation was shown to ameliorate some of the cognitive deficits arising from alcohol exposure during development (Akison et al., 2018). Supplementing choline to the offspring was also shown to be beneficial for neuronal plasticity, morphology, and cerebral cortex development (Derbyshire and Obeid, 2020).
Despite a wealth of preclinical studies on the topic and the critical role choline plays in healthy development, human research on choline was relatively scarce and a substantial gap could be identified in many health outcomes. In the same context, we did not identify many studies investigating different forms of choline (water-soluble or lipid-soluble) or the joint effects of choline and other one-carbon nutrients. We also note potential residual confounding in the observational studies.
This work has several limitations. First, we restricted the search to three databases which may have resulted in a smaller pool of identified studies. To address this limitation, we manually searched other resources and references. Second, the review was restricted to human studies due to resource constraints. Animal studies, though not free from biases, offer insights from rigorous experimental settings and may provide hypotheses for potential translation to human health. Future syntheses incorporating preclinical and mechanistic evidence may be well-positioned to inform the next steps in choline research.
Conclusion
Our review provides a comprehensive scan of the current evidence on choline and its relations to various health outcomes. The evidence predominantly consisted of observational studies and largely concentrated on cardiometabolic outcomes and infant/child neurodevelopment. Potential benefits of high maternal choline intake/status was indicated for neurodevelopment of the offspring; however, the evidence was insufficient or equivocal for most of the outcomes. A more comprehensive synthesis incorporating preclinical evidence will improve the current understanding of choline and its health effects.
Supplemental Material
sj-docx-1-nah-10.1177_02601060261430655 - Supplemental material for Choline status and related health outcomes in humans: A scoping review
Supplemental material, sj-docx-1-nah-10.1177_02601060261430655 for Choline status and related health outcomes in humans: A scoping review by Samantha Yoo, Andrew Aoun, Azita Montazeri, Erin Collins and Julian Little in Nutrition and Health
Footnotes
Acknowledgments
None.
Ethical approval
Not applicable.
Consent for publication
Not applicable.
Authors’ contributions
JL, SY, AM, and EC conceptualized the review. AA developed and executed the search strategy. AA, SY, and AM independently screened the articles. SY and AM independently extracted data and AA verified the extraction. SY performed the synthesis and drafted the manuscript. AA contributed to drafting the method section of the manuscript. AM, AA, EC, and JL provided critical inputs to the manuscript. All authors reviewed and approved the final draft of the manuscript.
Funding
This review was funded by the Canadian Institutes for Health Research (CIHR) Grant FRN-PJT-175263.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of data and materials
This review synthesized published articles that are publicly accessible.
Supplemental material
Supplemental material for this article is available online.
