Abstract
Background
Dietary patterns are linked to obesity and metabolic health.
Aim
To explore associations between dietary patterns, adiposity, and metabolic syndrome risk in Pacific and New Zealand (NZ) European women.
Methods
Pacific (n = 126) and NZ European (n = 161) women aged 18–45 years from Auckland were recruited based on BMI (normal weight and obesity) with approximately half in each ethnic group. Body fat percentage (BF%) was assessed with DXA and used to stratify participants into low (<35%) or high (≥35%) BF% groups. Dietary intake was assessed using a 5-day food record and semi-quantitative food frequency questionnaire, analyzed with the National Cancer Institute method. Dietary patterns were derived using principal component analysis. Fasting blood was analyzed for insulin, glucose, and lipids. Metabolic syndrome was defined using harmonized criteria.
Results
Four dietary patterns were identified: Vegetarian, Processed, Prudent, and Keto. Regression analysis controlling for ethnicity, age, socioeconomic deprivation, energy intake, and physical activity showed that higher adherence to Vegetarian and Prudent patterns characterized by “core” foods was inversely associated with BF% and visceral fat% (
Conclusion
Greater adherence to “core” food patterns was associated with lower adiposity and metabolic disease risk, while patterns high in processed “discretionary” foods and less diversity of “core” foods were associated with higher metabolic risk.
Introduction
In line with international trends, the prevalence of obesity in Aotearoa New Zealand (NZ) has increased considerably in the past three decades, with Pacific people (from different Pacific Island Nations in the South Pacific), Māori (the indigenous people of NZ), and those living in the most deprived neighborhoods disproportionately affected (Ministry of Health, 2024; World Health Organization, 2025). In the 2023/2024 NZ health survey, 65% of Pacific and 51% of Māori people were classified as having obesity, compared to 33% of NZ Europeans (Ministry of Health, 2024; World Health Organization, 2025). These disparities are reflected in the higher incidence of obesity associated noncommunicable diseases (NCDs), such as cardiovascular disease and type 2 diabetes, observed in these groups (Atlantis et al., 2017; Coppell et al., 2013; Health New Zealand Te Whatu Ora, 2023; Yu et al., 2020).
A healthy diet (that is nutritionally adequate and aligns with food based dietary guidelines) is protective against major NCDs (Afshin et al., 2019; World Cancer Research Fund/American Institute for Cancer Research, 2018), but how readily available and affordable healthy foods are to people is influenced by socioeconomic factors, income level, and their relative food environment (Darmon and Drewnowski, 2015; Herforth et al., 2020; Pearce et al., 2007). Healthier (nutrient-dense) foods tend to cost more on a per calorie basis than refined, energy-dense, and nutrient-poor foods (Darmon and Drewnowski, 2015; Herforth et al., 2020). Low-income neighborhoods are often underserved by supermarkets and fruit and vegetable stores, with a higher density of outlets selling fast-foods (Pearce et al., 2007; Sushil et al., 2017). People living in low income neighborhoods are also more likely to experience food insecurity (i.e., limited access to affordable nutritious foods) and therefore are less able to consume healthy diets, thus increasing risk of obesity and NCDs (Darmon and Drewnowski, 2015; Herforth et al., 2020). In NZ, Pacific people and Māori are more likely to live in low income neighborhoods and experience food insecurity (Futures, 2017; Health New Zealand Te Whatu Ora, 2023; Rush, 2009) which likely partially explains the observed differences in the burden of NCDs between Māori, Pacific, and NZ European people.
Nutrition advice and guidelines aim to promote dietary intakes that are compatible with good health. However, focusing nutrition recommendations on intakes of nutrients, individual foods, or food groups does not readily translate into practical and feasible recommendations for public health nutrition interventions. To achieve improvements in population health, they need to reflect the realities of the populations that they are intended for, taking into account that different groups consume different foods and nutrients, not in isolation, but rather as combinations in meals and snacks and the quality of which is influenced by factors including: food cost, income (Darmon and Drewnowski, 2015; Herforth et al., 2020), sociocultural factors, cultural preferences, and local food availability (Anderson, 2014; Robinson et al., 2021).
Dietary pattern analysis enables the assessment of the interrelationship between foods and nutrients consumed together, for which associations with disease and health outcomes can be assessed (Hu, 2002; Newby and Tucker, 2004; Tapsell et al., 2016).
Methods
Participants were part of the cross-sectional PROMISE (PRedictors linking Obesity and gut MIcrobiomE) study that aimed to characterize the gut microbiota of Pacific and NZ European women with different body fat profiles (normal weight and obesity). Details of the PROMISE study procedures and recruitment strategies, conducted between July 2016 and September 2017, have been published elsewhere (Kindleysides et al., 2019). In brief, eligible participants were premenopausal and postmenarche Pacific and NZ European women aged 18–45 years from the Auckland region who were free from any chronic illness. Participants were recruited using convenience sampling, with purposive stratification by ethnicity and BMI category to ensure balanced numbers of women with either a normal BMI (18–24.9 kg/m2) or a BMI in the obesity range (≥30 kg/m2) in each ethnic group. Self-reported BMI served as the initial screening tool and women with a BMI in the overweight range (25.0–29.9 kg/m2) were also included in the study and all analyses. As body fat percentage (BF%) was the primary variable of interest, participants were subsequently stratified into either the low-BF% (<35%) or high-BF% (≥35%) groups for all subsequent analyses, based on international BF% indicators of obesity (Dickey et al., 1998; Jo and Mainous, 2018; Kramer et al., 2013; Oliveros et al., 2014). The PROMISE study sample size was calculated to reach sufficient power to detect a difference in gene diversity between groups (Kindleysides et al., 2019). Participants attended two study visits and completed at home data collection between study visits.
The PROMISE study, from which the present analyses are derived, received ethical approval from the Southern Health Disability Ethics Committee (Reference: 16/STH/32). All procedures were conducted in accordance with the guidelines of the Declaration of Helsinki. The trial was registered at anzctr.org.au (ACTRN12618000432213). All participants received detailed information about the procedures, and study measurements, and gave written informed consent prior to their participation in the study.
Demographic, anthropometric, and biochemical sampling
Details of data collection have been published previously (Kindleysides et al., 2019; Renall et al., 2023, 2024). Relevant demographic information was captured with standardized face-to-face interviews. The NZ Deprivation Index (NZDep2013) was used to assign a socioeconomic deprivation score ranging from 1 “least deprived” to 10 “most deprived” (Atkinson et al., 2014). Blood pressure was measured with a digital blood pressure monitor (Omron HEM-907, Omron Healthcare Inc., Kyoto, Japan). Fasting weight was measured to the nearest 0.01 kg and stretched height to 0.1 cm and these values were used to calculate BMI in kg/m2 (Garrow, 1985). A whole-body scan using Dual-energy X-ray Absorptiometry (DXA) (Hologic QDR Discovery A, Hologic Inc, Bedford, MA with APEX V. 3.2 software) was performed to assess total body fat percentage (BF%) and visceral fat mass (VAT%). Blood was collected following an overnight fast, and metabolic markers (plasma glucose, insulin, glycosylated haemoglobin (HbA1c), and lipid profile) were assessed using standard diagnostic methods as described in detail previously (Kindleysides et al., 2019; Renall et al., 2024). Homeostasis model assessment (HOMA-IR) index for insulin resistance was calculated (Matthews et al., 1985). Presence of metabolic syndrome was determined with the harmonized definition, i.e., having ≥3 of the following: triglycerides ≥ 1.7 mmol/L, blood pressure ≥ 130/85 mm Hg, waist circumference ≥ 80 cm (Ministry of Health, 2017), fasting glucose ≥ 5.6 mmol/L, and HDL cholesterol < 1.3 mmol/L (Alberti et al., 2009).
Physical activity
As described in detail elsewhere (Slater et al., 2021), physical activity was assessed with a triaxial w-GT3X accelerometer (Actigraph, Pensacola, FL, USA) worn on the non-dominant hip for seven consecutive days. Data were collected in 60-s epochs and processed in ActiLife (Version 6.13.3). Nonwear time was defined as ≥60 min of zero counts, and a valid day required ≥10 h of wear. Participants with at least four valid days, including one weekend day, were included. Sleep periods identified from participant diaries were removed, and remaining waking-hour epochs were classified using standard cut-points to quantify time spent in different physical activity intensities, expressed as counts per min, (cpm) and minutes per day (min/day). In this analysis, a combined measure of moderate and vigorous-intensity physical activity (MVPA; >2020 cpm) was used, because current physical activity guidelines in NZ recommend at least 30 min of moderate intensity (or higher) activity 5 or more days of the week (Ministry of Health, 2020).
Dietary assessment
Details of dietary assessment and dietary data processing have been published elsewhere (Renall et al., 2023, 2024). In brief, each participant completed a 5-day, nonconsecutive, estimated food record (5DFR) at home, and a validated 220 item semiquantitative Food Frequency Questionnaire (NZWFFQ) (Beck et al., 2018a) during study visit 2. Both tools were self-reported; dietitians reviewed the completed records with participants to clarify entries and ensure plausibility prior to data entry. FoodWorks9 (Xyris Software (Australia) Pvt Ltd, Queensland, Australia) was used to estimate energy, macro- and micronutrient intakes of the 5DFR and NZWFFQ. Dietary data for those reporting energy intakes higher than 27,000 kJ/day (6453 kcal/day) or lower than 2100 kJ/day (502 kcal/day) were excluded from further analyses. The energy intake cut-offs used to identify potential misreporting were based on established recommendations (Willett, 2012) and were adapted for this population following detailed data reviews and prior studies involving similar ethnic minority groups (Chacko George et al., 2004; Gemming et al., 2014; Kolonel et al., 2000; Metcalf et al., 2008; Smith et al., 2015).
All reported foods items from the 5DFR (n > 2850) and the NZWFFQ were assigned to one of 55 food groups (in g/day) based on similar nutritional composition and food groupings used in relevant NZ population studies (Supplementary Table 1; Beck et al., 2018b; Ministry of Health, 2012). For mixed dishes and beverages, detailed entries in the 5DFR (ingredient weights, cooking methods etc.) were disaggregated and assigned to relevant food groups. For example, “spaghetti bolognaise,” was separated into pasta (“refined grains”), mince (“red meat”), tomatoes (“tomatoes”), and onion (“other nonstarchy vegetables”). When insufficient detail was provided, the entire dish was assigned to the most representative food group (e.g., “red meat” for spaghetti bolognaise).
For descriptive purposes, the 55 food groups were classified as “core” or “discretionary” based on the NZ (Ministry of Health, 2020) and Australian (National Health and Medical Research Council et al., 2006) dietary guidelines, with “tea” and “coffee” categorized as “other.” NZ guidelines recommend daily consumption from four “core” food groups: (a) vegetables and fruit; (b) whole grain cereals; (c) meat alternatives and lean proteins; and (d) low-fat milk products (Ministry of Health, 2020). “Discretionary” foods, as defined by Australian guidelines, include foods like cakes, biscuits, fried foods, processed meats, chocolate, and confectionary, that fall outside of the core food groups and are characteristically high in added sugar, saturated fat, and salt (Ministry of Health, 2020; National Health and Medical Research Council et al., 2006).
As published in detail elsewhere (Renall et al., 2023, 2024), the National Cancer Institute (NCI) method (National Cancer Institute, n.d.) was used to estimate the average daily intake of the 55 food groups (g/day). Intakes were estimated over a 1-month period because the semiquantitative FFQ used in this study assessed usual dietary intake over the previous month (Beck et al., 2018a). The 5DFR was used as the primary data in the NCI analyses and the covariates ethnicity, age, BMI, weekday/weekend, season, and the FFQ data were included in the estimation of the dietary data.
Principal component analysis (PCA) was conducted to identify dietary patterns. Intake estimates for each food group were logarithmically transformed (
Statistical analyses were conducted using SAS Enterprise Guide Version 7.1 (SAS institute, Cary, NC, USA) and SPSS 25.0 (SPSS inc., Chicago, IL, USA)). Non-normal data were reported as medians [25th, 75th] and Mann–Whitney
Multiple linear regression was used to assess the association between energy adjusted habitual dietary pattern scores and body composition and markers of metabolic health. Logistic regression was used to assess the association between dietary pattern scores and metabolic syndrome. Analyses were adjusted for ethnicity (due to selective recruitment), deprivation (NZDep2013; see above), and age based on their observed associations with dietary patterns in similar populations (Beck et al., 2018b). Time spent in MVPA was also included as a covariate due to its established association with metabolic health (Bradshaw et al., 2013; Warburton, 2006), and observed differences in group activity levels (Slater et al., 2021). Further adjustment for BF% group was conducted to account for study design and assess the independent association of adherence to dietary patterns and metabolic health markers. Regression coefficients (β) obtained from log-transformed data represent relative differences and were expressed as a ratio by exponentiating the regression coefficient (eβ). The association between adherence to dietary patterns and body composition and risk of metabolic syndrome were expressed per 1 unit of change in the dietary pattern score (which reflects adherence to the dietary pattern). Collinearity between the variables included in the model was assessed by computing the variance inflation factor and tolerance; no collinearity was detected.
Results
Seventeen participants were excluded based on implausible energy intake estimates in the NZWFFQ (> 27,000 kJ/day [>6453 kcal/day]), leaving a total of 287 participants, 126 Pacific, and 161 NZ European women, included in the analyses (Figure 1).
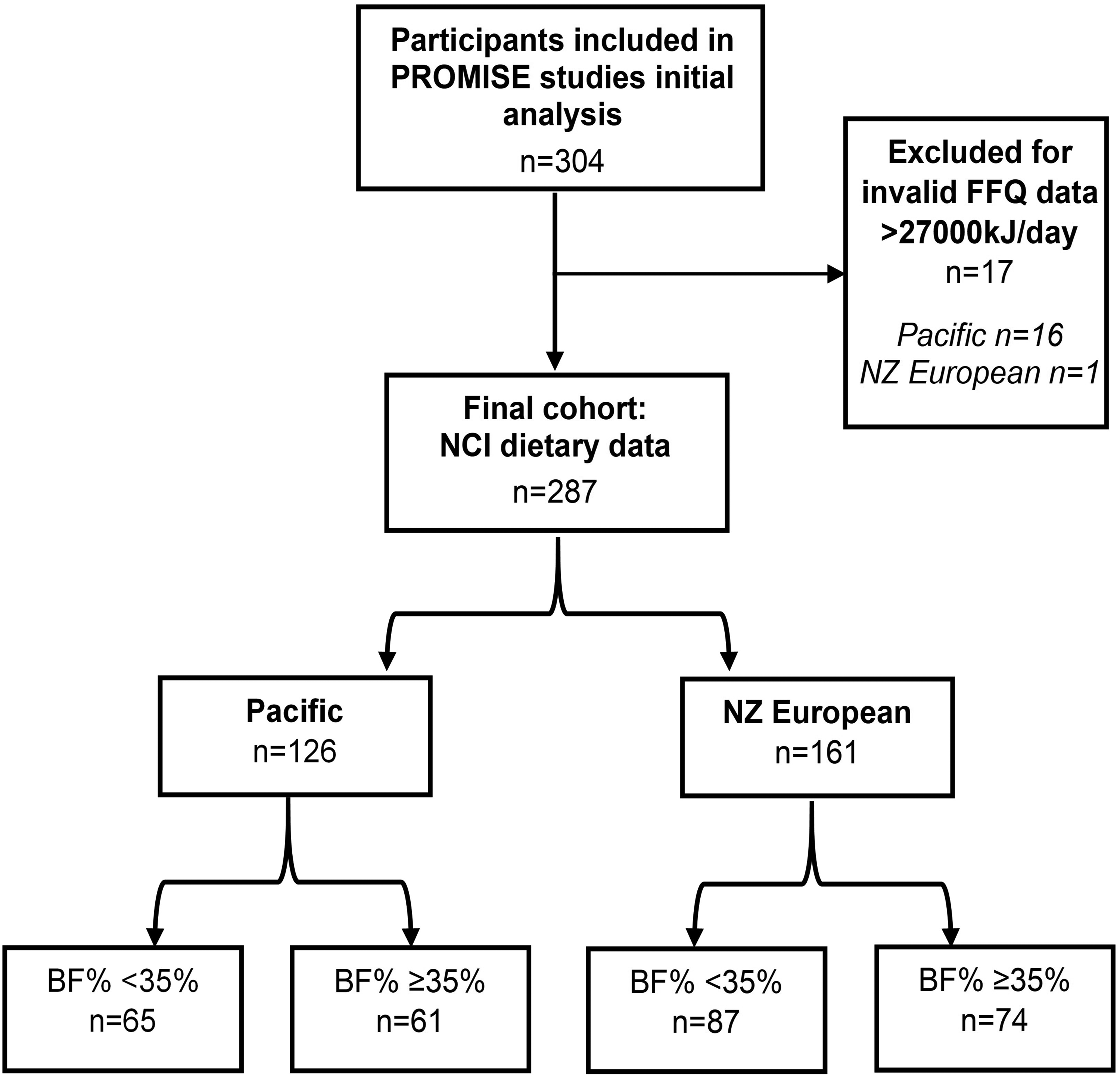
Flowchart of participants included in current analysis.
NZ European women were older than Pacific women with a median age of 32 years [25–75 percentiles: 25, 37,] and 23 years [25–75 percentiles: 20, 29], respectively. NZ European women also had lower NZDep2013 scores (Figure 2), including HbA1c, fasting insulin and HOMA-IR scores compared to Pacific women (
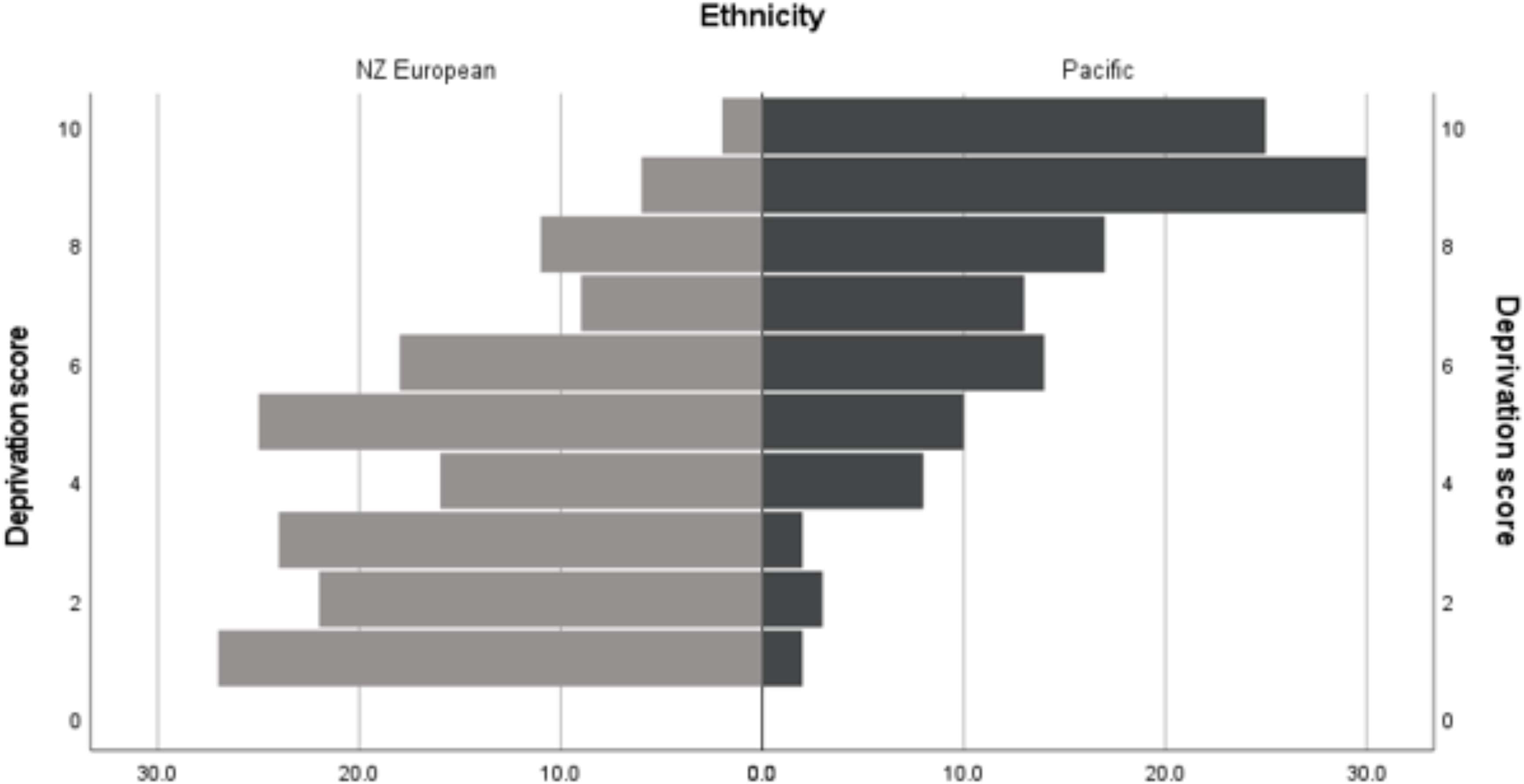
Percentage of NZ European and Pacific women in each score category of the NZ Deprivation Index 2013. Percentage of NZ European (left) and Pacific (right) women in each score category of the NZ Deprivation Index ranging from 1: “least deprived” to 10: “most deprived” (Atkinson et al., 2014).
Participant characteristics stratified by ethnicity and BF% group.
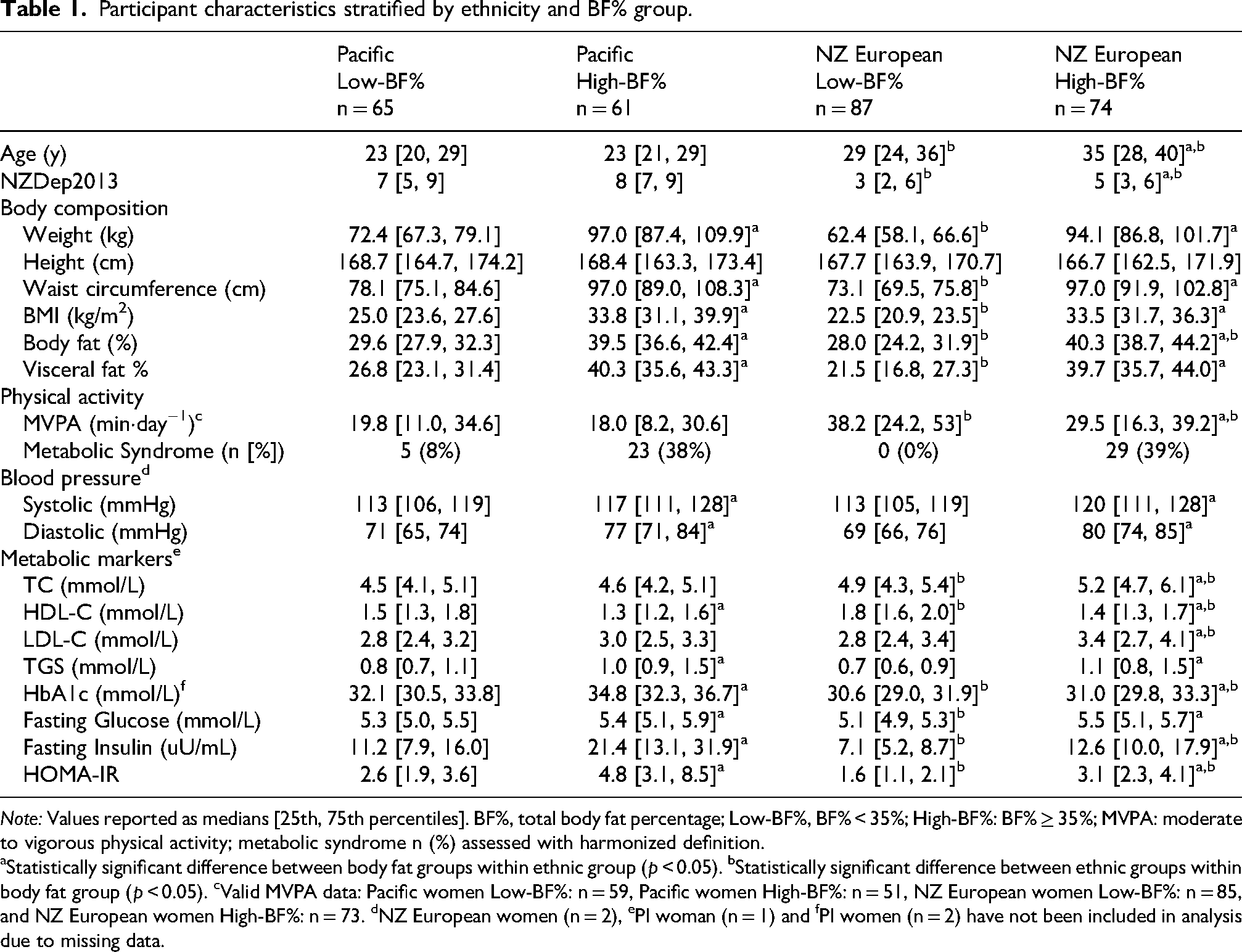
aStatistically significant difference between body fat groups within ethnic group (
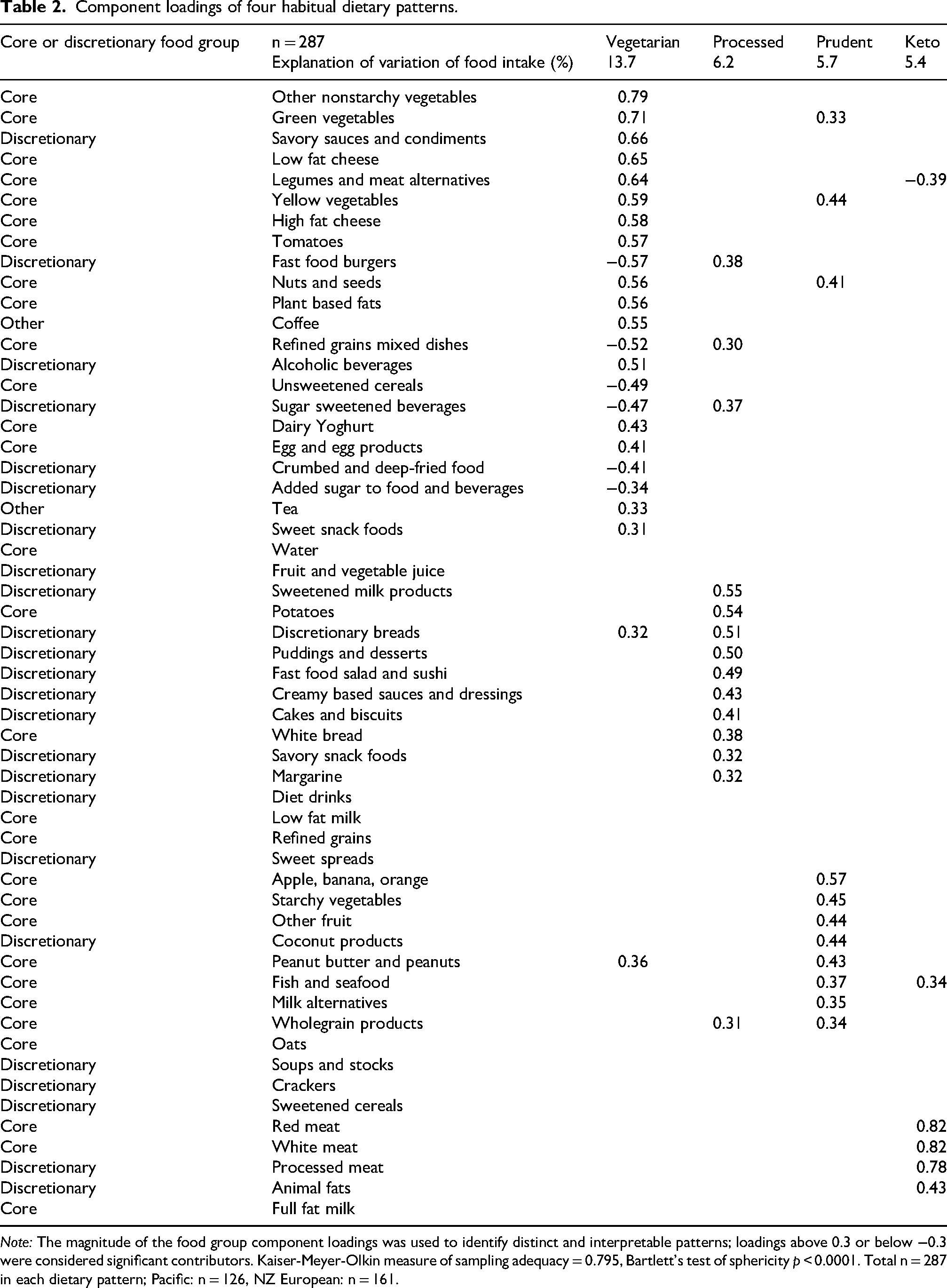
Four dietary patterns were extracted using PCA explaining 31% of the total observed variance of the habitual dietary intake data (Table 2). The first dietary pattern, referred to as the Vegetarian pattern, explained 13.7% of the variance and was characterized by high loadings on nonstarchy vegetables, plant-based proteins and dairy products, and negative loadings on fast foods, refined grains, and sugar sweetened beverages. The second dietary pattern, referred to as the Processed pattern, was characterized by higher loadings on processed foods such as sweetened milk products, discretionary breads, puddings, and cakes. The third dietary pattern, referred to as the Prudent pattern, was characterized by high loadings on fruit, starchy vegetables, and nuts. Finally, the fourth dietary pattern, referred to as the Keto pattern, was characterized by high loadings on meat and animal fat, and negative loadings on legumes and meat alternatives (Table 2).
Component loadings of four habitual dietary patterns.
Adherence to the Vegetarian dietary pattern was higher among NZ European women than Pacific women (Supplementary Figure 1). Higher adherence to Vegetarian and Prudent dietary patterns was inversely associated with multiple measures of adiposity. For example, every 1 unit increase in Vegetarian dietary pattern score (reflecting higher adherence) was associated with 0.9 kg/m2 lower BMI, 2.9% lower BF%, and 4% lower VAT% (
Association between adherence to dietary patterns and body composition and metabolic markers.
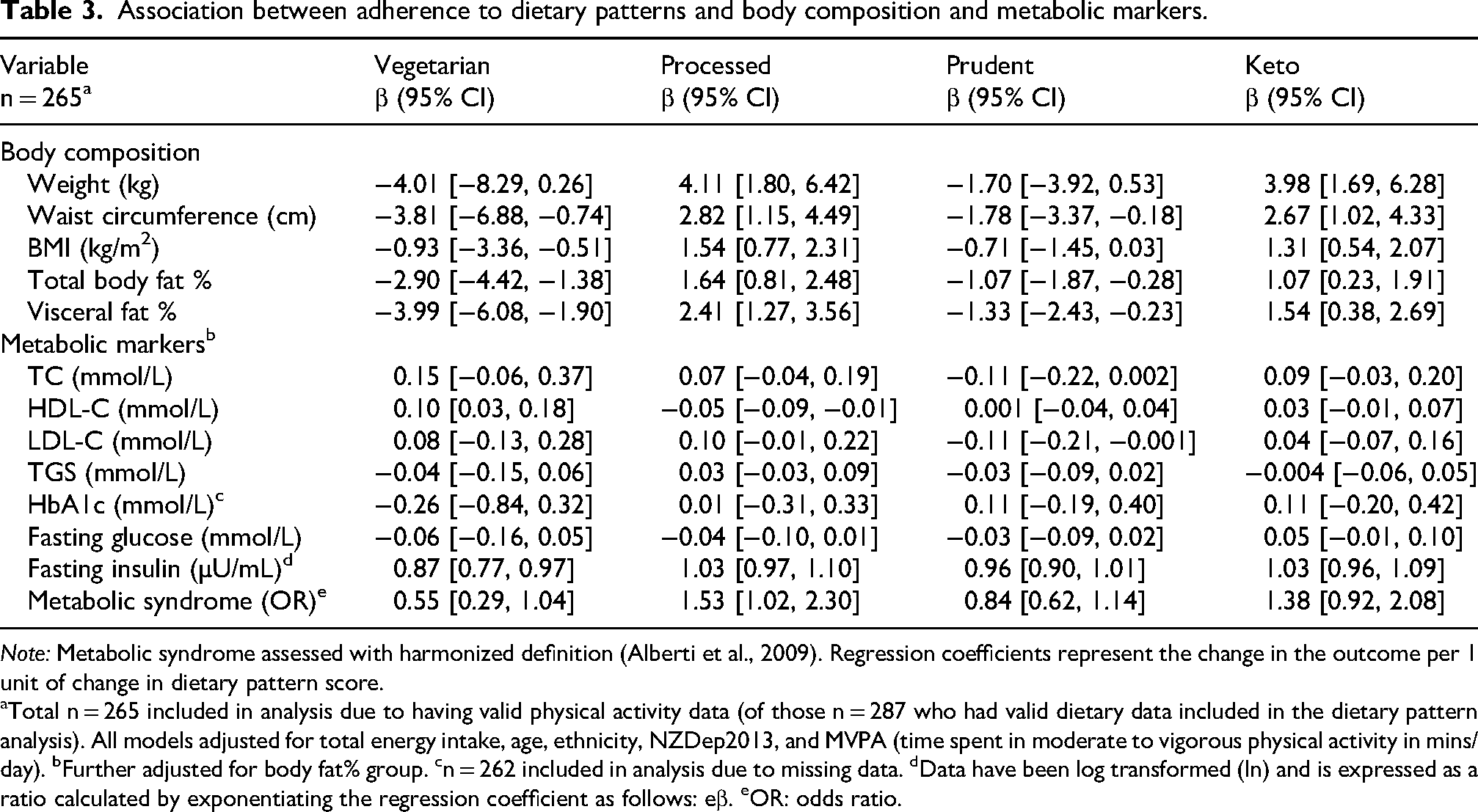
aTotal n = 265 included in analysis due to having valid physical activity data (of those n = 287 who had valid dietary data included in the dietary pattern analysis). All models adjusted for total energy intake, age, ethnicity, NZDep2013, and MVPA (time spent in moderate to vigorous physical activity in mins/day). bFurther adjusted for body fat% group. cn = 262 included in analysis due to missing data. dData have been log transformed (ln) and is expressed as a ratio calculated by exponentiating the regression coefficient as follows: eβ. eOR: odds ratio.
Higher adherence to the Vegetarian pattern was also positively associated with HDL-C and inversely associated with fasting insulin and HOMA-IR scores whereas higher adherence to the Processed pattern was inversely associated with HDL-C. Fasting insulin and HOMA-IR scores were also positively associated with higher adherence to the Processed pattern but negatively associated with higher adherence to the Prudent pattern (data not shown); however, these associations were no longer significant following further adjustments for BF% groups (Table 3). However, higher adherence to the Processed dietary pattern was associated with an increased likelihood of metabolic syndrome; there was a 53% increase in the odds (OR: 1.53 [95% CI: 1.02, 2.30],
Discussion
This study aimed to identify possible new insights for public health nutrition strategies for Pacific women by exploring the association between dietary patterns, adiposity, and metabolic disease risk in a NZ population of Pacific and NZ European women who differ in terms of body composition, metabolic disease risk, and relative socioeconomic deprivation. Similar to previous observations (Newby and Tucker, 2004; Rezagholizadeh et al., 2017; Tucker, 2010; United States Department of Agriculture, 2014), our study showed that higher adherence to vegetarian and prudent dietary patterns characterized by “core” foods were inversely associated with adiposity (i.e., BF% and VAT%), whereas higher adherence to a processed pattern characterized by “discretionary” foods (Schwingshackl and Hoffmann, 2015; Tucker, 2010; United States Department of Agriculture, 2014), and a keto pattern characterized by less diversity of “core” foods, were positively associated with adiposity (weight, BMI, BF%, and VAT%). Higher adherence to the Processed pattern was also associated with an increased risk of metabolic syndrome, independent of physical activity levels, suggesting a higher risk of T2DM. We also observed that NZ European women were older, less socioeconomically deprived, and more likely to follow healthier dietary patterns characterized by “core” foods compared to Pacific women who were younger and more likely to live in low-income neighborhoods. This is similar to previous observations where older wealthier women tend to follow healthier diets (Darmon and Drewnowski, 2008; Imamura et al., 2015). However, despite on average being younger, Pacific women in the high-BF% group had higher risk factors for diabetes (i.e., fasting insulin and HOMA-IR scores) than NZ European women in the high-BF% group. Taken together our findings suggest that socioeconomic factors may influence adherence to healthier dietary patterns, and higher intake of discretionary foods is associated with greater metabolic risks, especially for Pacific women. This underscores the importance of addressing socioeconomic disparities in dietary intakes as a public health priority in NZ.
Socioeconomic circumstances shape food choices and subsequently health outcomes. Higher SES is associated with more diverse, nutritious diets and lower obesity and NCD risk (Beck et al., 2018b; Darmon and Drewnowski, 2015; Newby and Tucker, 2004), conversely, lower SES and food insecurity are associated with higher intake of refined energy-dense and nutrient-poor “discretionary” foods and increased risk of excess adiposity (Darmon and Drewnowski, 2015; Health New Zealand Te Whatu Ora, 2023; Herforth et al., 2020; McLaren, 2007). In NZ, low-income neighborhoods tend to be characterized by a higher density of outlets selling fast food and greater exposure to marketing of such products, coupled with reduced availability of healthier retailers selling products like fruit and vegetables (Pearce et al., 2007; Sushil et al., 2017). Moreover, healthier “core” foods tend to cost more than relatively less healthy nutrient poor “discretionary” options (Darmon and Drewnowski, 2015; Herforth et al., 2020). This likely partially explains why fruit and vegetable intake is associated with deprivation level in NZ, for example, those living in areas characterized by higher deprivation are less likely to meet the daily recommendations for fruit and vegetable intake (Health New Zealand Te Whatu Ora, 2023; Ministry of Health, 2022). Therefore, our observations that Pacific women were less likely to adhere to the dietary patterns characterized by core foods are not surprising, and align with similar trends observed by Beck et al. where higher adherence to a “healthy” pattern, derived from the last NZ National Nutrition Survey data (from 2008/2009), was inversely associated with BMI and socioeconomic deprivation (Beck et al., 2018b). Socioeconomic deprivation may explain, at least in part, the observed differences in dietary pattern adherence between Pacific and NZ European women, that is, Pacific women may face greater barriers and less “choice” with regards to eating healthily than NZ European women (Robinson et al., 2021), which is likely contributing to the observed differences in nutrition-related NCD outcomes for these groups of women (Atlantis et al., 2017; Health New Zealand Te Whatu Ora, 2023; Yu et al., 2020).
Younger people's diets often include more cheap, energy-dense “discretionary” foods and less fruit, vegetables, and wholegrains, shaped by factors like: preferences, cost (Hebden et al., 2015), marketing, social influences, life transitions, and a focus on immediate priorities over long-term health outcomes (Munt et al., 2017). Although NZ European women in the low-BF% group were more active, possibly explaining the absence of metabolic syndrome in this group, the overall population was relatively sedentary (Slater et al., 2021). Nonetheless, higher adherence to the Processed pattern was associated with increased risk of metabolic syndrome independent of MVPA, underscoring concerns about the long-term impact of such diets. We observed that younger Pacific women in the high-BF% group had higher fasting insulin and HOMA-IR scores than NZ European women in the high-BF% group, suggesting lower adherence to healthier dietary patterns based on “core” foods as well as an increased risk of developing T2DM. Additionally, the association between greater adherence to the Processed pattern (characterized by “discretionary” foods) and increased adiposity and risk of metabolic syndrome raises public health concerns, because the observed trend for younger Pacific women to consume more of these foods suggests as this population ages, both adiposity and metabolic disease risk may continue to rise.
The PROMISE study aimed to explore whether differences in gut microbiota composition explained differences in metabolic risk observed between Pacific and NZ European women (Kindleysides et al., 2019). However, it appears more likely that differences in gut microbiota profiles are explained by differences in dietary patterns (Renall et al., 2023), which are socially determined. Public health nutrition approaches for Pacific women need to address the challenges of healthy eating when living in low-income neighborhoods and must also support younger adults to have healthier diets. Socioeconomic constraints often worsen food insecurity and limit the dietary choices available to individuals experiencing financial hardship, leading to a reliance on cheap energy dense processed foods (Robinson et al., 2021). Education and individual actions alone are not sufficient to address the numerous barriers to eating healthy young people and those living in high deprivation areas face (Munt et al., 2017; Sushil et al., 2017). Tailored and targeted policy-based interventions that focus on creating supportive environments and reducing barriers to healthy eating are required, such as: improving access to affordable healthy foods, increasing incomes, regulating unhealthy food marketing, and density of fast-food outlets. Such interventions would support both younger people and those living in low-income neighborhoods to have more food agency, and to have healthier diets (Mackay et al., 2022; Munt et al., 2017; Vandevijvere et al., 2019), which in turn, would contribute to improving long term health outcomes.
This cross-sectional study does have some limitations. Self-reported dietary data are prone to misreporting (Gibson, 2005; Willett, 2012); however, extensive methodological approaches were used to reduce this risk (Cade et al., 2017). While our misreporting cut-offs were higher than Willet's 2012 recommendations (Willett, 2012), they were considered appropriate for this population following detailed data reviews (Gemming et al., 2014; Metcalf et al., 2008; Smith et al., 2015), and align with other studies involving ethnic minority groups (Chacko George et al., 2004; Kolonel et al., 2000). The dietary patterns identified are study-specific and not generalizable, however they share characteristics and health associations with patterns reported elsewhere (Pachucki, 2012; Schulze et al., 2006; Tucker et al., 2015). Finally, causality cannot be inferred, as factors besides diet (like healthy lifestyle behaviors) can influence metabolic disease risk.
Strengths include the use of a prospective food record, which reduces misreporting (Park et al., 2018) and better captures culturally specific foods compared to retrospective FFQs (Gibson, 2005; Willett, 2012). The NCI method accounts for individual variation in day-to-day intake and allows for covariates to be incorporated to improve estimates of episodically consumed foods (Dodd et al., 2006; Tooze et al., 2006). Finally, this study's unique focus on healthy Pacific and NZ European women with different metabolic risk profiles and relative levels of socioeconomic deprivation, along with the assessment of numerous body composition and metabolic risk factors, enabled their association with dietary patterns to be explored.
Conclusion
Our study demonstrated that greater adherence to “core” food patterns was associated with lower adiposity and metabolic disease risk, while patterns high in processed “discretionary” foods and low in “core” foods were associated with higher metabolic risk for both Pacific and NZ European women. Socioeconomic deprivation appears to be a key driver of these diet-related risks, highlighting the need to improve access to and intake of healthy foods as a public health priority in NZ. While diet is important, obesity and metabolic disease risk are also shaped by wider social, economic, and environmental conditions that influence people's ability to live healthily. Our findings should be interpreted within this wider context. Nutrition strategies for Pacific women must also address the challenges of maintaining a healthy diet in low-income communities and support younger adults in adopting healthier eating habits. Longitudinal research is needed to assess the long-term impact of these dietary patterns on metabolic outcomes.
Supplemental Material
sj-docx-1-nah-10.1177_02601060261419742 - Supplemental material for Higher adherence to core food-based vegetarian and prudent dietary patterns is associated with lower adiposity and metabolic disease risk in Pacific and New Zealand European women
Supplemental material, sj-docx-1-nah-10.1177_02601060261419742 for Higher adherence to core food-based vegetarian and prudent dietary patterns is associated with lower adiposity and metabolic disease risk in Pacific and New Zealand European women by Nikki Renall, Benedikt Merz, Jeroen Douwes, Marine Corbin, Joanne Slater, Ridvan Firestone, Rozanne Kruger and Lisa Te Morenga in Nutrition and Health
Supplemental Material
sj-docx-2-nah-10.1177_02601060261419742 - Supplemental material for Higher adherence to core food-based vegetarian and prudent dietary patterns is associated with lower adiposity and metabolic disease risk in Pacific and New Zealand European women
Supplemental material, sj-docx-2-nah-10.1177_02601060261419742 for Higher adherence to core food-based vegetarian and prudent dietary patterns is associated with lower adiposity and metabolic disease risk in Pacific and New Zealand European women by Nikki Renall, Benedikt Merz, Jeroen Douwes, Marine Corbin, Joanne Slater, Ridvan Firestone, Rozanne Kruger and Lisa Te Morenga in Nutrition and Health
Footnotes
Acknowledgments
This manuscript is based on research conducted as part of NR's doctoral thesis, titled “New Pathways to Obesity Prevention and Metabolic Health: The Relationship Between Diet and the Gut Microbiome,” submitted to Massey University in 2020 and available at ![]() . The authors would like to thank all the volunteers who participated in the PROMISE study, including staff and students who participated in recruitment and data collection. Niamh Brennan, Sophie Kindleysides, and Moana Manukia, who played key roles in participant recruitment, coordination, and data acquisition of the PROMISE study.
. The authors would like to thank all the volunteers who participated in the PROMISE study, including staff and students who participated in recruitment and data collection. Niamh Brennan, Sophie Kindleysides, and Moana Manukia, who played key roles in participant recruitment, coordination, and data acquisition of the PROMISE study.
Ethical statement
Approval for the PROMISE study was provided by the Southern Health Disability Ethics Committee (16/STH/32) and conducted according to the guidelines of the declaration of Helsinki. The trial was registered at anzctr.org.au (ACTRN12618000432213).
Consent for publication
All authors have read and approved the final manuscript and consent to its publication.
Authors' contributions
Nikki Renall: data curation, formal analysis, and drafted the manuscript. Nikki Renall, Joanne Slater, and Rozanne Kruger: PROMISE data collection and entry. Benedikt Merz, Jeroen Douwes, Marine Corbin, and Lisa Te Morenga: supervised statistical analysis. Benedikt Merz, Lisa Te Morenga, and Rozanne Kruger: supervision of dietary analysis. Nikki Renall, Lisa Te Morenga, and Benedikt Merz: wrote and critically reviewed the manuscript with input from other authors. Rozanne Kruger, Jeroen Douwes, Ridvan Firestone, and Lisa Te Morenga: PROMISE study funding acquisition.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The PROMISE study was funded by the Health Research Council (HRC) of New Zealand (HRC 15/273). NR was supported by a PhD scholarship provided by the Riddet CoRE Institute, Massey University, Palmerston North, New Zealand.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data that support the findings of this study are available on reasonable request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Supplemental material
Supplemental material for this article is available online.
