Abstract
Introduction
Pulmonary hypertension (PH) is a progressive, well-defined mean pulmonary arterial pressure (PAP) of ≥ 25 mm Hg and normal pulmonary arterial wedge pressure of ≤15 mm Hg at rest as measured by right heart catheterization (Kramer et al., 2021). The definition has been modified recently, including patients with mean PAP > 20 mm Hg (Maron, 2023). PH occurs due to increased pulmonary vascular resistance and predisposes to premature death and right heart failure (Howard et al., 2021). Pathogenic mechanisms underlying PH are primarily driven by genetic mutations, such as those in the BMPR2 gene, which disrupt normal vascular cell signalling and function. Epigenetic shifts like DNA methylation further disrupt vascular function and facilitate disease progression. Inflammation and proliferative vascular adaptations also play crucial roles, leading to the hyper-proliferation and resistance to apoptosis observed in PH (Benincasa et al., 2023a, 2023b). Iron metabolism is a vital part of oxygen consumption in the body, and it also has essential functions in cell progression and DNA synthesis. Several outlines of evidence specify that abnormal iron handling often complicates PH. Iron deficiency in patients with PH has been shown to cause functional capacity limitation and poor outcomes, even in the absence of anaemia (Akaslan et al., 2022). Iron deficiency is common in patients with idiopathic and heritable PH in the absence of anaemia and is associated with a worse clinical outcome. High quantities of circulating hepcidin may hinder iron absorption through the mouth. Iron deficiency is frequently linked to the diagnosis of idiopathic PH. In the absence of overt anaemia, iron deficiency is linked to decreased functional ability and survival in patients with PH. The European Cardiac Society guidelines and European Respiratory Society guidelines for managing PH recommend that iron replacement be part of the treatment plan for PH patients (Howard et al., 2021).
Iron replacement's safety in idiopathic and heritable PH is still under question, and it is unclear how it will affect peripheral vascular resistance, cardiac function, and exercise tolerance (Howard et al., 2021). Hence, iron replacement has been shown to improve functional capacity in some subgroups. The benefits and safety of parenteral iron replacement in this patient population are still unclear (Akaslan et al., 2022). The current systematic review on iron replacement in PH comprehensively evaluated the impact on functional capacity, clinical outcomes, and safety in this patient population.
Methods
Protocol registration
This study followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Guidelines for reporting the findings (Page et al., 2021). The study protocol was registered in the International Prospective Register of Systematic Reviews (PROSPERO) with the registration number: CRD42024504460 before conducting the study.
Objectives
The research question for our systematic review is ‘What are the efficacy and clinical outcomes of iron replacement in patients suffering from PH?’ The research question was divided into ‘P’, ‘I’/ ‘E’, ‘C’, and ‘O’ (population, intervention/exposure, comparison, and outcome) format. The defined ‘population’ was patients with PH without age, sex, and ethnicity restriction. 'Intervention’ or ‘Exposure’ was patients replaced or supplemented with iron. The ‘comparator’ was any comparator, if available, including placebo. The ‘outcome’ measure was the efficacy and clinical outcomes.
Eligibility criteria
The following criteria were used to determine if the studies qualified for inclusion. Inclusion criteria: studies that addressed the investigation of iron replacement or supplementation in individuals with PH were considered for this review.
Exclusion criteria: any commentaries, reviews, non-retrievable articles, preclinical studies, newsletters, letters to the editor, abstract-only papers, and conference proceedings were excluded.
Information sources and search strategy
We have reviewed the literature to select appropriate articles published in the electronic bibliographic databases from the establishment until April 2024. We systematically implemented an advanced electronic search in PubMed, Scopus, Web of Science, and Embase to search for eligible studies. The search strategy in the above database was performed using the keywords and medical subject headings (MeSH) terms like ‘Pulmonary hypertension’, ‘Pulmonary arterial hypertension’, ‘PAH’, ‘Iron replacement’, ‘Iron supplements’, ‘Ferrous’, ‘Ferric carboxy maltose’ using ‘AND’ and ‘OR.’ We limited the search to English publications.
Study selection process and data extraction
The studies were screened by title and abstracts followed by full-text articles based on predefined criteria. Two independent reviewers (SJK and PM) performed the study selection, and differences of opinion were resolved by mutual consultation with another reviewer (TB). A well-defined data extraction sheet was employed for the data extraction. Data from the final selected studies included authors’ names, year of publication, study design, sample size, study groups, clinical outcomes, and adverse effects. Additional necessary data were obtained by accessing the trial registry using the unique trial registration ID (Table 1). One reviewer (TB) extracted the data in a standardized extraction sheet, and the other reviewers (RB, PM, and ZS) checked for appropriateness. Any disagreement was resolved by a mutual discussion or consultation with another reviewer (SJK).
Characteristics of included studies.

PH: pulmonary hypertension; RCT: randomized controlled trial.
Akaslan et al. (2022) have two study groups of groups 1 and 4 PH patients who were received with iron replacement.
Howard et al. (2021) is an RCT of two trials comprising European and Chinese trials.
Group 1 PH: refers to pulmonary arterial hypertension, which is caused when the arteries in the lungs become narrowed, thickened, or stiff; Group 4 PH refers to PH due to chronic blood clots in the lungs.
Risk of bias and quality assessment
The Newcastle – Ottawa Scale (NOS) was used to grade the cohort studies (Wells et al., 2024). The Cochrane Risk of Bias Assessment (CROB) tool was used for randomized control (RCT) studies (Higgins et al. 2011). The Methodological Index for Non-randomized Studies (MINORS) tool was used for non-RCT studies (Slim et al., 2003). Two independent reviewers (PM and ZS) performed the quality assessment, and any disagreements between the reviewers were settled through consensus or discussion with another reviewer (TB).
Data synthesis
All the extracted information pertaining to the study was synthesized qualitatively and presented in a narrative manner. The data which were extracted from the included studies are given in tabular form. The data synthesized in this review summarized the efficacy of current evidence for iron replacement in PH. Extracted data were in the form of mean ± standard deviation and median (interquartile range). Due to the lack of availability of maximum and minimum values for the range, we could not convert the values for uniformity and hence reported the values as mentioned. Meta-analysis could not be performed due to insufficient available data.
Results
Study selection
A total of 6926 records have been identified after a systematic search of the above-mentioned databases. A total of 2900 duplicate records have been removed before title and abstract screening. After removing duplicates, 4026 records were screened based on title and abstract. Of these, 4004 records were excluded because they focused on other supplementations or conditions unrelated to the scope of our review. Subsequently, 22 records were sought for full-text retrieval, out of which 20 records were successfully obtained. These 20 articles were subjected to full-text screening based on our review eligibility criteria. Finally, a total of five studies were incorporated into our review (Akaslan et al., 2022; Howard et al., 2021; Kramer et al., 2021; Ruiter et al., 2015; Viethen et al., 2014). Figure 1 depicts the PRISMA flow diagram.
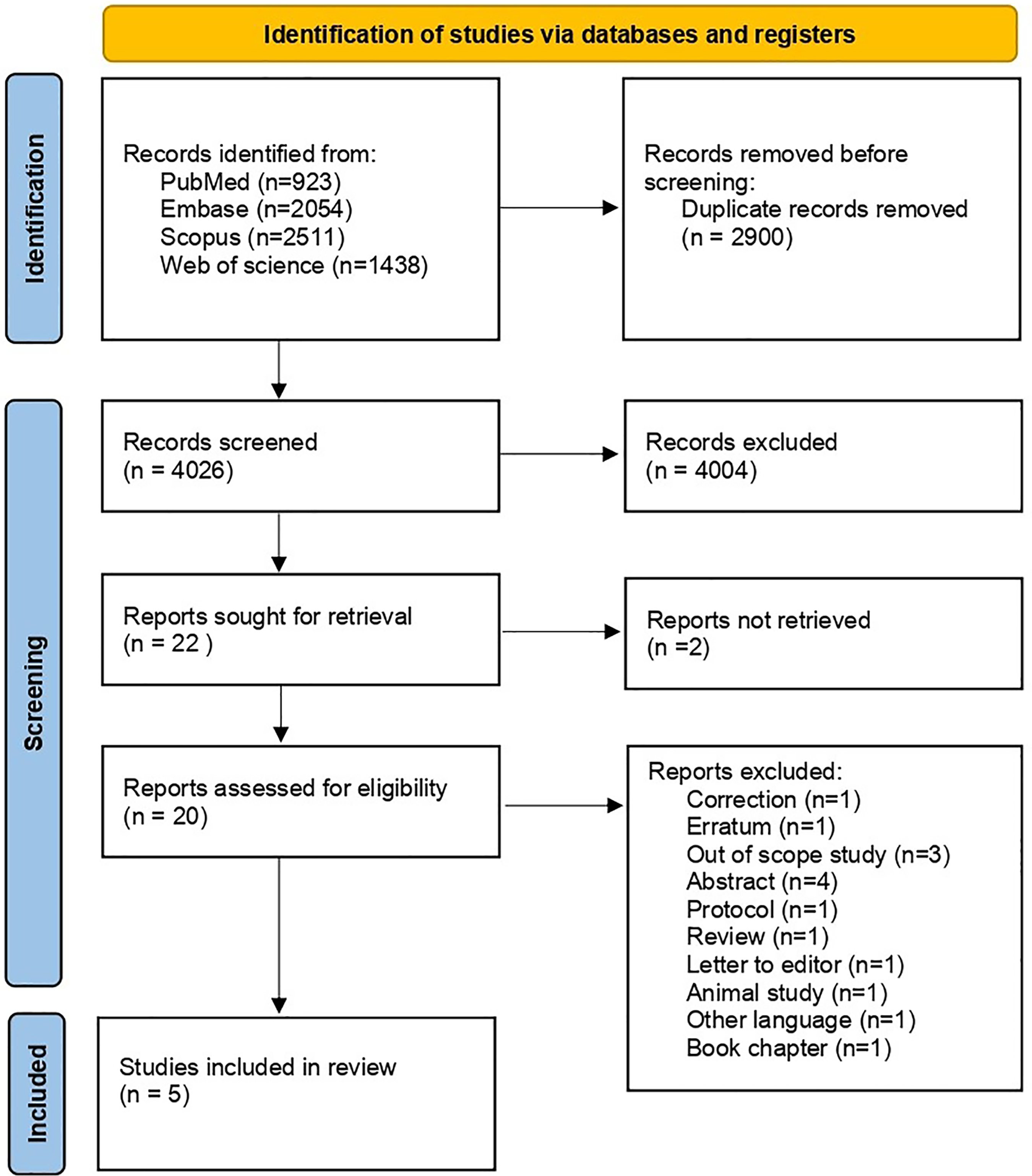
Preferred reporting items for systematic reviews and meta-analyses (PRISMA) flow diagram of the screening and selection process.
Study characteristics
Out of all final selected five studies, one was RCT (Howard et al., 2021), two were non-RCT (Ruiter et al., 2015; Viethen et al., 2014), and two were observational studies (Akaslan et al., 2022; Kramer et al., 2021). The characteristics of the included studies have been mentioned in Table 1. In Akaslan et al. (2022), the two study groups were diagnosed as groups 1 and 4 PH patients (based on diagnostic criteria), respectively, (Akaslan et al., 2022). Howard et al. (2021) is a multicentre trial comprised of European and Chinese trials with two different types of iron replacement (Howard et al., 2021). Ruiter et al. (2015) is a single-group assignment study without comparator groups (Ruiter et al., 2015).
In all the studies, the type of population was pulmonary hypertension with iron deficiency (Akaslan et al., 2022; Howard et al., 2021; Kramer et al., 2021; Ruiter et al., 2015; Viethen et al., 2014). The iron replacement was performed as an infusion in all included studies (Akaslan et al., 2022; Howard et al., 2021; Kramer et al., 2021; Ruiter et al., 2015; Viethen et al., 2014).
Ferric carboxymaltose has been used as iron replacement therapy in all included studies except for the Chinese trial of Howard et al. 2021 where iron dextran was used. Similar dosage followed in five studies using Ferric carboxymaltose (Akaslan et al., 2022; Howard et al., 2021; Kramer et al., 2021; Ruiter et al., 2015; Viethen et al., 2014) (Table 1).
Overall efficacy
We have extracted the efficacy data on (1) functional status: six-minute walk distance (6MWD), cardiopulmonary exercise test (peak oxygen intake and anaerobic threshold), (2) disease severity marker: N-terminal prohormone of brain natriuretic peptide (NT-proBNP), (3) iron indices (e.g. haemoglobin (Hb), mean corpuscular volume (MCV), serum Iron, serum ferritin, transferrin saturation, etc.), and (4) quality of life (QoL).
Functional status and disease severity marker
Except in the European trial of Howard et al. (2021), in all remaining included studies, there was a substantial increase in the 6MWD test after the iron replacement (Howard et al., 2021) (Table 2).
Functional and disease severity markers for included studies.
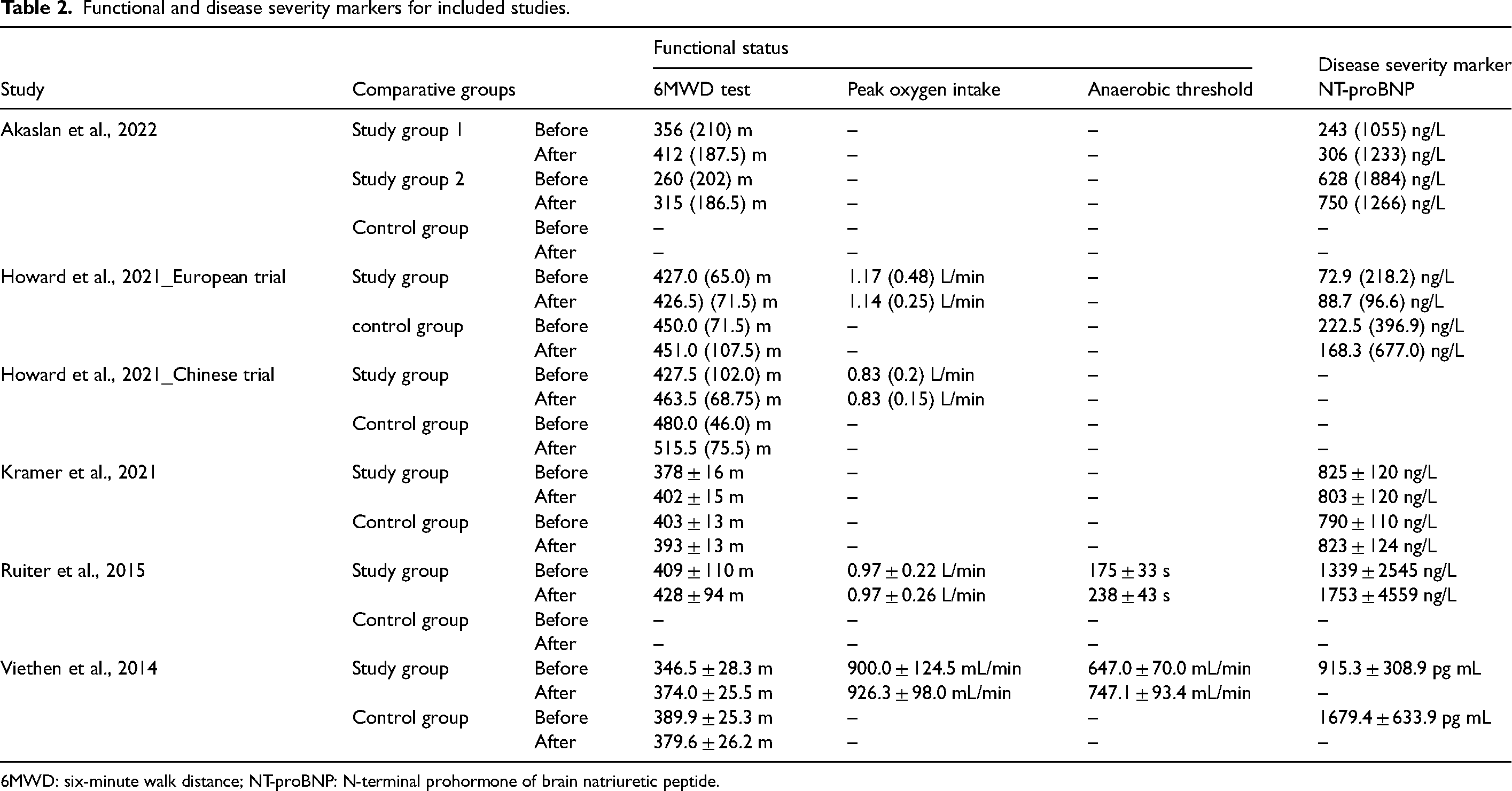
6MWD: six-minute walk distance; NT-proBNP: N-terminal prohormone of brain natriuretic peptide.
For the cardiopulmonary exercise test, we have extracted data on peak oxygen intake and anaerobic threshold (Table 2). Of all included studies, Howard et al. (2021), Viethen et al. (2014), and Ruiter et al. (2015) reported the peak oxygen intake (Howard et al., 2021; Ruiter et al., 2015; Viethen et al., 2014). Viethen et al. (2014) demonstrated a significant increase in the anaerobic threshold, with values of 647.0 ± 70.0 mL/min before and 747.1 ± 93.4 mL/min after iron therapy (Viethen et al., 2014). Similarly, Ruiter et al. 2015 observed a similar trend, reporting a significant increase in the anaerobic threshold from 175 ± 33 s before to 238 ± 43 s after iron therapy (Ruiter et al., 2015).
All included studies reported the NT-proBNP disease severity marker for PH (Akaslan et al., 2022; Howard et al., 2021; Kramer et al., 2021; Ruiter et al., 2015; Viethen et al., 2014) (Table 2). In Viethen et al. (2014), no significant differences were obtained in NT-proBNP after iron replacement (Viethen et al., 2014).
Iron indices
In all the included studies, serum iron, transferrin saturation, and serum ferritin levels increased after iron replacement (Akaslan et al., 2022; Howard et al., 2021; Kramer et al., 2021; Ruiter et al., 2015; Viethen et al., 2014) (Table 3). A similar trend was observed for Hb levels in all included studies (Akaslan et al., 2022; Howard et al., 2021; Kramer et al., 2021; Ruiter et al., 2015; Viethen et al., 2014). Akaslan et al. (2022), Kramer et al. (2021), Ruiter et al. (2015), and Viethen et al. (2014) reported that MCV increased after iron replacement (Akaslan et al., 2022; Kramer et al., 2021; Ruiter et al., 2015; Viethen et al., 2014).
Iron indices for included studies.
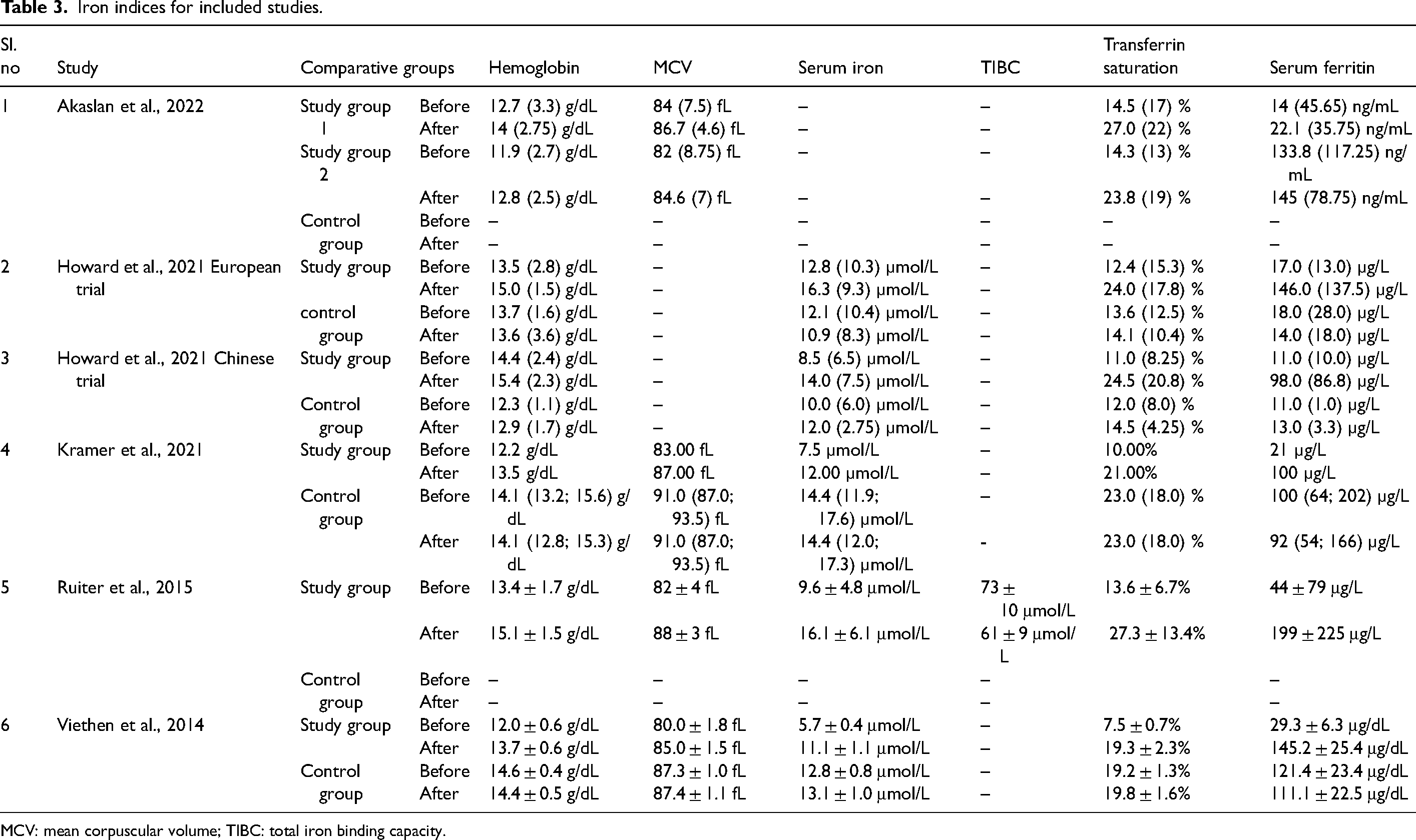
MCV: mean corpuscular volume; TIBC: total iron binding capacity.
Ruiter et al. (2015) also reported a change in total iron-binding capacity after iron replacement, which was 73 ± 10 μmol/L before replacement and 61 ± 9 μmol/L after replacement. This change was associated with increased serum iron levels (Ruiter et al., 2015).
Quality of life
Four studies have reported using the health-related QoL (HRQoL) questionnaire to assess the change in QoL in patients with PH after iron replacement. However, these four studies have used different types of QoL tools (Howard et al., 2021; Kramer et al., 2021; Ruiter et al., 2015; Viethen et al., 2014) (Table 4).
Quality of life for included studies.
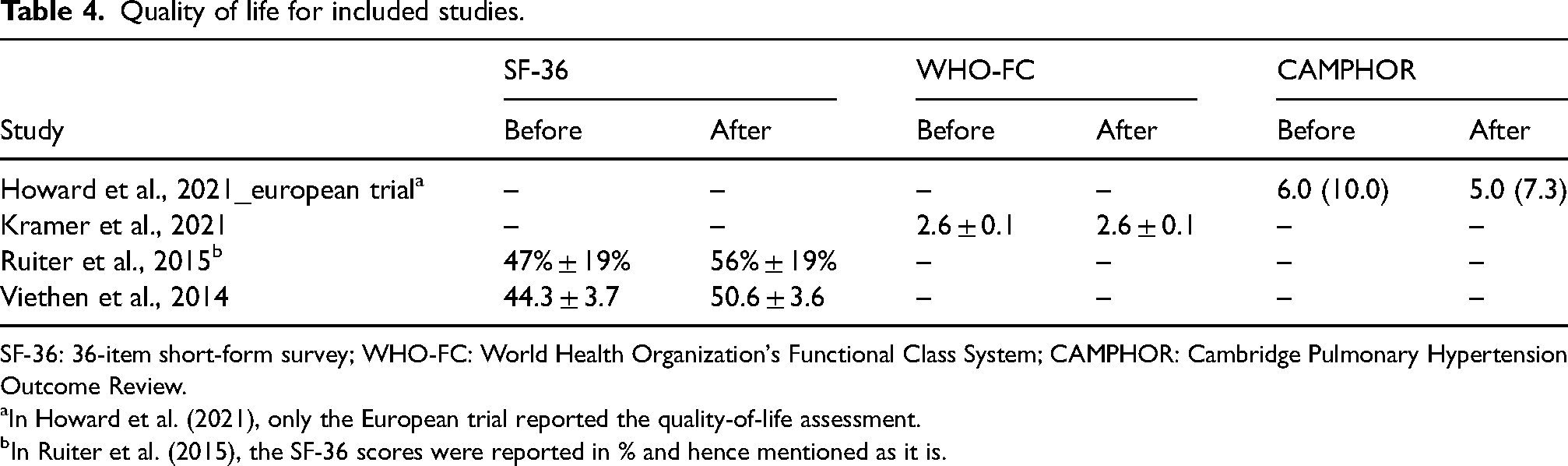
SF-36: 36-item short-form survey; WHO-FC: World Health Organization's Functional Class System; CAMPHOR: Cambridge Pulmonary Hypertension Outcome Review.
In Howard et al. (2021), only the European trial reported the quality-of-life assessment.
In Ruiter et al. (2015), the SF-36 scores were reported in % and hence mentioned as it is.
Howard et al. (2021) employed the Cambridge Pulmonary Hypertension Outcome Review (CAMPHOR), specifically utilized in their European trial. However, they did not find any significant difference concerning iron replacement. The CAMPHOR scores were as follows: before iron replacement: 6.0 (10.0) and after replacement: 5.0 (7.3). These values indicate no substantial change in QoL following iron replacement (Howard et al., 2021).
Kramer et al. (2021) used the World Health Organization's Functional Class System (WHO-FC) to assess patients’ QoL with PH. They found no change in the QoL, with scores of 2.6 ± 0.1 reported before and after iron replacement (Kramer et al., 2021).
Ruiter et al. (2015) and Viethen et al. (2014) used the 36-item short-form survey (SF-36) to measure the difference obtained in QoL with respect to iron replacement (Ruiter et al., 2015; Viethen et al., 2014). These two studies reported an increase in QoL score after the iron replacement. Ruiter et al. (2015) reported the overall SF-36 score after the iron replacement was 56 ± 19%, compared to a baseline score of 47 ± 19% (Ruiter et al., 2015). Similarly, Viethen et al. (2014) also reported the overall SF-36 score after iron replacement to be 50.6 ± 3.6, compared to a baseline score of 44.3 ± 3.7 (Viethen et al., 2014).
Adverse effects
In all included studies, the iron replacement was tolerated well with no serious adverse events (Akaslan et al., 2022; Howard et al., 2021; Kramer et al., 2021; Ruiter et al., 2015; Viethen et al., 2014). However, Kramer et al. (2021) and Viethen et al. (2014) reported minor side effects in one or two patients following iron infusion. These side effects included transient flu-like symptoms and minor skin discoloration at the infusion site (Kramer et al., 2021; Viethen et al., 2014).
Risk of bias within the studies
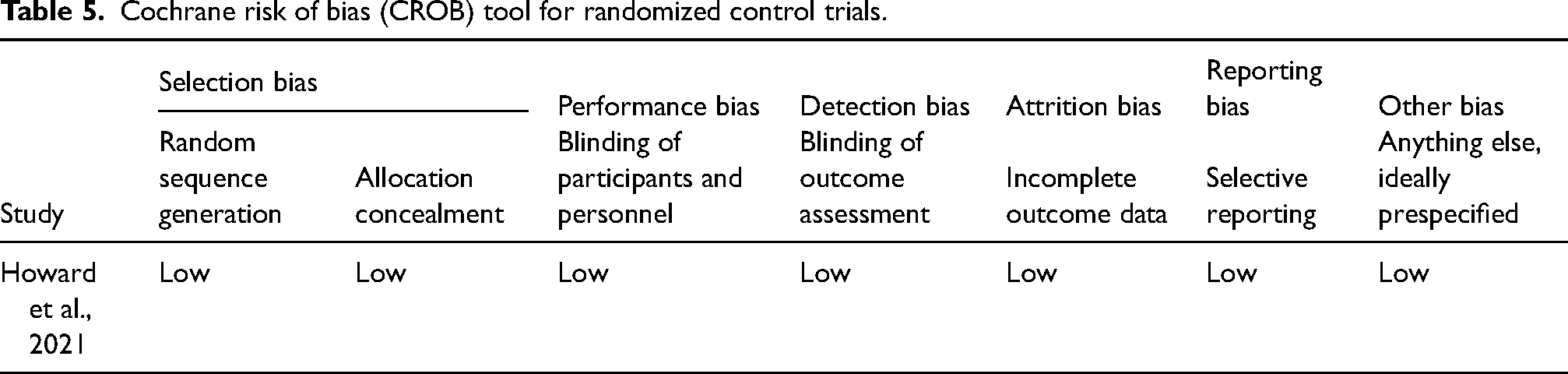
The methodological quality assessment of all five studies (Akaslan et al., 2022; Howard et al., 2021; Kramer et al., 2021; Ruiter et al., 2015; Viethen et al., 2014) has been mentioned in Tables 5 to 7. We have conducted the quality assessment for CROB for Howard et al. (2021) focused on the following domains: random sequence generation, allocation concealment, incomplete outcome data, selective reporting, and other biases (Howard et al., 2021). The risk of bias in the mentioned domain in all included studies was categorized according to the following: high risk of bias, low risk of bias, unclear risk of bias, and not applicable.
Cochrane risk of bias (CROB) tool for randomized control trials.
Methodological items for non-randomized studies (MINORS).

The items are scored 0 (not reported), 1 (reported but inadequate), or 2 (reported and adequate). The global ideal score being 16 for non-comparative studies and 24 for comparative studies.
12 items were applied as this is a comparative study.
Eight items were applied as this is a non-comparative study.
Newcastle Ottawa quality assessment scale (NOS) for cohort studies.

One point for each numbered item within the selection and exposure categories. A maximum of two points are given for comparability.
The methodological assessment for MINORS has been mentioned in Table 6. In this tool, items 9 to 12 are the additional items used by Viethen et al. (2014), which is a comparative study (Viethen et al., 2014). For Ruiter et al. (2015), eight items were used as this is a non-comparative single-arm study (Ruiter et al., 2015).
The quality assessment for NOS in Akaslan et al. (2022) and Kramer et al. (2021)studies focused on the following criteria: selection, comparability, and exposure (Akaslan et al., 2022; Kramer et al., 2021).
Discussion
This systematic review is a first-of-a-kind review where we comprehensively evaluated the efficacy and safety of iron replacement in patients with PH. Our findings revealed promising results about iron replacement's efficacy in improving functional status in patients with PH. Specifically, we observed significant improvement in the 6MWD test in the included studies (Akaslan et al., 2022; Howard et al., 2021; Kramer et al., 2021; Ruiter et al., 2015; Viethen et al., 2014), except for the European trial by Howard et al. 2021 (Howard et al., 2021). Furthermore, cardiopulmonary exercise tests showed favourable outcomes, with an increase in peak oxygen intake and anaerobic threshold (Howard et al., 2021; Ruiter et al., 2015; Viethen et al., 2014). This suggests that iron replacement may enhance functional performance regarding exercise tolerance and cardiopulmonary function in patients with PH, a crucial aspect of their management.
According to Berghaus et al. (2017), serum NT-proBNP levels were found to be significantly correlated to disease severity in treatment-naive patients with precapillary PH (Berghaus et al., 2017). In high-risk groups like those with connective tissue diseases, portal hypertension, congenital systemic-to-pulmonary shunts, or a family history of idiopathic pulmonary arterial hypertension, NT-proBNP levels are a useful biomarker for detecting PH. In patients with PH, NT-proBNP levels positively correlate with measures of disease severity such as mean PAP, peripheral vascular resistance, and right ventricular end-diastolic pressure. Increased NT-proBNP concentrations are suggestive of more advanced PH and may influence prognostication and treatment choices (Casserly and Klinger, 2009). NT-proBNP levels are inversely correlated with functional capacity, as evidenced by measures such as the 6MWD (Leuchte et al., 2004). This could explain why we observed increased NT-proBNP levels after iron replacement (Tables 2 and 3).
According to a study by Ruiter et al. (2011), iron deficiency accounts for about 43% of idiopathic PH and is linked to reduced exercise capacity (Ruiter et al., 2011). Iron is an essential cofactor in the body's oxygen transport process. Iron also affects the respiratory chain and other mitochondrial oxidative enzymes. As a result, reduced oxygen delivery to the heart and skeletal muscles occurs in iron deficiency cases and impairs exercise capacity (Abbaspour et al., 2014). This emphasizes how crucial it is to treat iron deficiency in PH to maximize oxygen delivery and enhance functional ability.
Intriguingly, despite variations in study design and patient population, our findings consistently showed the effects of iron replacement on iron indices (Table 3). These observations highlight the importance of addressing iron deficiency in PH patients, as it may burden disease progression and symptoms.
We also focused on assessing the impact of iron replacement on QoL in PH. Studies utilizing the SF-36 reported increased QoL scores, indicating enhanced physical and mental well-being in PH patients receiving iron replacement (Ruiter et al., 2015; Viethen et al., 2014). However, it is notable that the WHO-FC and CAMPHOR did not establish any significant changes in QoL scores by Kramer et al. (2021) and Howard et al. (2021), respectively (Howard et al., 2021; Kramer et al., 2021). This emphasizes the need to investigate further patients’ subjective experiences of receiving iron replacement.
Our review findings summarized that iron supplements generally exhibit good tolerability with no serious adverse effects across the studies, but minor side effects like transient flu-like symptoms and temporary skin discoloration at the infusion site, as reported by Kramer et al. (2021) and Viethen et al. (2014) (Kramer et al., 2021; Viethen et al., 2014). These side effects are typically manageable and not severe. Despite this, the overall safety profile of iron supplements remains promising, enabling them to continue to be used in clinical practice.
We recognize the limitation in our review. Due to heterogenicity in the study design, we could not be able to perform the meta-analysis. There were only five studies comprising 206 PH patients who had undergone iron replacement (Akaslan et al., 2022; Howard et al., 2021; Kramer et al., 2021; Ruiter et al., 2015; Viethen et al., 2014). Further controlled trials are needed to enable better treatment group comparisons. Exploring long-term impacts on comorbidities, mortality, and disease progression would provide valuable insights for managing pulmonary hypertension.
Conclusion
Our systematic review highlights the potential efficacy of iron replacement in PH patients. Regardless of the variation in study design, positive effects were observed on multiple outcome measures like the 6MWD test, cardiopulmonary exercise test parameter, and iron indices. Moreover, carrying out more controlled trials in this field would improve the validity of the results and enable better comparisons between treatment groups. Future studies may prioritize the effects of iron replacement on patients’ outcomes, with a particular emphasis on HRQoL. Using standardized and validated QoL assessment instruments in well-designed controlled trials would offer important new insights into patients’ subjective experiences taking iron supplements. Additional research on the long-term impacts of iron replacement on patient outcomes, such as comorbidities, hospitalization rates, mortality, and disease progression, would provide valuable insights into managing PH.
Supplemental Material
sj-docx-1-nah-10.1177_02601060241303814 - Supplemental material for Efficacy of iron replacement in pulmonary hypertension: A systematic review
Supplemental material, sj-docx-1-nah-10.1177_02601060241303814 for Efficacy of iron replacement in pulmonary hypertension: A systematic review by Tejaswini Baral, Pravachana Malakapogu, Zuha Shyma, Shilia Jacob Kurian, Ruby Benson, Mohan K. Manu, Debasis Bagchi and Sonal Sekhar Miraj in Nutrition and Health
Footnotes
Acknowledgements
The authors gratefully acknowledge the help and facilities provided by the Manipal Academy of Higher Education, Manipal, India.
Authors’ contributions
SSM and TB conceptualized the work. SJK, PM, and TB performed a systematic literature search. TB extracted the data which PM, ZS, and RB validated. The risk of bias assessment was performed by ZS and PM which was validated by TB. Manuscript draft written by TB and PM. All the authors participated in the review of the manuscript. All the authors approved the final draft of the article.
Availability of data and materials
Not applicable.
Consent for publication
Not applicable.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
