Abstract
Keywords
Introduction
While globally, childhood undernutrition rates are falling, they continue to be prevalent in low and middle-income countries (Black et al., 2008, 2013). The 2015–2016 Demographic and Health Survey in Myanmar found undernutrition continues to be a public health problem with 29% of children under 5 years of age being stunted (Hong, 2020; Kang and Kim, 2019; Ministry of Health and Sports Nay Pyi Taw Myanmar, 2017); down from 35.1% in 2009 (Ministry of National Planning and Economic Development and Ministry of Health, 2011). Childhood undernutrition has major consequences for children's growth and development that can impact later adult health (Blankenship et al., 2020; Mericq et al., 2017; Mutunga et al., 2020).
According to the Food and Agriculture Organization of the United Nations (FAO), a growing number of developing countries shoulder a double burden of malnutrition, caused not only by the persistence of undernutrition, particularly among children, but combined with a rapid rise in excess body weight, obesity and diet-related chronic diseases (Valencia et al., 2012). Despite accurate and precise measurements of body composition providing valuable information on nutritional status (Jackson et al., 2013; Nielsen et al., 2007), body composition data for children in Myanmar are lacking. Lwin and colleagues estimated lean body mass and percentage body fat (%BF) in 12–14-year-old children from skin-fold thicknesses according to the Durnin and Rahaman formula which is of questionable applicability in this population (Durnin and Rahaman, 1967; Lwin et al., 2006).
It is important to have a reliable and accurate estimate of body composition when studying the various health correlates of disease (Wootton et al., 2014). Several methods are available, each with advantages and limitations (Pietrobelli et al., 2003). These included the underwater weighing technique, air displacement plethysmography, dual-energy X-ray absorptiometry, deuterium dilution technique and bioelectrical impedance analysis (BIA). Many of these methods are expensive and technologically complex and not available in low-resource countries. The measurement of body water from deuterium dilution is used to predict the fat-free mass (FFM), based on the principle that FFM contains a relatively constant proportion of water (Schoeller, 2005). The method while safe to use in children is relatively expensive and requires laboratory analysis of deuterium, therefore, is more appropriate for research rather than daily clinical application. On the other hand, BIA is a simple, rapid and inexpensive method for the assessment of total body water and hence FFM and body fat mass (FM). The BIA equipment is easily carried to the community for population-based studies (Sen et al., 2010). The International Atomic Energy Agency (IAEA) has had a long-standing program to establish the deuterium dilution technique in low-resource countries to support research in body composition to aid public health and nutrition programs (International Atomic Energy Agency, 2010) while fostering the development of more generally practical techniques such as BIA (Shepherd et al., 2016).
The BIA method is based on the principle that body water (containing electrolytes) unlike fat conducts electricity. The opposition to the flow of an electric current, impedance (Z) is quantitatively related to the total body water volume (TBW). Predictive equations are required that relate measured impedance to TBW and FFM. Such equations are empirically derived and tend to be population-specific (Ward et al., 2006). Prediction equations are commonly produced through validation studies of BIA against reference measures of TBW from deuterium dilution (Kourkoumelis et al., 2021; Valencia et al., 2012).
The purpose of this study was to measure the body composition (FFM and FM) of children aged 4–8 years by deuterium dilution and to develop a BIA prediction equation for the determination of TBW in children, aged 4 to 8 years, in Myanmar.
Materials and methods
Setting and participants
A total of 102 healthy children (57 boys and 45 girls), with ages ranging between 4 and 8 years, were recruited from six villages in different parts of the Htan ta Pin Township in the Yangon region of Myanmar. The township is located about 50 km northwest of the center of Yangon City, Myanmar. It is predominantly rural with rice-based agriculture being the main livelihood for agricultural laborers. There is no significant in or out-migration. The majority of people live on paddy farms and are farm laborers. Although the population relies primarily on farming for income, only 38% of the population is land-owners. There is limited home gardening because the general topography is low-lying swampy paddy land with little or no elevation and floods regularly. Purchased meat (chicken), fruits, and vegetables are primarily from market vendors.
The study villages, namely Kyar Hone, Kyein Pike and Yaeso, Thanpayarkhon and Lamutangyi were purposely selected based on the adequacy of the study children and accessibility from a total of 230 villages comprising the township. From school lists of four to eight-year-old children (25 children in each five age group), a total of 102 children were opportunistically recruited into the study, with both sexes selected randomly (57 boys and 45 girls). There were no stratifications on sex and ethnicity for subject recruitment.
A detailed history for each child was obtained by interview with parents and child, questions including name, age, education, birth history, family history, drug history and past medical and surgical history. The age of the children was confirmed from birth records. Children with parentally reported congenital heart disease, delayed development and severe illness were excluded from the study.
The study was approved by the Ethics Committee of the Department of Medical Research (31 May 2016). The study was explained to parents and children and written informed parental consent was obtained.
Study design
The study was cross-sectional with opportunistic participant recruitment and was conducted between late 2016 and 2017. Measurements were obtained at school by trained staff supervised by authors, AAM and TT. In order to develop a prediction equation relevant for use in children under normal free-living conditions, no requirements for fasting or limiting exercise were imposed although the TBW assessment protocol required no food or drink for 30 min prior to assessment (described in detail below).
Anthropometry
Body weight was measured with a digital electronic scale (Model874, seca gmbh & Co., Ltd, Germany) calibrated with standardized weight. Participants were weighed barefoot wearing a minimum of clothing. Weight was recorded to the nearest 0.1 kg in duplicate and the mean values were used.
Standing height, with the participant standing in the Frankfort plane, was measured using a Harpenden stadiometer (Holtain Ltd, UK) to the nearest 0.1 cm. Measurements were made in duplicate and the mean value was used.
Body mass index (BMI) was calculated as body weight (kg) divided by height squared (m2). Overweight and obesity were classified according to the World Health Organization (WHO) BMI cutoffs for children (De Onis et al., 2007).
Bioelectrical impedance analysis (BIA)
Whole body, wrist-to-ankle, impedance was measured using a tetrapolar single frequency (50 kHz) impedance device (BIM 4, ImpediMed Ltd, Brisbane) following recommended standardized procedures (Brantlov et al., 2017a, 2017b). Impedance (Z, ohm), resistance (R, ohm), reactance (Xc, ohm), and phase angle (θ, °) were measured. During measurement, the child was lying relaxed in a supine position on a bed for 5 min prior to measurement. The current drive electrodes were fixed to the dorsal surface of the third metacarpal of the hand and the dorsal surface of the third metatarsal of the foot. The voltage-sensing electrodes were placed on the dorsum of the wrists and ankles with the mid-line of the electrode midway between the styloid process at the wrists and malleoli at the ankle. The separation between current and sense electrodes was 5 cm. Electrode sites were cleaned with alcohol wipes prior to attachment of electrodes (Ag/AgCl EKG-style gel electrode, ImpediMed Ltd., Brisbane). All measurements were obtained on the right side of the body. Measurements were obtained at least in duplicate with the mean values used for analysis. The mean difference between duplicate measurements was 0.25% ranging from 0.03% to 0.4%. The resistance index (RI, H2/R) was calculated as height squared (H2, cm) divided by resistance (R, ohm).
Total body water
TBW was determined from enrichment of deuterium in saliva samples 3–4 h following an oral dose of deuterium oxide (D2O) according to the IAEA protocol (International Atomic Energy Agency, 2010). The participant was instructed not to eat or drink anything for at least 30 min before the procedure. Briefly, each child received an oral dose (0.5 g/kg body weight) of 99.8 atom% D2O (Sercon Ltd, UK). Doses were weighed to the nearest 0.01 g. Saliva samples (2 mL) were obtained pre-dose and two further samples at 3 and 4 h post-dose. It was often necessary to stimulate salivation to collect a sufficient sample. Participants were given a dry cotton wool ball and to move it around their mouth for 2 min or until it was sodden. The plunger was removed from a 20 mL plastic syringe. Participants were asked to move the cotton wool to the front of their mouth and transfer it into the syringe barrel. The plunger was replaced in the syringe barrel, and saliva was squeezed out of the syringe into the appropriately labelled sample tubes. All samples were stored at −20 °C to await analysis. Sample analyses were undertaken locally by author AAM following IAEA training and quality control assessment in the technique.
Enrichment of deuterium was quantitatively determined by Fourier transform infrared spectrometer (FTIR IR Affinity 1, Shimadzu Corporation, Kyoto, Japan) (International Atomic Energy Agency, 2010). TBW was calculated from the deuterium in the post-dose saliva samples according to the standard operating procedure described by the IAEA (International Atomic Energy Agency, 2013). Calculation of TBW assumed that the dilution space of 2H is 4.1% higher than TBW due to the exchange of H with non-aqueous H in the body. FFM was calculated from TBW assuming age- and sex-specific hydration values of Fomon et al. (Fomon et al., 1982).
Data analysis
Summary statistics are reported as mean and standard deviation (SD) to describe TBW, FFM, FM, and percentage FM (%FM) for all participants and each sub-group. Anthropometric Z scores were calculated using the WHO reference standards for 0–19 years old (De Onis et al., 2007) using the online calculator (https://apps.cpeg-gcep.net/quickZ_WHO/, last accessed 17/05/2024). Prediction equations for TBW and FFM were developed using stepwise multiple linear regression in the prediction group. TBW and FFM derived from deuterium dilution were used as the dependent variables. Potential predictor variables included age, sex (males coded 1 and females coded 0), height weight, and RI. All variables were checked for normality and the presence of outliers. Multi-collinearity between variables was assessed using regression diagnostic of variance inflation factor (VIF). A VIF < 10 was deemed acceptable. The goodness of fit of the regressions were assessed using Malow's Cp, the Akaike information criterion (AIC) and the predicted residual error sum of squares (PRESS) statistic.
In the absence of an independent population in which to assess prediction validity and predictive power, two approaches were used (Guo et al., 1996; Tronstad and Pripp, 2014). Firstly, single-fold leave-one-out cross-validation (LOOCV) was used to determine the overall prediction error. Secondly, a 50:50 split-sample approach was used. The total study population was randomly allocated to either a prediction-generating group (prediction) or a cross-validation group (validation) maintaining sex proportionality in each group to achieve an approximately 50:50 split. The predictive performance of generated prediction equations in the validation group was assessed using Lin's concordance correlation (Lin, 1989) and limits of agreement analysis (Bland and Altman, 1986). In addition, the performance of the final prediction equation was compared to that of similar population-appropriate published equations (Table 1) using correlation, limits of agreement, median absolute percentage error (MAPE) (Seoane et al., 2015) and equivalence analysis for paired data (Lakens, 2017).
Published prediction equations for body composition in children aged 5–18 years.
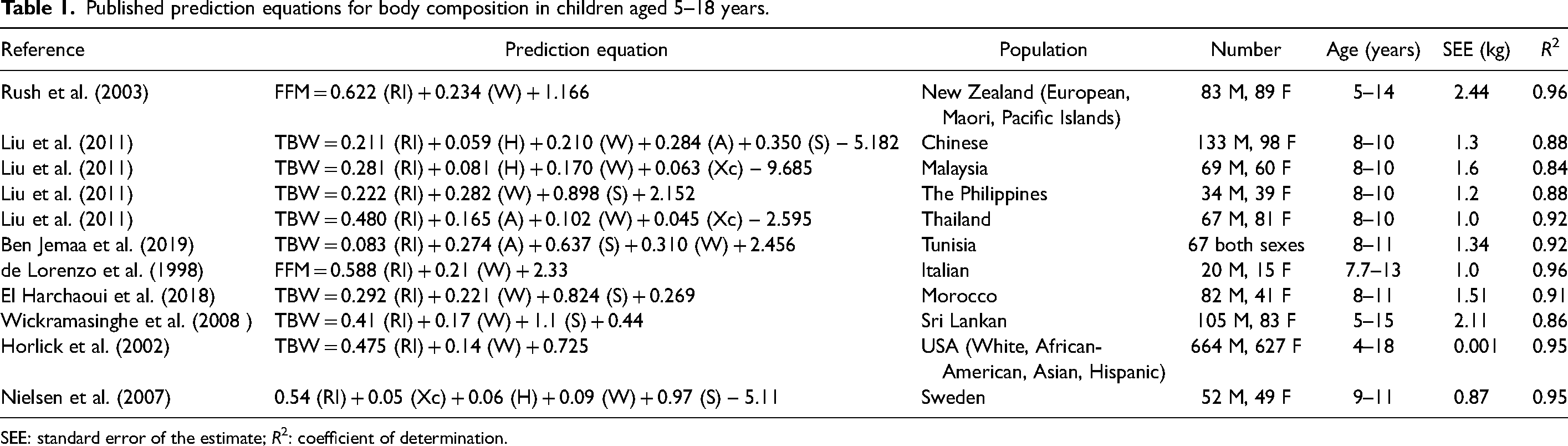
SEE: standard error of the estimate; R2: coefficient of determination.
Statistical analyses were performed using Medcalc version 22.007 (MedCalc Software Ltd, Ostend, Belgium; https://www.medcalc.org; 2022). The LOOCV procedure was performed using Solverstat 2019 R0 (Comuzzi et al., 2003); software available at https://solverstat.wordpress.com/, last accessed 17/05/2024). Statistical decision for significance was made at 0.05 level of type I error.
Results
Participant characteristics: age; body size; body composition and BIA variables of the subgroups, and sex and group differences are shown in Table 2. There were no differences in age or anthropometric variables between boys and girls although all children exhibited weight-forage and height-for-age Z-scores below WHO standards. There were no significant differences between the validation and cross-validation groups among either the boy or girl sub-sets. Impedance was significantly higher in girls than boys (P = 0.0008). Similarly, resistance was significantly higher in girls than in boys (P = 0.0006) yielding a significantly lower resistance index (P = 0.008).
General characteristics of the study sample, Myanmar children aged 4–8 years.
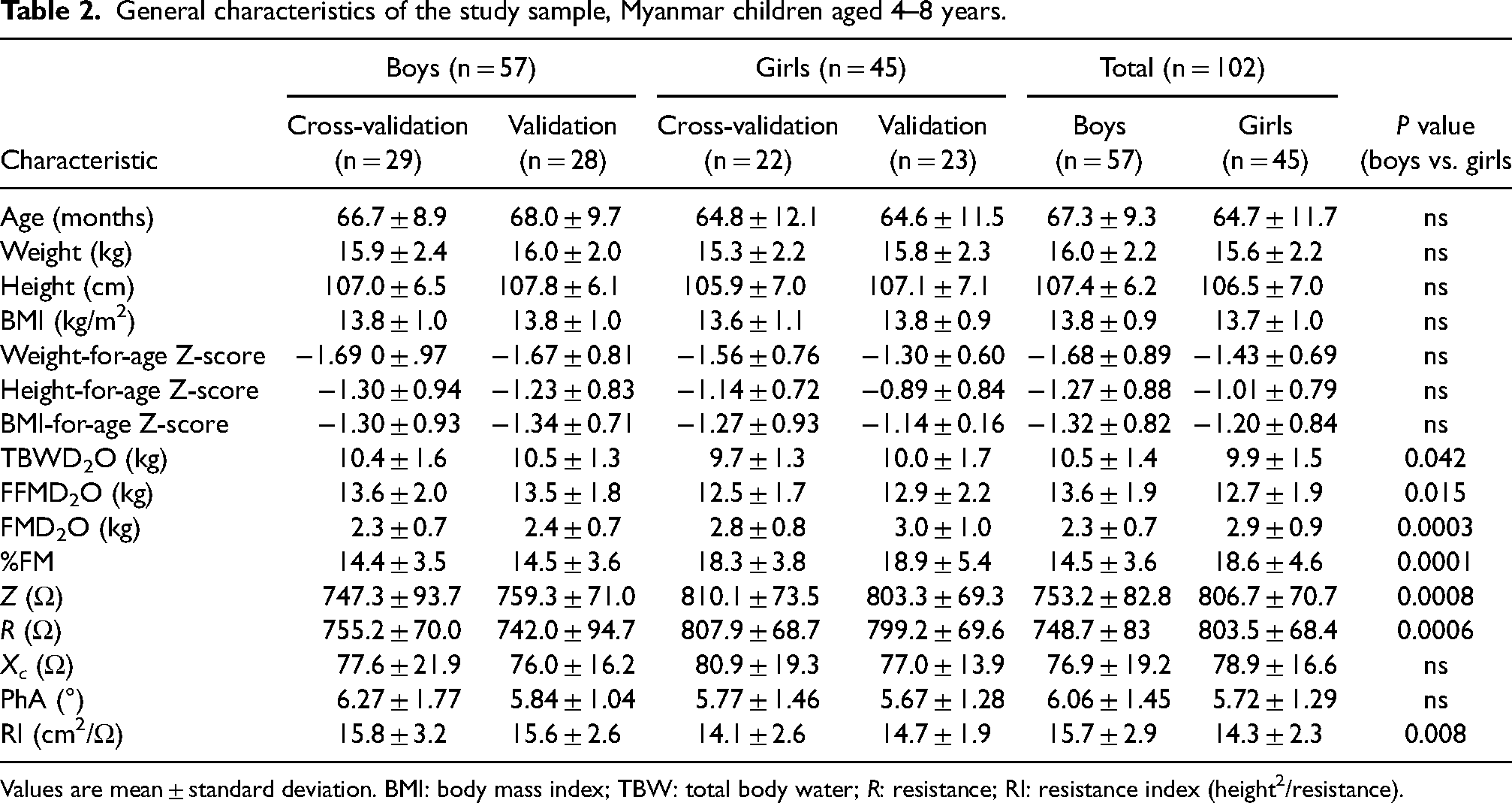
Values are mean ± standard deviation. BMI: body mass index; TBW: total body water; R: resistance; RI: resistance index (height2/resistance).
Boys had significantly higher TBW (from deuterium dilution) (P = 0.042) and FFM (P = 0.015) but lower FM (P = 0.0003) compared to the girls. Percentage FM was significantly higher (18.6%, P < 0.0001) in girls compared to boys (14.5%).
Different tested predictive models for TBW are presented in Table 3. All VIF values were < 10 indicating the absence of multi-collinearity. Age, sex, height and reactance were not found to be significant predictive variables in any of the models in which they were included; TBW being significantly predicted by weight and RI alone. Coefficients of determination were high being greater than 0.84 for all models except for model 1 using RI as the sole predictor. On the basis of the predictive power criteria (RMSE, C(p), PRESS, and AIC) model 3, using RI and weight as predictors, exhibited the best predictive power.
Predictive models for total body water in Myanmar children aged 4–8 years.
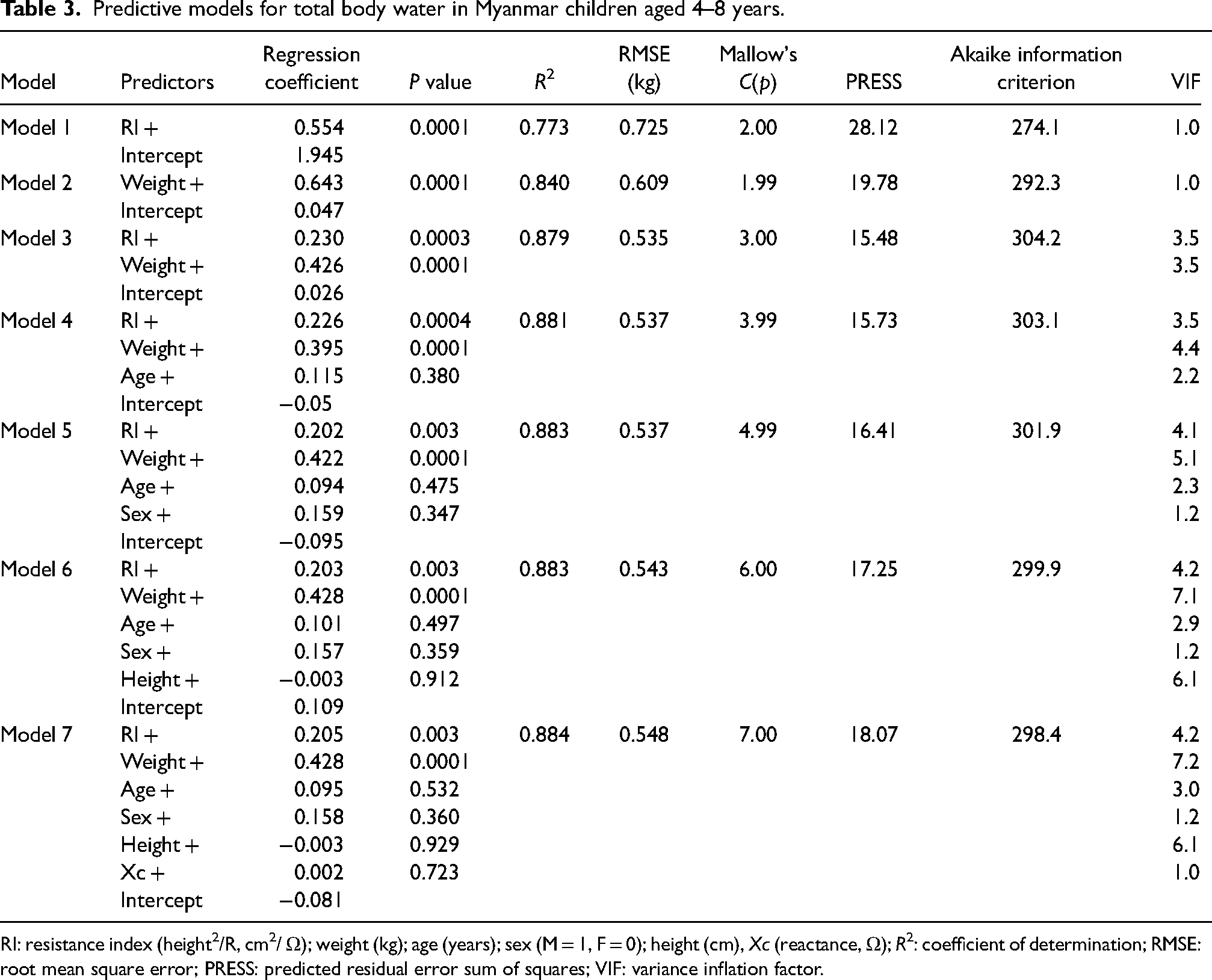
RI: resistance index (height2/R, cm2/ Ω); weight (kg); age (years); sex (M = 1, F = 0); height (cm), Xc (reactance, Ω); R2: coefficient of determination; RMSE: root mean square error; PRESS: predicted residual error sum of squares; VIF: variance inflation factor.
The outcome of the validation study and comparison with published prediction equations are presented in Table 4. Model 3 prediction performed well in predicting TBW in split-group cross-validation with only small group bias (0.02 kg, 0.2%) with 1.96SD limits of agreement of ± 1.0 kg (9.8%). The prediction equation of Horlick et al. (Horlick et al., 2002) performed almost as well closely followed by the equations of Jemaa et al. and Liu et al. in a Filipino population (Ben Jemaa et al., 2019; Liu et al., 2011).
Comparison of predictive equations for total body water in Myanmar children aged 4–8 years.
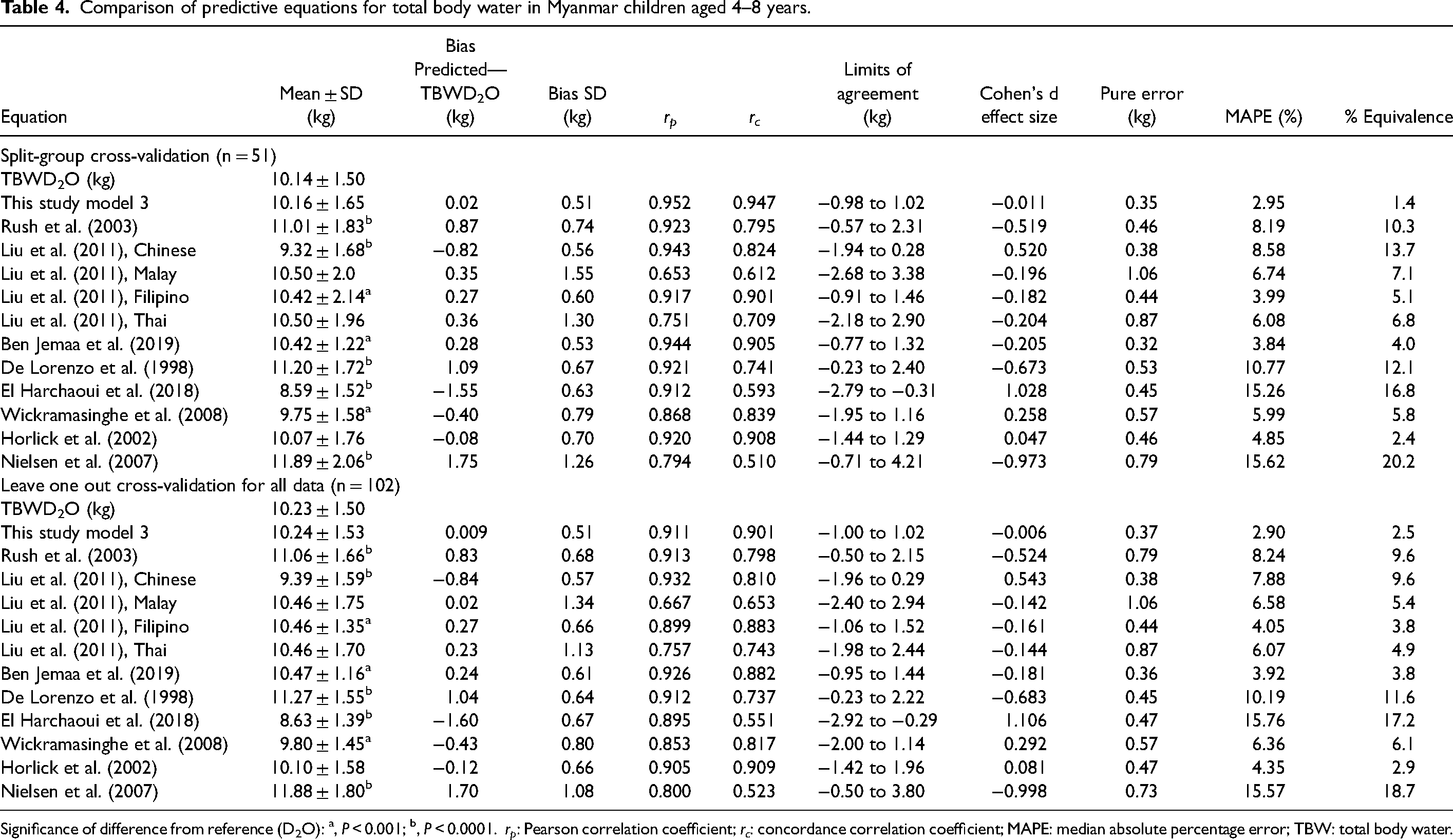
Significance of difference from reference (D2O): a, P < 0.001; b, P < 0.0001. rp: Pearson correlation coefficient; rc: concordance correlation coefficient; MAPE: median absolute percentage error; TBW: total body water.
These results were mirrored for Model 3 in the whole population when assessed using the LOOCV method (Table 4, bottom panel). Similarly, the Jemaa and Liu equations performed almost as well. The final prediction equation based on all participants was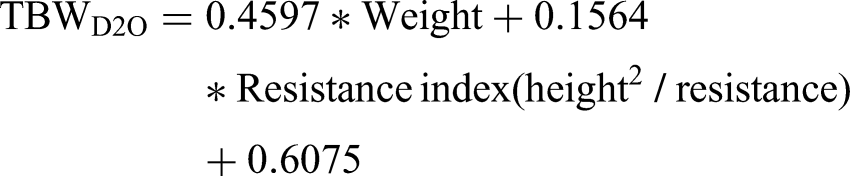
Discussion
The assessment of body composition can provide useful information in public health nutrition since body composition represents the time-integrated outcomes of preceding nutritional challenges (Ward, 2018). This is particularly important in the growing child, where adverse nutritional events in childhood can have life-long lasting effects. The measurement of body composition in vivo represents logistical and cost difficulties, particularly in children (Wells and Fewtrell, 2006). Bioelectrical impedance analysis presents a simple and inexpensive method for assessment of body composition suitable for widespread population use. The present study developed a BIA prediction algorithm suitable for use in young children (4–8 years old) in Myanmar. The algorithm was generated and validated against the reference method of TBW measurement by deuterium dilution. The study also produced from these reference data body composition data for this age group.
Very little body composition data exists for the Myanmar population in general and is further limited in children. Data is generally restricted to simple anthropometric information such as weight and height or predictions based on skin-fold measurements (Hlaing et al., 2017; Lwin et al., 2006). Data are also frequently limited to infants or those under 5 years of age (Blankenship et al., 2020; Hong et al., 2018). A common observation is that some degree of stunting based on height-for-age < −2SD below the WHO reference median is present (Blankenship et al., 2020; Hong et al., 2018). In the current study, the overall median height-for-age z-score was −1.01 although 14 children (13.7%) exceeded the < −2SD threshold. Generally, children were lean; the median %FM was 16.2%. These observations contrast with the findings of persistent stunting in Myanmar children from population-wide surveys (Hong, 2020; International Food Policy Research Institute, 2015).
The deuterium dilution method while widely used and applicable in children is not ideal for large-scale population-based assessment of body composition. Compared to other techniques such as dual-energy X-ray absorptiometry it is relatively inexpensive but does require a laboratory for sample analysis. In contrast, BIA is rapid, and simple, and uses highly portable equipment but requires calibration against a reference method such as deuterium dilution. It is generally recognized that BIA prediction algorithms exhibit population specificity and dedicated algorithms are required in children (Nguyen et al., 2020). This study has produced an algorithm suitable for use in young Myanmar children. At a group level, accuracy was extremely high with a very small bias between BIA-based prediction and reference measurements of TBW. This makes the equation eminently suitable for use in epidemiological population-wide surveys. Limits of agreement are used to assess suitability for prediction in an individual. There is no consensus on acceptable limits for body composition parameters (Ward, 2018) but the limits observed here of approximately ± 10% for BIA-based predictors are generally deemed to be acceptable for a method to be used in clinical practice (Ward, 2019). This study provided the opportunity to assess the relative predictive power of alternative published prediction equations. Most of the published equations performed well with small biases but with LOA approximately twice those of the new equation. Of particular note was the poor performance of the Nielsen equation (Nielsen et al., 2007). This is perhaps not surprising since it was generated in a quite different population, Caucasian children but of a similar age. Contrastingly, the equation of Horlick et al. (Horlick et al., 2002) performed almost as well as the new equation despite being generated from a large mixed ethnic-origin population of American children and adolescents This predictor would be a suitable alternative to the current equation particularly in older children since the population studied covered a much wider age range (4–18 years).
The study has strengths and limitations. This is the first study to assess body composition in Myanmar children using a reference method and the first to produce a BIA prediction equation specific to Myanmar children. The age range studied was, however, limited (4–8 years) and research should be undertaken to extend this to include adolescents and young adults. In the interim, the Horlick equation may provide an acceptable alternative. Although it is not possible to determine a priori an acceptable sample size when generating prediction equations it is generally recognized that the larger the sample size the more precise predictions will be (Guo et al., 1996). The sample size in the present study exceeds that recommended by Guo and colleagues (Guo et al., 1996) and is similar to (Ben Jemaa et al., 2019) or exceeds that of comparable studies (Rush et al., 2003) but verification of the prediction equation in a larger sample would be valuable. Alternatively, Bayesian statistical analysis may be used as it is less susceptible to sample size effects (McNeish, 2016) but has rarely been used for this type of method comparison (Lakmali et al., 2022).
Summary
In conclusion, this study has demonstrated the feasibility and suitability of both deuterium dilution and BIA for the assessment of body composition in young children. The derived BIA prediction equation, the first for Myanmar children, should prove useful for this purpose in future epidemiological studies.
Future directions
The present study may act as a study template for the development of BIA-predictive equations in other age groups. The IAEA-supported establishment of the deuterium dilution method in Myanmar can provide a regional reference center for this purpose.
Footnotes
Acknowledgements
The authors gratefully acknowledge the participation of all children and their parents in this study.
Authors’ contributions
Author Maw contributed to the study design, undertook data collection, conducted laboratory analyses and contributed to manuscript writing and final review. Author Thwin contributed to the study design obtained participant consent, provided medical oversight, assisted with data collection and analysis, and prepared original manuscript drafts and subsequent writing and final review. Author Owino provided IAEA liaison, deuterium analysis protocols, manuscript writing and review. Author Ward contributed to the study design, took primary responsibility for data and statistical analyses and contributed to manuscript writing and final review.
Availability of data and materials
Original data will be made available to bona fide researchers upon reasonable request to the corresponding author.
Consent for publication
All authors and where required institutions have approved the manuscript for submission to Nutrition and Health for publication.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Author Ward provides consultancy services to ImpediMed Ltd, manufacturer of impedance devices. ImpediMed had no input into the design, conduct or reporting of this study. All other authors have no conflicts to declare.
Ethical approval
The study was conducted in accordance with the guidelines laid down in the Declaration of Helsinki. The study was reviewed and approved (3 June 2013, extended 31 May 2016) by the Ethics Review Committee, Department of Medical Research, Ministry of Health, Republic of the Union of Myanmar. All children, their parents and teachers were informed about the aims and procedures of the study and written informed parental consent was obtained.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported, in part with the financial support from the International Atomic Energy Agency (IAEA) under project Using Nuclear Techniques to Assess the Role of Nutrition-Sensitive Agri-Food Systems in Improving Diet, Health and Nutritional Status of Vulnerable Populations (E4.30.29.-CR-1). Author Maw received a short-term training fellowship from the IAEA (C6/MYA/12005).
