Abstract
Introduction
According to the Women's Cancer Research Fund (WCRF), breast cancer (BC) is the most common form of cancer and the leading cause of cancer related death in women worldwide (WCRF, 2018b). Prevention of cancer is one of the most significant public health challenges of the 21st century. The incidence of BC is increasing in low- and middle-income countries (Parkin and Fernandez, 2006; Villarreal-Garza et al., 2013; WCRF, 2018b). In México, BC has surpassed cervical cancer and is the leading cause of death in women of reproductive age (Centro Nacional de Equidad de Género y Salud Reproductiva, 2016). In 2014, 28.75 new cases of BC per 100,000 were registered in 20 + year old women (Mexico, 2015) whilst in 2019, incidence was reported to increase to 35.24 new cases per 100,000 women (INEGI, 2020). According to the Global Cancer Observatory, in 2020 the estimated age-standardised incidence rate of BC in women reached 40.5 per 100,000 women in Mexico (GLOBOCAN, 2020). Within Mexico, BC incidence is most highly observed in the north, in the state of Sonora, which is a border state with the United States (US) (INEGI, 2014).
The main risk factors for BC include age at menarche, age at menopause, age at first full-term pregnancy and number of births (Fioretti et al., 1999; Hamajima et al., 2002; Collaborative Group on Hormonal Factors in Breast Cancer, 2001). However, these factors do not explain the increase in BC rates and imply that lifestyle factors can modify the risk of the disease (Heitz et al., 2018; Parkin and Fernandez, 2006; Cappellani et al., 2012; Iwasaki and Tsugane, 2011). Several studies have focused on the role of diet as a potentially modifiable risk factor over the risk of BC (Michels et al., 2007). Apart from a consistent positive association between alcohol intake and BC (Liu et al., 2015; Thomson, 2012; Ellison et al., 2001), most of the associations between food or nutrients and BC remain controversial (American Cancer Society, 2019; Michels et al., 2007; Edefonti et al., 2009; Mannisto et al., 2005). Given the complexity of human diets, the correlation and effect modification of nutrient intake, and the synergistic interactions of chemical compounds in food, conclusions about the effect of consumption of a single nutrient or food on BC may be misleading (Edefonti et al., 2009; Velie et al., 2005).
During the past two decades, interest has shifted to analyse food consumption as dietary patterns, as it offers a different perspective from the traditional single nutrient approach and may provide an understanding of the complexity of nutrient combination, their interaction (Hu, 2002) and their association with BC (Brennan et al., 2010). Thus, the patterns of dietary behaviours represent a broader picture of food and nutrient intake, which may provide evidence for public health recommendations (Edefonti et al., 2009; Cottet et al., 2009; Buck et al., 2011).
Several studies conducted in Europe (Edefonti et al., 2009; Mannisto et al., 2005; Buck et al., 2011; Sieri et al., 2004; Sant et al., 2007; Demetriou et al., 2012; Bessaoud et al., 2012; Baglietto et al., 2011), US (Adebamowo et al., 2005; Fung et al., 2005; Velie et al., 2005; Agurs-Collins et al., 2009; Murtaugh et al., 2008; Hirose et al., 2007; Link et al., 2013), Asia (Hirose et al., 2007; Zhang et al., 2011; Cui et al., 2007), Africa (Jordan et al., 2013), and South America (De Stefani et al., 2009; Ronco et al., 2006; Tumas et al., 2014), have explored the influence of dietary patterns on BC risk. These studies suggest that unhealthy type of dietary patterns, characterised by foods high in fat, salt, sugar and calories as well as ultra-processed foods (i.e. products developed from substances extracted from foods such as fats, starches, added sugars and hydrogenated fats (Gibney, 2018), pose a higher risk for BC and other types of cancer (Steck and Murphy, 2020). However, most of these studies were conducted in countries with diets that differ from the Mexican diet.
To our knowledge, no previous studies have evaluated the association between dietary patterns, cooking methods and BC risk in Mexican women. Therefore, the aim of this exploratory case-control study was to identify dietary patterns in women of Northwest Mexico, to evaluate the relationship of dietary patterns and cooking methods with BC risk. In addition, we evaluated the aforementioned associations by oestrogen exposure categories.
Materials and methods
Study population
This exploratory case-control study included 120 women from Northwest Mexico, 25 years or older. Sample size was determined considering a case control study design, an odds ratio (OR) of 3.01 based on the literature (Zheng et al., 1998), a proportion of 50% of control exposure to meat consumption, an α-value = 0.05 and a power of 80%. Cases (n = 60) were diagnosed with incident, histologically confirmed BC within the past 2 years (since 2010), with no history of diabetes, heart disease or cancer at other sites who were admitted to two of the major general hospitals in the region. Eighty-five percent of cases were selected from the State Oncology Hospital, since only cancer patients go to that hospital. In addition, 15% of patients were recruited from the State Security and Worker Social Services Institute (ISSSTESON). Controls (n = 60) were also recruited from the major general hospitals in the region; they were selected from the general medicine ward area of hospitals (e.g. physical therapy) excluding the oncology department. Controls were comparable on all observed covariates (e.g. age, sex, geographic area) and were not diagnosed with cancer or any other non-communicable disease that could have modified their dietary behaviour at the time of data collection. The study protocol was approved by the Ethical Committee of the Food and Development Research Centre (Centro de Investigación en Alimentación y Desarrollo, CIAD), in Mexico. All participants gave a written informed consent prior to their participation in the study.
Data collection
The data collection of this study took place in the year 2012 for a two-year period. The participants were interviewed at their homes by a trained health professional, by using a standardised questionnaire to collect demographic and risk factor data. The questionnaire included information on age, level of education, marital status, and socioeconomic status (SES). The latter was evaluated using the questions included in the AMAI 8X7 instrument (Mexican Association of Market Research Agencies). This index classifies households into seven levels, considering seven characteristics or household possessions and the education of the head of the family or person who contributes the most to spending. The questionnaire also assessed smoking status, alcohol consumption, family history of cancer, hormone replacement therapy, oral contraceptive use, age at menarche and menopause, pregnancies, and breastfeeding history.
Anthropometric and body composition data were undertaken in cases and controls. Weight was measured on a flat surface using a digital scale (AND FG-150K) with 0.05 kg of precision. Height was measured with a transportable field stadiometer (Holtain Limited, 0.695–2.10 m) with the participant's head in the Frankfort horizontal plane, and it was recorded to the nearest 0.1 cm. Body mass index (BMI) was calculated by dividing weight by height squared (kg/m2). Waist and hip circumference were measured with a measuring tape. Body composition was determined by electric bioimpedance using a portable electric bioimpedance device with a single 50 kHz frequency (IMPEDIMED, San Diego, CA). The reactance (X) and resistance (R) data obtained were entered into the corresponding anthropometric equation validated in adults (Macias et al., 2007), and total body water, fat-free mass, and fat mass were calculated.
Dietary patterns
Dietary patterns were determined in the year prior to diagnosis for the cases, and in the year before the interview for the controls, using a semi-quantitative food frequency questionnaire (FFQ) validated for women in the region (Quizán-Plata and Ortega-Vélez, 2000) and modified for this particular study. A trained professional interviewer applied the questionnaire that included 162 food items under five categories: fruits and vegetables; dairy products; bread, tortillas, and snacks; fish and seafood; and candy and desserts. Frequency of consumption and portion sizes of each food item were evaluated on a daily, weekly, or monthly basis. Daily intake quantification was performed using a food dictionary that included data from the United States Department of Agriculture (USDA) (USDA, 2011), the National Institute of Nutrition in Mexico, and the food composition database from our Institution (Campa-Siqueiros, 2012).
Cooking methods
Cooking methods were assessed through the validated Computerized Heterocyclic Amines Resource for Research in Epidemiology of Disease (CHARRED) questionnaire designed by the National Cancer Institute (National-Cancer-Institute, 2006). The questionnaire was applied by a trained data collector to each participant who self-reported their cooking methods. The CHARRED questionnaire was linked with the FFQ in which consumption of meat was reported as never, daily, weekly, or a monthly basis. If participants confirmed consumption of any type of meat, including fish or poultry, questions on cooking methods and cooking intensity were assessed.
The questions focused on the cooking method used, and included: fried, cooked without oil, broiled, oven-broiled, cooked in microwave, roasted, barbecued, and smoked. In addition, the cooking method frequency of use and estimated portion size of cooked food consumed were evaluated. Cooking intensity of meat was also assessed using the CHARRED method. The intensity of meat cooking included categories such as “very well done”, “well done”, “just until done”, “juicy and mostly pink”, and “almost raw” (Tasevska et al., 2009; Cross et al., 2007; National-Cancer-Institute, 2006).
Statistical methods
To compare descriptive variables between cases and controls, Student's t-tests and Chi-squared tests were used as appropriate. K-mean cluster analysis (CA) was used to define dietary patterns (Hu, 2002). This approach overcomes the limitation of the commonly used principal component analysis (PCA) method, which implies that the patterns, by definition, must be uncorrelated (Bessaoud et al., 2012). First, the frequency of food consumption and the average quantity in grams of each food consumed by each participant was calculated. Second, participants with similar diets were grouped into homogenous subgroups (i.e. clusters), using K-means analysis with a maximum of 600 iterations and 60 random starts. Foods with closer distances were grouped into the same cluster or split into a different cluster when the K-mean distance between clusters was greater. Three main clusters, which represented different dietary patterns among participants, were identified. Pattern ‘2’ and ‘3’ were both characterised by similar food groups and a high calorie content; however, pattern ‘3’ only had n = 5 participants. Therefore, patterns ‘2’ and ‘3’ were combined due to their similar characteristics, which resulted in two main dietary patterns.
To determine the association of dietary patterns and BC risk, Odds ratios (ORs) and 95% confidence intervals (CIs) were calculated by using multivariate, unconditional logistic regression. The healthier pattern was used as a reference to interpret results. A second model was generated to assess the association between the intensity of cooking methods, as an independent variable, and BC, as a dependent variable. Cooking intensity was categorised as 0 for lower intensity cooking and 1 for intense cooking. Stepwise regression was used to determine the selection of confounders in the statistical models. Tested variables which were evaluated as possible confounders included age, age at menarche, menopause stage, family history of cancer, number of children, oral contraceptive use, physical activity, smoking, alcohol intake, poultry and meat cooking intensity (for the dietary patterns model), total energy consumption, BMI and SES. Lastly, logistic regression analyses were undertaken considering oestrogen exposure (age at menopause – age at menarche) strata. Analyses were performed using STATA version 16, and a p ≤ 0.05 was considered as statistically significant.
Results
Characteristics of the sample
General characteristics of cases and controls are presented in Table 1. Ages ranged from 25 to 77 years and 12% of BC patients were diagnosed before the age of 40. Compared to controls, cases had a significantly lower age at menarche (p < 0.05), were more likely to have a family history of cancer (p < 0.05), and more likely to have used oral contraceptive drugs (p < 0.05). Body fat exceeded 45% in cases and was higher than in controls. Exposure to oestrogens was higher, although non-significant (p < 0.1) in cases than in controls. The two groups did not differ in terms of age, age at menopause, number of children, breastfeeding, weight, height and BMI, from which both groups had a BMI above 25 kg/m2.
General characteristics of cases and controls, N = 120.
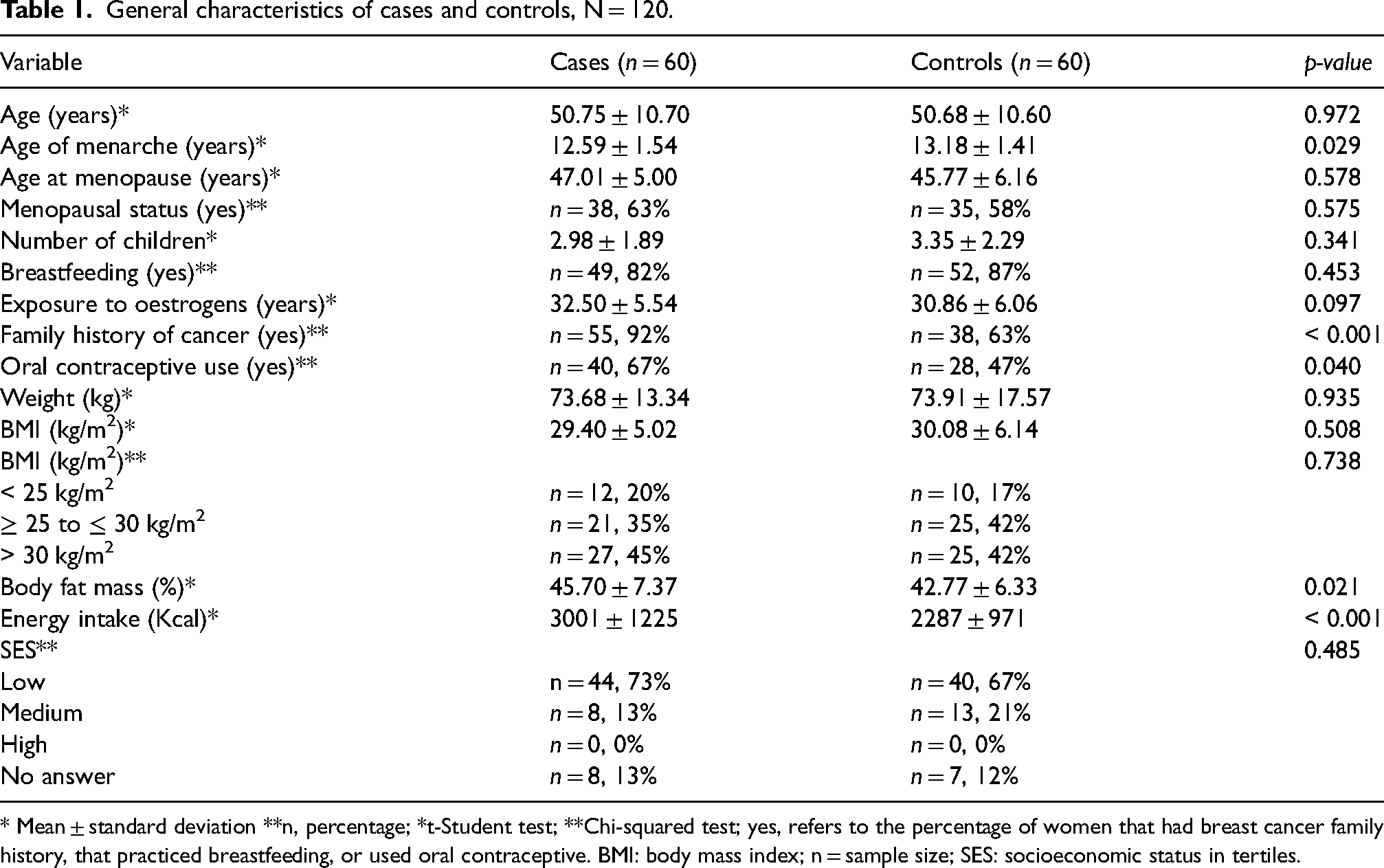
* Mean ± standard deviation **n, percentage; *t-Student test; **Chi-squared test; yes, refers to the percentage of women that had breast cancer family history, that practiced breastfeeding, or used oral contraceptive. BMI: body mass index; n = sample size; SES: socioeconomic status in tertiles.
Dietary patterns and cooking intensity
The most consumed foods by cases, in terms of grams, were soft drinks, instant coffee, fried beans, whole milk, brewed coffee, tomato sauce, corn tortillas, stewed tomatoes, oranges and pasta soup. Controls consumed high quantities of instant coffee followed by powdered instant drinks, orange juice, pasta soup, and tomato sauce (Figure 1 and Supplementary Table S1).
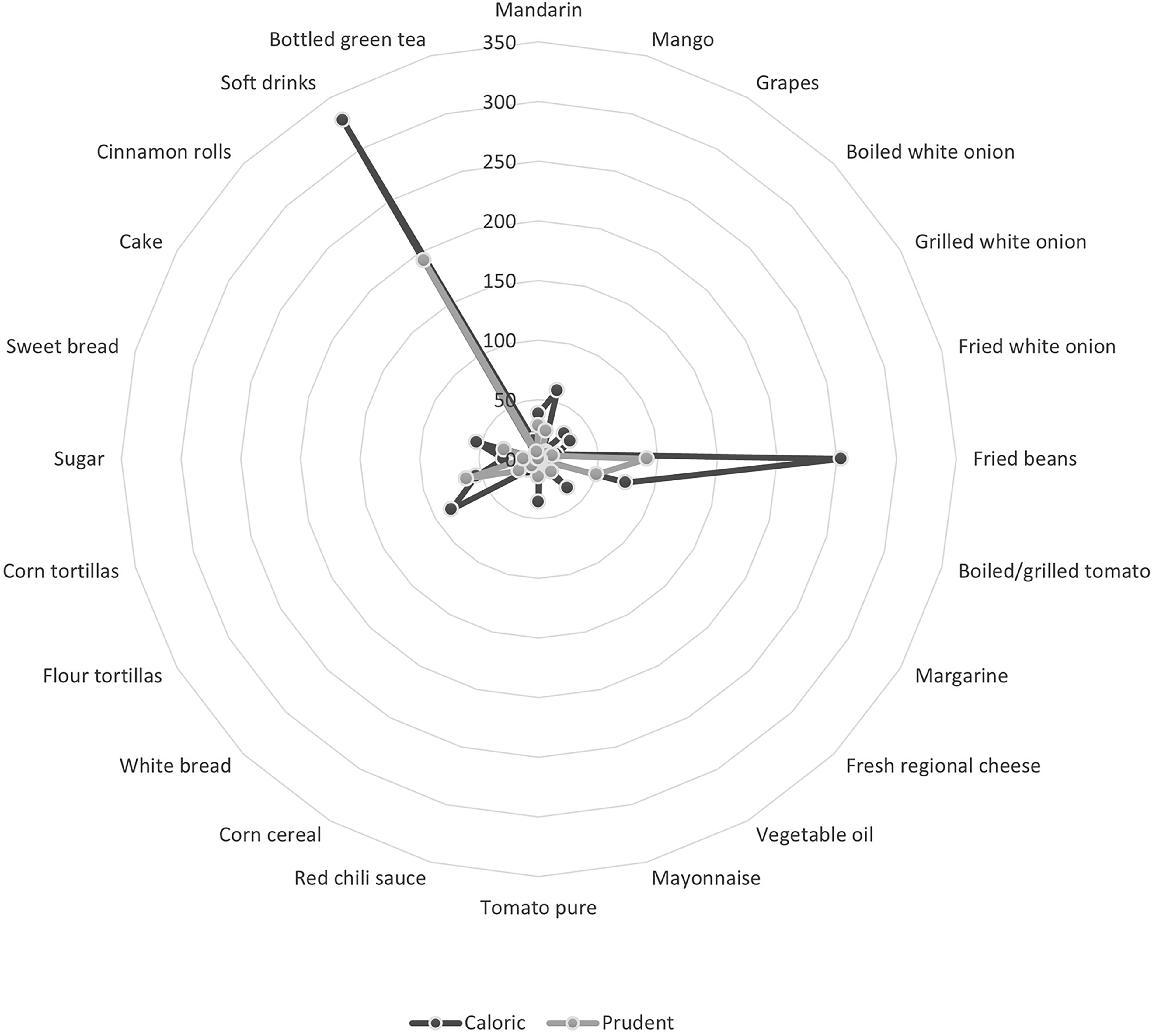
Prudent and caloric dietary pattern characterisation of cases and controls. Food consumption is presented in grams per day (50–350 grams).
Using the K-means analysis, three types of dietary patterns (clusters) were identified and two of them were collapsed together due to the low sample (n = 5) identified for pattern 3. Pattern 1 (prudent) was characterised by a low intake of saturated fats, refined grains, or fruits with a high glycaemic index (e.g. grapes and oranges), and a high intake of corn tortillas compared to the control group. Pattern 2 (caloric) consisted of an abundant consumption of saturated fats, especially fried beans with lard, flour tortillas (with added fat), refined grains and fruits with high glycaemic index as well as a high consumption of added sugar and desserts (Supplementary Table S2).
Table 2 shows the dietary patterns consumed by cases and controls and the percentage of individuals in each classification. Regarding cases, 57% (n = 34) consumed a type 1 (prudent) dietary pattern, and 43% (n = 26) consumed a type 2 pattern. In contrast, 93% (n = 56) of controls consumed a type 1 or prudent dietary pattern, and 7% (n = 4) consumed a caloric type of dietary pattern.
Dietary pattern definition for cases and controls, n = 120.
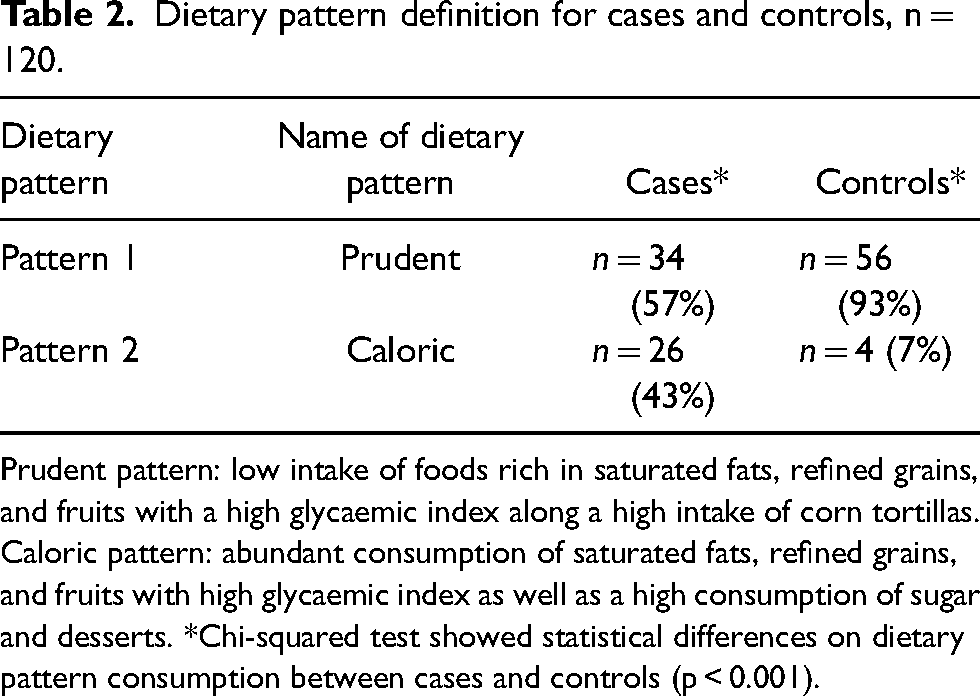
Prudent pattern: low intake of foods rich in saturated fats, refined grains, and fruits with a high glycaemic index along a high intake of corn tortillas. Caloric pattern: abundant consumption of saturated fats, refined grains, and fruits with high glycaemic index as well as a high consumption of sugar and desserts. *Chi-squared test showed statistical differences on dietary pattern consumption between cases and controls (p < 0.001).
Caloric dietary pattern showed a higher content of energy and macronutrients compared with prudent dietary pattern, although differences were not significant (Supplementary Table S3). Cooking intensity of meat and poultry (Table 3) was more prevalent in cases, since 44% compared to 17% of controls consumed well-done poultry with burned sections; also, 48% of all cases and only 8% of all controls consumed well done grilled beef.
Proportion of cases and controls that used different cooking intensity of beef and poultry, N = 120.

Results indicate number and percentage of participants that used each cooking method. Three participants from the cases group did not eat poultry.
*Chi-squared tests showed statistical differences on the cooking intensity level between cases and controls (p < 0.001).
The combination of dietary pattern and cooking intensity for the participants is illustrated in Figure 2. Only 20 cases compared to 50 controls, had a combination of prudent dietary pattern and a low level of cooking intensity; 14 cases and 6 controls had a prudent pattern-high cooking intensity. A total of 12 cases and 2 controls had a caloric pattern-low cooking intensity, and a higher number of cases (n = 12) compared to only two controls, had a caloric dietary pattern combined with a high intensity of cooking.
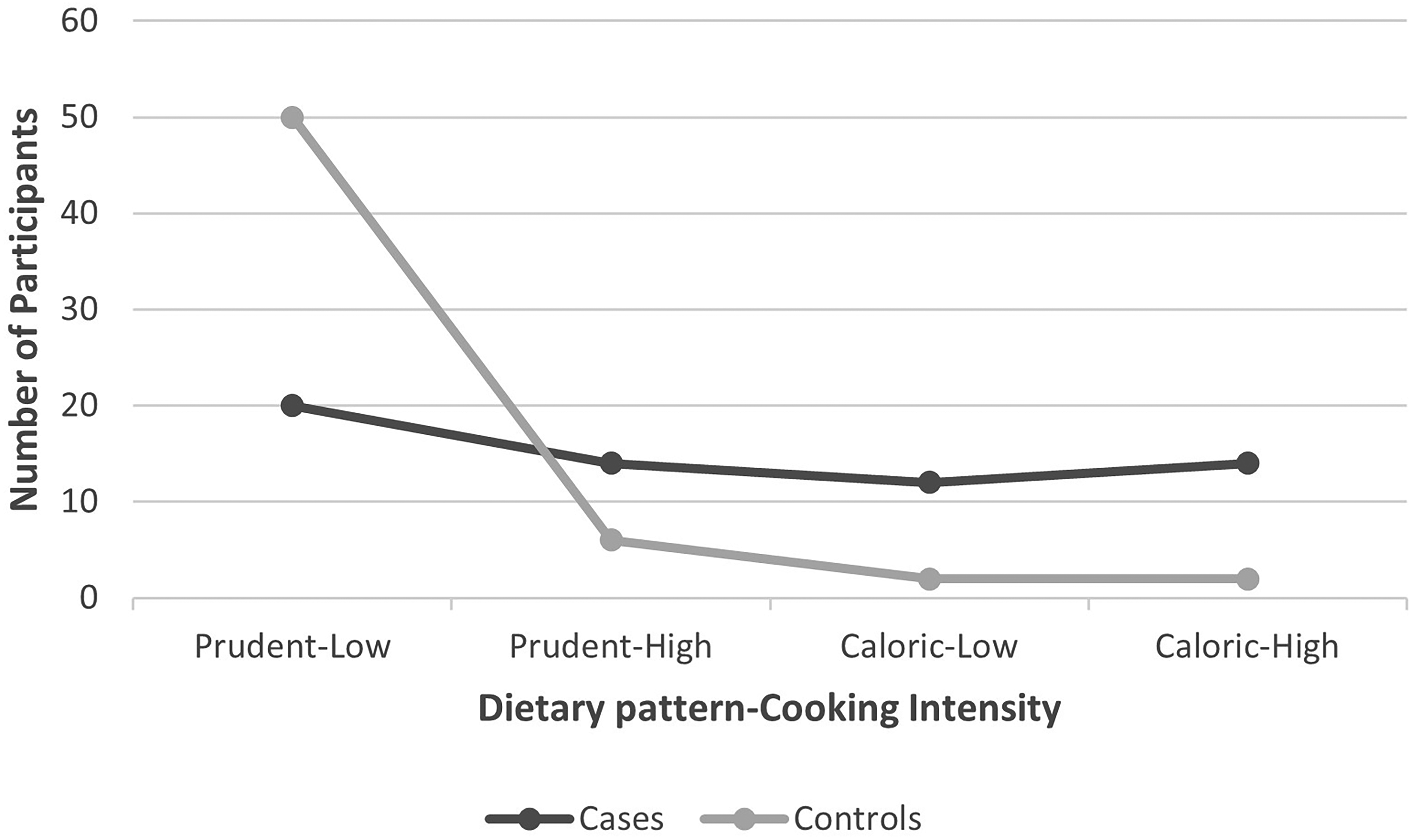
Dietary patterns and cooking intensity of case and control participants. Prudent and caloric represent the type of dietary patterns consumed by study participants while low and high represent the levels of meat cooking intensity. For example, prudent-low represents a prudent type of dietary pattern in combination with a low level of meat cooking intensity while a caloric-high represents a caloric type of dietary pattern with a high intensity level of meat cooking.
Association of dietary patterns and cooking methods with breast cancer risk
BC risk was significantly associated with a caloric type of dietary pattern (Table 4). Crude and adjusted models showed that participants who consume a caloric dietary pattern have a higher risk for BC than women who consume a prudent dietary pattern (Model 1: ORadj: 5.07,9 5% CI: 1.44–17.84, p = 0 .011; Model 2: ORadj: 9.18 (95% CI: 1.51–55.81, p = 0.016). A higher cooking intensity of meat was also found to be associated with a higher risk of BC. Table 4 also shows that a higher intensity of cooking is associated to the risk of BC (Model 1: ORadj: 13.91, 95% CI: 4.66–41.48), p < 0.001; Model 2: ORadj: 14.70, 95% CI: 4.48–48.22, p < 0.001), in relation to a lower intensity of cooking. Both associations remained after adjusting the statistical models for potential confounders including sociodemographic characteristics (e.g. SES) (Table 4).
When the exposure to oestrogens was less than 32 years (Table 5), the caloric dietary pattern was associated with breast cancer risk (ORadj: 4.69, 95%CI: 1.01–21.92, p = 0.049), and a higher intensity of cooking was associated with the risk of breast cancer in both categories of oestrogens exposure (< 32 years and ≥ 32 years).
Association of breast cancer risk with type of dietary pattern consumption, and with cooking intensity level, N = 120.
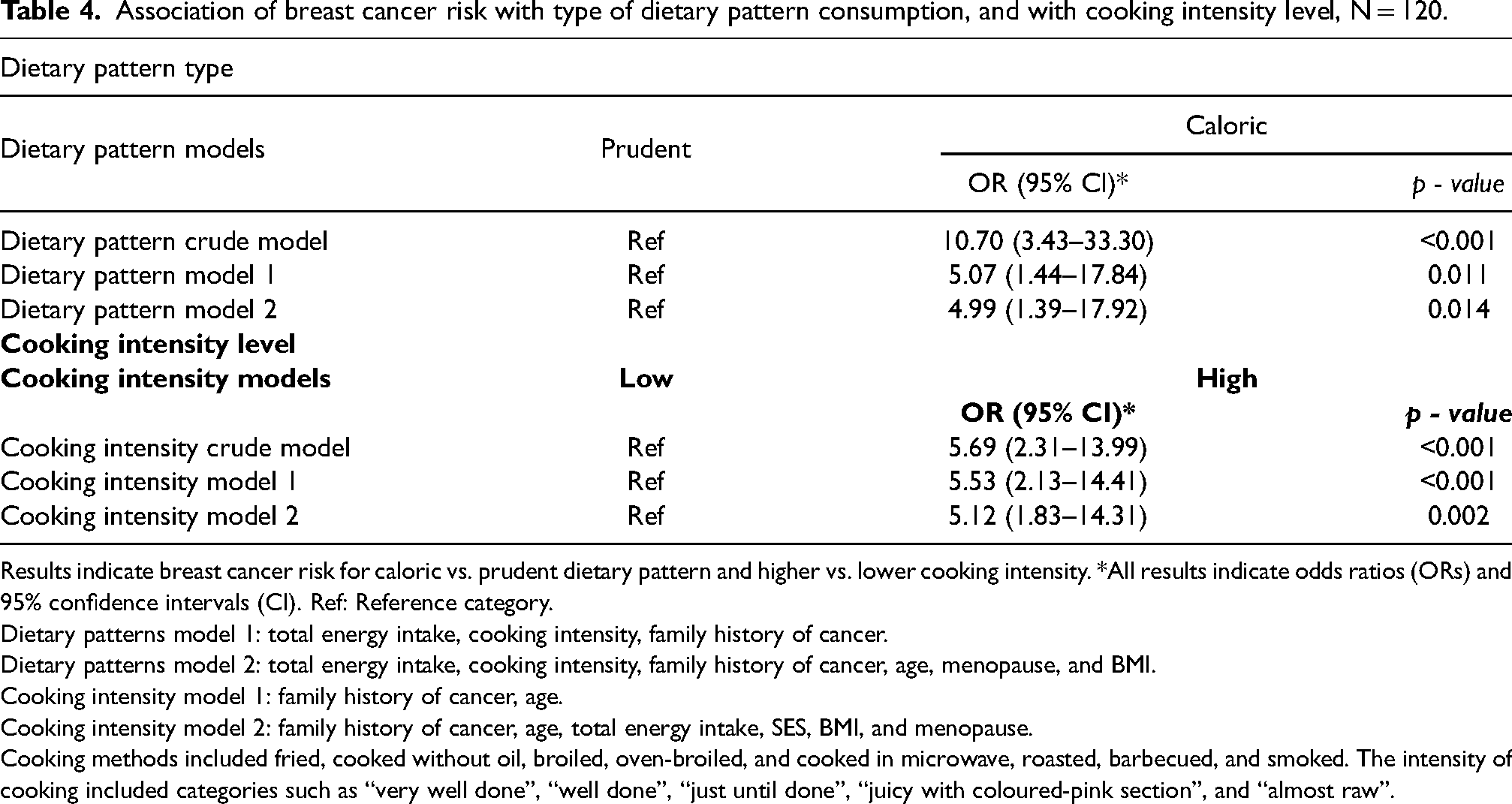
Results indicate breast cancer risk for caloric vs. prudent dietary pattern and higher vs. lower cooking intensity. *All results indicate odds ratios (ORs) and 95% confidence intervals (CI). Ref: Reference category.
Dietary patterns model 1: total energy intake, cooking intensity, family history of cancer.
Dietary patterns model 2: total energy intake, cooking intensity, family history of cancer, age, menopause, and BMI.
Cooking intensity model 1: family history of cancer, age.
Cooking intensity model 2: family history of cancer, age, total energy intake, SES, BMI, and menopause.
Cooking methods included fried, cooked without oil, broiled, oven-broiled, and cooked in microwave, roasted, barbecued, and smoked. The intensity of cooking included categories such as “very well done”, “well done”, “just until done”, “juicy with coloured-pink section”, and “almost raw”.
Odds ratios (ORs) and 95% confidence intervals (CI) for breast cancer risk of the caloric vs. prudent dietary pattern and the higher vs. lower cooking intensity, stratifying by oestrogen exposure, N = 120.
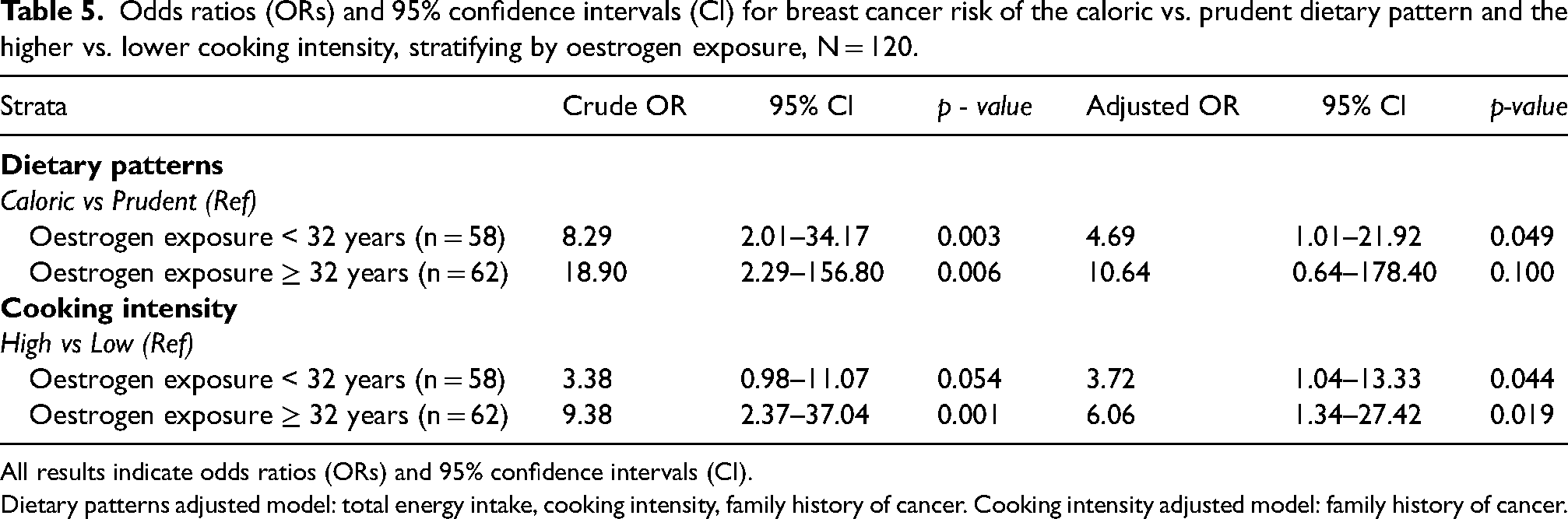
All results indicate odds ratios (ORs) and 95% confidence intervals (CI).
Dietary patterns adjusted model: total energy intake, cooking intensity, family history of cancer. Cooking intensity adjusted model: family history of cancer.
Discussion
In this study, our aim was to evaluate dietary patterns and cooking methods as potential risk factors for BC in women from northwest Mexico. According to our results, a high caloric pattern, rich in saturated fat and refined sugar, was found to be a risk factor for BC. In addition, an excessive cooking of poultry and beef was associated with a higher cancer risk. The study findings support our hypothesis that a caloric dietary pattern and a high intensity of cooking are associated with a higher risk for BC. The associations that we found in this study were more pronounced when exposure to oestrogens was 32 years or more, compared to the associations found in the strata < 32 years. This makes sense, as lifetime exposure to oestrogens is a risk factor for BC (Lippman et al., 2001).
The results found in this study agree with studies related to the association of dietary patterns and BC risk in other countries. According to a meta-analysis by Brennan et al. (2010), a larger risk of BC was associated with an occidental or non-healthy diet (OR = 1.31; 95% CI: 1.05, 1.63; p = 0.02). Another study in Tanzania (Jordan et al.), indicates that a fatty type diet, characterised by a high consumption of milk, vegetable oils, fats, butter, lard and red meat was found to be associated to a higher risk of BC (OR: 1.42, IC 95%: 1.08, 1.87, p = 0.01). Dietary patterns rich in saturated fat and red and processed meats were consistently found to be associated with increased BC risk (Dandamudi et al., 2018). On the other hand, an Italian cohort study by Sieri et al., highlights that a dietary pattern rich in vegetables lowers the risk of BC (Sieri et al., 2004). Similarly, a meta-analysis study by Brennan et al., indicates that a healthy or prudent dietary pattern is less likely to be associated with the risk of BC (OR = 0.89; 95% CI: 0.82, 0.99; p = 0.02) (Brennan et al., 2010).
In the present study, we found that soft drinks were highly consumed by the participants that had a caloric dietary pattern. An average of 350 grams were consumed per day, which is equivalent to a 12 oz can of soft drink in the US. This could suggest that these types of beverages may play an important role in the development of cancer. Previous studies have identified an association of sugar-sweetened beverage consumption with cancer (Chazelas et al., 2019; Anderson et al., 2020). Results from the NutriNet-Santé prospective cohort reported that the consumption of sugary drinks was significantly associated with the risk of overall cancer (n = 2193 cases, sub-distribution hazard ratio (HR) for a 100 mL/d increase: 1.18, 95% CI 1.10–1.27, p < 0.0001) and BC (HR:1.22, 95%CI: 1.07–1.39, p = 0.004) (Chazelas et al., 2019). Similarly, sugar-sweetened beverage consumption was found to be associated with higher mortality, of which the most common cause was cancer (Anderson et al., 2020). This issue is relevant since in Mexico, sugar sweetened beverages contribute 10% of the total caloric intake of children and adults (FAO, 2020; Aburto et al., 2016), and 70% of the total added sugar in the diet (Sánchez-Pimienta et al., 2016).
The WHO guidelines recommend that free sugar intake should be <10% of total energy intake (WHO, 2015). According to Braverman-Bronstein et al. (2020), 19% of diabetes, cardiovascular disease and obesity-related cancer mortality is attributable to sugar sweetened beverages, representing 37,000 excess deaths/year in Mexican adults of 20 years or older.
Mexico, like most middle-income countries in Latin America, has undergone a rapid epidemiological transition featuring a shift in dietary habits and higher obesity prevalence (Popkin, 2006; INSP, 2018). In 2018, 77% of women in Mexico presented overweight or obesity (INSP, 2018). Specifically in the North-western region of the country, prevalence of obesity in adults is 77% (Sonora, 2020), and dietary intake has its own unique features, being characterised by a high intake of saturated fat, grilled red meat, refined sugars, beans, corn and wheat tortillas with a high fibre content, along with a low consumption of fruits and vegetables (Gonzalez-Siqueiros, 2008). In addition, the consumption of well cooked meat and poultry as well as the excessive use of oil and lard, may be potential risk factors for BC in the region, since associations have been found between the presence of heterocyclic amines, which are potent mammary carcinogens, and the duration and temperature of cooking (Zheng and Lee, 2009).
Most of the women who participated in this study had a low SES (73%) and a low level of education (mean of 10.7 years of education). Their consumption of poultry and meat was lower than intakes in higher income women. Due to the higher price of meat, low-income populations could decide on processed meat alternatives or other protein choices. Interestingly, we found that the intensity of cooking of poultry and meat consumed by the participants, but not the frequency of consumption, was associated with cancer risk. Therefore, the caloric pattern was found to be high in fried foods prepared with large amounts of oil or butter, making it a high-fat pattern, and a higher risk of BC was observed when poultry and meat were highly cooked. Similar findings have been observed in other countries. In Iran, a higher risk of BC was observed when consuming unhealthy dietary patterns rich in fried foods (OR: 4.5 (95% CI, 2.1, 9.4) (Marzbani et al., 2019). Similarly, In the US, a high intake of red meat was associated with a higher risk of BC. The intake of well-done red meat was associated with an almost twofold increased risk of BC (OR: 1.5, 95% CI: 1.3, 1.9, p < 0.001) (Fu et al., 2011). Due to a consumption of solid and semi-solid fats from more than half of the participants in our study, the effect of fried food on BC was elevated. In this regard, it is necessary to carry out studies on the methods of frying, the temperature used and the duration of frying.
According to the World Cancer Research Fund and the American Institute for Cancer Research (Wiseman, 2008; WCRF, 2018a), the proposed mechanisms for the link between red meat consumption and cancer are based on the production of N-nitrous compounds and the release of iron. N-nitrous compounds are generated during digestion, due to the presence of bacteria in the stomach and large intestine, as well as during the cooking process, in which high temperatures favour the generation of heterocyclic amines and polycyclic aromatic hydrocarbons. Additionally, during the cooking process, the heme group releases iron, which favours the production of free radicals, activates the transcription factors responsible for the oxidative response, pro-inflammatory cytokines and hypoxia signals (Wiseman, 2008).
We chose CA to determine dietary patterns. This technique reduces complex multivariate data into smaller clusters (Leonard and Droege, 2008). CA assigns individuals to clusters in which food consumption is relatively homogeneous. The intra-individual variability inside the cluster is small, whilst the variability between-groups is significant due to the differences in food intake among subgroups. Therefore, the implementation of CA to identify dietary pattern refers to clustering among individuals rather than among variables, therefore considering the regularity of food intake (Cunha et al., 2010; Sauvageot et al., 2017). However, a potential limitation of this method is that the selection of clusters is largely subjective. Therefore, we carried out the analysis with varying number of clusters, finding similar results. Among the limitations of this study is the addition of foods to the FFQ, which could have overestimated the consumption of nutrients and energy. Since this is an exploratory study, sample size is another limitation that may affect the results, especially those related to the association of dietary patterns and BC risk in the adjusted models. The low number of participants reduces the external validity of the study; therefore, we could not generalise the results to the entire population of Mexican adult women. Exposure to oestrogens was calculated by subtracting age at menarche to age at menopause, without considering the number of pregnancies or time of breastfeeding. Among the strengths of the study are that the dietary evaluation was assessed using dietary patterns and not nutrients or isolated food components. We analysed and considered cooking methods and cooking intensity, which contributes to the limited knowledge that exists in this area. Another strength of this study is that only one trained interviewer collected the data, which reduces potential interviewer bias. Due to the lack of studies in relation to BC, cooking methods and dietary patterns in Mexico, this exploratory study brings a closer approach to the understanding of diet and cooking methods as potential risk factors for BC risk in women. Further research and interventions that examine the role of dietary patterns and BC risk are warranted.
Conclusions
The results of this exploratory study provide evidence of a strong association between a caloric dietary pattern and risk of BC in Mexican women. A dietary pattern that exceeds the recommended caloric intake coupled with a high content of saturated fats and refined carbohydrates, turned to be a risk factor for BC in women from Northwest Mexico who participated in this study. Likewise, the consumption of burnt meat products by high intensity cooking proved to be a risk factor for the disease. To our knowledge, this is the first study in Latin America that evaluates the relationship of dietary patterns and cooking methods on BC risk. We recommend carrying out further research that analyses the role of dietary patterns and cooking methods on BC risk, to sustain and expand the information presented in this exploratory study.
Supplemental Material
sj-docx-1-nah-10.1177_02601060221119260 - Supplemental material for Dietary patterns, cooking methods and breast cancer risk in Mexico: An exploratory case-control study
Supplemental material, sj-docx-1-nah-10.1177_02601060221119260 for Dietary patterns, cooking methods and breast cancer risk in Mexico: An exploratory case-control study by Elisa Pineda, María I. Ortega-Vélez and Martin Preciado-Rodríguez, Socorro Saucedo-Tamayo, Graciela Caire-Juvera in Nutrition and Health
Footnotes
Acknowledgements
Our thanks to the women that participated in the study and to the members of the research team. Greatly appreciated is the input of Ms Miriam Ramos López and Ms Diana Luna for their contribution to the study.
Authors’ contributions
E.P. carried out the data collection and analyses, wrote the manuscript, and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
M.I.O.V. revised the manuscript, approved the final version, and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
M.P. revised the manuscript, approved the final version, and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
S.S.T. revised the manuscript, approved the final version, and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
G.C. conceived and designed the work that led to the submission, revised the manuscript, approved the final version, and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Availability of data
Data is available upon request through the corresponding author.
Consent for publication
A consent for publication form was signed by each participant.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical statement
Ethical clearance was obtained from the Food and Development Research Centre [details omitted for double-anonymized peer review]. Informed consent was obtained from all individual participants included in the study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Department of Nutrition, Food and Development Research Center, [Centro de Investigación en Alimentación y Desarrollo, A.C].
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
