Abstract
Background
Dietary guidelines are intended to inform and aid the general public, with the aim of improving healthy diets and reducing health risk. The effectiveness of these guidelines, however, is rarely investigated.
Aim
This work investigates the effects of three different types of dietary recommendations for reducing free sugars, on free sugar intakes over 12 weeks. Secondary aims will also investigate how these different recommendations affect secondary outcomes, outcomes in subsets of the trial population, and identify barriers and facilitators to dietary change.
Methods
Using a randomised controlled parallel-group trial with three intervention and one control arms, 240 individuals consuming >5% total energy intake from free sugars will be randomized to receive: nutrient-based, nutrient- and food-based, nutrient-, food- and food-substitution-based recommendations or no recommendations, with outcomes assessed for the following 12 weeks. Our primary outcomes are free sugar intakes and adherence to the recommendations. Secondary outcomes are daily energy intake, dietary composition, anthropometry, sweet food perceptions and preferences, sweet food choice, attitudes towards sweet foods, eating behaviour and food choice, knowledge and lifestyle variables, quality of life, adverse events, and barriers and facilitators towards intervention adherence.
Results
Data will contribute to three distinct analyses: 1) Analyses to investigate the effects of the three different dietary recommendations versus control; 2) Analyses of the effects of the dietary recommendations in different population subgroups, and 3) Investigation of the barriers and facilitators to success.
Conclusion
This work offers new perspectives on the effects of different dietary recommendations to enact behaviour change.
Keywords
Introduction
Extensive epidemiological evidence supports a relationship between dietary intakes and the incidence, prevalence and severity of non-communicable diseases (Afshin et al., 2019). Thus, the prevention and treatment of non-communicable diseases may be aided through greater population-based adherence to healthy diets (Brunner et al., 2007; Eriksen et al., 2018).
One strategy for improving diets on a population-wide basis is the provision of dietary guidelines. With the evidence for benefit primarily based on nutrients, early dietary recommendations focused on nutrient intakes, e.g., salt, fat, folate. These guidelines adapted nutrient daily reference intake values into simple messages for the public (Public Health England, 2016). The UK's salt reduction programme, for example, relied on simple nutrient-based recommendations (He et al., 2014). In combination with industry reformulation, and a straightforward monitoring programme, successful reductions in salt intakes and population blood pressure were observed (He et al., 2014), but progress has stalled since 2011 (He et al., 2019; National Diet and Nutrition Survey, 2020).
In 1998, the World Health Organization (WHO) and Food and Agriculture Organization (FAO) recommended that consumer guidelines should be based on foods, as foods, not nutrients, make up dietary choices and that encouraging changes to whole dietary patterns would benefit multiple single nutrient goals (World Health Organisation et al., 1998). National food-based dietary guidelines (FBDG) now exist in over 90 countries (Herforth et al., 2019).
The majority of FBDG are produced from scientific data, versions of previous guidance, and guidelines in other countries (Blake et al., 2018). For example, Public Health England (PHE) recently reformulated the UK's ‘Eatwell guide’ in response to a national free sugar reduction programme (Public Health England, 2014) and revised dietary recommendations (Scientific Advisory Committee on Nutrition, 2015). Linear programming of intake and food composition data were used to identify foods and food groups, and models were created to identify the fewest number of dietary changes required to meet national daily reference intake values (Public Health England, 2016). The new guidance was then evaluated for understanding in consumer research interviews (Public Health England, 2016), but no real-world field testing was undertaken. This lack of testing may contribute to low uptake and adherence to FBDG (Leme et al., 2021; Yau et al., 2019), suggesting a need for greater rigour in assessing the efficacy and effectiveness of FBDG (Brown et al., 2011).
Limited evidence also suggests that the use of food substitutions may aid FBDG. Pilot evidence suggests that campaigns around snack substitutions may enact dietary change (Juszczyk & Gillison, 2018), and online supermarket-based studies report benefits to the contents of shopping baskets by altering the order of the offered food and suggesting lower saturated fat options (Koutoukidis et al., 2019). However, other studies have found little benefit from food substitution strategies (Forwood et al., 2015).
The proposed study will investigate the effectiveness of nutrient-, food-, and food-substitution-based recommendations for reducing free sugar intakes. Increased consumption of dietary sugars and associations with increased risk of dental carries, non-communicable diseases and excess weight (Ahmad et al., 2020; Monnard and Grasser, 2018; Scientific Advisory Committee on Nutrition, 2015; World Health Organisation, 2015) have resulted in the recommendation that intakes of ‘free sugars should not exceed 5% of total dietary energy’ (Scientific Advisory Committee on Nutrition, 2015: page 196). Effects of the differing types of recommendation will be assessed on free sugar intakes, dietary profiles and sweet taste outcomes in adults consuming >5% total energy intake (TEI) from free sugars.
Methods and design
This trial is a randomised controlled parallel-group trial with three intervention arms and one control arm. The primary purpose is to assess the effects of three different types of dietary recommendations for reducing free sugars, on free sugar intakes over 12 weeks, in individuals consuming >5% TEI from free sugars. Secondary aims will investigate how these different recommendations affect secondary outcomes, outcomes in subsets of the trial population, and will identify potential barriers and facilitators to dietary change.
Ethical considerations
The trial received ethical approval from the Research Ethics Committee of Bournemouth University, UK (ref: 30612) on 28.04.20 (with amendments approved on 29.03.21) and was registered on Clinicaltrials.gov (ID: NCT04816955) on 24.03.21. The trial will be run in accordance with the Declaration of Helsinki (1983), the Ethical Guidelines of the British Psychological Society, and Bournemouth University's Research Ethics Code of Practice. All participants will provide written informed consent prior to participation.
Participants
Recruitment and eligibility
We aim to recruit 240 male and female participants from the general community residing in the South of England. Individuals will be eligible for trial inclusion if they are: aged 18–65 years, consuming >5%TEI from free sugars, able to provide informed consent, and complete all trial measures, including those to be completed via mobile phone or computer. Exclusion criteria are pregnancy or breastfeeding; underweight (BMI<18.5 kg/m2); pre-existing medical conditions affecting swallowing ability, taste and/or smell perception; currently, or within the previous three months of starting the trial, following a specific dietary programme; currently, or within the previous three months, smoking; pre-existing clinical conditions, including allergies, diabetes mellitus, eating disorders, Crohn's disease, leading to the use of external nutritional advice and dietary restrictions.
Sample size
Sample size equations are powered at 80% for an alpha of 0.05 to test for a 2% change in percentage free sugar intakes from baseline to trial end (Whitley and Ball, 2002). Due to a lack of literature on the use of dietary recommendations for reducing free sugar intakes at trial conception, sample size equations were based on the reported effects of a trial on the use of dietary recommendations for reducing saturated fat content (Smith et al., 2015). The highest standard deviation calculated from these data (SD = 2.4) was used to calculate a required group size of N = 46 per trial arm (Whitley and Ball, 2002). Allowing for a 20% drop-out rate and unequal recruitment across trial arms, we aim to recruit 240 people in total.
Randomisation
Participants will be allocated into one of four trial arms following baseline assessments, using stratified randomisation (Suresh, 2011), based on gender, BMI, and free sugar intakes (%TEI) at baseline, as variables that may affect our outcomes. Randomisation will be undertaken by a researcher with no direct contact with participants using a random number generator, before the trial start. Group allocations will be concealed using opaque sealed envelopes.
Intervention/control
There will be four trial arms: three arms delivering different types of recommendations for reducing free sugars and one control arm. All dietary recommendations have been gained from current publicly available information (NHS, 2018, 2019; Diabetes UK, 2021). All intervention / control instructions will be delivered to participants in a sealed envelope, alongside a user guide for Nutritics Libro App (Nutritics, 2019), and an instruction ‘to keep an accurate diet diary using the Nutritics software’. All groups will receive the same instructions regarding the diet diaries, but this instruction has been carefully worded, such that for participants in the control group, this instruction can be construed as a dietary recommendation. On receipt of their sealed envelope, all participants are also informed that we consider a dietary recommendation to be anything from simply recording your diet to the provision of specific instructions.
Group N: Nutrient-based Guidelines
The nutrient-based recommendations begin with the instruction: ‘Your dietary recommendation is to reduce your intake of free sugars to less than 5% of your total energy intake’. This sentence will be followed by one page of nutrient-based information, including the different names for sugars and how to identify the sugar content of foods, e.g., ‘high in sugar – 22.5 g or more of total sugar per 100g’. Current recommendations from PHE (NHS 2018) have been amended to provide only the nutrient-based information that relates to sugars, through the deletion of non-nutrient based information. Diet diary instructions then follow.
Group NF: Nutrient- and Food-based Guidelines
These recommendations begin with the instruction: ‘Your dietary recommendation is to reduce your intake of free sugars to less than 5% of your total energy intake. To aid with this, reduce your intake of foods high in free sugar’. Participants will then be provided with the same nutrient-based information as Group N plus four additional pages detailing which foods are commonly high in free sugars and examples of how much sugar is included, e.g. ‘A bowl of sugary breakfast cereal could contribute 70g of sugar (up to 22 sugar cubes) to your diet over a week’. Current recommendations from PHE (NHS 2018, 2019) have been amended to provide the nutrient- and food-based information that relates to sugars, through the deletion of nutrient and food-based information that relates to other nutrients. Diet diary instructions then follow.
Group NFS: Nutrient-, Food- and Food-Substitution-based Guidelines
These recommendations begin with the instruction: ‘Your dietary recommendation is to reduce your intake of free sugars to less than 5% of your total energy intake. To aid with this, reduce your intake of foods high in free sugar and replace these with low sugar versions’. Participants will be provided with the same nutrient- and food-based information as Group NF, plus five additional pages on low-calorie sweeteners (LCS) and low sugar versions of foods. This information details what LCS are, where they are found, their different uses, and suggests low sugar substitutions for high sugar products, e.g., ‘biscuits – swap for oatcakes, oat biscuits, or unsalted rice cakes’. This information has been gained from Diabetes UK (Diabetes UK, 2021) and includes only details on LCS with removal of all references to diabetes. Diet diary instructions then follow.
Control group
Participants in the control group will be given no dietary recommendations. These participants will receive only the instructions to keep an accurate diet diary.
Intervention delivery
Participants will be provided with their intervention envelopes following baseline measures. Participants will not be permitted to ask questions on their recommendations. This mirrors the current scenario for the UK public where dietary recommendations are provided, e.g., via government slogans and TV advertisements, without the opportunity to ask questions. An inability to ask questions will also ensure that the same information is provided to all participants, maintaining intervention fidelity. Activities undertaken to adhere to the recommendations by participants, e.g., information gathering, LCS use, will be assessed among the outcomes of the study.
Blinding
All envelopes containing the intervention and control recommendations will be identical, sealed and coded by the researcher undertaking the randomisation (KMA). All envelopes will be packaged to include the same number of pages regardless of intervention / control (through the addition of blank pages) to maintain researcher blinding. The researcher in direct contact with participants (LRB) will be kept blinded to treatment allocation throughout data collection. It is impossible to blind participants to group allocation, however, participants will be blinded to the true purpose of the trial and to all interventions other than their own. To further disguise the purpose of the trial, all participants will complete several questionnaires as part of the trial, in addition to those focusing on sugar.
Trial outcomes
Our primary outcomes are percentage changes in free sugar intakes and adherence to the dietary recommendations over a 12-week period. Secondary outcomes are: daily energy intake, dietary composition, anthropometry, sweet food perceptions and preferences, sweet food choice, attitudes to sweet foods, attitudes towards eating behaviour, motives for food choice, knowledge and lifestyle variables, quality of life and adverse events. Secondary qualitative outcomes are: barriers and facilitators towards intervention adherence and success / failure in achieving the recommendations. Sweet liker status, 6-n-Propylthiouracil (PROP) status, and demographic variables will also be assessed to aid in the interpretation of all outcomes.
Free sugar intakes
Change in free sugar intakes, as %TEI, will be assessed using diet diaries, undertaken using the Nutritics software platform and ‘Libro’ App (Nutritics, 2019). Usual dietary intakes will be calculated from three days of diet diaries (1 weekend day and 2 weekdays) (National Cancer Institute, 2021) at baseline and 12 weeks.
Alongside changes in free sugar intakes, participants will also be recorded as ‘successful’ in achieving the dietary recommendation, where free sugar intake is ≤5%TEI, or ‘not successful’ where free sugar intake remains >5%TEI.
Adherence
Adherence to the dietary recommendations will also be assessed by diet diaries. Eighteen daily diet diaries, in addition to the three diaries at baseline and at 12 weeks, will be undertaken over the 12-week intervention, as identified in
Scheduled diet ndiary recording to measure compliance.
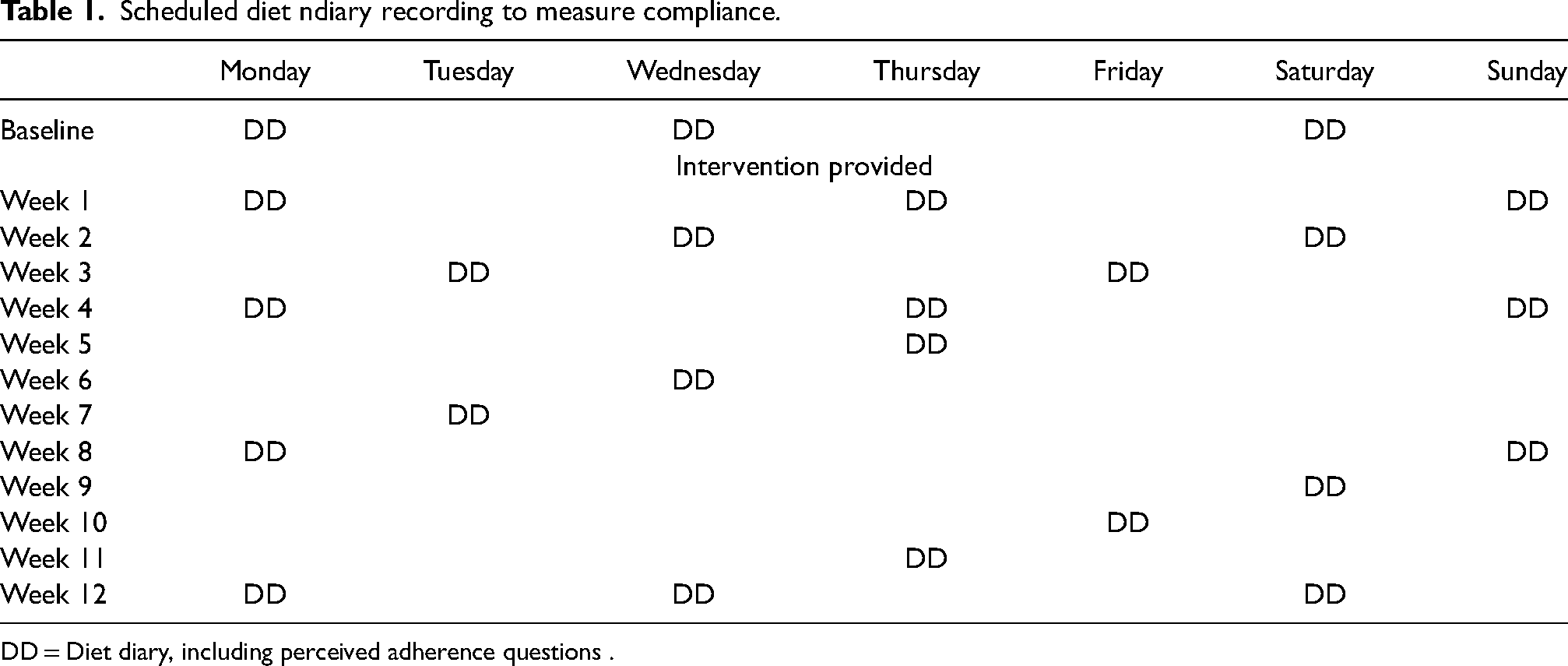
DD = Diet diary, including perceived adherence questions .
Adherence will be assessed from every diary, then classified five times across the 12-week intervention. During the first two weeks of the trial, adherence will be based on participant ability to reduce free sugars by ≥2%TEI from baseline or not, classified as ‘adherent’ or ‘non-adherent’ respectively. Participants will then be classified at weeks 4, 8 and 12 using data on their ability to reduce free sugars by ≥2%TEI from previous assessment (baseline for week 4), and their answers to the following adherence question: ‘Are you currently following the dietary recommendations you were given?’ Reductions of free sugar intakes ≥2%TEI and an answer ‘YES’ will result in a classification of ‘active adherent’, reductions of free sugar intakes ≥2%TEI and an answer ‘NO’ will result in a classification of ‘passive adherent’, reductions of free sugar intakes <2%TEI and an answer ‘NO’ will result in a classification of ‘active non-adherent’, and reductions of free sugar intakes <2%TEI and an answer ‘YES’ will result in a classification of ‘passive non-adherent’.
Secondary outcomes
Details for assessing all secondary outcomes, including the qualitative outcomes are given in the Supplementary Materials.
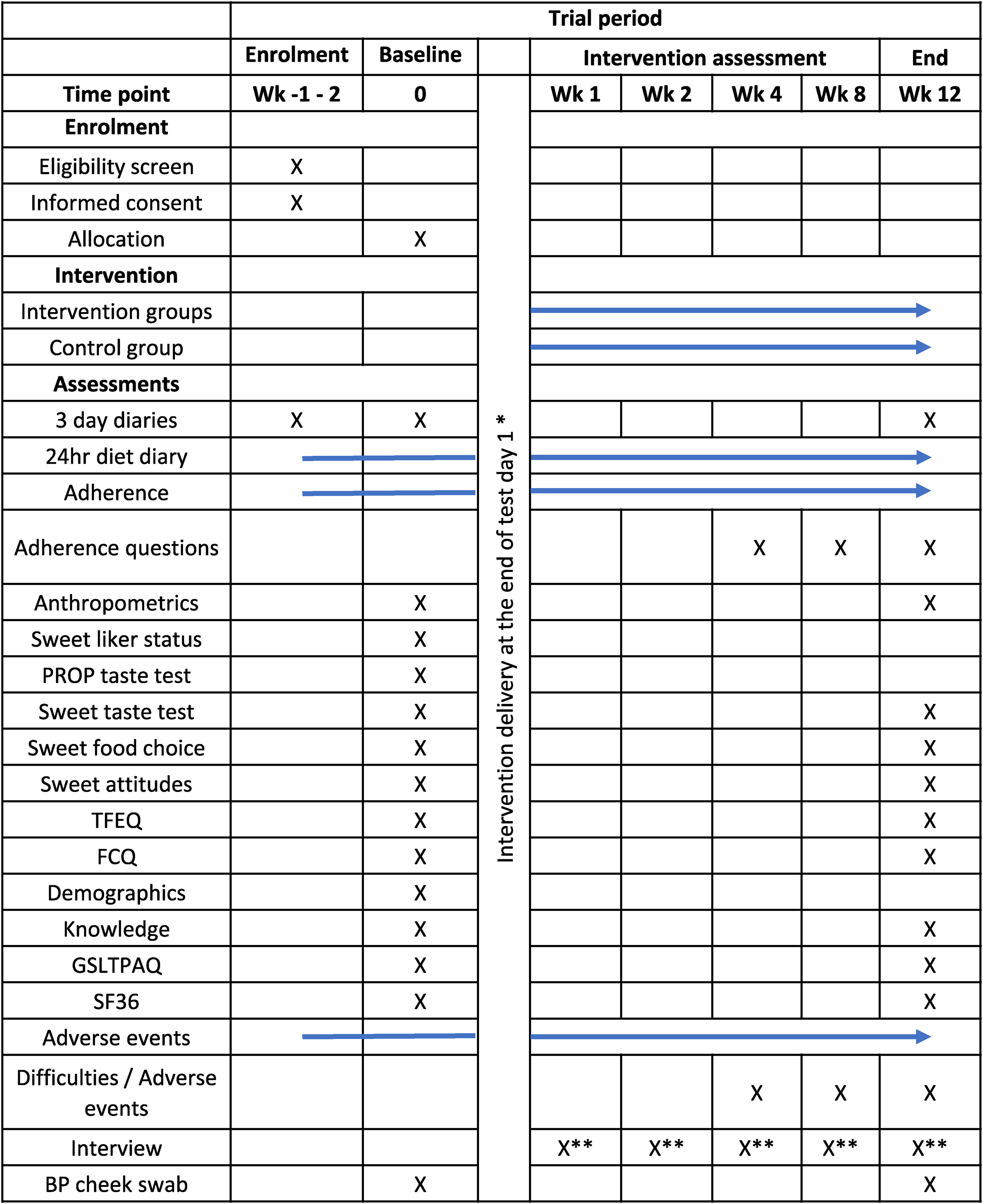
Outcome assessment schedule
An overview of the outcome assessment schedule is given in Table 2.
Schedule of enrolment, interventions and assessments
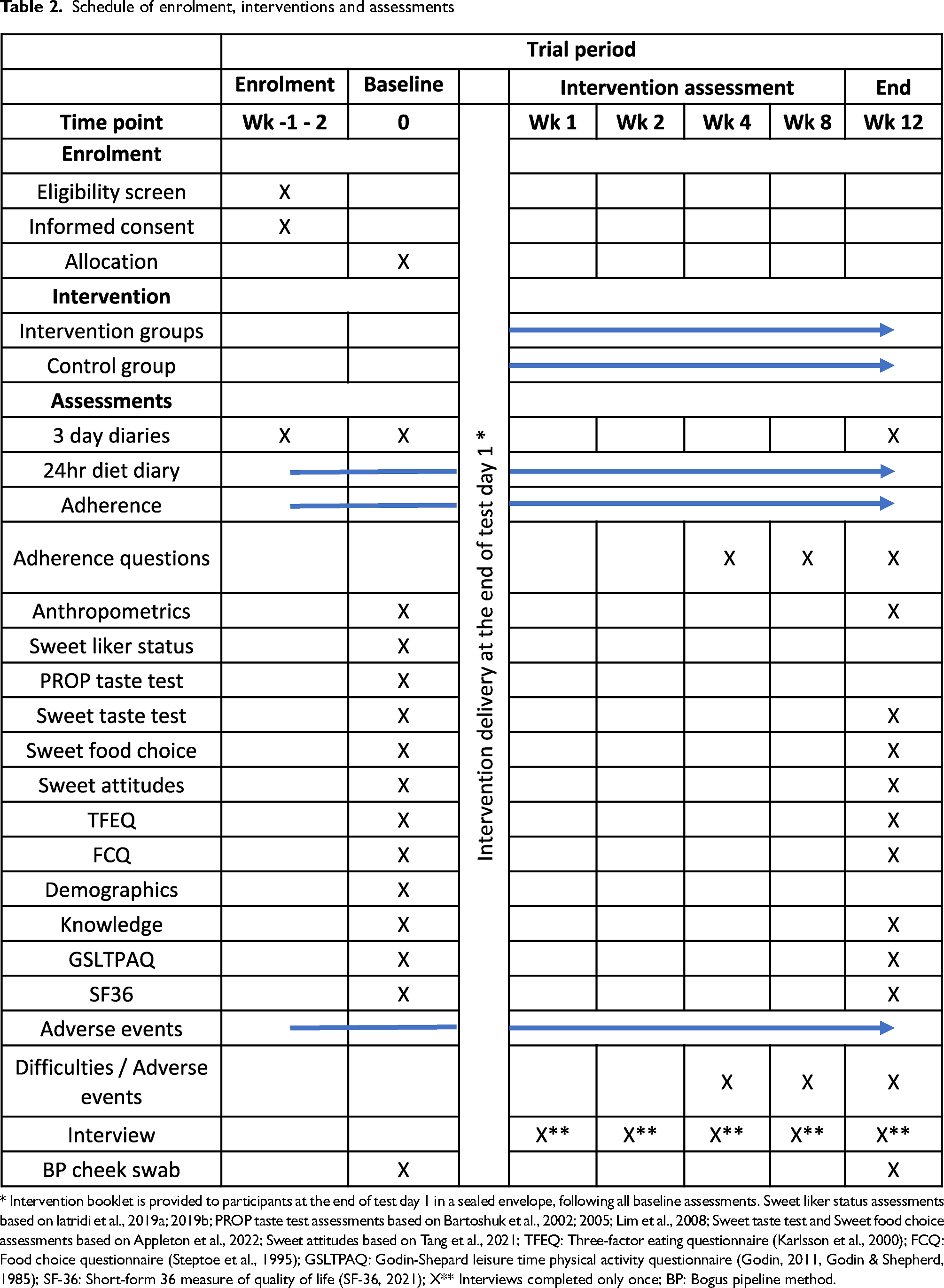
* Intervention booklet is provided to participants at the end of test day 1 in a sealed envelope, following all baseline assessments. Sweet liker status assessments based on Iatridi et al., 2019a; 2019b; PROP taste test assessments based on Bartoshuk et al., 2002; 2005; Lim et al., 2008; Sweet taste test and Sweet food choice assessments based on Appleton et al., 2022; Sweet attitudes based on Tang et al., 2021; TFEQ: Three-factor eating questionnaire (Karlsson et al., 2000); FCQ: Food choice questionnaire (Steptoe et al., 1995); GSLTPAQ: Godin-Shepard leisure time physical activity questionnaire (Godin, 2011, Godin & Shepherd, 1985); SF-36: Short-form 36 measure of quality of life (SF-36, 2021); X** Interviews completed only once; BP: Bogus pipeline method.
Free sugar intakes and adherence will be assessed throughout the intervention period as given in Table 1. All other outcomes will be assessed at baseline and at trial end, with the exception of the following: sweet liker status, PROP taste sensitivity and demographic characteristics will be measured only at baseline; questions on adherence and difficulties will be asked at weeks 4, 8 and 12.
All participants will undertake all measures, in the same manner, regardless of intervention arm. Dietary assessments, questions on adherence and difficulties will be undertaken via the Nutritics software. Self-report questionnaires will be administered online via Qualtrics. Measures associated with taste status and appetite will be presented to participants using a paper format to be completed during a test session.
Compliance with trial measures will be enhanced using a bogus pipeline method (Hugh, 2013; Muhlheim et al., 1998; Reid et al., 2014); participants will be asked to provide a saliva sample at baseline and at trial end, for the supposed purpose of examining salivary enzymes that may vary with dietary change. In reality, samples will be discarded. Only at the end of the trial will participants be informed that their samples have not been analysed.
Procedure
Trial setting
The trial is based in the United Kingdom and run from Bournemouth University. Recruitment started in April 2021 with testing commencing in May 2021. The trial will run for a total period of 18 months, over the year to ensure against seasonal effects, but we anticipate increased recruitment at certain times of the year (January - February and May – July). The trial will not continue over the Christmas period to avoid potential effects as a result of unusual dietary intakes over this period.
Recruitment and enrolment
Potential participants will be recruited via: personal contacts; University contacts and outlets, including a participant pool; contacts with local groups, e.g. church groups, adult education groups; social media advertising and flyers in local public buildings, e.g. libraries; advertising in local news outlets; flyers at local eating establishments and delivered in residential areas. The study will be marketed to participants as ‘An investigation of the impacts of different dietary recommendations on diet’.
All potential participants will be asked to complete the informed consent, eligibility form and a 3-day diet diary before being invited to participate. Eligible participants will then be scheduled for a baseline assessment, and randomised following completion of all baseline measures. The process from recruitment to enrolment is shown in Figure 1. The 3-day diet diary for eligibility will also provide an opportunity to train participants, to allow participants to gauge the commitment required for the study, and to ensure participants are competent in the diet diary data collection methods prior to their completion of baseline measures. Participants will not be recruited into the trial until they are comfortable with the commitment and diet diary data collection methods.
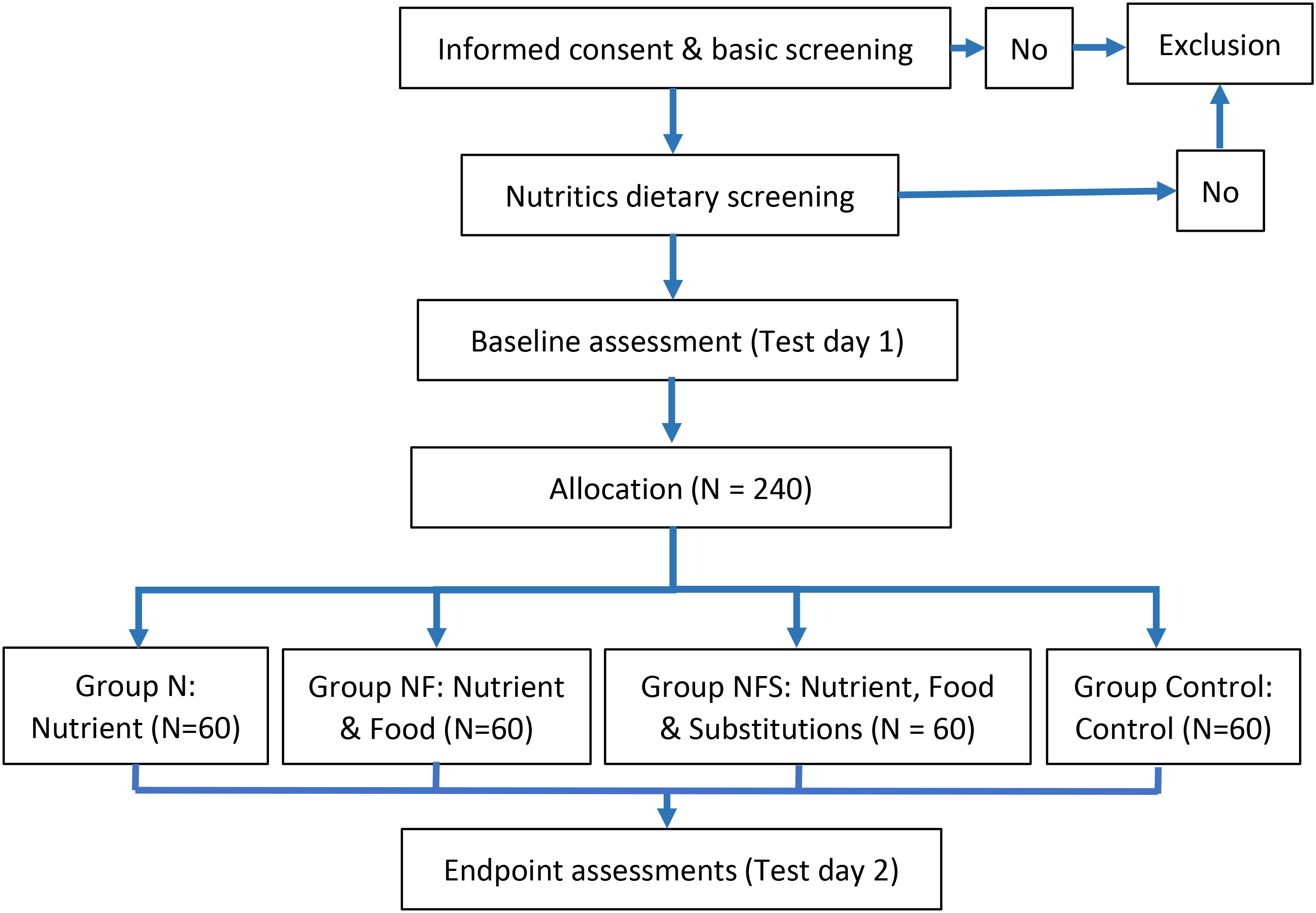
Participant flow diagram.
Participant testing
Baseline and end assessments will be conducted in a single session for all participants. Sessions will last approximately 30–60 min, and will be conducted at Bournemouth University where possible, or in the participant's home via Zoom. ‘At-home’ test sessions will be used if participants are unable or unwilling to come to the University and were intended primarily to allow the trial to continue during the COVID-19 pandemic when National lockdown measures and precautions were recommended in the UK (March 2020 - July 2021). These home test sessions may also open the trial to participants who would otherwise be unable to take part, primarily due to location, enhancing study inclusivity. Participants will be tested in the same location at both baseline and trial end, as far as possible.
All participants will complete the same measures regardless of their completion of test sessions at the University or ‘at-home’, with a few exceptions: Participants who are tested ‘at-home’ will not undertake the sweet taste perception and preference tests, the sweet food choice test, nor the solution-based measures of sweet liker status. Participants who are tested ‘at-home’ will also complete their own anthropometric measurements while the trial researcher observes via Zoom. Comparability across measurements will be facilitated by involvement of the same researcher whether at the University or ‘at-home’, and will be investigated once the trial is complete.
All sessions will commence before 11am to allow individuals to undertake the measures in a fasted state and will begin at the same time at baseline and trial end. The day before testing participants will also be asked to consume no alcohol, to consume nothing after 10pm, and to undertake no heavy exercise. Measures will be undertaken in the same order during each test session, as follows, or simply omitted: anthropometry; saliva sample; sweet liker status; PROP taste test; sweet taste test; and sweet food choice test. All participants' questionnaires will be completed and checked for completion before the test session. Incomplete questionnaires will be completed on the test day. Missing diet diaries throughout the trial will result in an automatic reminder. This reminder will ask participants to complete the diaries, but make no reference to dietary recommendations.
Withdrawal and debriefing
Participants will be considered as having withdrawn from the trial if they either request to withdraw or do not complete the final diet diaries. If individuals fail to complete diet diaries during the trial, they will be sent reminders; data will be noted as ‘missing’ while the participant continues in the trial.
Individuals will be debriefed on exit, or at their original 12-week intervention end time-point if other household members are partaking. During the debrief session, participants will be asked for their understanding of the trial purpose, to investigate the success of the methods to disguise the trial aims, and will then be debriefed on the true purpose of the trial. Following the debrief session, participants will be offered a consultation on their diet, by a Registered Associate Nutritionist, as a thank you for taking part.
Analyses
The data gathered will contribute to three distinct analyses: 1) Analyses of the population as a whole to investigate the effects of the three different dietary recommendations versus control; 2) Analyses of the effects of the dietary recommendations in different population subgroups, and 3) Investigation of the barriers and facilitators to success. Quantitative data will be analysed using SPSS, on an Intention-to-Treat basis, following checks for the assumptions for parametric data. Some checks for accurate diet diary recording based on Schofield equations will also be made. Exclusion of participants with missing data and high levels of likely inaccurate recording will result in the completion of additional per-protocol analyses. Qualitative data will be analysed as detailed below.
Analyses one: Effects of the different dietary recommendations
To test the effects of the different dietary recommendations, a series of multiple regression analyses will be run. A separate analysis will be run for each outcome variable, where the outcome at week 12 will be predicted by trial arm (intervention/control) and outcome variable at baseline. Additional independent variables will also be included in each analysis as possible, to include: demographic variables, total energy intakes, sweet liker status; PROP taster status; and attitudes to sweet foods, eating behaviour and food choice.
Analyses two: Effects in different population subgroups
The above analyses will be repeated in specific population groups, assuming appropriate numbers, based on demographic variables and other variables identified as important in analyses one.
Analyses three: Barriers and facilitators toward dietary change
Qualitative data will be transcribed and analysed using thematic analysis following Braun and Clarke's 6-stage methodology (Braun and Clarke, 2006). These analyses will be aided by the use of NVIVO software, and reported using the Consolidated Criteria for Reporting Qualitative Research (COREQ) (Tong et al., 2007). Themes will be gained from the population as a whole, at different time points, and interpreted in combination with the data on free sugar intakes and adherence. Comparisons will be made between those who are successful and not successful at changing their free sugar intakes, and those who are adherent and not adherent to the recommendations.
Discussion
Population estimates suggest that the majority of individuals do not achieve multiple nutrient or food-based dietary goals (Leme et al., 2021; Yau et al., 2019). Despite national public health programmes, individuals continue to overconsume nutrients, such as free sugars (Public Health England, 2020) with the health and budgetary benefits from dietary change (Public Health England, 2015) unlikely to come to fruition. This study seeks to extend the limited literature on the effectiveness of nutrient-, food-, and food-substitution-based recommendations using current PHE free sugar reducing advice. It will offer a new perspective on the effects of different dietary recommendations to enact behaviour change. The research has international relevance given widespread links between diet and disease (Roth et al., 2020) and low adherence to national dietary guidelines (Leme et al., 2021; Yau et al., 2019).
Supplemental Material
sj-docx-1-nah-10.1177_02601060221111234 - Supplemental material for Protocol: The effects of nutrient- vs food- vs food-substitution-based dietary recommendations for reducing free sugar intakes, on free sugar intakes, dietary profiles and sweet taste outcomes: A randomised controlled trial
Supplemental material, sj-docx-1-nah-10.1177_02601060221111234 for Protocol: The effects of nutrient- vs food- vs food-substitution-based dietary recommendations for reducing free sugar intakes, on free sugar intakes, dietary profiles and sweet taste outcomes: A randomised controlled trial by Lucy R. Boxall, Emily Arden-Close, Janet James and Katherine M. Appleton in Nutrition and Health
Footnotes
Acknowledgements
Grateful thanks are extended to the funders of this work.
Funding
This trial will be funded by Bournemouth University, UK, and the International Sweeteners Association (ISA), BE. The funders have offered limited comment on the trial design and materials but have had no role in the finalisation of the trial and will take no part in running the trial or interpreting the trial findings. The funders have had and will have no direct contact with the PhD student.
Availability of data and materials
Materials are available from the corresponding author on request.
Authors contributions
KA conceived the trial idea; LB and KA designed the trial with input from EAC and JJ. LB wrote the first draft of the trial protocol. All authors critically reviewed and revised the final version.
Declaration of interests
This trial is part of a PhD studentship, funded by Bournemouth University, UK, and The International Sweeteners Association (ISA), BE.
Ethical approval
Ethics approval was gained from the Research Ethics Committee of Bournemouth University (Ethics ID: 30612). The standard protocol item recommendations for intervention trials (SPIRIT) guidelines (Chan et al., 2013) were used in the writing of this protocol and production of study documents.
Consent for publication
All authors approve the final manuscript. Consent for publication has been obtained from all necessary parties.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This trial is part of a PhD studentship, funded by Bournemouth University, UK, and The International Sweeteners Association (ISA), BE.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Bournemouth University, International Sweeteners Association (ISA), BE, (grant number N/A).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.