Abstract
Background
The benefits of a healthy lifestyle in reducing risk of cancer and chronic disease are well-documented. Many individuals who have had head and neck cancer (HNC) report complex social situations with a history of poor dietary habits, smoking and alcohol abuse. Survivorship can be a strong motivator to make positive lifestyle changes, reducing risk of cancer recurrence and ill-health. Research investigating whether HNC survivors adopt healthy lifestyle recommendations is lacking.
Aim
To explore the health-related practices of post-treatment HNC patients, seeking to identify barriers and motivators to following recommended health guidelines.
Methods
Tape-recorded interviews were conducted with 20 HNC survivors, and comparisons made to Department of Health recommendations.
Results
80% of participants made lifestyle changes following HNC treatment. The most prevalent changes were to diet and alcohol intake. Key motivators were reducing cancer risk and ill-health; barriers included lack of motivation, support and misinformation. Treatment side-effects presented both motivators and barriers. There was widespread recognition of the “5 a day” message, and harm caused by smoking. Other public health recommendations were less well-known; 98% were unaware of current alcohol guidelines, physical activity was overestimated, and only one participant took vitamin D.
Conclusion
In this study HNC survivors were highly motivated to make healthy lifestyle changes. Further work is required to increase awareness of Government guidelines, as health messages are not always reaching the public or are misinterpreted.
Introduction
The benefits of a healthy lifestyle to help prevent cancer and chronic disease are well-documented (WHO, 2022). Research indicates as many as 71% of deaths globally are caused by lifestyle choices (Tomlinson, 2020) and adopting a healthy lifestyle may prevent up to 40% of cancer cases and 50% of cancer deaths (Song and Giovannucci, 2016; WHO, 2022). Individuals who have had HNC present some of the most significant post-treatment morbidities of any group of patients with cancer (Ninu et al., 2016). Even before diagnosis, many individuals report complex social situations with a history of poor dietary habits, smoking and alcohol abuse (Lees, 1999). The side-effects of treatment further impede proper nutrition (Ninu et al., 2016; Wilson et al., 1991) impacting on rehabilitation and quality of life (QoL). Smoking, drinking excess alcohol, poor diet and lack of exercise have been shown to decrease overall survival in HNC survivors (Duffy et al., 2009; Maino et al., 2019; Freedman et al., 2008; Pavia et al., 2006; Parkin, 2011), whilst the increasing prevalence of H&N cancer from human papilloma virus (HPV) affecting a younger cohort, makes QoL ever more significant (Young et al., 2015). Following treatment, survivors remain at high risk of developing recurrent disease, second primary cancers, and chronic diseases arising from treatment (Baxi et al., 2014; Simpson et al., 2018). The personal lifestyle choices made by HNC survivors may therefore have far-reaching implications on the effective management of co-morbidities, disease progression and cancer recurrence.
Department of Health recommendations are that individuals recovering from HNC follow established guidelines for good health, with a view to improving physical and psychological well-being, reducing risk of recurrence, and improving survival (Department of Health, 2010). Survivorship can be a strong motivator to make positive lifestyle changes, helping survivors feel more self-sufficient and in control of their lives. In essence, this means considering behaviours, such as the options of what food to eat, whether to exercise, smoke, or drink alcohol, maintain a healthy body weight, and taking action to implement strategies to achieve optimum health.
To date most research on HNC survivorship has focused on the supportive needs, concerns and challenges experienced by survivors, using quantitative methodology and data collected via questionnaires, mostly in relation to QoL. Whilst useful, they do not allow for indepth exploration of patients’ practices or attitudes towards following a healthy lifestyle after cancer treatment. Other qualitative studies have interviewed HNC survivors with a view to investigating survival times. To our knowledge, no studies have assessed the proactive changes made by HNC survivors in response to their cancer diagnosis, with a view to reducing recurrence risk and other co-morbidities. Our aim was to ascertain the nature of health-related behavioural changes, if any, adopted by HNC survivors, further identifying barriers and motivators to achieving health recommendations. Such information may help determine need and direction of resources and services, guide future rehabilitation services, and assist in reducing risk of cancer recurrence and other co-morbidities, thereby enhancing survival outcomes and QoL for HNC survivors.
Methods
Study design and recruitment
Following a pilot study on a service user, a purposive sample of 20 post-treatment HNC patients were recruited to this prospective cross-sectional study, between March and July 2018. HNC is a rare cancer hence recruitment was through volunteer sampling from a regional UK HNC specialist unit. Candidates meeting set criteria were identified from a central database, 143 invitation letters sent, and recruitment made from those agreeing to participate; 29 individuals responded, 3 did not fulfil criteria, 3 declined participation, and 2 were declined as research quota was achieved. A target of 20 participants was sought to provide meaningful insight into the study aims, whilst mindful of the time involved conducting and transcribing tape-recorded interviews. The qualitative approach enabled participants to expand upon data specific to the study aims, providing insight into barriers and enablers to healthy living. Participants eligible for inclusion were
Data collection
Following informed consent, interviews were conducted at the patients’ homes, or the cancer unit, by either a HNC specialist Dietitian or Speech & Language Therapist. To reduce bias, individuals known to a particular Allied Health Professional (AHP) were interviewed by peers. Interviews were tape-recorded, using a Phillips digital pocket memo recorder LFH9620. An interview guide comprising questionnaire was employed to maintain focus, record demographic information and quantitative data central to the study aim (Appendix 1). Dietary intake was assessed via study specific questions integrated into the questionnaire. Closed questions were used to ascertain frequency and quantity across behavioural categories, however participants were encouraged to talk freely throughout the interview, sharing thoughts and insights regarding their lifestyle choices, barriers and motivators to change. For reporting purposes, where participants provided a range in response to a question, eg 10-20, the average amount was calculated; if two consecutive answers were given, eg, 2 or 3, the higher value was used. Interviews lasted up to one hour, were fully transcribed word-for-word by an independent administrator, and verified for accuracy by participants prior to analysis. No compensation or remuneration was provided. Participants received a health resource pack as a gesture of gratitude on interview completion. A pilot interview was conducted on a service user to identify flaws or other weaknesses within the interview design, and minor adjustments made prior to study commencement.
Data analysis
Content analysis was employed to interpret qualitative data from transcriptions, and responses collated and categorised under pre-determined sub-headings of Smoking, Alcohol, Diet, Weight, Exercise and Activity. Each transcription was read and re-read, coded, and common themes and patterns identified for each behavioural category, with further review by a second author. Data obtained from closed questions in the interview guide were quantified, and descriptive statistics conducted using Microsoft Excel SPSS V.27. Data sets from transcriptions and interview guides were analysed separately, then merged for each behavioural category. Results were compared to Department of Health recommendations.
Results
Twenty participants completed the study. Participant characteristics are outlined in Table 1.
Demographic characteristics, HNC site, treatment and co-morbidities of subjects.

*Mean age 64yrs (male) and 70yrs (female).
** Retirement age at time of study was 65yrs (male), 63yrs (female).
***Median time post-treatment of 4 years [range 2-8 years].
Findings
Lifestyle changes
“it just opened my eyes, gave me a completely different outlook on life… because you never know now do you?”[P5]
Sixteen (80%) participants made lifestyle changes following HNC treatment. Those who did not reported healthy lifestyles prior to diagnosis. Participants obtained health information from a range of sources, including newspapers, TV documentaries, the internet, social media, friends and family, as well as health professionals. Figure 1 shows the nature and prevalence of lifestyle modifications made. Key motivators for change were reducing cancer risk and ill-health. Chronic treatment side-effects (dysphagia, dysgeusia, fatigue, odynophagia, xerostomia) presented barriers to change for 95% of participants across all behaviour categories, and facilitated positive change for 50% of individuals, “The reason I don’t drink is not because I don’t want to, but that it’s like paint-stripper going down my throat!”[P16]. Table 2 displays participants’ reported barriers and motivators to behavioural change.
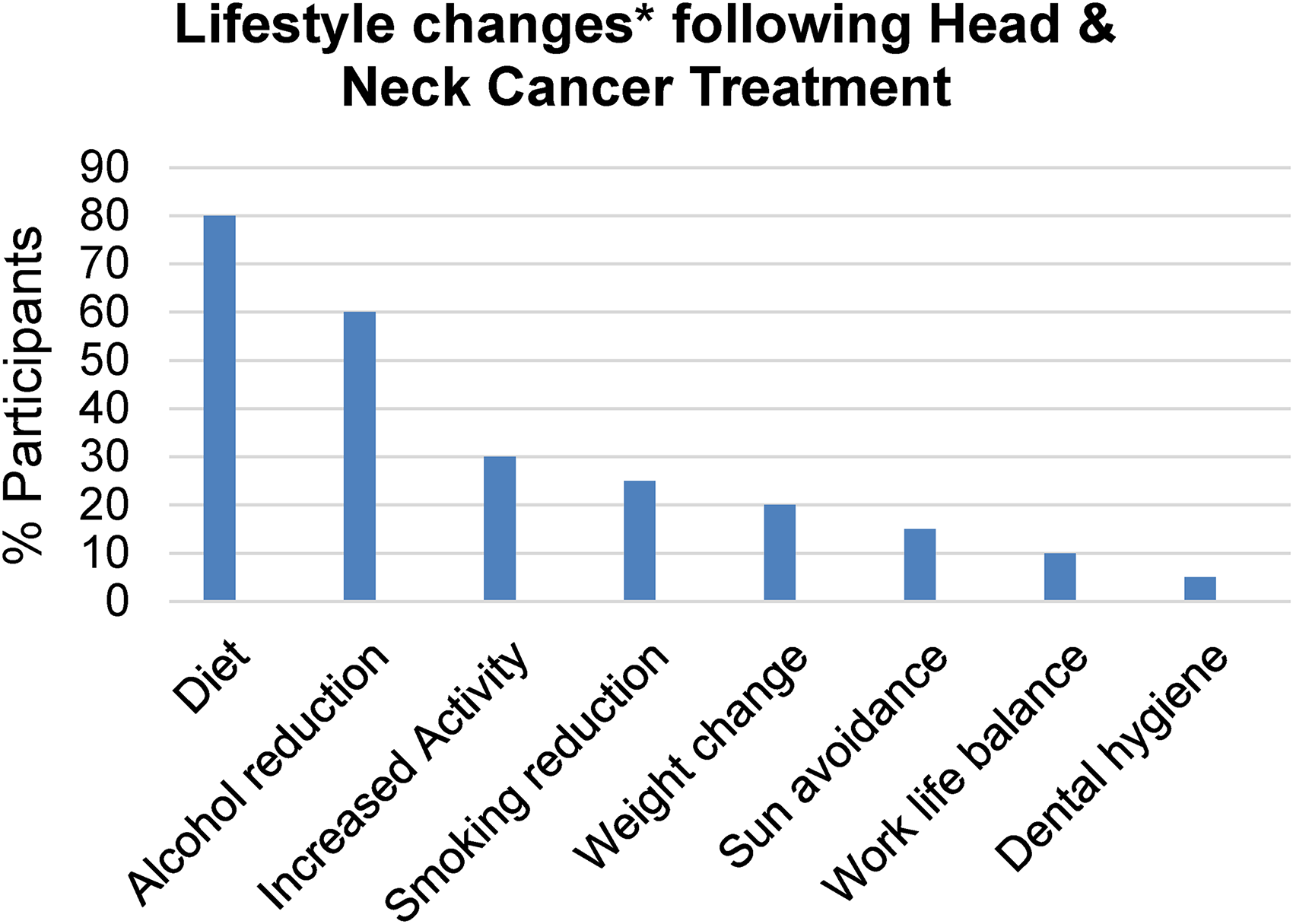
Lifestyle modifications following HNC treatment. *> 1 change made by participants.
Barriers and motivators to behavioural change after HNC.
“There could be more help afterwards - I felt quite isolated really”[P13]
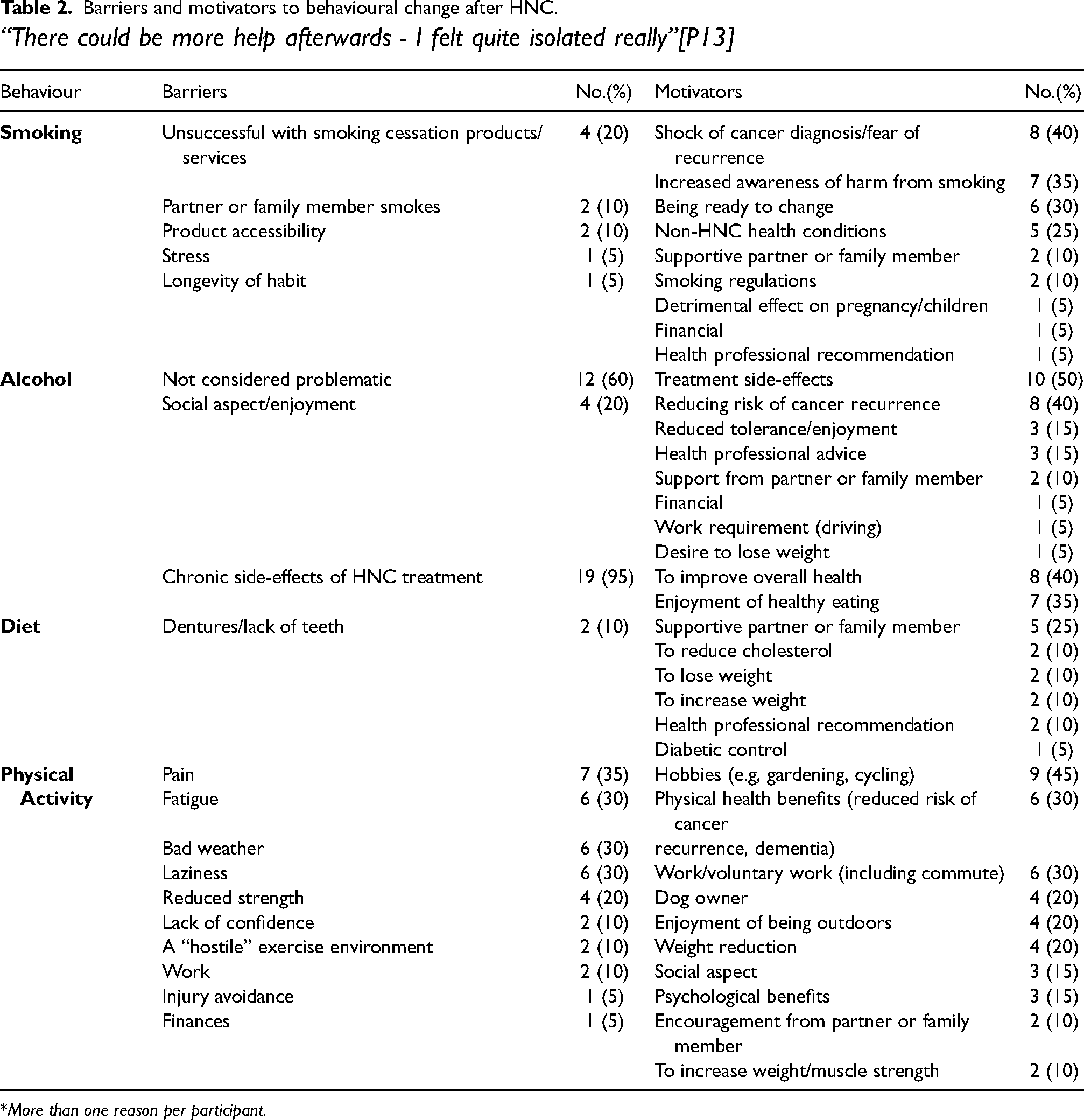
*More than one reason per participant.
Smoking
“If I’d known what smoking was doing to me when I was younger I would never have done it!”[P14].
The main catalyst for smoking cessation was receiving a cancer diagnosis, “It was such a shock to the system…I thought ‘right that’s it!’”[P6]. Changing public attitudes towards smoking influenced participants’ habits,”Everybody smoked! You had cigarettes around just like sweets or biscuits”[P14]; “I’m horrified that I did it now”[P4]. Recurrent themes from ex-smokers were being ready to change and will-power. The high cost of cigarettes and legislation banning smoking in public places (Health Act, 2007), encouraged participants to quit, “We weren’t allowed to smoke at work, so I wouldn’t go outside with all the others”[P15]. Seventy-five percent of participants could name
Alcohol
“I’m not going to resent things which I enjoy!”[P11]
Figure 2 displays participants’ reported alcohol units before and after HNC treatment. Mean(sd) alcohol units consumed were 18.2(14.9) and 8.2(7.6) per week respectively
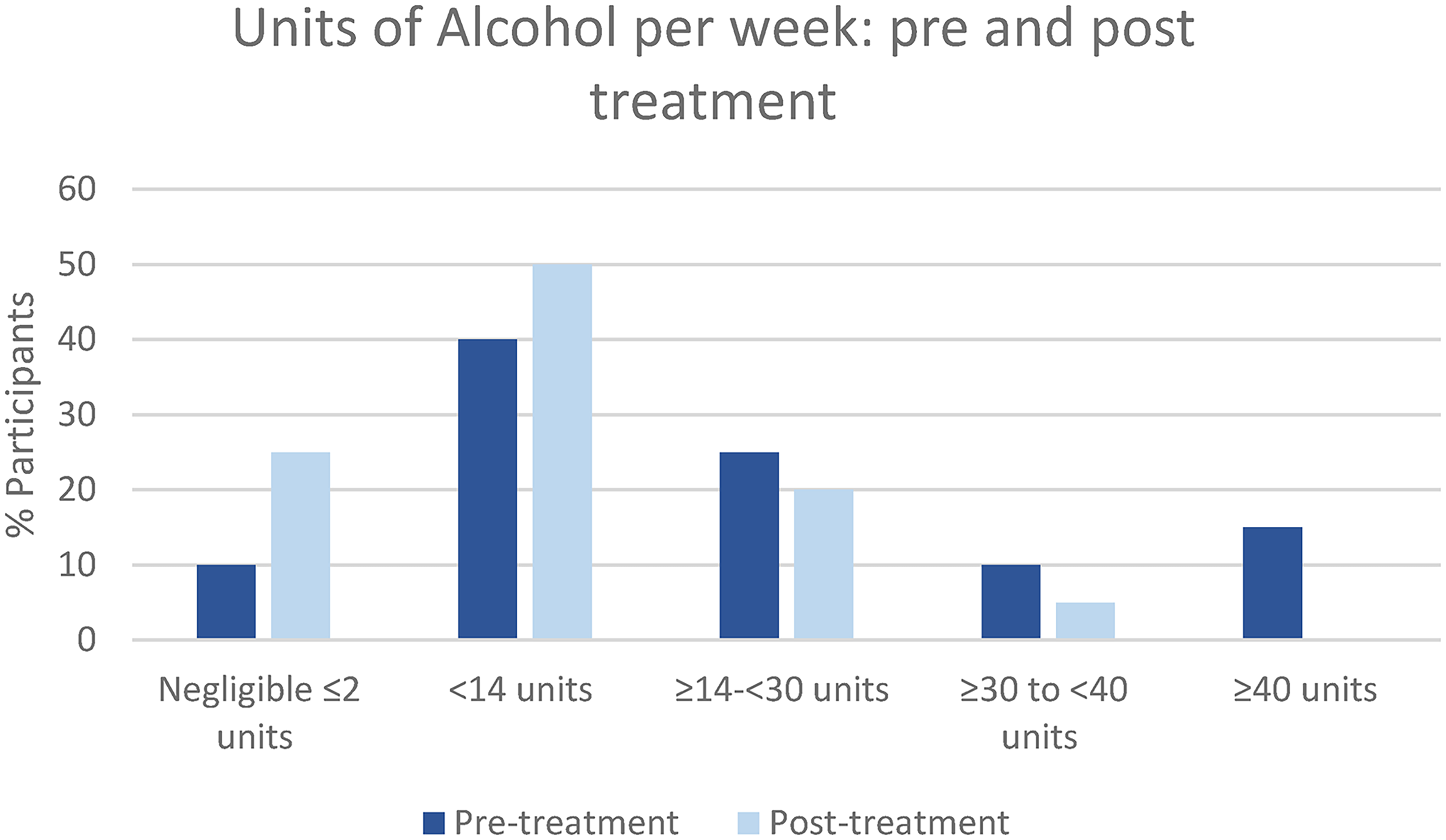
Participants’ alcohol consumption pre and post HNC treatment.
Following treatment, 60% of participants reduced alcohol consumption and 15% abstained from alcohol, resulting in a 50% reduction in alcohol units per week. Treatment side-effects influenced drinking behaviour, “…it burnt my throat like acid, put me right off drinking!”[P12], particularly in the early post-treatment period, “I couldn’t face a drink for about 6 to 9 months”[P3]. Despite the reduction in alcohol consumption, 25% of participants continued exceeding Government recommendations. Nineteen (95%) participants did not know Department of Health guidelines for alcohol, whilst two (10%) cited old recommendations. Uncertainty surrounded what constituted one unit of alcohol, for example, participants considering a pint or a can of beer to be one unit of alcohol regardless of alcohol by volume (ABV), or querying how many units a bottle of wine or spirits contained, “I measure things out but I don’t know the units”[P9]. Some continued consuming significant amounts of alcohol regardless of negative health risks, “Would I rather live another 6 months but not have alcohol at all for that period? The answer is no!”[P3]. Research taken out of context, media reports exaggerating the benefits of alcohol, and anecdotal stories were alluded to in support of continuing alcohol consumption, “The last thing I remember is John Major saying that a couple of pints a day was good for you!”[P7]. Those exceeding Department of Health guidelines after treatment did not perceive their alcohol intake as harmful, “We’re not heavy drinkers”[P21]; “I don’t think I have an alcohol problem”[P18]. Few participants could name any alcohol support resource, with just one service cited by those who could (Alcoholics Anonymous).
Diet
“…I do feel better for it”[P21]
Sixteen (80%) participants made dietary changes following treatment. Eight (40%) effected change to benefit health, whilst 11(55%) reported enforced dietary changes, due to treatment side-effects affecting tolerance to certain foods or beverages, making it challenging to follow health recommendations,
Range and rationale for OTC supplements* taken by participants.
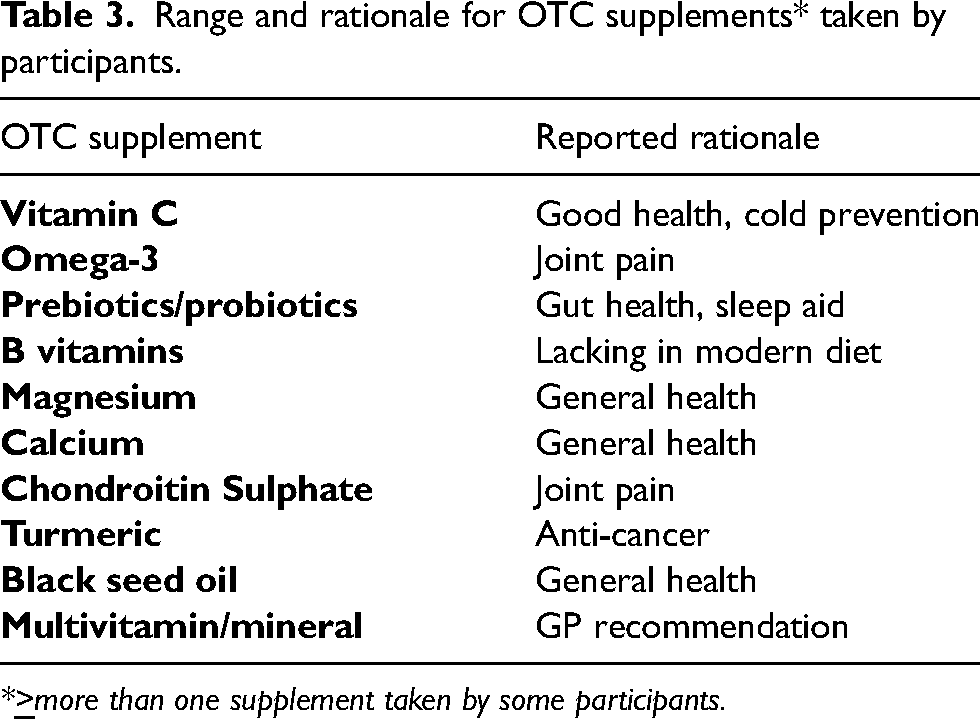
*
Weight
“I lost 19kgs – that I remember!”[P11]
Following treatment, 13(65%) participants had a Body Mass Index (BMI) within the healthy range, 10(50%) men and 3(15%) women. Four (20%) participants remained obese and 1(5%) became underweight. Figure 3 displays participants’ BMI before and after HNC treatment. The mean (sd) BMI pre and post treatment is 27.7 (4.5)kg/m2 and 25.3 (4.1)kg/m2 respectively.
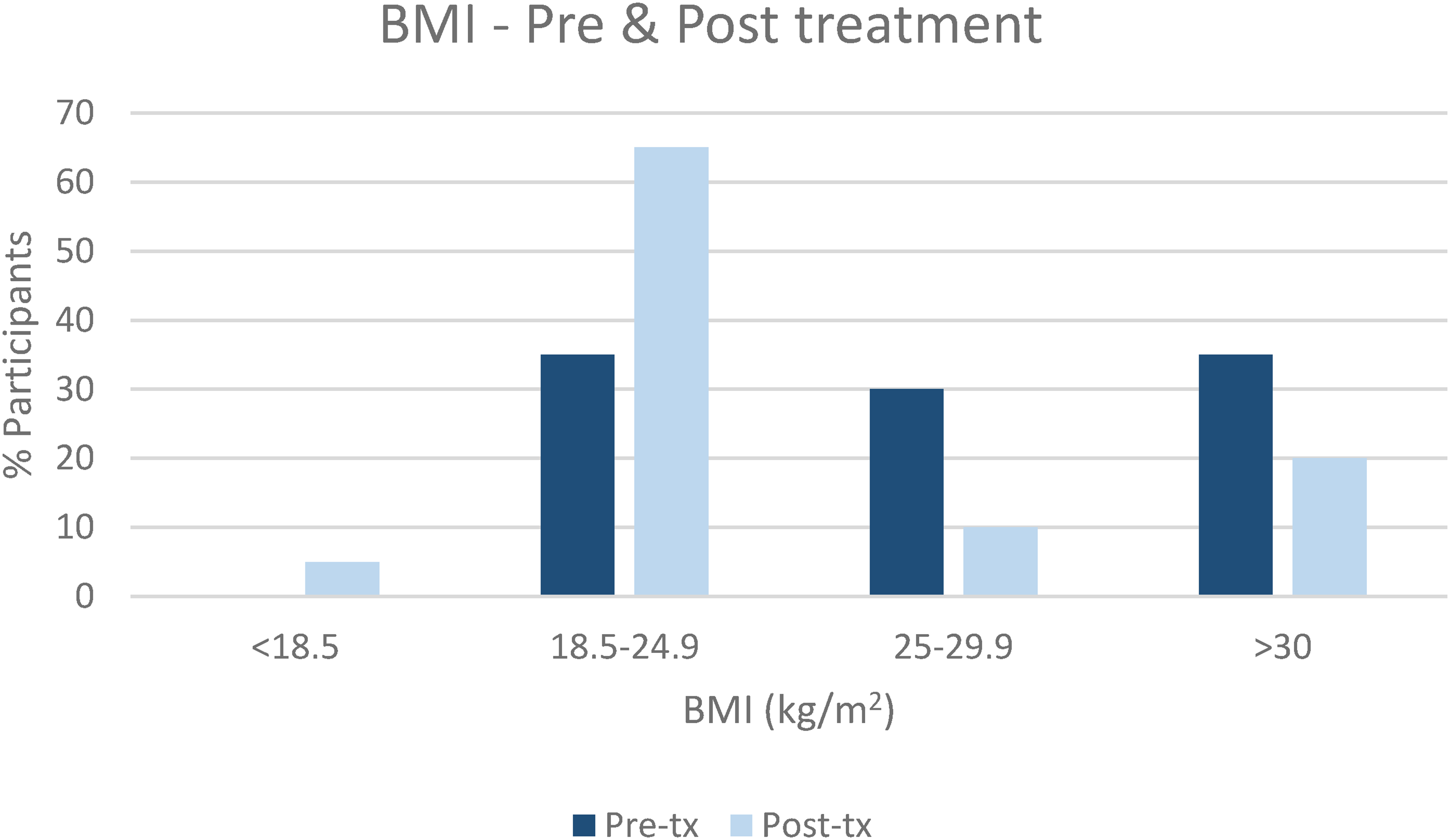
Participants’ BMI pre and post-treatment.
Physical activity
“We got given legs and lungs for a reason didn’t we?”[P6]
Participants rated self-perceived activity levels on a Likert scale, where 1 = sedentary and 5 = very active. Activity levels were recorded as “active” to “very active” by 80%; once compared with Department of Health (2019) guidelines of 150 min of moderate exercise per week, 40% achieved this target. Only one person reported a completely sedentary lifestyle, “I do have problems with my knees!”[P16]. Physical injuries and pain adversely affected activities, resulting in loss of confidence and muscle, “I’ve lost my strength…coming up the stairs from the car park is enough!”[P10]
Discussion
“If it comes back at least I’ve done everything I can“[P5]
In this study HNC survivors were highly motivated to make lifestyle changes to reduce their risk of cancer recurrence, and benefit long-term health. Treatment side-effects presented barriers to following health recommendations across all behavioural categories, yet also facilitated change, particularly for alcohol consumption and dietary intake, with consequential weight reduction for those seeking weight loss. However, effectiveness of behavioural change depends on the quality of information available. Despite an enormous amount of health information, available and accessible in various formats, we found Government health messages were not always reaching participants, were not fully understood, or were being misinterpreted. This appeared mainly due to conflicting or unclear information, presenting a barrier to participants knowing what advice to follow, or how to implement it, resulting in some health practices based on anecdotal, unnecessary, outdated or incorrect advice.
This was particularly evident for alcohol consumption. Research shows that the more an individual drinks the greater the risk of developing cancer, especially HNC (Talamini et al., 2002; Bagnardi et al., 2001) with increased recurrence risk for survivors continuing to drink (Christensen et al., 1999). Despite the proactive reduction in alcohol after treatment, several participants continued exceeding Government recommendations. Of note is that only one participant knew the recommendations for alcohol. One possible explanation for this is that guidelines were altered in recent years (Department of Health, 2016), subsequently older resources remain in circulation, and updated recommendations may not have reached everyone. Nonetheless, the wide range of different container sizing, ABV, non-standardised labelling of alcoholic products and hesitancy surrounding what constitutes a unit of alcohol further contributed to confusion. An array of professional support services exist in the UK to help individuals reduce alcohol consumption to low risk levels, yet few participants could name any alcohol support service. This could reflect participant uncertainty regarding what represents low risk drinking, therefore such services may be perceived irrelevant. However, with escalating rates of post-pandemic alcoholism, increased awareness and accessibility of these services is warranted (Public Health England, 2021). Our findings correspond with previous research highlighting that, although people are known to underreport alcohol consumption by as much as 40-60%, this may be due to uncertainty, lack of knowledge or inaccurate recall rather than an attempt to deceive (Livingston and Callinan, 2015).
Historically HNC has been associated with smoking and drinking, commonly in older adults (Cancer Research, 2017), and evidence demonstrates increased risk of disease progression and mortality for survivors who continue smoking tobacco or chew betel nut (Chaturvedi et al., 2019; Parsons et al., 2010). More recently, an emerging demographic shows a decline in smoking-related HNC and an increase in HNC of the oropharynx due to HPV, particularly in younger adults aged 40-50 years (Young et al., 2015). Studies show an improved prognosis for HPV positive over HPV negative HNC individuals, making QoL increasingly important (Fakhry et al., 2008). Data was incomplete in our study, since routine recording of HPV status only commenced at the Cancer Unit in 2015, however, where known, the ratio of HPV positive to HPV negative participants was threefold greater. Participants were aware of the risks to health from smoking, nonetheless ex-smokers recalled their shock at receiving a HNC diagnosis, fear of recurrence and heightened perception of the dangers of passive smoking. Our findings reiterate previous studies highlighting how a new diagnosis of HNC motivates patients to stop smoking, presenting a constructive opportunity whereby smokers can be effectively encouraged and supported to quit, thereby improving long-term QoL and survival outcomes (Komra and Hegarty, 2006; Semple et al., 2008). However, limited success was reported with smoking cessation products. Whether participants who tried cessation merchandise simultaneously accessed specialist guidance and smoking cessation services was unknown, however research by the British Lung Foundation (National Centre for Smoking Cessation and Training, 2012) demonstrates people are up to four times more likely to successfully quit using a combination of smoking cessation medication and specialist help. With a rise in media stories reporting detrimental effects of vaping, clear guidance and reassurance on the use and safety of smoking cessation products is essential to allay public concerns, and enable health professionals to confidently offer support and advice for those seeking help.
Support was a recurring theme amongst participants, who recognised the influence of family, friends and partners on their behaviours. Research shows that cancer patients also want more direct support with health professionals at treatment end (Laugesen et al., 2015; Tan and Goonawardene, 2017), and this was echoed in our study, especially for individuals living alone who reported feeling “abandoned” and “isolated” after treatment. Certainly, participants were keen to seek health knowledge from the specialist AHPs interviewers. Health professionals are well-placed to guide choice in line with government health recommendations, yet research highlights such opportunities are often lost due to insufficient engagement by healthcare staff in discussing strategies with patients (Lawson and Flocke, 2009; Austin et al., 2005), possibly due to heavy caseloads and inadequate time. Brief interventions, such as Making Every Contact Count (Local Government Association, 2014) can assist behaviour change, however, the rise of dedicated specialist rehabilitation teams at treatment-end may offer a more effective way to provide ongoing support to survivors. Facilitating equality of access to rehabilitation teams for all HNC survivors does, of course, require additional time and reallocation of resources.
Nonetheless, where health messages were clear and concise, participants were confident of the advice they should be following and tried to adhere to recommendations. A good example of this is the 5-a-day campaign; even when participants were unsure whether they were consuming adequate intake of fruit and vegetables, they knew what they should be aiming for and the amount was attainable. Older studies report a poor intake of fruit and vegetables by HNC patients, with a third consuming < 4 servings of fruit per month (Duffy et al., 2009). Intake was significantly higher in our cohort, possibly a result of the high profile World Health Organization (2009) “5-a-day” campaign, which has been successfully embraced by UK supermarkets and schools. However, the inclusion of numerous daily smoothies by participants with dysphagia made accurate interpretation of consumption challenging, increasing potential for overestimation. Moreover, Public Health England (2015) only recognise one 150ml smoothie as counting towards the 5-a-day target due to the high sugar content. Conversely, there appeared limited awareness of the carcinogenic risk associated with red and processed meat intake, with consumption of the latter particularly commonplace. Current guidelines recommend limiting or avoiding processed meats, and reducing red meat to a maximum of 18oz/500g a week (World Cancer Research Fund (WCRF), 2018), however quantification of products may be difficult for people to visualise in practical terms. Further, some participants displayed hesitancy in distinguishing between the two types of meat, posing difficulties for individuals trying to comply with government recommendations. Indeed, studies do not typically report data separately for red and processed meat, and there is the additional confounding factor regarding cooking methods, given the association with potential carcinogenic compounds linked to grilling and barbecuing meat (Sinha et al., 1999), which were not assessed in this study.
The WCRF (2018) recommend fish as a good alternative to processed and red meat, however many participants disliked fish so ate none at all. Omega-3 supplements can be a useful substitute, yet those who took them already met recommended intakes for fish, or used them for pain relief, highlighting uncertainty regarding the role of OTC supplements. Indeed, the rationale behind participants’ use of OTC supplements was sometimes unclear, and evidence for associated health claims questionable. Rather than nutritional supplements, WCRF recommend a varied diet to provide essential nutrients, as some evidence indicates high-dose supplements may increase cancer risk (SACN, 2016). Nonetheless, HNC patients can be at risk of nutritional deficiencies, since eating a balanced diet can pose difficulties, both during and after treatment. Of note is that only one participant took vitamin D (multivitamin). Government recommendations are that everyone
Dietary intake is frequently compromised by HNC treatment side-effects, with resulting weight loss. Substantial research demonstrates an association between excess bodyweight and increased risk of several cancers, including HNC (Bogers et al., 2007; Calle et al., 2016); however, being underweight also increases mortality risk when compared to people with an average BMI (Roh et al., 2014). Despite some participants remaining obese, we observed a significant shift in numbers moving from a higher to a lower BMI category following treatment. These weight changes likely reflect the negative effects of HNC treatment on eating and drinking rather than a proactive response to weight correction; indeed, several participants directly attributed weight loss to treatment side-effects. At the time of study, 33% of men and 40% of women in England were within the BMI healthy weight range (Health Survey for England, 2018); our results were above the national average for men (50%), but significantly less for women (15%). Compared to a healthy population this was not unexpected, although it was surprising this was not equally reflected across genders. One possible explanation may be the comparatively older mean age of our female cohort and associated post-menopausal weight gain. Moreover, men generally have greater skeletal muscle mass than women, therefore muscle loss may also be a contributory factor affecting body weight, due to inactivity associated with treatment side-effects (Janssen et al., 2000). Physical activity supports a healthy weight, and research shows the more an individual exercises, the greater the risk reduction for many cancers, including HNC (Moore et al., 2016). Despite recognising the health benefits of exercise, participants reported the psychological barriers, of getting and keeping motivated, as well as physical barriers, including fatigue and pain associated with cancer treatments, affected motivation and ability to exercise. We found participants tended to over-estimate self-reported physical activity, and significantly underestimate sedentary time. Such misperceptions present barriers to change, echoing previous research highlighting the complex nature of assessing activity levels (Schaller et al., 2016). Studies show that fitness trackers or pedometers can boost motivation to increase activity levels (Cairns et al., 2018) and, where appropriate, should be encouraged to offer insight into how active someone actually is rather than how active they perceive themselves to be. Nonetheless, our results compare favourably with previous literature which estimates <30% of people living with cancer meet recommended physical activity levels (NICE, 2018). Of note is that 90% of our cohort were married or had a partner, which may positively influence motivation and activity levels. Moreover, despite half achieving retirement age, most participants continued to work, boosting activity levels. Raising awareness of GP exercise prescription and social prescribing schemes could further encourage survivors to participate in exercise and increase physical activity levels.
Conclusion
“And I do quite proudly say: I’m a Survivor!”[P14]
This study demonstrates HNC survivors are highly motivated to make healthy lifestyle changes to reduce risk of cancer recurrence and other co-morbidities. Our findings highlight some of the challenges encountered by survivors when trying to follow health recommendations. Chronic treatment side-effects affected behavioural choice and the extent to which participants felt able to implement positive lifestyle change. Further work is necessary to increase awareness of Government guidelines, since health messages are not always reaching the public or are misinterpreted. Timely access to health professionals at treatment-end could guide lifestyle decisions, help survivors navigate potential barriers, interpret health messages into practical terms, and encourage sustained behavioural change, thereby increasing patient self-efficacy whilst advancing achievement of government health recommendations, to deliver substantial health benefits and improve survival outcomes.
Limitations
This was a small study and respondents who chose to participate may reflect a more motivated cohort than individuals who did not engage in the study. Further, mean cohort age was younger than national data reports for people with HNC, which may positively affect motivation. There will therefore be unidentified barriers to making health-related lifestyle changes which remain unaccounted for in our study. Self-selection in volunteering for the study may influence qualitative data but was unavoidable due to ethical need for individual choice and avoidance of coercion. Information was collated and conclusions drawn from the interview responses supplied by HNC survivors, and consideration should be given to the subjective nature of reporting on lifestyle factors and the inevitable potential for over or underestimations.
Footnotes
Availability of data and materials
Upon request and after application to the Principal Investigator, Claire Hanika (Claire.hanika@nhs.net).
Authors’ contribution
CH conceived and designed the study. CH, NP and KB recruited participants, collected data, analysed data and interpreted the results; CH drafted the manuscript; NP, CH and KB edited and revised the manuscript; CH and NP approved the final version of the manuscript and take public responsibility for the content. JM conducted quantitative statistics for the study.
Acknowledgements
We thank the participants, and Ms Nicola Tilley, administrative support, for transcribing the tape-recorded interviews.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study protocol conformed to the ethical guidelines of the 2013 Declaration of Helsinki and Medical Research Council (NHMRC) National Statement on Ehtical Conduct in Human Research, as reflected in the approval by the Health Research Authority and South Central Berkshire local Research Ethics Committee (17/SC/0562). Written informed consent was obtained from all participants.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Macmillan Cancer Support, (PS@Mac no. 6275734).
