Abstract

Currently, the world has come to a complete standstill as it battles the pandemic caused by the spread of novel coronavirus (COVID-19). Given the global efforts, one is hopeful that a solution to this problem would be found sooner than later. Whenever the world has seen the possibility of an epidemic of this proportion, the reaction has been similar: pump in huge resources; develop solutions faster; and hope to contain this in the quickest possible time to mitigate the severity to human lives and social and economic determinants. On the other end of the spectrum to these pandemics lie the genetic diseases, popularly termed as rare diseases (RDs). As the name suggests, very few people suffer from these diseases within a population. Whatever might be the causation of a disease, the mission of governments of various countries has been that no one is left behind when it comes to saving lives. The World Health Organization (WHO) Sustainable Development Goal 3 emphasizes on a vision of inclusive healthcare access. ‘Just because a disease affects a small number of people does not make it irrelevant or less important than diseases that affect millions’ (WHO, 2018).
Definitions of Rare Diseases in Various Geographies.
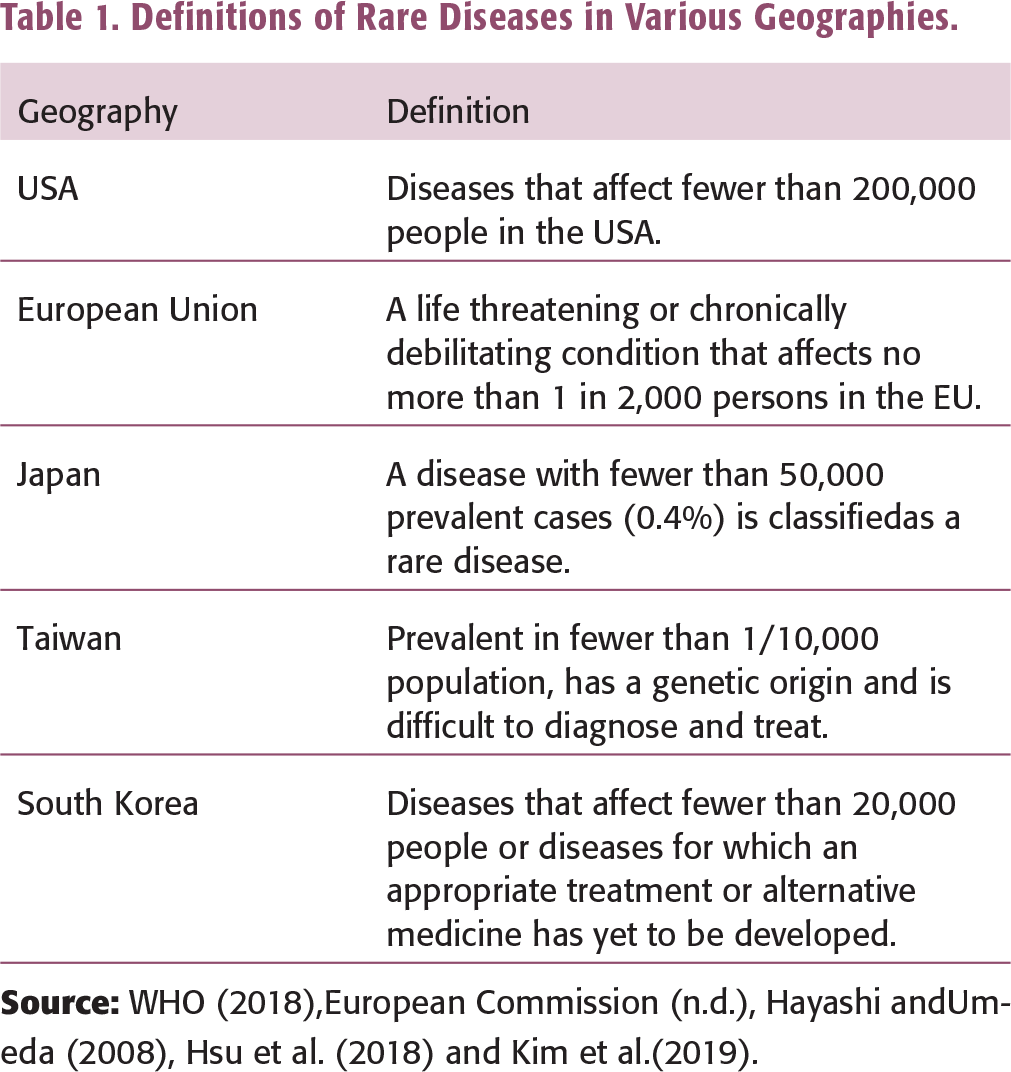
In India, while there is no accepted definition of RDs, the Drugs Controller General of India (DCGI), in March 2019, came out with a definition of orphan drugs, which essentially are used to treat RDs, as the one which addresses a potentially treatable population of 500,000 in India. RDs include inherited cancers, autoimmune disorders, congenital malformations and certain endemic infectious diseases due to their rarity and low prevalence (Orphan et, n.d.). It is estimated that there are 7,000 RDs, of which 80% are genetic. Among the 7,000 RDs only 5% are treatable (National Institutes of Health, n.d.). According to the WHO estimates, 1 in 15 people worldwide suffer from one RD or the other (WHO, n.d.). In India, over 70 millionpeople suffer from one RD or the other. About 700 rare or ultra-rare diseases are currently present in India (Rare Diseases India, n.d.). The prevalence of consanguineous marriages is often cited as the reason for the higher density of RDs in the South Asian region.
Despite discoveries of diseases and efforts to bring early treatment in the market, most healthcare systems that exist globally lack agility in absorbing medical interventions on an equitable basis;for instance, most of the RDs are not tracked except in certain developed economies;hence, it is hard to determine the exact number of RD patients or how many people are affected. Lacunae in maintaining electronic health records or registries of the cases observed in poor and vulnerable sections of society lead to government’s indecisiveness in providing healthcare access.
In India, health is a state subject, and the state government is accountable for the successful implementation of healthcare programmes. However, the central government is also an equal partner in healthcare governance according to Indian Constitution Article 14 (Right to Equality) and Article 21 (Right to Life). After all, health is not a luxury for the sole possession of privileged few. Just because someone is poor, the centre/state cannot allow him to die (Md Ahmed vs Union of India, 2014). It is the constitutional obligation of the government, whether centre or state, to provide access to treatment if the patient is suffering from a RD or a chronic disease or in case of a pandemic, maintaining the overall well-being of its citizens.
Given the small number of people suffering from these diseases, individually, each disease may not be contributing much; however, the burden on the affected family is quite substantial. While speaking to a few families with patients suffering from RDs, we noticed that it takes a long time to arrive at the correct diagnosis. The costs incurred by the family keep adding up in the interim. Furthermore, the cost to the governments is also not trivial, especially when we consider the patient lawsuits and the cost of defending them;for example, in a recent court case, the Delhi High Court ordered All India Medical Sciences to treat a young patient with Gaucher’sdisease (PTI, 2020). Similarly, in April 2021, the Karnataka High Court ordered the state government to release ₹59.5 million for the next 4 months for the treatment of 45 existing patients from its unutilized corpus fund (The Hindu, 2021). In January 2020, The Madras High Court called upon the State Government of Tamil Nadu to allocate ₹50 million per annum and directed the central health ministry to allocate ₹44 million as an annual budget for the treatment of 11 patients (Imranullah, 2020).
On 23 September 2019, there was a political declaration by the UN on the inclusion of Rare Diseases within Universal Health Coverage. As India is one of the member states, it is important for The Ministry of Health and Family Welfare (MOHFW), Government of India to develop actionable items for alleviating the grievances of RD patients. Recently, MOHFW came up with the draft National Policy for Rare Diseases 2020 (GOI, 2021). Some of the key recommendations include
Identifying high-risk families and couples of reproductive age and screening them for any genetic abnormalities and potential preventive mechanisms; popularizing screening for newborns and improving diagnostic equipment; and providing better supporting infrastructure to the suffering population.
Currently, the acute treatment interventions, as mentioned in Group 1 of the Policy, can be covered under Ayushman Bharat Pradhan Mantri Jan Arogya Yojana 1 or Rashtriya Arogya Nidhi (RAN) 2 commitment. Ideally, while the policy mentions the government’s commitment to RDs, in totality, there is a definite need to allocate funds for RD treatments (wherever available). The policy is clearly silent about the sustainable funding mechanism to provide treatment for RDs that need chronic treatment support.
It should also be noted that several industry associations such as Confederation of Indian Industry (CII), Federation of Indian Chambers of Commerce & Industry (FICCI), Organisation of Pharmaceutical Producers of India (OPPI) and US-India Strategic Partnership Forum (USISPF) and patient advocacy groups such asLysosomal Storage Disorders Support Society (LSDSS), Organization for Rare Diseases in India (ORDI), Indian Alliance of Patient Groups (IAPG) have also provided several recommendations to the Indian government on how to solve the problem of RDs. While we do not touch upon the scientific aspects of RDs, the purpose of this article is to understand why market failure happens in the case of RDs and what steps can the Indian government undertake to assuage the problem, even if partly.
CAN MARKET FORCES SOLVE THE PROBLEM OF RARE DISEASES?
At present, several approved therapies exist globally for a few RDs. Moreover, over a hundred clinical trials are underway on over a hundred drugs. Having said that, a significant majority of RDs, at present, have no therapies; for some other diseases, therapies exist but they are prohibitively expensive. An important question that arises at this juncture is, can the free market, in itself, solve the problem faced by these diseases? More specifically, if there is no governmental intervention, is it possible for a solution to emerge?
The scenario of RDs is a prime example of a case where a free market will not solve the problem. Even if it does provide a solution, the solution is not feasible enough. This is because of a significant amount of sunk cost involved in discovering and manufacturing the drug. Simple economic theory suggests that sunk cost plays a vital role in a firms’entry decision into a particular market. To elaborate further, consider the following:
Research and development in a pharmaceutical market involves substantial risk. According to recent estimates, it costs well above US$2 billion to bring one molecule into the market (DiMasi et al., 2016). Out of several thousand molecules discovered, a handful of them see the light of the day. 3 In order for pharmaceutical firms to invest in research and development, it is imperative that the successful molecules cover not only their own research and development costs but also the sunk cost involved in molecules that have not been successful. Since there is so little consumption in the case of RDs, each unit will have to account for not only the cost of manufacturing but also a significant part of overheads associated with the discovery process.
As Grabowski (2007) points out, clinical trials are a significant part of drug discovery. Clinical trials happen over several phases, and several potential molecules keep dropping out at every stage. These trials (phase III and, in some cases, phase IV) involve identifying several patients with certain medical conditions and designing a robust experimental design to prove that the drug is efficacious. In the case of RDs, this problem is significantly exacerbated. Search cost for locating suitable patients for clinical trials tends to be huge. Since the patients are rare, and many doctors may not be aware of these diseases, it is likely that the pharmaceutical companies need to spend significant resources to identify patients for clinical trials, making them very expensive.
While visiting a manufacturing unit, which manufactures similar medicines, we realize that an exclusive facility (in some cases, facilities) must be developed for manufacturing some of these medicines. Furthermore, unlike a typical pharmaceutical plant (especially in the case of smallmolecule drugs) where a manufacturing facility can be used to produce multiple products, these enzyme therapies require dedicated plants for manufacturing. Therefore, the limited number of doses to be manufactured necessarily have to account for the cost of setting up the plant.
As an aside, please note that many of these medicines are not patent protected. Given the complexity in the manufacturing technology involved and lack of substantial demand, it makes sense to protect these drugs as business secrets, as against patenting them in the first place. The sunk cost involved in research and development plus set-up costs would automatically deter entry. This implies that we only have a single brand available in the market most of the time. Furthermore, since the number of patients is low in any given region, these production facilities are planned with an intention to cater to global demand. Hence, localized manufacturing is not likely to yield any gains.
To gain a better understanding of the problem faced by the patients, we spoke to three patients who are currently suffering from one of the recognized RDs. This highlighted quite a few problems. First, it took a significantly a long time to arrive at the right diagnosis. This is probably because general physicians (first recourse for any patient) are not sufficiently wellversed with RDs. Second, once the patient is diagnosed appropriately, the cost of treatment is often prohibitive for the patient. While some organizations (and in some cases, pharmaceutical companies themselves) fund the treatment, the cost from patients’ side is still significant. Third, since health is a governmental responsibility, some patients/organizations have approached courts requesting financial and other assistance. In several cases, courts have ruled in favour of patients mandating governments to pay. Understanding patient characteristics and problems faced by patients should be a strong topic for future research.
Given these issues discussed above, it is clear that significant markups are needed to account for the sunk cost. Furthermore, since the production is at a lower scale, these sunk costs need to be distributed across the limited number of doses manufactured. Therefore, medicines associated with therapies for RDs tend to be priced high. 4 Another massive problem is identifying the right patient population to conduct a feasible clinical trial. Therefore, conducting clinical trials become very expensive. Since many patients (especially those in emerging economies) are from weaker economic conditions, affordability becomes very difficult, resulting in a less than efficient market. Further more, in some cases, pharmaceutical companies may not even have any incentive to invest in the drug discovery process (especially for more complex therapies) because of insufficient return, resulting in another kind of market failure. In sum, an inefficient market happens for two reasons: high costs of innovating medicine and prohibitive costs of affording the existing therapies.
HOW COULD PROBLEMS WITH RARE DISEASES BE MITIGATED?
Since the free markets are not likely to provide a solution directly, it is imperative for the governments to intervene and set up mechanisms to avert prohibitive costs. There have been some successful interventions, which provided pharmaceutical companies to innovate in the RD category;for example, the Orphan Drug Act of 1983 in the USA provides extra privileges such as tax incentives, clinical trial subsidies, patent life extension and subsidizing R&D through government funding for innovation in RD categories (Wikipedia, n.d.). Similarly, in 2007, the US government introduced a ‘priority review voucher program’ as a form of incentive to innovate for RDs. Under this programme, a pharmaceutical company innovating medicines approved by the Food and Drug Administration (FDA) wins a priority review voucher for any drug of its choice. This may mean that the pharmaceutical company with a voucher could allow medicines intended for common diseases to reach the market faster. Furthermore, this creates a vibrant marketplace to trade these vouchers. Several studies outlining the actual benefits of this programme have been highlighted in the literature. 5
While the extension of patents and voucher programme may address one kind of market failure (incentives to invest in drug development), they may still not avert market failure related to the affordability of medicines. In developed economies where a significant population is insurance covered, this problem may not be as significant. In emerging economies where a large part of the population is not covered by any kind of health insurance (and with income levels being relatively low), market failure becomes very important due to affordability reasons. Since most of the production facilities of these diseases are located abroad (especially in developed economies like the USA) where the cost of labour is on the higher side, an immediate solution that comes to mind is shifting the production base to a cheaper location. However, this is not likely to work because, as already discussed, set-up costs tend to be huge. While one may save on labour expenses, the overheads associated with setting up a production unit are likely to be so huge that it would not result in any substantial savings. Furthermore, discussions with a firm that specializes in biologics (complex molecules with huge atomic weight) show that despite incurring set-up costs, the end-product need not be similar. Therefore, since many medicines are to be imported, eliminating customs duty on these products can reduce disease burden, even if marginally.
As already indicated, it is not realistic to expect free markets to solve the problems associated with RDs. Moreover, since these diseases will never be a priority from a public health perspective, it is difficult to expect various governmental departments to allocate any part of their healthcare budgets towards provisioning for RDs. In other words, since these departments are hard-pressed for funds to combat other health exigencies, the allocation for RDs would never be a priority. Therefore, the only feasible solution is for the governments to allocate a fixed portion of the healthcare budget to treat RDs. It can be argued that for a country like India, which suffers significantly from a paucity of funds for healthcare, can the states afford to earmark some funds exclusively for RDs? Since free markets will not be able to solve these problems, a small fraction of healthcare budgets needs to be allocated, especially because healthcare is a universal right.
While such allocation solves a part of the problem, it does not solve the entire problem of affordability. The pharmaceutical companies that manufacture these drugs should be willing to subsidize the patient significantly so that the shortfall, if any, in governmental allocation can be filled;that is, it has to be a joint effort on the part of pharmaceutical companies and the government to tackle the issue of RDs,and a fixed allocation by the government allows all the concerned parties to estimate the commitment required. In this argument of the government versus pharmaceutical companies, another factor comes into the picture. Since several RDs have no cure currently available, the actual number of patients who would require government funding could be quite small. While more accurate estimation is required, our understanding is that even a very small allocation could be significant in alleviating the problem. Such an approach is not without precedence in India;for example, the Government of Kerala has set up such fund exclusively for the treatment of RDs.
As already discussed, the US government provides incentives to pharmaceutical companies for innovations related to combating RDs. Incentives like patent life extension and priority review voucher programme have gone a long way in facilitating research for medicines for RDs. Similar incentives could be considered in the Indian context, albeit with sufficient modifications that deal with the local conditions. While the exact nature of tax breaks requires a significant amount of research, incentives ought to be linked with the volume of discount provided. A simpler mechanism could be to provide tax waiver up to the value of lost sales (defined as the original price of the drug minus the sale price of the drug multiplied by total sales). Further incentives like expedited approval processes priority approval for other drugs not meant for RDs (in case of reasonable reduction in price), etc., could go a long way in helping the cause of RDs.
Firms spend a significant amount of resources on activities related to corporate social responsibility (CSR). In fact, as per the Indian Companies Act, 2013, it is mandatory for companies of a certain size to contribute 2% of their net profit towards CSR activities. The Act also specifies the list of activities that are acceptable towards spending (India Filings, n.d.). At this point, supporting RD patients is not on the list. We suggest a certain portion of the overall CSR spend (actual number requires in-depth study) be allocated for RDs and channelled to a Ministry of Health fund, which gets replenished annually. This will go a long way in ensuring that the therapy becomes accessible to the end-user.
Can price regulation in the lines of drug price control order (DPCO) work in the case of RDs? Already, the incentives to invest in RDs are low. It takes firms significant time before they recover these expenses. Price regulation is likely to reduce these incentives further, dissuading firms from investing in any therapies for the diseases for which cures do not exist. Additionally, literature also suggests that entry by pharmaceutical firms in countries with heavy price restrictions is significantly delayed (Kyle, 2007). Since most of the innovator firms are located abroad, there could be a possibility of adverse health outcomes. This raises another important question: can health insurance be a solution to the affordability issue? Currently, several health insurance plans do not cover RDs. Moreover, many health insurance plans cover a limited amount of expenses. While that amount may seem ample in most cases, the cost of medicines for RDs makes it insufficient. Another important consideration, in this case, is the problem of the sustainability of premiums. Unfortunately, since health insurance coverage is often incomplete, even for more common diseases, coverage for RDs does not seem like a feasible solution anytime soon (Choudhury & Saberwal, 2019).
Finally, a preliminary discussion with a few medical practitioners reveals that the awareness related to these diseases and the available curative regimes is quite low. Therefore, significant resources need to be spent on educating the medical community in particular and the general public at large on these RDs. In our discussions with a few patients, we found that the correct diagnosis was arrived at after a series of misdiagnoses on the part of general physicians they visited in the initial stages. This is arising partly because of a lack of knowledge among general medical practitioners. One can imagine that these misdiagnoses are fairly expensive; furthermore, they rob patients of a chance to get on an appropriate therapy sooner, thereby denying them of leading near-normal lives much sooner. Therefore, it is not only sufficient to make the primary healthcare system more responsive to RDs, but also, the entire system should be overhauled so that a systemic and systematic detection of RDs is possible.
This leads to another important question: Who bears the expenses pertaining to physician education? Here again, a public–private–partnership model could be the solution. Since there are three major stakeholders—governments (state and central), patients and patient organizations and pharmaceutical companies—these expenses should also be borne by all three players. However, since health is ultimately a government responsibility, we feel that the government should clearly articulate its contribution and expect support from the other two players. Pharmaceutical companies could also be incentivized, for instance, by giving them exclusive rights to selling medicines.
CONCLUSION
While diseases that afflict a significant population attract significant attention from pharmaceutical companies and governments worldwide, RDs seldom attract attention. However, the problems faced by the patients are real and require significant attention from governments all over the world to assuage the problem. A more direct question could be, if not the government, where else could these patients look for support? From allocating fixed budgets to working closely with pharmaceutical companies, to raising more funds through innovative policies, there are some solutions that are worth trying. In this article, we endeavour to put together a series of suggestions that could potentially mitigate the problem of people who suffer from RDs. While we identify a few ways, there are still several out there, which we hope future researchers will work on.
Footnotes
DECLARATION OF CONFLICTING INTERESTS
FUNDING
The authors received no financial support for the research, authorship and/or publication of this article.
