Abstract

Brain fog (BF) is a common neurocognitive phenomenon often associated with fatigue in post-COVID infection.1,2 In COVID-19 infections, this may result from hypoxia-induced microstructure changes or hyperinflammatory responses. 1 Stimulants such as methylphenidate have been reported to be successful in treating fatigue due to non-COVID causes and have been shown to improve associated attention difficulties in a few cases in the literature. 3 There is only one observational study of the successful use of bupropion in COVID-19-associated BF syndrome, which is limited due to non-quantitative subjective reporting. 4 Here we describe four such cases, which were treated with methylphenidate shown to improve the symptoms of BF as evidenced by objective quantitative measures.
Case Series
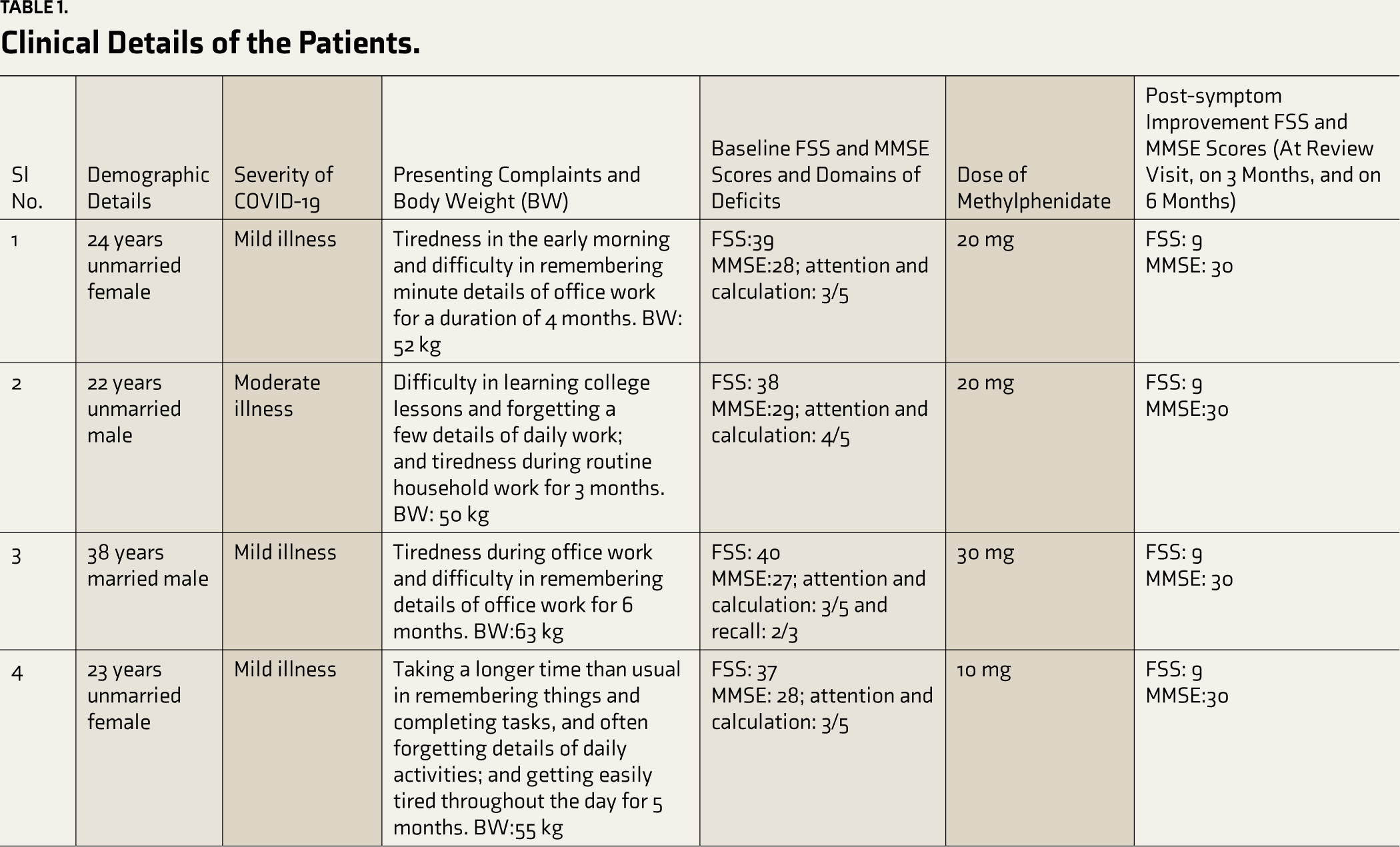
All of our patients were assessed by two psychiatrists in liaison with medical specialists under collaborative care. As there are no standard criteria for BF, we used an operational definition used in Asadi-Pooya et al. 2 which stated BF as patients’ subjective feeling of worsening of ability to concentrate and think compared to pre-COVID, that developed during or after COVID-19 infection and persisted for more than 12 weeks post-infection, not attributable to other causes. Their post-COVID presentation in a psychiatric facility is briefed in Table 1, including demographics, phenomenology, assessment, and treatment. The COVID-19 infection in cases 1, 3, and 4 was managed with home isolation, rest, adequate hydration, and paracetamol 650 mg SOS for fever. Case 2 was treated with ward admission, paracetamol 650 mg SOS, oxygenation, prone positioning, oral methylprednisolone 16 mg BD, and enoxaparin 60 mg sub-cutaneous for 7 days. He showed improvement after 3 days of hospital admission and was discharged after 7 days. Their symptoms of BF and fatigue started within a week of the onset of the acute phase of the COVID-19 infection and continued until 3–6 months later with socio-occupational dysfunction (Table 1). They did not meet the criteria for any mood, anxiety, psychotic, or major neurocognitive disorder. There was no history of any substance use, any past medical illness, a past cognitive disorder, or a family history of any mental illness. In general and systemic examinations, no remarkable sign was elicited. All patients were evaluated with a complete blood count, blood glucose, blood urea and creatinine, liver function tests, electrolytes, thyroid function test, lipid panel test, vitamin D3, vitamin B12, and folate levels, HIV-I and II antibodies, VDRL, anti-HCV and anti-HBsAg antibodies from accredited laboratories, and the results of all blood reports were within their biological reference level. A computed tomography scan of the brain was done in cases 1 and 3, and there was no parenchymal abnormality found; in cases 2 and 4, it was not done due to the economic issues of the patients. EEG was also not done in our cases for the same reason. Due to the invasiveness of the procedure, the associated inadvertent risk, and the absence of any overt indications like any focal neuro-deficit or meningeal sign, cerebrospinal fluid analysis was not considered in our cases. The patients were evaluated with the Mini Mental Status Examination (MMSE) 5 and Fatigue Severity Scale (FSS) 6 at baseline, at post-intervention on the next review visit of patients, (3 weeks for case 1, 2 weeks for cases 2 and 4, and 4 weeks for case 3) and at 3- and 6-month follow-up. During the administration of MMSE, we used different words for recall tasks, different common objects for naming, and different tasks for attention and calculation domains (e.g., serial 7s, spelling WORLD backward, asking names of days in a week backward, etc.) on each follow up to avoid practice effect. The FSS scale is a 9-item scale, with item scores ranging from 1 to 9 and a median score >4 for each item signify the presence of fatigue in the Indian population. 6 Methylphenidate was tried for all subjects for their fatigue symptoms, in the hope that it might improve their associated attention deficit, also as supported by a few past evidences 3 and was continued for 1 month after the review visit and was then tapered down by 5 mg every week and then stopped for cases 1, 3, and 4. Case 2 stopped taking methylphenidate after 1 month.
All patients showed improvement in symptoms subjectively as well as objectively, as evident in the reduction of FSS and MMSE scores compared to baseline within 2–4 weeks (Table 1). No relapse of symptoms was observed in all cases at follow-ups. Written informed consent was obtained from all the patients before including their data in the manuscript.
Clinical Details of the Patients.
Discussion
The onset of symptoms immediately following COVID-19 infection, the absence of any obvious explainable cause in the diagnostic workup, the presence of similar reporting, 4 and the presence of explainable mechanisms behind the phenomenon in association with COVID-19 1 points toward COVID-19-related BF in our cases. 7 Norepinephrine and dopamine both interplay in attention, working memory, and executive functioning by regulating the signal-to-noise ratio and the selection of information in the prefrontal cortex, hippocampus, and subcortical neuronal network.8,9 Methylphenidate, by adjusting the dopamine and norepinephrine levels in the prefrontal cortex, 10 yielded an improvement in cognitive functioning in our cases. Bupropion, having a similar mechanism of action as methylphenidate, has been shown to be effective in a previous study, 4 which supports our arguments. Moreover, long COVID cases with cognitive deficits have been reported in the literature are mostly at their 40 or older,1,2 in the contrary our cases are younger, which may be a factor for their faster recovery and little decline in MMSE score at baseline evaluation due to their cognitive reserve. Apart from objectivity, cognitive improvement following intervention in our cases is within weeks (Table 1), which is in contrast to the usual chronicity of non-intervened COVID-19-related BF. 1 Though natural remission cannot be overruled, given the drastic improvement after MPH use, the presence of its pro-cognitive mechanism of action, and similar phenomena with other stimulants, favor our argument regarding cognitive improvement in our cases due to MPH use. 7
Further, our cases completely recovered with methylphenidate and improvement persisted at follow-up without any extended treatment. Our study has many limitations, like its observational nature, small sample size, absence of a control group, and non-availability of neuroimaging for cases 2 and 4, but it provided objective insight about the effect of stimulants on COVID-19-associated BF syndrome, unlike previous studies. 4
Conclusion
Despite the numerous shortcomings of our findings and the cautionary view of the necessity of replication of our findings in future research, we can opine on methylphenidate as a potential option for the treatment of COVID-19-related BF.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Declaration Regarding the Use of Generative AI
None used.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Written consent was obtained from all the patients for publication of this manuscript.
