Abstract
Background:
Being a nonbenzodiazepine, zolpidem is believed to have a favorable side-effect profile and is widely prescribed for insomnia. However, in the past few years, numerous neuropsychiatric adverse reactions, particularly complex sleep behaviors (CSBs), have been reported with zolpidem.
Objective:
To conduct a systematic review of zolpidem-associated CSBs.
Data Sources:
An electronic search was conducted using MEDLINE, Embase, PubMed, and Cochrane database of systematic reviews to extract relevant articles till July 2020.
Study Eligibility Criteria:
Any type of literature article (case report, case series, and observational or interventional study) reporting CSBs associated with zolpidem.
Results:
In this review, we present aggregate summarized data from 148 patients presenting with zolpidem-induced CSBs (79 patients from 23 case reports and 5 case series; 69 patients out of 1454 taking zolpidem [4.7%] from three observational clinical studies). Various types of CSBs associated with zolpidem were reported, most common being sleepwalking/somnambulism and sleep-related eating disorder. On causality assessment, around 88% of cases were found to have a probable association with zolpidem.
Limitations:
Extraction of data from observational studies and spontaneous reports, due to nonavailability of any randomized controlled trials relevant to the study objective.
Conclusion and Implication of Key Findings:
Zolpidem-induced CSBs, although not very common, may develop when the drug is used at therapeutic doses for insomnia. Doctors need to be alert to monitor such adverse effects of zolpidem and exercise caution while prescribing it.
Keywords
Zolpidem is a one of the widely prescribed drugs for insomnia due to its better safety profile compared to benzodiazepines. However, various neuropsychiatric adverse events like hallucinations, sensory distortions, delirium, parasomnias or complex sleep behaviours (CSBs), amnesia, etc associated with the use of zolpidem are a matter of serious concern. Health care professionals need to be aware of such adverse events and exercise caution while prescribing the drug.Key Messages:
Zolpidem, an imidazopyridine, was approved by USFDA in 1992 for the short-term treatment of insomnia. It is a rapid and short-acting sedative-hypnotic drug, with only minor anxiolytic, anticonvulsant, and muscle-relaxant properties. Being a nonbenzodiazepine, it is considered to have a favorable side-effect profile, particularly with respect to rebound insomnia, amnesia, and dependence, compared to benzodiazepines. Hence, it is nowadays widely used in clinical practice for the management of insomnia. 1
Numerous postmarketing studies and case reports have shown an association of zolpidem with various neuropsychiatric adverse events like hallucinations, sensory distortions, delirium, parasomnias or complex sleep behaviors (CSBs), amnesia, etc. A majority of these cases comprise CSBs like somnambulism or sleepwalking, sleep driving, sleep eating, sleep cooking, sleep shopping, sleep sex, etc. A literature review published in 2008 identified ten case reports of nonbenzodiazepine receptor agonists induced CSBs involving 17 patients. Out of the 17 patients, 15 had consumed zolpidem, while the other 2 had taken zaleplon and zopiclone, respectively. 2 The incidence of somnambulism associated with zolpidem has been reported as 1% (1 in 96 patients) and 0.3% (7 in 192) in two postmarketing studies conducted in 1988 and 1995, respectively.3,4
CSBs associated with zolpidem have been a focus of attention, particularly after the USFDA requested a label change in 2007. In 2006, substantial media coverage was observed on zolpidem-associated CSBs in conjunction with a class-action lawsuit and the high-profile vehicle collision of Congressman Patrick Kennedy. 5 The death of Heath Ledger, a famous Australian actor, falsely implicated to be related to zolpidem use, intensified the controversy surrounding the drug in both the United States and Australia. Furthermore, around 700 reports of impaired driving ability and roadside accidents associated with zolpidem use were received by USFDA, as a result of which the agency recommended addition to the “warnings and precautions” section of zolpidem about CSBs, including sleep driving.6,7
In January 2013, the USFDA released a safety announcement regarding the risk of next-morning cognitive impairment and residual daytime effects with zolpidem. The risk was higher with extended-release (ER) forms of the drug and in females. Later, in May 2013, another recommendation was added to the drug label to avoid driving or other activities requiring mental alertness the day after taking the drug’s ER form. 8 In April 2019, based on accumulating data in FDA adverse event reporting system database and medical literature, USFDA added a boxed warning with zolpidem regarding the risk of serious injuries and deaths from CSBs, even at the lowest recommended dose and after just one dose. 9
In light of the accumulating data, it was thought prudent to conduct a systematic review on the safety of zolpidem with respect to CSBs when used therapeutically for approved indications in adult patients.
Materials and Methods
Search Strategy and Study Selection
A systematic literature search was conducted using MEDLINE, Embase, PubMed, and Cochrane database of systematic reviews, using keywords “zolpidem,” “complex sleep behaviors,” “nightmares,” “parasomnias,” “sleep eating,” “sleep talking,” “sleep walking,” “somnambulism,” “sleep terror,” and “sleep driving,” to extract all types of relevant articles until July 2020. All the searches used Boolean operators. Two investigators conducted the searches independently, which were later compiled. A manual search was also carried out by going through the reference lists of retrieved articles.
Criteria for Study Inclusion
Publications on zolpidem-associated CSBs, namely nightmares, parasomnias, sleep eating, sleep talking, sleepwalking/somnambulism, sleep terror, etc., were included in the present review. Publications of any type, including case reports, case series, conference abstracts, reviews, observational studies, and clinical trials relevant to the objective of the current review, that is zolpidem-associated CSBs, were included. Articles in languages other than English were also included, provided relevant information could be extracted from the abstract if available in English.
Data Extraction
Data extraction forms were filled to gather information on patient details, the indication for zolpidem use, dose and duration of intake of zolpidem, sleep behavior experienced, the outcome of the event, and any other relevant points.
Quality Assessment
The quality of case reports was assessed using the case report (CARE) guidelines and 13-item checklist.10,11 Each item was assigned a weighted score as 1, 0.5, or 0 for every Yes, Partly, or No recorded response, respectively, and a total score was calculated. Based on percent of the maximal total score, the quality of case reports was classified as excellent (90%–100% score), good (70%–89%), average (50%–69%), or poor (≤49%). 12
The case series were assessed for quality using Joanna Briggs Institute (JBI) tool having ten criteria. 13
Newcastle Ottawa Scale was used to assess the quality of observational studies.14,15 In this, quality of studies is assessed in the following three domains: (a) “selection,” that is representativeness of the exposed cohort, selection of nonexposed cohort, ascertainment of exposure, absence of outcome of interest at the start of the study; (b) “comparability,” namely comparability of cohorts based on the design or analysis; and (c) “outcome,” namely method of outcome assessment, follow-up long enough for the outcome to occur, and the adequacy of the follow-up of cohorts. A star system is used to assess study quality. Each criterion in selection and outcome domains is allotted 1 star, whereas 2 stars are assigned for comparability, making a total maximum score of 9. Depending on the overall quality score thus obtained, a study can be categorized as of good/fair/poor quality.
Study Outcome
The primary outcome was to evaluate the safety and causality of therapeutically used zolpidem with respect to CSBs in adult patients.
Causality Assessment
The causality of zolpidem with CSBs was ascertained using the WHO causality assessment scale. 16
Data Analysis
Data collected were entered in Microsoft Excel and analyzed using descriptive statistics. No specific hypothesis was tested.
Results
Study Selection
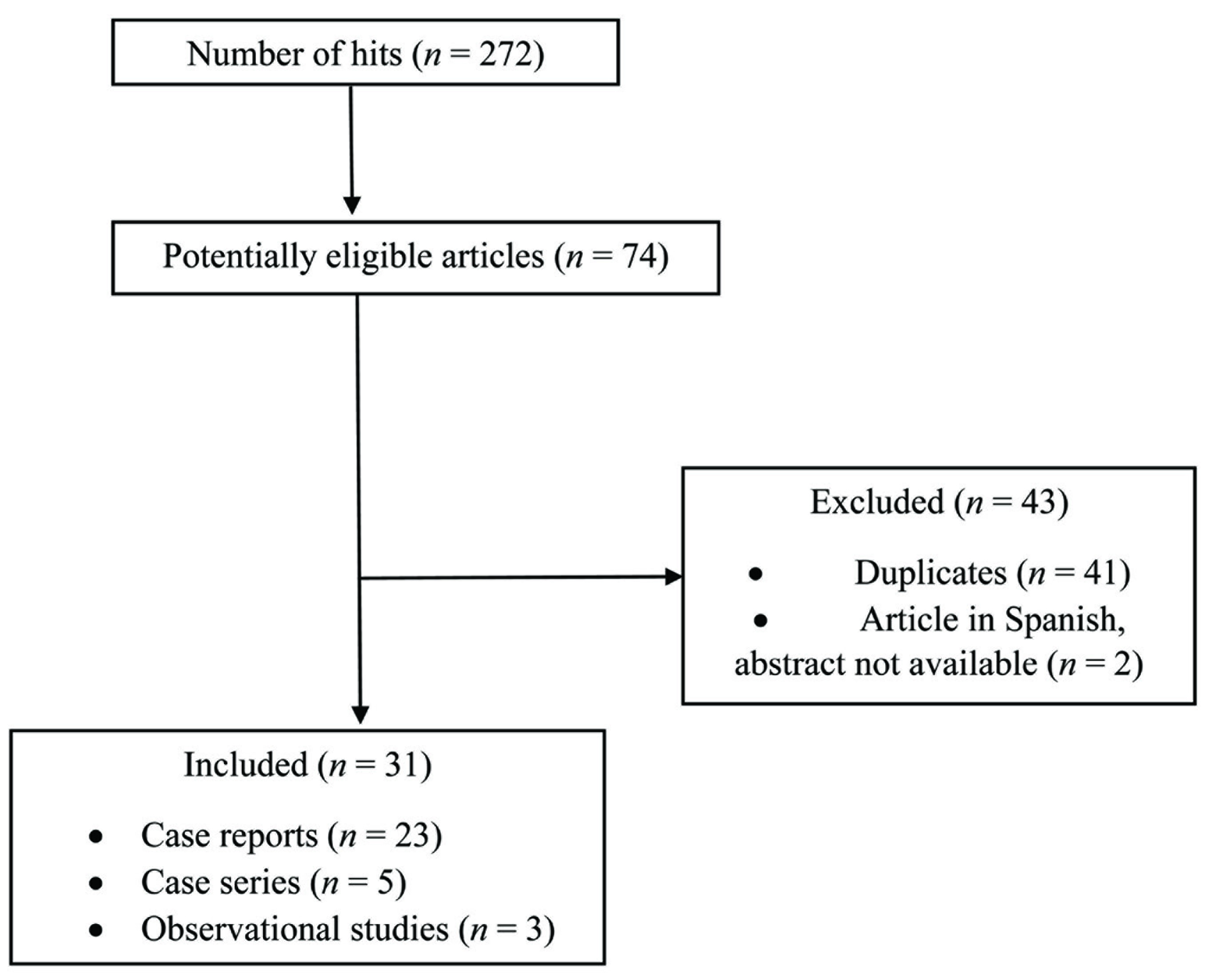
During the initial search, 272 hits were obtained using various keywords. Out of 74 articles considered to be potentially eligible, 31 were included in this systematic review (Figure 1). Of these, there were 23 case reports,17–39 five case series,40–44 and three observational studies.45–47
Study Characteristics
Table 1 depicts the characteristics of various literature articles reporting zolpidem-associated CSBs.
Flowchart Depicting the Study Selection Process
Characteristics of Literature Articles Included in the Systematic Review
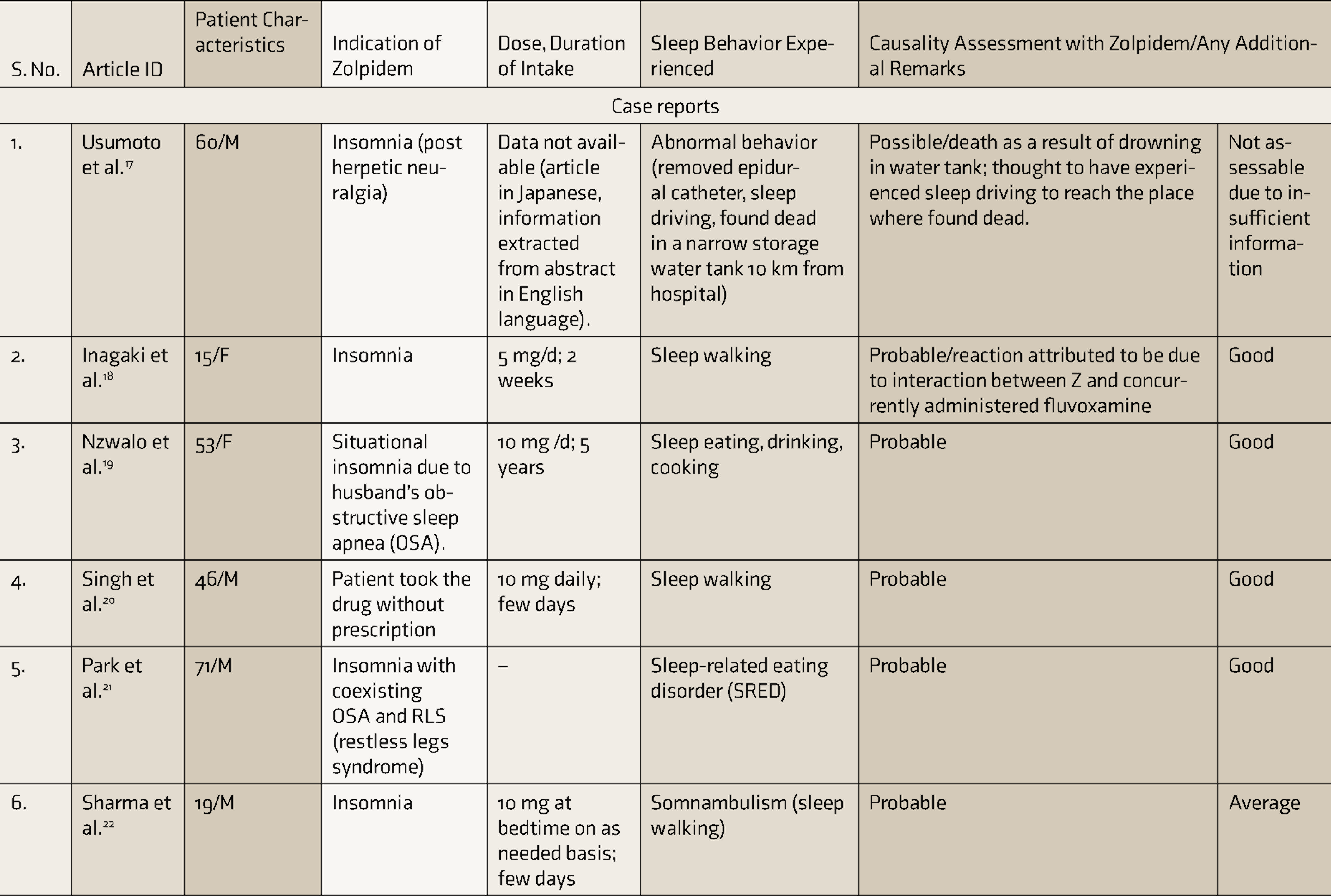
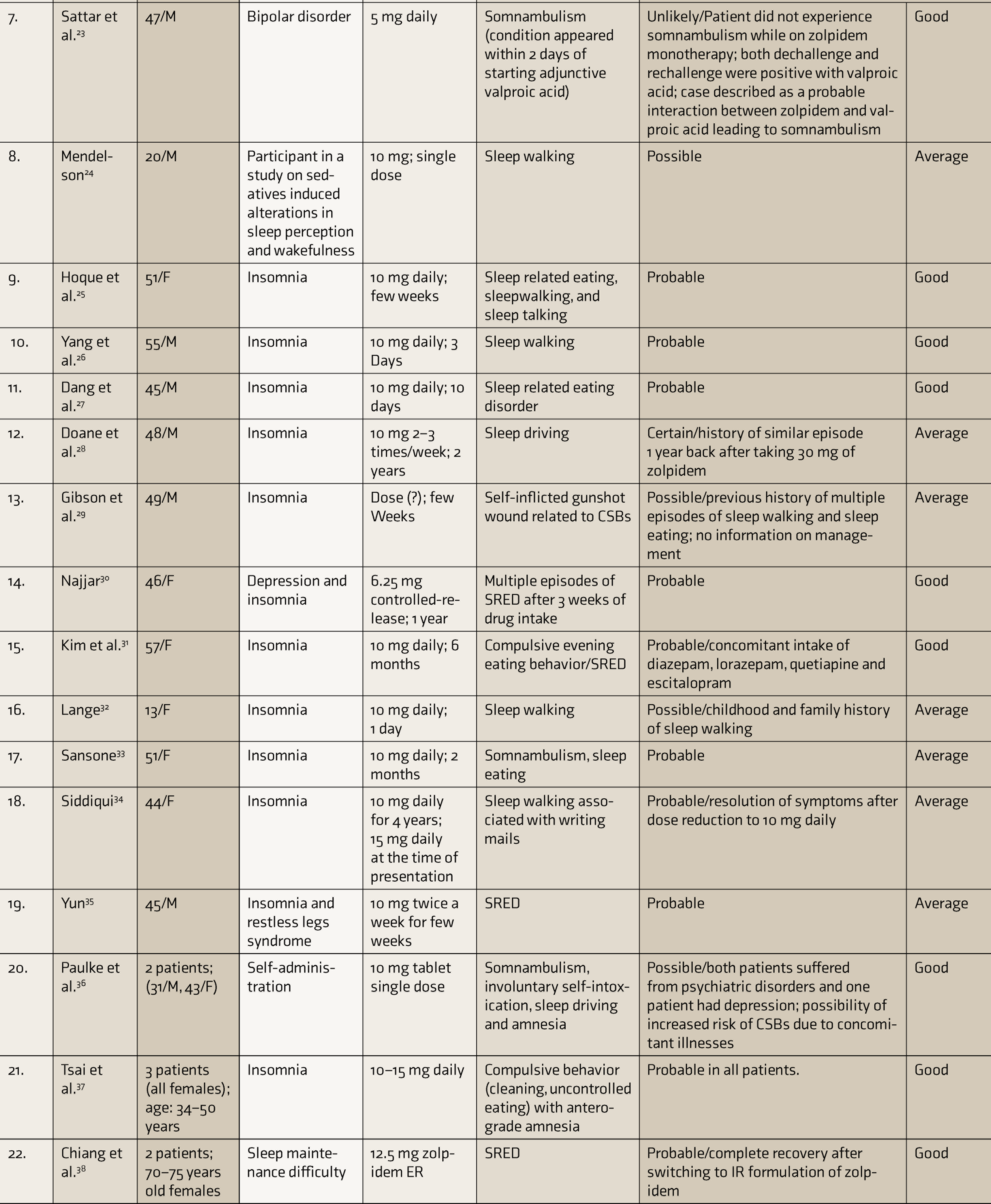
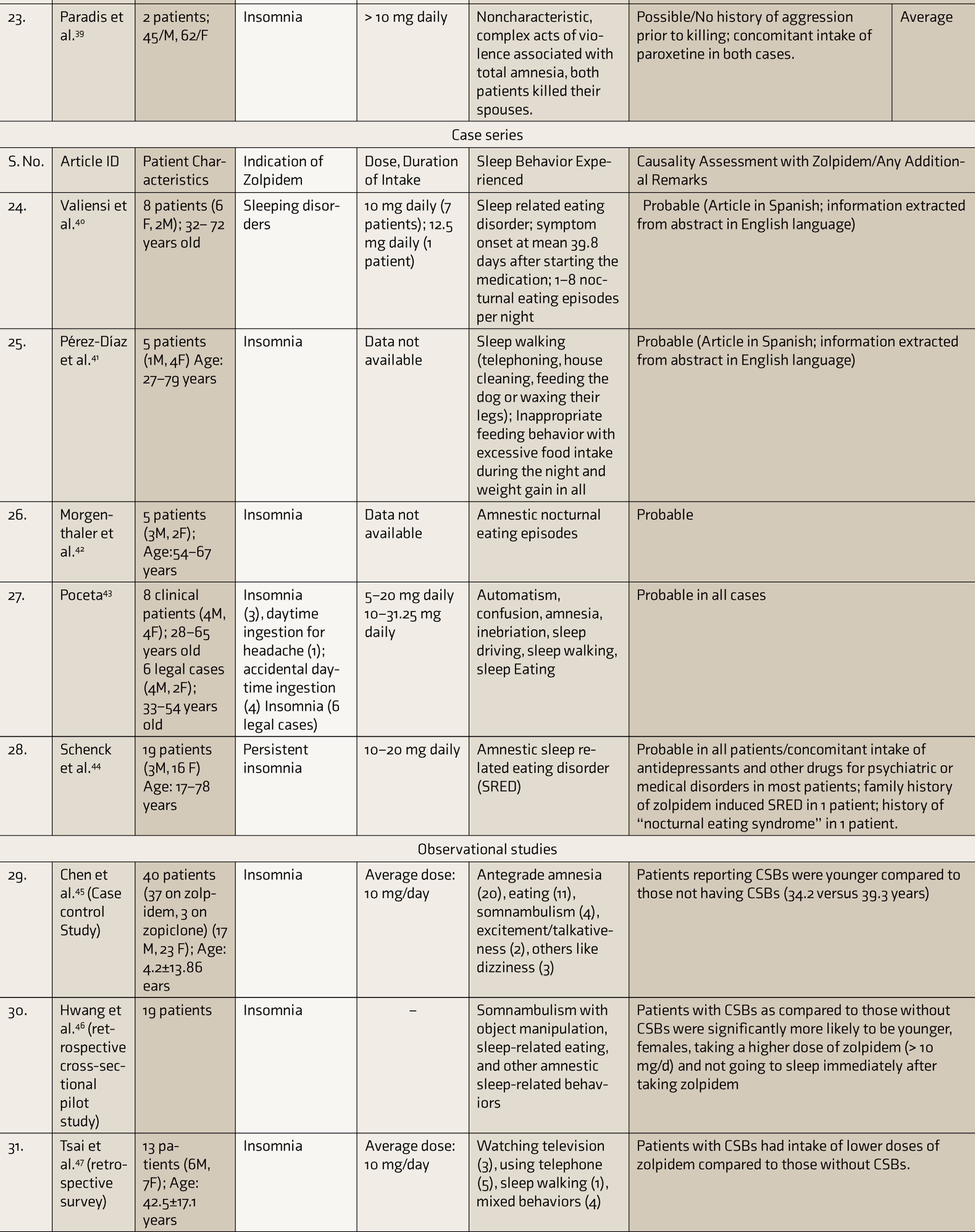
CARE: case report, SRED: sleep related eating disorder, ER: extended release, CSBs: complex sleep behaviors.
Quality of Included Studies
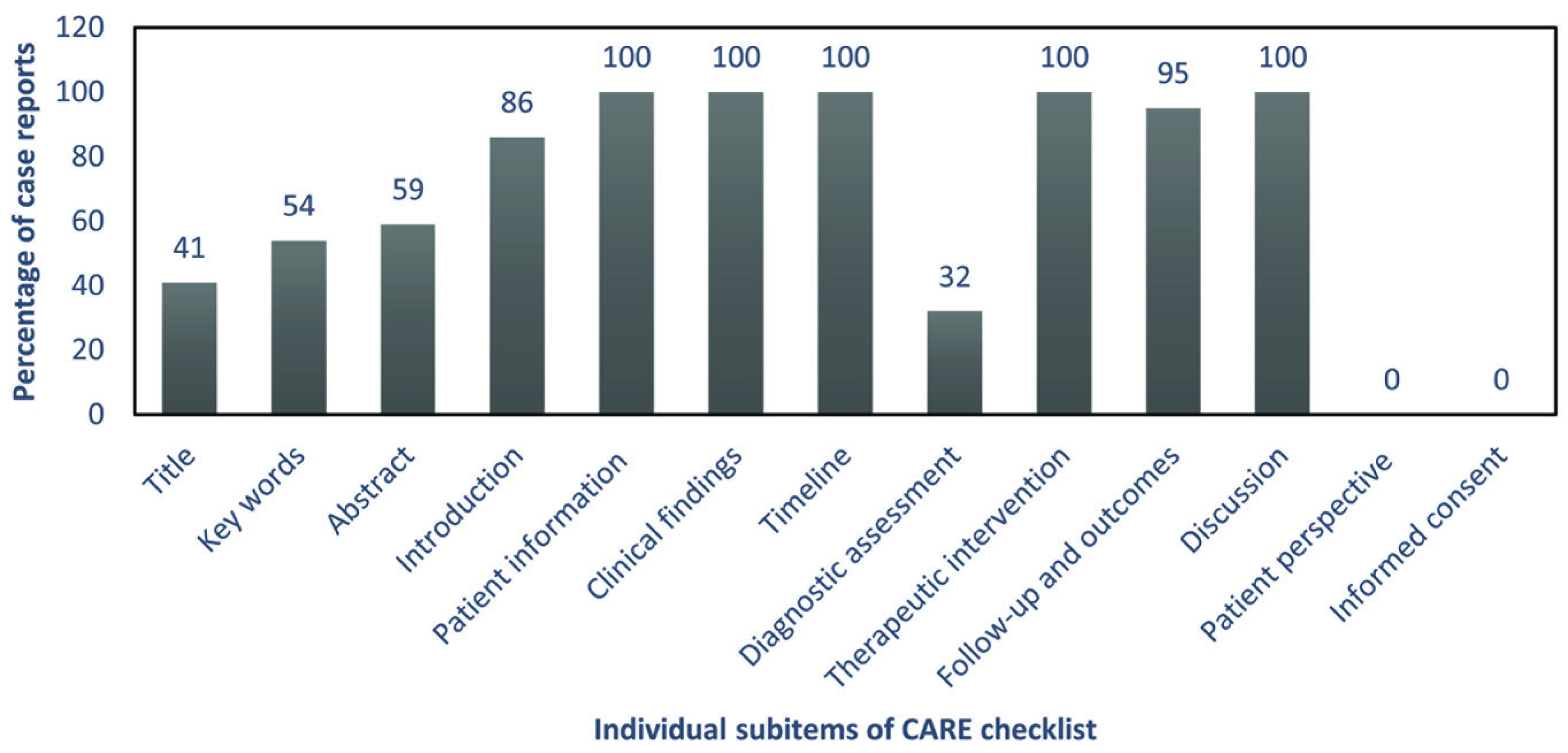
Quality assessment was done for 22 case reports and three case series (full-text articles published in non-English languages in 1 case report 17 and 2 case series39,41). The overall quality of case reports was rated as good (59%) or average (41%). None of the reports fell in excellent or poor category. The adherence of the reports to the individual items of the CARE checklist is depicted in Figure 2; all reports showed adherence to patient information, clinical findings, timeline, therapeutic intervention, and discussion; however, none exhibited adherence for two subitems, namely patient perspective and informed consent. All but one (appropriate statistical analysis) quality criteria on the JBIs tool were adhered to in most case series (Figure 3).
Among observational studies, all the three studies had scores >6 on the Newcastle Ottawa Scale and were, therefore, categorized as of good quality.
Percentage Adherence to Individual Subitems of CARE Checklist Among the Included Case Reports
Adherence to Various Criteria of JBIs Tool Among the Included Case Series

Study Outcomes
Table 2 enlists various types of CSBs reported with zolpidem in the literature.
Types of Complex Sleep Behaviors Reported with Zolpidem in Literature
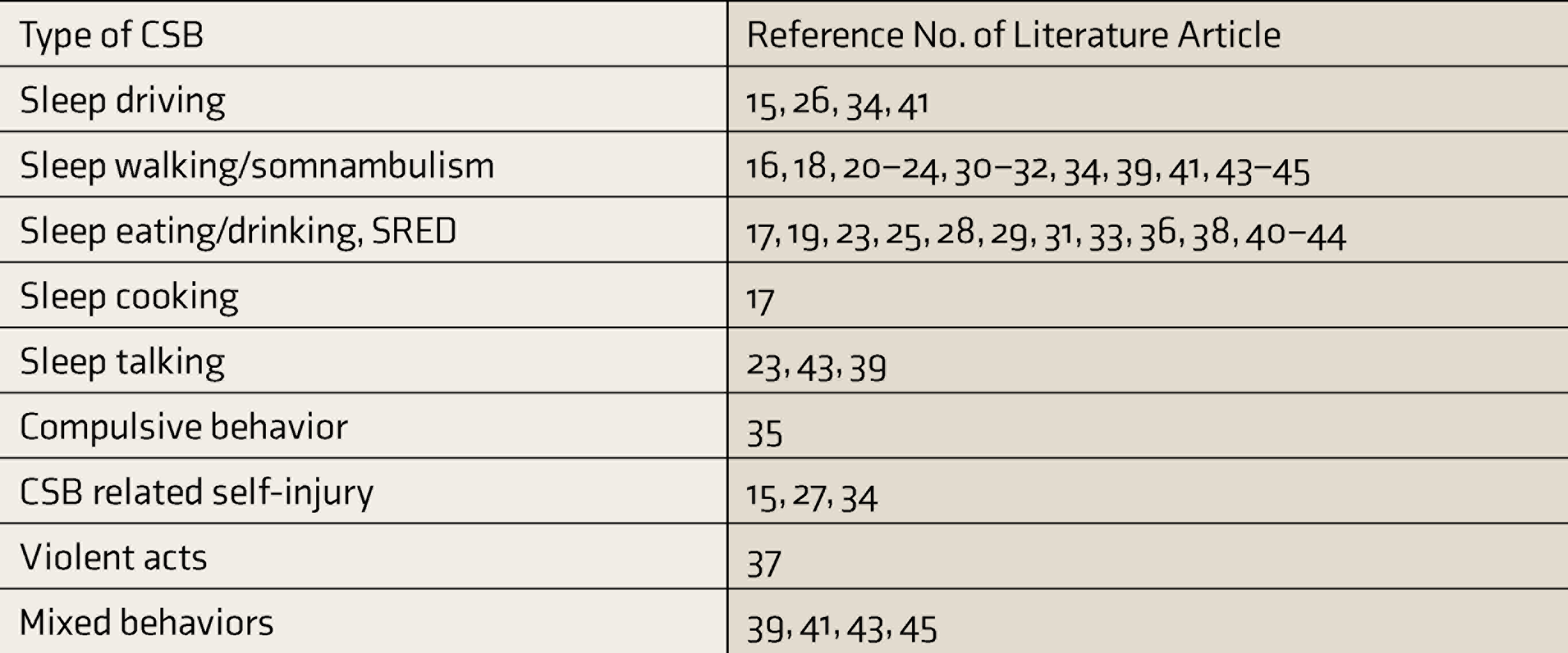
CSBs: complex sleep behaviors, SRED: sleep-related eating disorder.
Causality Assessment
Among the included case reports and case series, the causality association of zolpidem with CSBs was ascertained as mainly probable (69/79, 88%) and possible (8, 10%). Very few reports had certain (1, 1%) and unlikely (1, 1%) causal association with zolpidem intake.
Discussion
Various types of CSBs have been reported with zolpidem. Major variables identified as risk factors for zolpidem-induced CSBs are female sex, advanced age, and >10 mg daily dose. 48
A higher incidence of CSBs in females was reported in case reports (more than 60%) and a cross-sectional study. 46 One possible explanation is the slower elimination of the drug in females, based on which USFDA has recommended a 50% dose reduction in females. Chen et al., however, did not observe a similar pattern of occurrence of CSBs. 45 In another study, Greenblatt et al. concluded that the lesser dose recommendation in women needs scientific evidence and may, in fact, lead to underdosing and inadequate treatment of insomnia in women. 49
This review found very few cases of CSBs reported in the elderly. Hence, there is a lack of evidence on advanced age as a risk factor for zolpidem-induced CSBs.
Dose relatedness of zolpidem-associated CSBs has been hypothesized. At low doses, zolpidem exhibits selective binding to α1subunit of GABAA receptor, which is responsible for its hypno-sedative and amnesic properties. At higher doses (>10 mg), the drug displays additional binding to α2, α3, and α5 subunits (like benzodiazepines), leading to pharmacological effects and complex behaviors as noted with benzodiazepines.45,50 Chen et al. reported that a higher dose of zolpidem (>10 mg daily) was associated with CSBs in only adult patients (20–55 years) and not in older adults (≥65 years); alcohol consumption was also observed as a risk factor. 51 Nevertheless, there is an imminent demand for evidence-based medicine to ascertain the association of such neuropsychiatric adverse events with dose and blood concentration levels of zolpidem.
Some role of drug interactions has also been implicated in increasing the risk of CSBs with zolpidem. Since zolpidem is mainly metabolized by CYP3A4, concomitant administration of CYP3A4 inhibitors may increase the risk of CSBs with zolpidem. 48 Paradis et al. reported two cases of zolpidem-associated homicide. 39 Both the patients were concomitantly taking paroxetine, and it was hypothesized that, being highly protein-bound, SSRIs like paroxetine might cause displacement reactions and elevate blood levels of free zolpidem. Apart from pharmacokinetic interactions, pharmacodynamic drug interactions (concomitant administration of two or more GABAergic agents) may also increase the risk of CSBs with zolpidem. Sattar et al. reported somnambulism in a 47-year-old male taking valproate and zolpidem. 23 Valproic acid has agonistic activity at GABAA receptor, and the additive GABAergic effect of zolpidem and valproate was postulated to be the cause of somnambulism. Valproate, in addition, has pharmacokinetic interaction with zolpidem by virtue of its CYP-inhibiting property.
The pathophysiology of zolpidem-induced CSBs remains unclear. A postulated hypothesis is that after taking zolpidem, a patient may inadvertently or intentionally remain awake and experience disinhibited behaviors or hallucinations, with associated amnesia. 5 Another possible explanation is that there is a partial arousal from sleep under the influence of the drug, during which the nocturnal event occurs, with no subsequent recall due to the associated amnesia. 25 A mechanism hypothesized for zolpidem-induced somnambulism is the suppression of REM sleep 52 and increased duration of slow-wave sleep, serving as a predisposing factor for somnambulism. 53 It has been suggested that zolpidem produces a physiologic state during slow-wave sleep that manifests clinically as somnambulism. 22 An increased susceptibility for zolpidem-induced somnambulism has also been suggested in subjects with a history of sleepwalking during childhood. 24 There is also the “theory of cerebral pattern generators (CPGs),” which are postulated to be neuronal collections in the central nervous system and have a role in controlling innate motor behaviors like walking, driving, eating, etc. It is hypothesized that diffuse binding of zolpidem in the cortex releases CPGs, leading to disorders of arousal such as sleepwalking, SRED, and sleep driving.54,55
A “serotonergic model” for hypno-sedative-induced CSBs has also been proposed. 56 According to this, increased activity of serotonergic neurons in the dorsal raphe nuclei is associated with short periods of microarousals during slow-wave sleep. The binding of hypno-sedatives to GABAA causes an initial activation of the receptors, followed by their desensitization. As a result of the desensitization, the spontaneously firing serotonergic neurons display a postinhibitory rebound phenomenon, causing a transient increase in serotonergic activity, which in turn manifests as an overt behavioral reaction. Later, autoregulatory mechanisms lead to a compensatory decrease in the serotonin release, which is responsible for the termination of the serotonin-dependent behavior. Hence, the time window for CSBs comprises the delay between the GABAA receptors’ desensitization and the compensatory decrease in the serotonin levels. Factors like individual variability in receptor desensitization, serotonin autoregulatory mechanism, and drug pharmacokinetics may determine the risk of developing CSBs. For example, if the concentration of a hypno-sedative drug is very low during the time window for CSBs, there is an increased serotonergic activity due to persistent receptor desensitization, leading to CSBs. On the other hand, higher drug concentration during this period may surpass the receptor desensitization and suppress serotonergic neurons, preventing CSBs. Hence, the ER formulation of zolpidem, producing a higher drug concentration during the 3–6 hours postadministration period, is assumed to be associated with a lower incidence of CSBs. 56 In our literature search, we observed only four cases of CSBs associated with the ER formulation of zolpidem. Chiang et al. 38 reported SRED associated with ER preparations of zolpidem in two elderly females. Both the patients had been taking the immediate-release (IR) form of the drug (10 mg daily) for a few years; due to inadequate sleep maintenance, they were shifted to the ER form of zolpidem (12.5 mg daily), after which they developed amnestic SRED. In both patients, the condition totally resolved after switching back to the IR form of the drug. A possible explanation for such behavior, as given by the authors, is the higher peak blood levels and greater blood levels achieved later in the night with the ER formulation. This, however, contradicts the hypothesis explained by the serotonergic model. The evidence for an association of zolpidem-induced CSBs with the formulation of the drug, hence, needs to be substantiated by a systematic comparison between ER and IR forms of the drug.
Limitations
A major limitation of our review is the extraction of data from observational studies and spontaneous reports, due to the nonavailability of any randomized controlled trials relevant to the study objective. Due to the lack of any data on the number of patients exposed, the exact incidence of zolpidem-associated CSBs could not be commented upon. Also, there is a paucity of clinical information on concomitant disorders, such as epilepsy, restless legs syndrome, depression, and dementia, which may also be associated with such neuropsychiatric adverse events. Information on concurrent consumption of alcohol and other recreational or illicit substances, too, is especially crucial because of their potential interference with sleep architecture.
Conclusion
The presence of a clear causal association of zolpidem with CSBs, when used therapeutically, demands the need for physicians and healthcare professionals to be vigilant while prescribing this drug. Certain risk factors, including female sex, advanced age, a higher dose of the drug, and the possibility of drug interactions, need to be duly considered to reduce the incidence of such adverse behaviors. Also, well-designed prospective randomized clinical trials need to be planned to provide quality evidence in this direction.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
