Abstract
Background:
Sleep state misperception (SSM) is seen among patients with obstructive sleep apnea (OSA) as well as those having insomnia. Moreover, OSA and insomnia can also be comorbid. This study aims at finding the proportion of SSM and “Comorbid Insomnia with OSA” (COMISA) among patients of OSA and chronic insomnia. Macroachitecture of sleep was also compared across groups.
Methods:
This study utilized the retrospective laboratory and medical records of two groups of patients: chronic insomnia and OSA. Sleep disorders were diagnosed according to standard criteria. Daytime sleepiness was examined using the Epworth Sleepiness Scale. Diagnosis of SSM was based on the difference between subjective and objective sleep onset latency (Subjective SOL > 1.5 × Objective SOL).
Results:
Sixteen adult subjects were included in each group. Based on the difference between subjective and objective sleep onset latency, SSM was reported by 62.5% subjects of chronic insomnia and 56.25% subjects having OSA (OR = 1.29; 95% CI = 0.31–5.33; P = 0.79). The proportion of COMISA in subjects with chronic insomnia was 18% and among subjects with OSA, it was 43%. Effect size for the proportion was calculated as odds ratio (33.96; 95% CI = 7.48–154.01; P < 0.0002). Thus, the odds for COMISA were higher among subjects with OSA than those with chronic Insomnia. The three groups (OSA, COMISA and Chronic Insomnia) were comparable with regard to the macro-architecture of sleep.
Conclusion:
SSM is common among subjects with OSA and chronic insomnia. COMISA was commoner among patients with OSA compared to those with chronic insomnia. Macro-architecture of sleep is comparable among groups.
Sleep state misperception is common among subjects having OSA and chronic insomnia. Patients having OSA and treatment-refractory chronic insomnia should be examined for the presence of other sleep disorders. Patients having OSA, chronic insomnia, and COMISA have comparable macro-architecture of sleep.Key Messages:
Insomnia and obstructive sleep apnea (OSA) constitute the top two most common sleep-related disorders. These disorders are frequently comorbid, necessitating that a clinician should be able to recognize both. 1 Insomnia is primarily diagnosed on the basis of clinical history, while for the diagnosis of OSA, overnight polysomnography (PSG) is required in addition to clinical features. 2 According to the prevailing guidelines, PSG is not routinely indicated in the diagnosis of chronic insomnia (CI), leaving space for missed diagnosis of comorbid insomnia with OSA (COMISA).3–5
Many patients having insomnia and OSA misperceive their sleep as wakefulness. 6 This is called sleep state misperception (SSM) or paradoxical insomnia, which was considered a distinct clinical phenotype of insomnia until 2014. 7 These patients often present with complaints of nonrefreshing sleep or dreamy sleep or “no sleep at all” rather than the difficulty in initiating or maintaining sleep or waking up earlier than the desired wake time.
Polysomnography is a useful tool to identify SSM, where a discrepancy between subjective and objective sleep parameters becomes evident in these patients. SSM can be reported by patients with insomnia as well as OSA because of hyperarousal and recurrent microarousals, respectively. Choi et al. 6 reported that subjects with insomnia differ from those with OSA on the demographic variables and sleep architecture. However, another study reported contradictory results wherein no difference was found with regard to the microarchitecture of sleep between subjects with OSA and insomnia. 8 Khan et al. 9 extended the scope by comparing sleep architecture between subjects with varying severity of insomnia and reported that sleep architecture was comparable among groups.
However, literature addressing this issue is scanty, and after a careful search, we could find only three studies that compared sleep architecture between subjects with insomnia and OSA.6, 8, 9 A recent systematic review and meta-analysis suggested that though a decent amount of literature is available from other regions regarding COMISA, literature from the South-East Asia region is scarce.
5
There is a need for filling this void considering the difference in body-mass index, craniofacial architecture, and sleep patterns of the Asian population vs. Caucasian population.
5
Moreover, these studies have diagnosed insomnia using
Thus, this study was planned to find out the proportion of subjects with SSM and COMISA among subjects with CI and OSA. The further aim of the study was to compare sleep architecture between patients with OSA, CI, and COMISA.
Materials and Methods
Institutional Ethical Committee approved this study. Because of the retrospective nature of this study, consent was not required to investigate the data obtained for clinical indications and stored in our database. In the present study, the sample size was not calculated a priori because polysomnography in the patients with CI is indicated only among subjects not responding to treatment.3, 4
Sleep studies of (a) patients fulfilling clinical criteria for CI, not responding to usual medical and behavioural management, and (b) sleep studies of subjects meeting clinical and polysomnography criteria for OSA according to International Classification of Sleep Disorders 3rd edition (ICSD-3) were included in this study. 2 Records of subjects who underwent a sleep study for treatment-resistant CI between October 2019 and March 2020 were considered. We included an equal number of patients having polysomnographically confirmed OSA from the same period, selecting consecutively, starting from the most recent PSG study, backward. However, subjects who had clinical or polysomnography diagnosis of other sleep disorders (i.e., restless leg syndrome, periodic limb movements during sleep, or parasomnias) or other neurological or medical disorders that could have influenced sleep architecture (e.g., congestive heart failure, myopathy, dementia, and Parkinson’s disease) at the time of the sleep study were excluded. Similarly, subjects diagnosed with substance use disorders (including hypnotics) and who have had withdrawal symptoms within six months of study were also excluded.
Sixteen polysomnography studies of subjects with CI could be identified abiding the criteria mentioned above during the study period. We reviewed the patients’ case record forms for gathering the necessary subjective complaints.
Collection of Clinical Data
Demographic and anthropometric data gathered at the time of attending the sleep study were collected from the patient’s record form. It included age, sex, body mass index, clinical diagnosis, presence of other medical disorders or substance use disorders, daytime sleepiness, and subjective parameters of sleep after the diagnostic polysomnography. Past medical and treatment history was confirmed from the available medical records.
Diagnosis of Insomnia
Insomnia was diagnosed following the ICSD-3 criteria that include difficulty in falling asleep, difficulty in maintaining sleep, early morning awakening, or a combination of these that occurs three or more nights a week along with daytime symptoms, for example, heaviness of head, irritable mood, poor concentration, and fatigue, to name a few. 2 For the present study, records of patients diagnosed with CI, that is, symptoms persisting beyond three months, were included. 2 The diagnosis was based on the report from the patients themselves and corroborated by the bed partner.
Obstructive Sleep Apnea
OSA was diagnosed following the ICSD-3 criteria that define it by the presence of night-time clinical symptoms, for example, snoring, witnessed pauses in breath, daytime clinical symptoms, fatigue, and sleepiness, along with apnea-hypopnea index (AHI) greater than five as measured by polysomnography. 2 If the AHI is greater than 15, it can be diagnosed even in the absence of daytime symptoms. Clinical diagnosis was based on the report from the patients themselves along with corroboration by the bed partner, and it was confirmed by polysomnography.
Excessive Daytime Sleepiness
Excessive daytime sleepiness was measured using Epworth Sleepiness Scale (ESS). 12 It contains eight items that are scored on a three points Likert scale and assess the propensity to fall asleep during daily activities in the daytime. A score >10 is indicative of excessive daytime sleepiness. It has a test-retest reliability of 82% and internal consistency of 88%. 12
Diagnosis of COMISA
COMISA was diagnosed when patients with CI met the criteria for the diagnosis of OSA and vice-versa. 2
Diagnosis of SSM
All subjects were asked about subjective sleep quality (compared to their usual sleep: as usual/worse/better) in the morning after diagnostic PSG. They were also asked about subjective sleep onset latency (SOL), the number of awakenings at night, and time to fall asleep after the awakenings, if any. The responses of the patients were kept in their medical record forms, which were later accessed for the collection of these data. The discrepancy between objective (PSG) and subjective numbers in any of the following parameters, SOL (Subjective SOL > 1.5 × Objective SOL), was considered as SSM. 13
Overnight Attended Polysomnography
Overnight attended PSG has been done after the patients were given adequate wash-out period from any of the hypnotic medication/ medications with hypnotic potential were taking. PSG was started after providing time to acclimatize to sleep laboratory (Somnoscreen, Somnomedics GMBH). All parameters recommended by the manual for scoring of polysomnography data were recorded. 14 The sensitivity of channels, sampling rates, and filters for all channels were set according to prevailing guidelines. 14 The sensitivity of electroencephalogram (EEG) was 7 microV/mm, whereas that of electrooculogram (EOG) and electromyogram (EMG) were adjusted according to the individual subject. The High Frequency (HF) filter and Low Frequency (LF) filter for EEG were placed at 35 Hz and 1 Hz, respectively; for EOG and EMG, the HF and LF filters were set at 10 Hz and 100 Hz, respectively. The sampling rate was 200 Hz for all three parameters. 14
Electrophysiological sleep parameters included frontal (F3/M2 and F4/M1), central (C3/M2 and C4/M1), and occipital (O1/M2 and O2/M1) EEG; right and left side EOG; and sub-mentalis (right and left) EMG. Limb movements were observed using anterior tibialis EMG from both legs (leg EMG). Cardiac electrical activity was observed using lead II electrocardiogram (ECG). Patients were tested while breathing room air. Airflow was detected using thermistors and a pressure transducer. Respiratory motion was detected using chest and abdomen respiratory inductance plethysmography (RIP) belts. Arterial pulse oximetry was measured using an oximeter set in fast response mode using a finger probe averaging three seconds. Body position was assessed using a body position sensor. Manual scoring of raw data was done following guidelines of the American Academy of Sleep Medicine. 14 Each study was first scored by one of the two authors (KK and GS), and later on, it was jointly scored by RG and LKS. Any difference of opinion was discussed and sorted out according to the manual. 14 During attended PSG, only sweating artefacts and major body movement artefacts were identified in the recordings. Sweating artefacts were dealt with by raising the low-frequency filter to 1 Hz, and the epochs containing major movements were scored according to American Academy of Sleep Medicine (AASM) guidelines. 14
All indicators and indices recommended by the manual for scoring of polysomnography data were calculated. 14 Polysomnography provided the following data: total sleep time (TST), SOL, wake after sleep onset (WASO), time spent awake after the first epoch of sleep till the end of recording, sleep efficiency (SE), arousal index (AI, number of micro-arousals per hour of sleep), number of awakenings (major arousals during sleep study), the proportion of different sleep stages (N1, N2, N3, and rapid eye movement [REM] sleep), apnea-hypopnea index (number of apnea/hypopneas per hour of sleep), desaturation index (number of times the blood oxygen desaturation fell below 3% from the baseline per hour of sleep), and periodic limb movement index during sleep (number of muscle twitches in anterior tibialis muscles per hour of sleep).
Statistical Analysis
Statistical analysis was done using the SPSS 23.0 (IBM Corp. Released 2015. (IBM SPSS Statistics for Windows, Version 23.0. Armonk, NY: IBM Corp). Descriptive statistics was calculated. Chi-squared test was used to find out differences between the groups. Shapiro-Wilk test for normality was applied to assess normality of continuous variables. Mean (+ Standard Deviation) and Median (Interquartile Range) of the numerical variables were calculated for variables having normal and skewed distribution, respectively. Independent sample t-test was utilized to compare the means of age, body mass index (BMI), TST, time in bed (TIB), and SE, between the OSA and CI groups. All the other PSG parameters, ESS scores, and the subjective-objective SOL were compared between these groups with the help of the Mann–Whitney U test. For comparing continuous variables in three groups (OSA, CI, and COMISA), Kruskal–Wallis test was applied, owing to the small sample size in each group.
Results
This study included 16 subjects each in the CI and OSA groups. The proportion of subjects with a history of substance use disorders was comparable between groups (two subjects in the CI group versus one in the OSA group). The proportion of subjects having other medical disorders that are not known to interfere with sleep architecture (e.g., hypertension and diabetes) was higher in the OSA group compared to CI group (6 vs. 1 subject; P = 0.01). Table 1 shows the comparison of demographic, clinical, and polysomnography parameters between subjects with OSA and CI.
Comparison of Subjects With Chronic Insomnia and Obstructive Sleep Apnea (OSA)
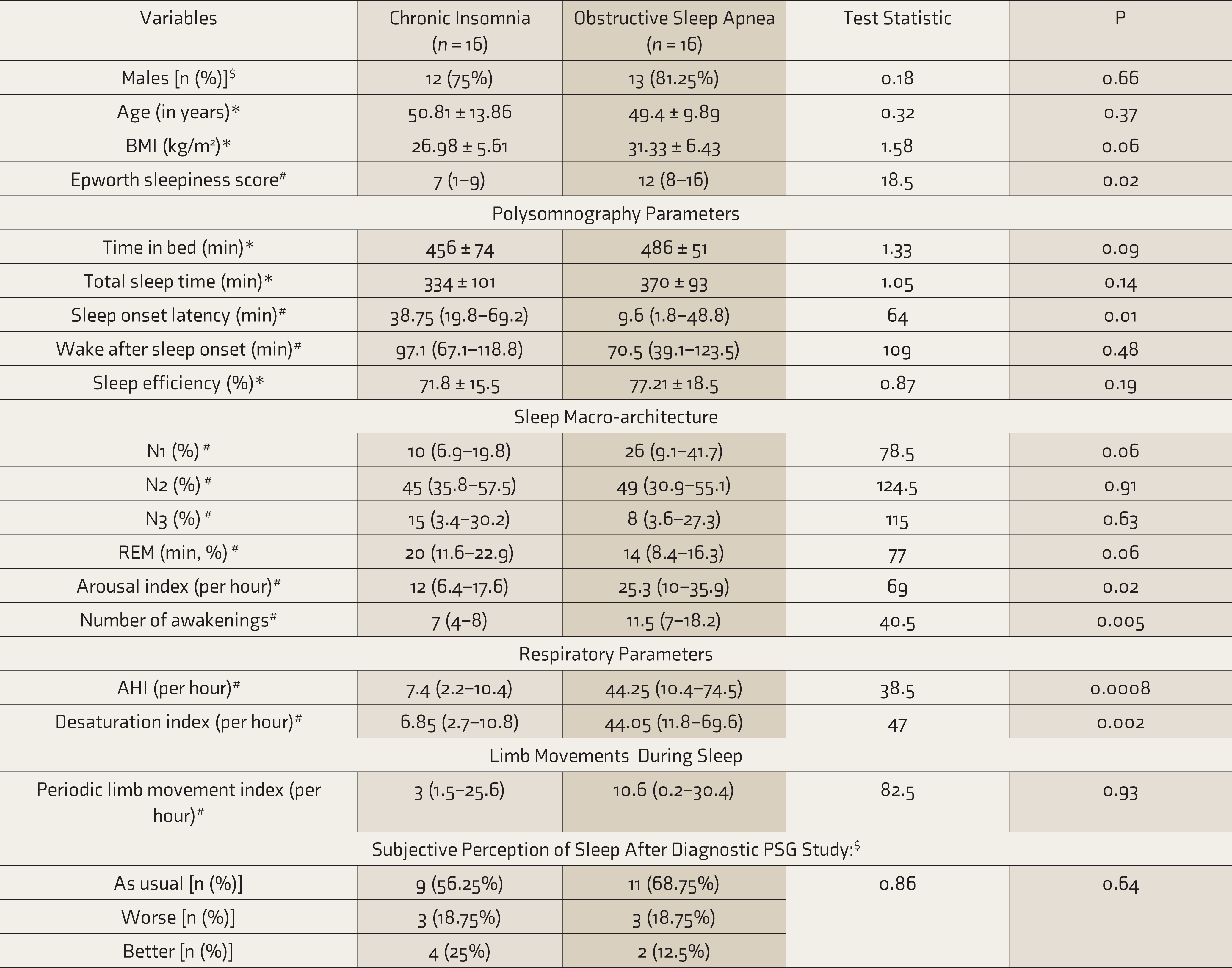
BMI: Body mass index, N1,N2, N3: Non Rapid Eye Movement sleep stages 1,2 and 3 respectively, REM: Rapid Eye Movement sleep, AHI: Apnea Hypopnea Index, * Values represented as Mean ± SD, *Independent sample
Sleep State Misperception
Based on the difference between subjective and objective SOL, SSM was reported by 62.5% subjects of CI and 56.25% subjects having OSA (OR = 1.29; 95% CI = 0.31–5.33; P = 0.79). The proportion of SSM was comparable despite greater arousal index and the number of awakenings in patients with OSA (Table 1). This finding remained unaltered even when subjects with COMISA were removed from either group (Table 2). Subjects with CI had longer subjective SOL than objective (polysomnography ascertained) SOL (P = 0.04). Similarly, subjective SOL was longer in CI group (P = 0.003). The presence of insomnia increased the mismatch between subjective and objective SOL (Table 2). SOL, awakenings, and arousal index did not influence SSM.
Comparison of Subjects With Pure Insomnia, Pure Obstructive Sleep Apnea (OSA) and Comorbid Insomnia with Sleep Apnea (COMISA)
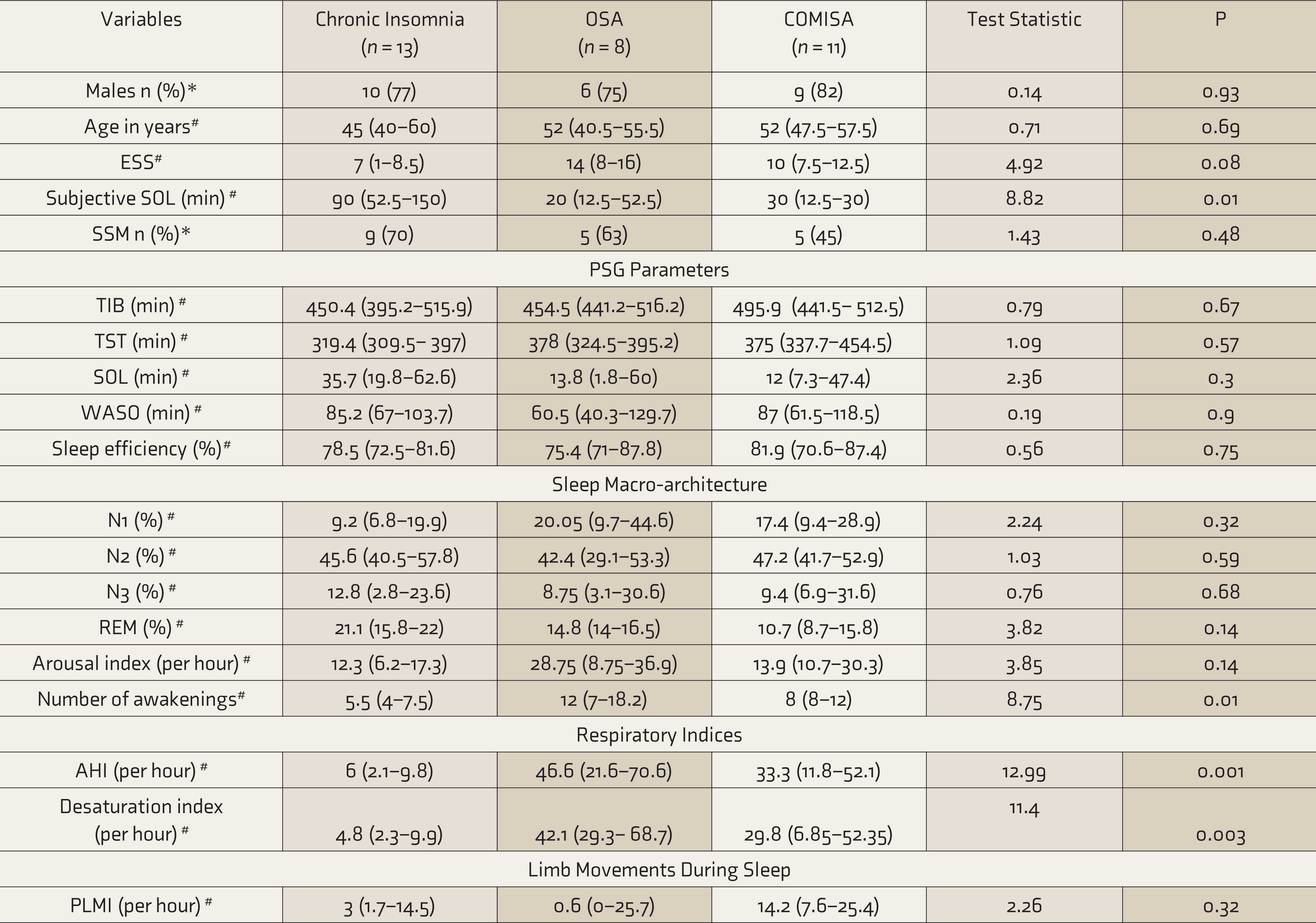
ESS: Epworth Sleepiness Scale, SSM: Sleep State Misperception, TIB: Time in Bed, TST: Total sleep time, SOL: Sleep Onset latency, WASO: Wake after sleep onset, N1,N2, N3: Non Rapid Eye Movement sleep stages 1,2 and 3 respectively, REM: Rapid Eye Movement sleep, AHI: Apnea Hypopnea Index, PLMI: periodic limb movement index, * Values represented as n (%), *Chi Square Test applied, # Values represented as Median (Interquartile range), # Kruskal Wallis Test applied.
COMISA
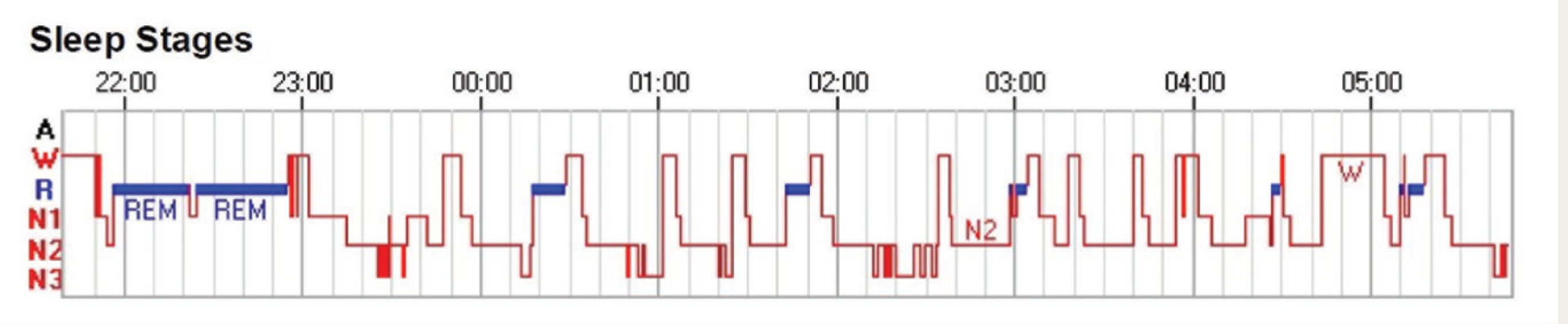
Although a total of 62% of subjects with CI had AHI >5 but <15, they were not diagnosed with COMISA owing to the absence of clinical symptoms. 2 Following the ICSD-3 criteria, the proportion of COMISA in subjects with CI was 18% and among subjects with OSA was 43% (OR = 33.96; 95% CI = 7.48–154.01; P < 0.001). 2 Thus, the odds for COMISA were higher among subjects with OSA than those with CI. The comparison of clinical and sleep-macro-architecture between OSA, CI, and COMISA groups is depicted in Table 2. Figure 1 shows the proportion of subtypes of insomnia complaints across the three groups, while Figure 2 depicts the representative hypnogram from each group.
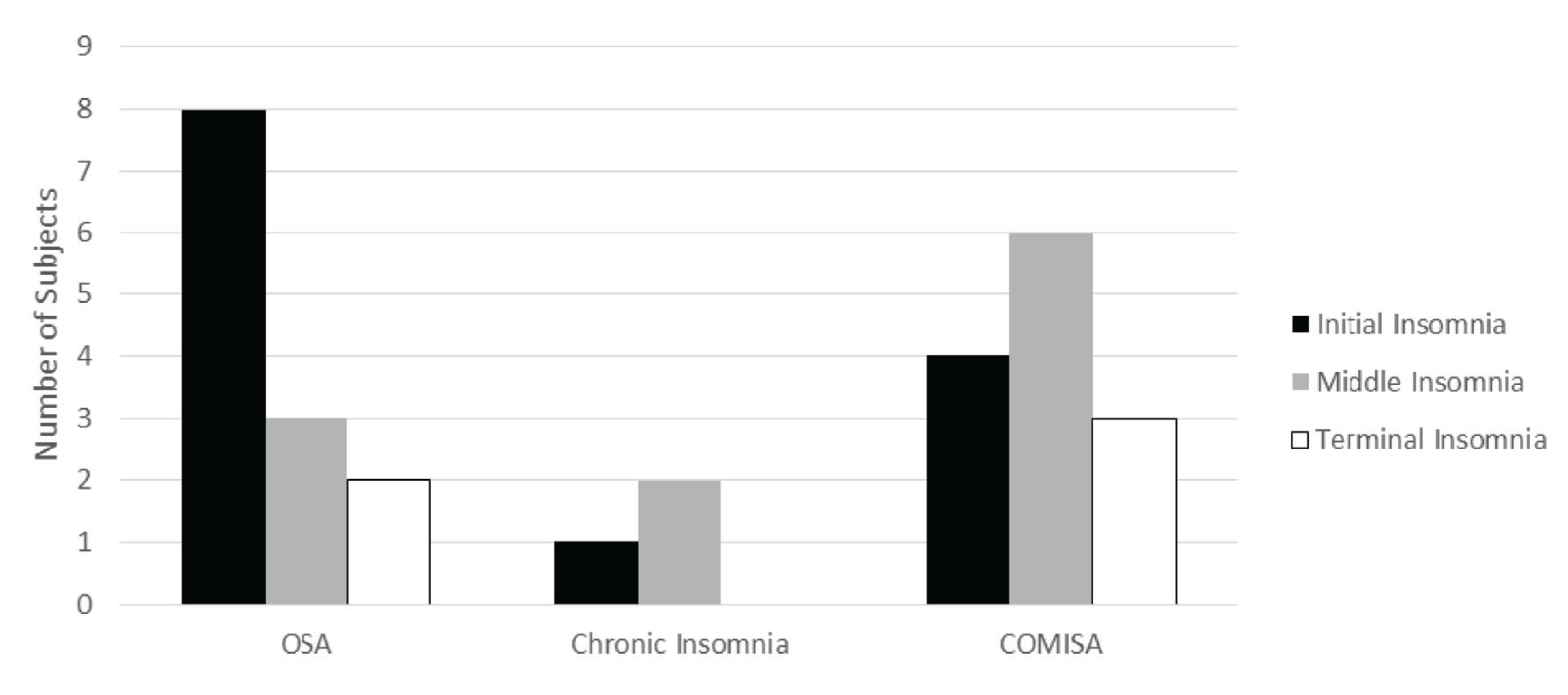
*Many parients reported more than one subtype
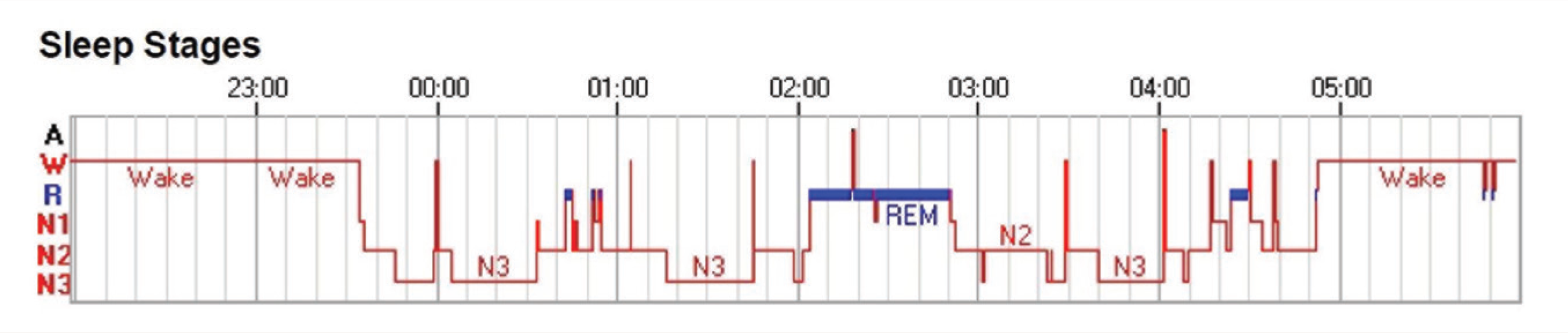
Representative Hypnogram From a Patient with Obstructive Sleep Apnea (OSA; Repeated Short Arousals and Lack of a Prolonged Period of Any Continuous Sleep Stage)
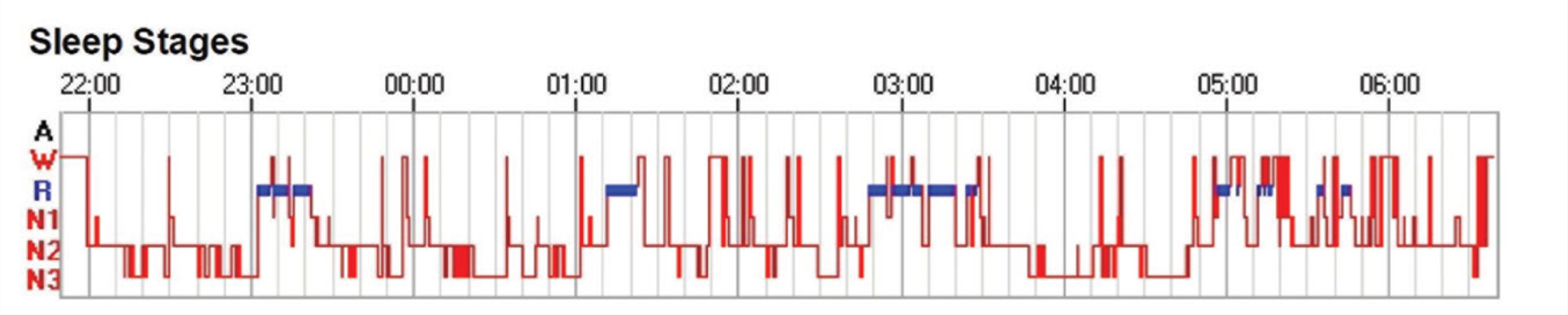
Representative Hypnogram From a Patient with Comorbid Insomnia with Sleep Apnea (COMISA; Almost Similar to That of Obstructive Sleep Apnea [OSA])
Discussion
Our results show that SSM is common among patients with OSA as well as CI. Arousals at night and SOL do not contribute to SSM. Despite having a lower number of awakenings at night and arousal index, sleep difficulty was more frequently reported by patients with CI. COMISA was more common among subjects with OSA compared to CI. Lastly, one-fifth of the patients with treatment-resistant CI had OSA, while two-fifth of subjects with OSA had CI.
Nearly half of the subjects having OSA and CI were found to have SSM. However, when subjects having COMISA were removed from both groups, the proportion increased further (Table 3). Whether these many subjects had SSM is a question worth discussing. As of now, the definition of SSM remains controversial. Castelnovo et al. 13 examined 16 definitions that have been used by different scientists and reported that none of them could provide reliable results. They reported that definitions including the discrepancy between subjective and objective TST and sleep efficiency should be preferred to those that include the discrepancy between subjective and objective SOL (as used in the present study). 13 The latter definition tends to overestimate the proportion of SSM, which could have occurred in the present study. However, using the definition containing the former parameters has multiple issues and these parameters are influenced by environmental factors, which are difficult to overcome. 13 In short, as of now, a universally applicable, acceptable, and reliable definition of SSM is not available. Considering the impact of untreated SSM and the proportion of patients having SSM as per available definitions, further research in this area is required.13, 15
The next important issue that needs to be examined is the clinical and polysomnographic variables associated with SSM. It has been argued that sleep disorders (CI and OSA) and psychiatric disorders (depression, schizophrenia, and posttraumatic stress disorder) can be risk factors for SSM. 15 Further, it has been hypothesized that SSM may be linked to arousals during sleep or continued cortical activity during sleep. 15 Some of the previous studies have attributed SSM to the “micro-arousals associated with breathing events” that occur frequently in patients with OSA. However, other studies provide conflicting data on this issue. 15 Similarly, it is considered that subjects having insomnia may have higher rates of SSM owing to hyperarousal associated with insomnia. 16 Considering these factors, we expected that proportion of SSM should be the largest among subjects with COMISA. However, contrary to the expectation, we did not find any significant change in the proportion of SSM even after removing COMISA from both groups (Table 2). Even though the arousal index and the number of arousals were higher in the OSA group than CI (Table 1), the proportion of SSM was comparable. Further, removal of subjects having COMISA from both groups (Table 2) showed that though the arousal index and the number of arousals at night were higher in the OSA group than the other two groups, still the proportion of SSM was the same. This shows that SSM is not related to micro- or complete arousals at night. Perhaps, SSM is related to the absence of “local sleep” and having “local wakefulness” that was not examined in the present study. 13 This is another area that requires further research.
A significant number of subjects (18% with CI and 43% having OSA) were diagnosed as having COMISA according to standard criteria in the present study.
2
These findings do not entirely match the available pooled prevalence of comorbid insomnia in OSA (38%; 95% CI = 15%–64%) and comorbid OSA in insomnia (29%; 95% CI = 18%–41%).
5
This could have occurred due to the differences in the definitions used to diagnose insomnia between present and previous studies. The definition of insomnia has been revised in ICSD-3, and “poor quality/ nonrefreshing sleep” has been removed, and frequency criterion (≥3 nights per week) has been added, which was followed in the present study.
2
On the other hand, studies included in the meta-analysis by Zhang et al. diagnosed insomnia using the earlier versions of
Finding COMISA is pertinent to the optimal management of patients with OSA as well as chronic unresponsive insomnia. Patients having comorbid insomnia with OSA have poor compliance to positive airway pressure (PAP) therapy and a greater proportion of suicidal ideation.17, 18 Similarly, in patients with CI, the management of the undiagnosed OSA with continuous positive airway pressure (CPAP) is known to improve insomnia. 19 The reported proportion of COMISA, together with the stated management complexities, necessitates identifying OSA and insomnia in subjects of CI not responding to treatment and OSA, respectively.
In the present study, patients with OSA reported difficulty in falling asleep (DFA) as the most common problem, while those with COMISA reported difficulty maintaining sleep (DMS; Figure 1). This is in concordance with the pooled prevalence that showed DMS as the most common complaint reported by subjects having COMISA. 5 Patients with OSA may be divided into different clusters depending upon clinical features. 20 Nearly one-third of patients with OSA mainly complain of sleep difficulties, the other third have excessive daytime sleepiness, and the rest have prominent respiratory symptoms. 20 Among subjects having sleep difficulties, the proportion of DFA has been reported as 71%, DMS as 96%, and early morning awakening as 61%. 20 Although the proportion of DFA was comparable with this data in the present study, reasons for the lower proportion of DMS are not known. 20 However, because of the small sample size, further analysis could not be done.
This study showed that polysomnography-related variables were comparable between groups (
This study has some limitations. First, the small sample size and the well-defined inclusion and exclusion criteria preclude generalization of findings to all patients. Second, owing to the retrospective nature, consecutive PSG studies were selected. This could have resulted in selection bias. Third, we could have benefitted from taking a healthy control group to compare these two groups with, giving us a clearer picture of correlates for SSM. Fourth, more subjective scores of TST and WASO could have been gathered to understand the subjective–objective mismatch. This could have resulted in having the diagnosis of SSM according to the most reliable definitions available. 13 Lastly, a number of environmental and temporal factors influence sleep. 13 Sleep is considered better in a familiar environment, while for this study, subjects were sleeping in a sleep laboratory. Although acclimatization was encouraged, there is still a possibility that some of them did not have sleep as usual (Table 1), which could have affected our results. SSM is difficult to diagnose based on a single-night study, because of the night-to-night variability of sleep.13, 22 This could have also affected our results.
Nevertheless, the study’s strength lies in having attended polysomnography, assuring inter-rater reliability in the scoring of data, and using prevailing definitions (clinical and laboratory parameters) for diagnosing OSA and insomnia.
Conclusion
SSM is common among subjects with OSA and CI. Two-thirds of patients with OSA have CI, while nearly 20% of treatment-resistant CI subjects have OSA. Although the macro-architecture of sleep appeared comparable among subjects with CI, OSA, and COMISA, further studies with a larger sample size are required to examine these findings.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
