Abstract
Background
Retrosternal gastric tube reconstruction is a common surgical technique for managing esophageal cancer, but it complicates acute type A aortic dissection repair and raises concerns about gastric tube damage.
Case presentation
A 73-year-old female, who underwent esophagectomy with retrosternal gastric tube reconstruction 6 months ago for esophageal cancer, presented with severe chest pain. Acute type A aortic dissection was confirmed by contrast-enhanced computed tomography, and emergency hemiarch replacement through a median sternotomy was performed, preserving the gastric tube without injury. The patient recovered well and was discharged after 3 weeks, showing no gastrointestinal symptoms or signs of mediastinitis.
Keywords
Background
Acute type A aortic dissection (AADA) is a life-threatening condition that requires emergency surgical treatment, which becomes more challenging when it occurs in patients with a retrosternal conduit. This report presents our initial experience with hemiarch replacement for AADA in a patient who had retrosternal gastric tube reconstruction after esophageal cancer.
Case presentation
A 73-year-old female with a previous history of esophageal cancer (T1bN0M0) underwent video-assisted thoracoscopic esophagectomy, and retrosternal gastric tube reconstruction 6 months ago. A surveillance computed tomography (CT) scan showed no cancer recurrence or metastasis. The patient presented with sudden severe chest pain and a contrast-enhanced CT confirmed AADA, with narrowing of the true lumen along the descending aorta to the abdominal section. A pericardial effusion measuring 1 cm in maximum thickness at the anterior site was identified. Additionally, a gastric tube was found adhered to the posterior surface of the sternum (Figure 1).
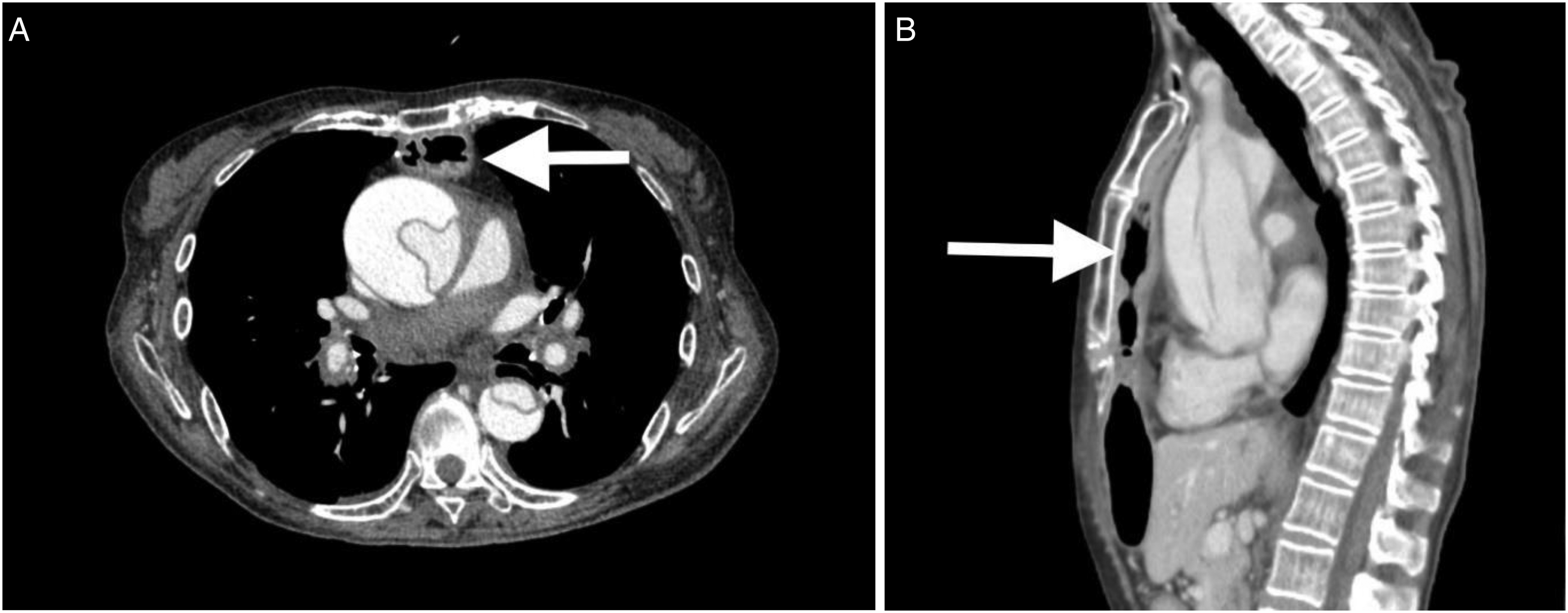
Preoperative contrast-enhanced CT scans, as shown in (A) axial view and (B) sagittal view, illustrate Stanford type A aortic dissection and the gastric tube adhered to the posterior surface of the sternum (arrow).
Given the favorable cancer prognosis and the urgency of the AADA, immediate surgical intervention was chosen. A median sternotomy was performed using an oscillating saw. Meticulous dissection and mobilization of the retrosternal gastric tube to the left side were achieved without complications, providing a clear and optimal operative view. Cardiopulmonary bypass (CPB) was established using transapical cannulation and bicaval drainage, which is implemented as a standard practice at our institution. Due to the patient's previous esophagectomy, transesophageal echocardiography was not used to confirm apex cannula position or assess aortic valve regurgitation. Nonetheless, successful CPB was achieved. After attaining deep hypothermic circulatory arrest at a tympanic temperature of 20 °C, the CPB was stopped. The primary entry tear was identified in the middle of the ascending aorta, with no re-entry site found in the aortic arch. We proceeded with our standard hemiarch replacement procedure, using the adventitial inversion technique for the distal anastomosis. Retrograde cerebral perfusion was given for cerebral protection, followed by selective antegrade cerebral perfusion to the brachiocephalic artery. Following the completion of the anastomosis, the patient was successfully weaned off CPB, and the gastric tube returned to its original position.
After four days, the patient was extubated and resumed oral intake on postoperative day 6. A follow-up CT scan revealed residual aortic dissection with a thrombosed false lumen and an increased size of the true lumen (Figure 2). Three weeks after surgery, the patient was discharged from the hospital without gastrointestinal symptoms, gastric tube complications, or mediastinitis.
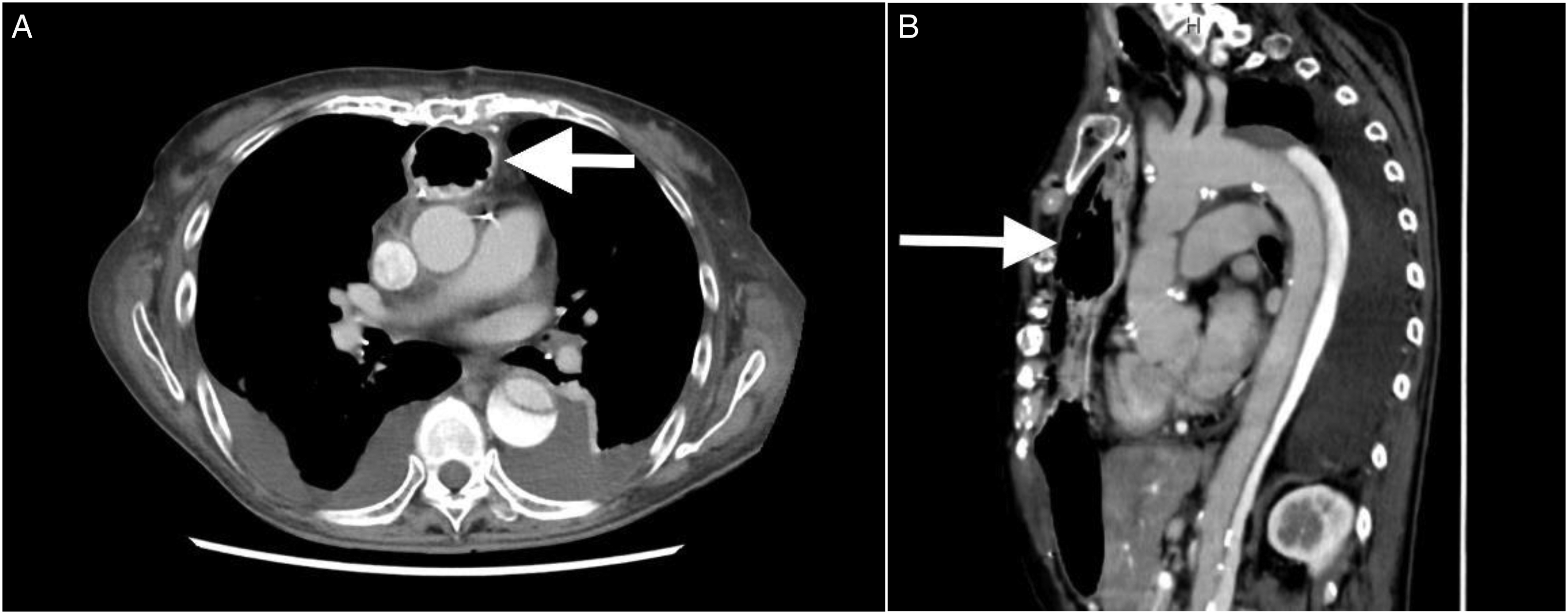
Postoperative contrast-enhanced CT scans, as shown in (A) axial view and (B) sagittal view, revealed residual aortic dissection with an enlarged size of the true lumen and the gastric tube remaining in its original position (arrow).
Discussion
Performing cardiovascular surgery on patients with retrosternal conduit reconstruction following esophagectomy presents obvious challenges that necessitate careful consideration. Assessing the patient's conduit anatomy, such as its position, length, and the presence of scarring or adhesions, is crucial in deciding the optimal surgical approach. Preoperative contrast-enhanced CT scans are particularly valuable for assessing the extent and severity of aortic dissection and providing essential information about the anatomy of retrosternal gastric tube reconstruction.
Selecting the appropriate technique to prevent conduit damage relies on the clinical diagnosis and the planned procedure. Left thoracotomy is commonly recommended for coronary artery bypass grafting to minimize conduit injury. 1 Another safe option for aortic valve replacement is the right parasternal approach. 2 Additionally, anterolateral right thoracotomy has been reported as a viable option for mitral valve replacement. 3 While median sternotomy carries a risk of conduit injury, successful repair of AADA without conduit damage has been achieved using this approach.4–8
Therefore, in this case, the median sternotomy approach was chosen due to its ability to provide a clear operative field and its suitability for various types of aortic dissection surgeries, including aortic root and/or aortic arch replacement. In this scenario, the sternum was carefully divided using an oscillating saw to protect underlying structures. Prompt repair or total removal of the gastric tube should be considered if damage occurs during surgery to prevent postoperative mediastinitis. We meticulously dissected the gastric tube from the posterior surface of the sternum using electrocautery. The dissection began from the subxiphoid region and extended towards the jugular notch. Minimal dissection of the gastric tube was performed on the left side of the sternum to create space for sternal retractor insertion. This cautious approach aimed to minimize the risk of damaging the gastroepiploic artery, which is the main vascular supply to the gastric tube usually located on the left side. As for the right side, we accessed the right pleural space while taking precautions with the staple line on the right side of the gastric tube. Following the placement of pericardial stay sutures, the conduit was secured beneath the left side of the sternum, providing an excellent view of the standard operative field (Figure 3).
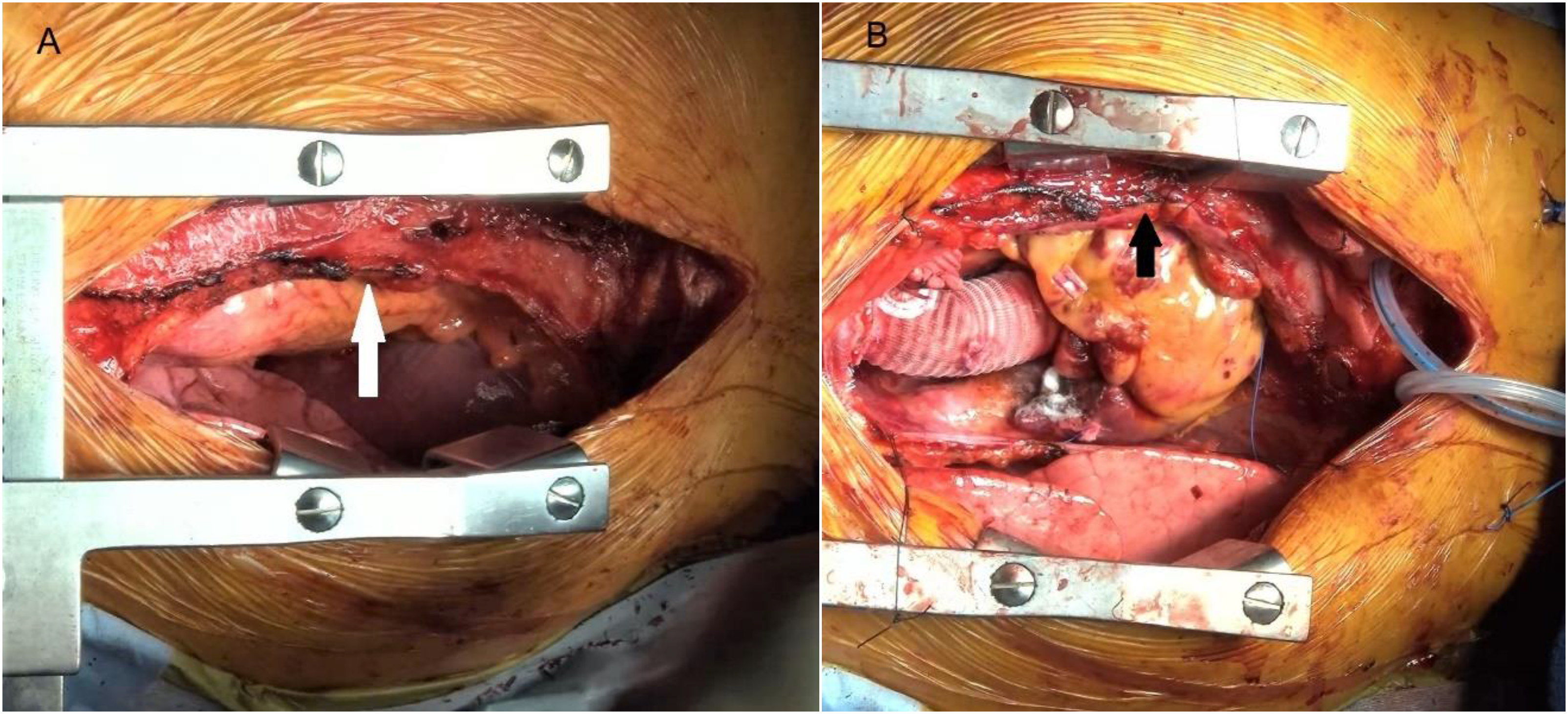
(A) The preoperative image and (B) the postoperative image depicted the secure placement of the gastric tube beneath the left side of the sternum (arrow), enabling access to the standard operative field.
Another interesting point is that we did not utilize transesophageal echocardiography due to concerns about potential rupture of the gastric tube. Preoperative transthoracic echocardiography can be considered to assess aortic valve regurgitation severity in future cases. Additionally, epiaortic ultrasound effectively guides apex cannulation, providing real-time visualization for identifying the optimal site and reducing the risk of complications during the procedure.
In summary, successful management of AADA in patients with retrosternal conduit reconstruction requires an optimal surgical approach. Careful planning, meticulous dissection, and the use of alternative imaging modalities are vital for achieving favorable outcomes in these complex cases.
Footnotes
Acknowledgments
The authors would like to extend our heartfelt appreciation and gratitude to the exceptional multidisciplinary staff of the Kawasaki Aortic Center. Their unwavering commitment, expertise, and compassionate care have played an indispensable role in providing exceptional healthcare services to our patients.
Author's contributions
NL wrote and KO, SO, and SY supervised the writing of the report. KO, TH, and NL performed the surgery. All authors read and approved the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
This study was approved by “The Research ethics committee of Kawasaki Saiwai Hospital,” Kawasaki Saiwai Hospital, Kawasaki, Kanagawa, Japan.
Informed consent
Written informed consent was obtained from the patient for scientific activity, including the publication of this case study.
