Abstract
Introduction
The incidence of pneumonectomy for lung cancer in the UK is continuing to decline in the era of minimally invasive thoracic surgery totalling approximately 3.5% of lung cancer resections annually. Literature is lacking for long-term survival of pneumonectomies. This study updates our previous results. Between 1998 and 2008, 206 patients underwent pneumonectomy compared to 98 patients between 2009 and 2018.
Methods
From January 1998 until December 2018, 304 patients underwent pneumonectomy. This was a retrospective study; data was analysed for age, gender, laterality, histology and time period.
Results
Operative mortality was 4.3% overall which was lower than the national average of 5.8%. In the last five years, there were no in-hospital, operative or 30-day mortality. During this period, 90-day mortality was 9.2%. Left-sided pneumonectomies had significantly better overall survival (3.00 vs. 2.03 years; p = 0.0015), squamous cell carcinoma (3.23 vs. 1.54 years; p = 0.00012) as well as those aged less than 70 (2.79 vs. 2.13 years; p = 0.011). There was no significant difference in survival between gender (p = 0.48). Intervention from 1998 to 2008 had significantly greater survival compared to the latter 10 years (2.68 vs. 2.46 years; p = 0.031). The Cox model shows that laterality, age, histology and time period remain significant with multivariate testing. No patient survived after 16 years.
Discussion
Our updated retrospective study has built on our previous results by reinforcing the success of pneumonectomies. The incidence of pneumonectomies is likely to decrease with the deployment of nation-wide lung cancer screening in the UK due to earlier detection.
Introduction
In the UK, annually, over 50,000 cases of lung cancer are recorded. 1 As the third most common cancer and accounting for approximately 13% of all new cancer cases, a significant mortality of just over 35,000 deaths per year is conferred. 1 Lung cancer can be broadly categorised into small cell and non-small cell carcinoma (NSCLC), with the latter consisting of adenocarcinomas (AD), squamous cell carcinomas (SCCs), carcinoid tumours and large cell cancers. 2 Although most patients will undergo chemo and radiotherapy, with such a significant physical burden, it is important to choose the best surgical approach for optimal results.
First performed in 1933, pneumonectomy is now a much rarer practice and is typically used for malignancy of central origin, infection and trauma. It has been noted to be associated with a number of complications such as bronchopulmonary fistula, pulmonary oedema, cardiac herniation and arrhythmias. 3 The former is a significant issue with regards to prognosis, with a morbidity of 25%–71% and mortality of 18%–67% identified. 4
Pneumonectomy is an extensive surgical operation and is associated with multiple risk factors. 5 This can often be complicated when tumours invade the pericardium through the pulmonary vein, pulmonary artery and left main stem bronchus or involve the fissure between lobes.
The latest National Lung Cancer Audit run in the United Kingdom in 2018 and published in 2021 highlights that 3.5% of all lung resections were pneumonectomies, a decrease from 5.8% in 2013. 6 Furthermore, survival has been cited to be lower than that of lobectomies at 30 days (94.2% vs. 98.6%) and 1 year (76.8% vs. 90.5%). 4
Recent trial results of lung cancer screening in the UK showing a significant mortality reduction of 20% with three annual low dose CT-scans. 7 With the recent implementation of lung cancer screening in UK since June 2023, 8 we can estimate that the incidence of pneumonectomies will continue to drop due to earlier detection. Furthermore, a recent meta-analysis including five lung cancer screening programmes has shown that reported harms, false positives and benign resection rates are low. 9 These outcomes have been noted to be on-par and in some cases, superior to published trials.
Overall, there is a lack of literature when it comes to long-term survival and outcomes with pneumonectomy patients. Our unit published a 15-year outcome study of patients undergoing pneumonectomy in 2016. 10 This paper will serve as an update with regards to outcomes after 20 years and visions for future practice in pneumonectomies.
Methods
Patients who underwent pneumonectomy from January 1998 to December 2018 were identified from a tertiary centre database, and the collected data was analysed for short and long-term outcome. This was a retrospective study and all patients were followed up until August 2021. All data was collected from departmental databases and hospital records. There were no inclusion criteria aside from lung cancer patients who underwent pneumonectomy for resection. Patients who underwent an extra-pleural pneumonectomy for mesothelioma were excluded.
All patients were admitted to a high-dependency unit following surgery. Furthermore, for pre-operative staging and evaluation, all patients were required to have had a recent CT or PET scan, mediastinoscopy and thoracoscopy. Surgical technique and management remained the same throughout the study period. Post-operatively, regular physiotherapy was provided twice a day by trained staff in addition to meticulous fluid management in the 48 h post-surgery.
All patients were assessed for fitness for surgery according to our departmental protocol. To measure post-operative risk, the Thoracoscore model was used prospectively and retrospectively; the mean predicted risk of mortality was 8%.
Statistical analysis
The primary endpoint of analysis was mortality. Survival was defined from the point of surgery to the date of death. Univariate survival analysis was conducted using Kaplan–Meier curves to determine differences in survival with regards to pathological and demographical factors. The log-rank test was used to identify statistical significance between curves. Our continuous variables are presented as mean ± standard deviation and median ± interquartile range (IQR). A Cox proportional-hazards model was also constructed using significant factors from univariate testing for multivariate testing. A p-value of 0.05 or less was deemed significant. The model was reported using hazard ratios with 95% confidence intervals. All statistical analysis was undertaken on R 3.6.3 using the survival and ggplot2 package.
Results
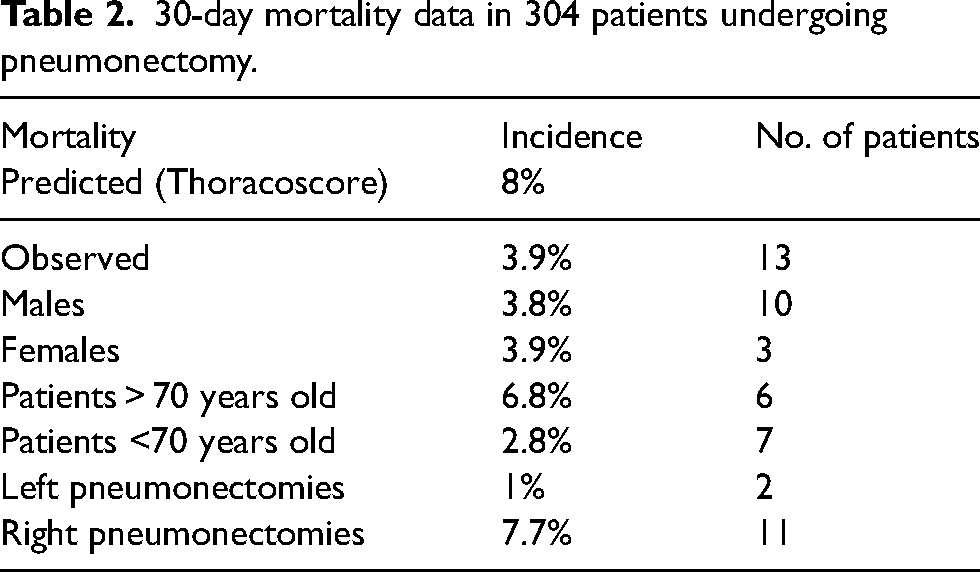
Between January 1998 and December 2018, 304 patients underwent pneumonectomy for neoplasia. A summary of pre-operative characteristics is presented in Table 1.
Pre-operative and operative characteristics of 304 patients undergoing pneumonectomy.
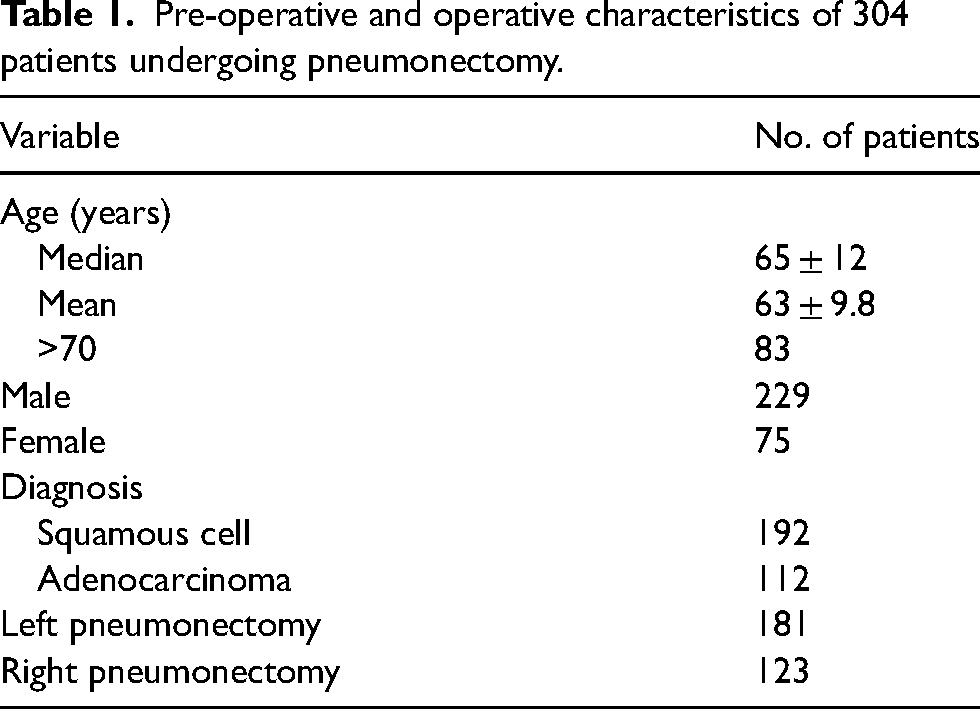
30-day mortality data in 304 patients undergoing pneumonectomy.
The overall predicted Thoracoscore was 8% which was higher than our 30-day mortality of 4.2% (13 patients in total). This is demonstrated in Table 2. The median hospital stay was 11 ± 9 days.
The overall survival rate is 0.3% at 16 years and is illustrated in Figure 1. The overall 1-, 2-, 3-, 5-, 10- and 15-year survival was 73.4%, 56.3%, 35.5%, 28.0%, 10.9% and 1.3%, respectively.
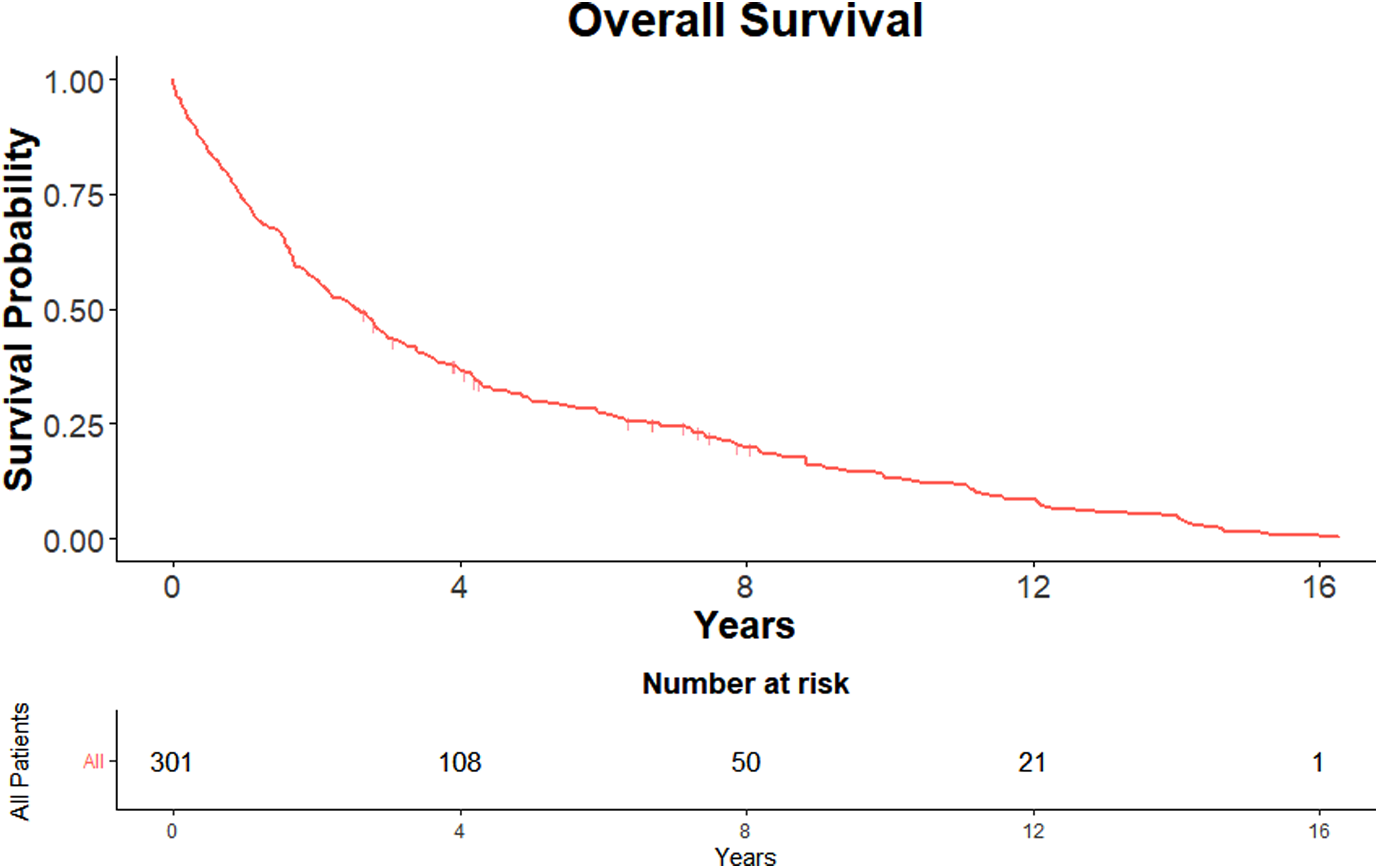
KM curve showing overall survival.
There was no significant difference in survival between gender (p = 0.48) with the overall median survival for females being 2.9 years with an IQR of 1.56–7.79 and a 95% confidence interval (95% CI) of 1.98–4.16 years, and males being 2.40 (IQR: 0.87–6.3; 95% CI: 1.87–2.96). Similarly, there was no significant difference in survival between gender at 1-, 2-, 3-, 5-, 10- or 15-years.
Furthermore, in our population, those <70 years of age had a significantly better survival (p = 0.011) with a median of 2.79 years (IQR: 0.66–7.84; 95% CI: 2.12–3.41) than those >70 years with a median of 2.13 (IQR: 0.66–4.71; 95% CI: 1.51–2.96). This significant difference persisted when comparing 5-year survival (p = 0.012), 10-year survival (p = 0.0025) and 15-year survival (p = 0.013). This is demonstrated in Figure 2.
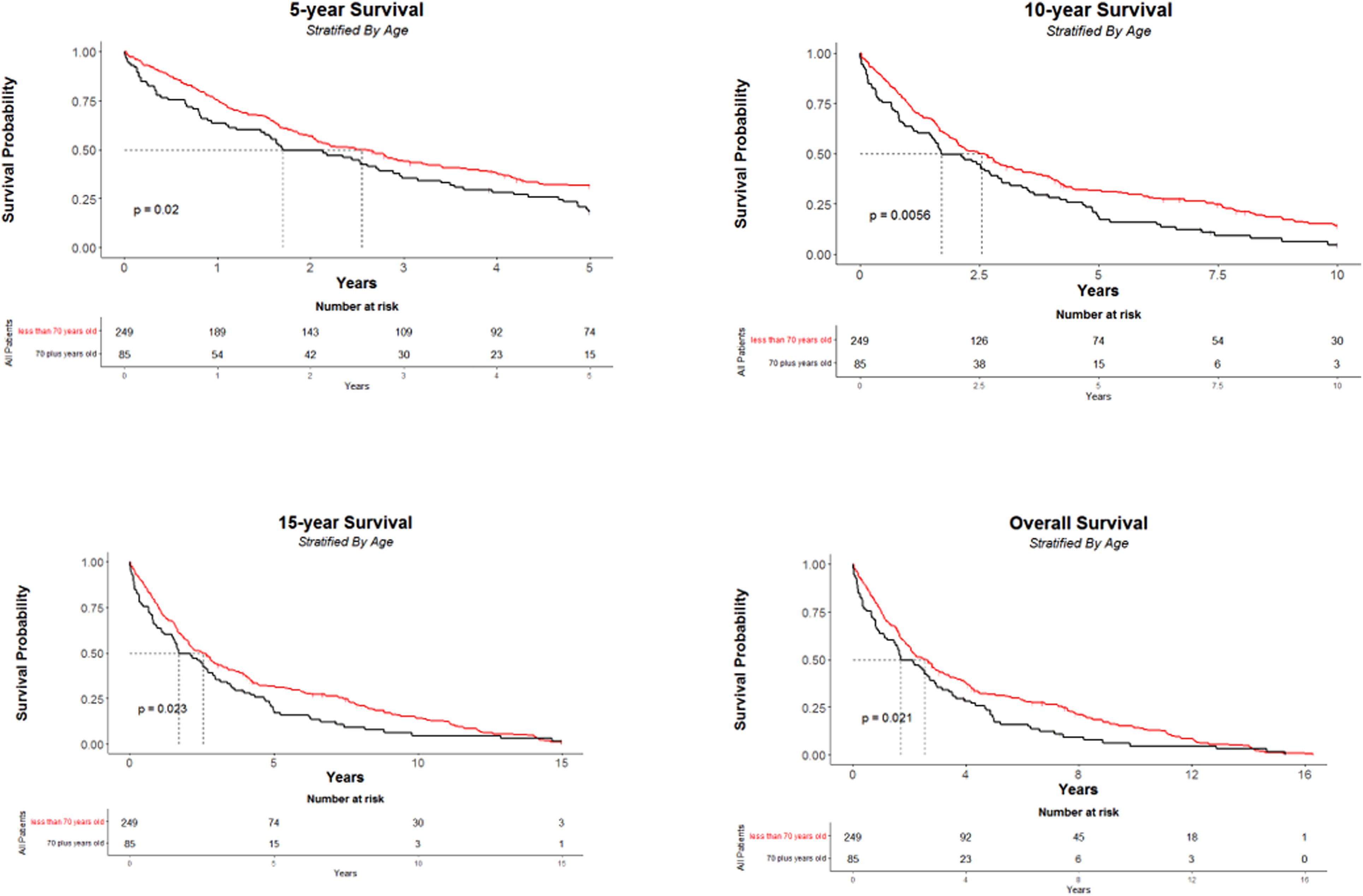
KM curves showing (1) 5-year overall survival, (2) 10-year survival, (3) 15-year survival, (4) overall survival, stratified by age.
Similar results were seen with regards to the side of the pneumonectomy. Left-sided pneumonectomies had significantly better overall survival than right pneumonectomy's (p = 0.0015) with a median of 3.00 years (IQR: 1.09–7.84; 95% CI: 2.17–3.68) compared to a median of 2.03 (IQR: 0.8–4.67; 95% CI: 1.56–2.79). This continued to be the case for 5-year (p = 0.02), 10-year (p = 0.0063) and 15-year (p = 0.0015) (Figure 3).
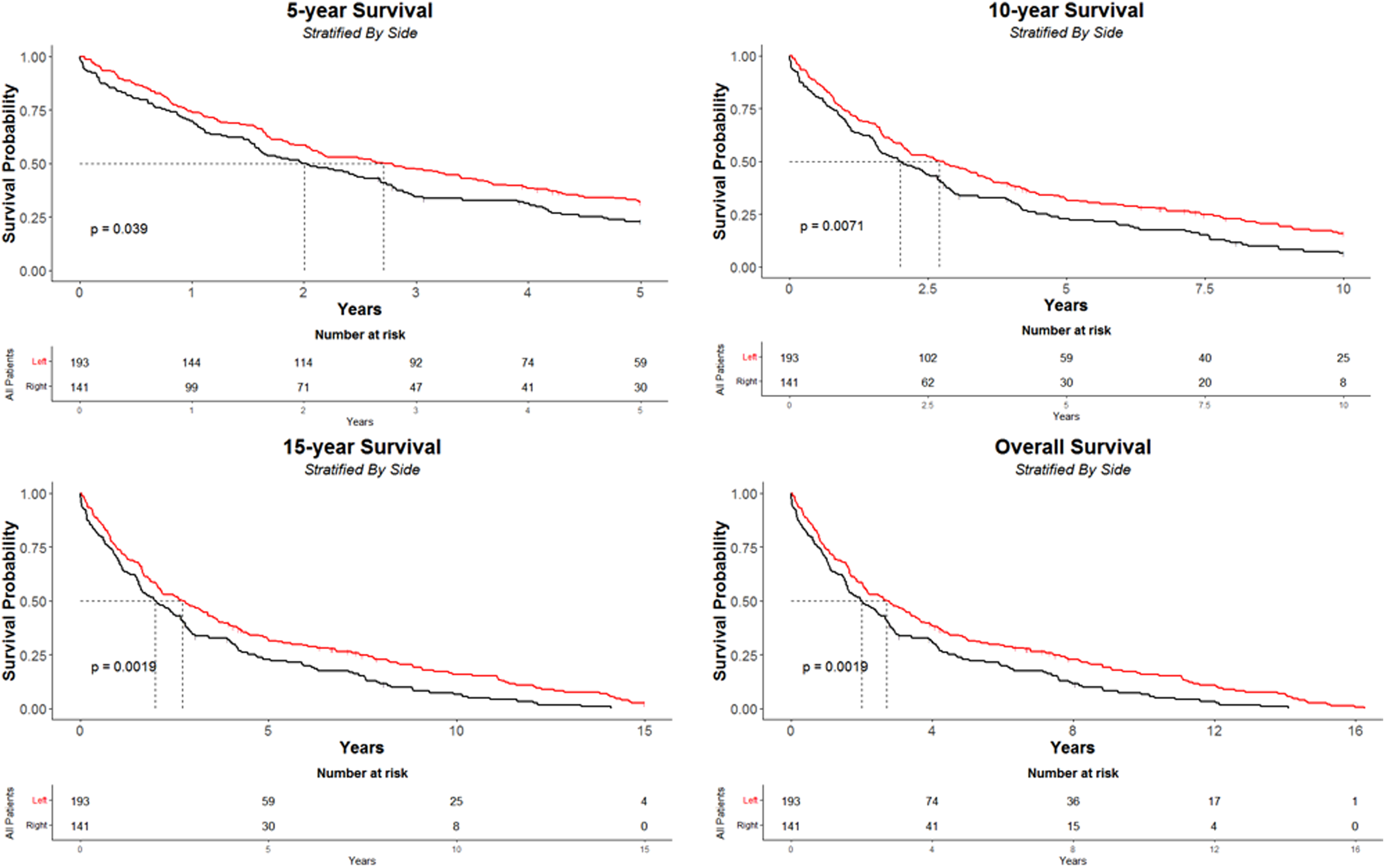
KM curves showing (1) 5-year overall survival, (2) 10-year survival, (3) 15-year survival, (4) overall survival, stratified by side.
The mortality for cases operated between the first 10 and last 10 years of the cohort was collected, that is, 1998–2008 and 2009–2018. With the former, 206 patients underwent pneumonectomy whereas 98 underwent pneumonectomy with the latter. Those who underwent intervention in the earlier time period had a significantly greater survival compared to the latter ten years (p = 0.031) with a median of 2.68 years (IQR: 0.87–7.84; 95% CI: 1.92–3.54) compared to 2.46 years (IQR: 1.01–4.26; 95% CI: 1.87–3.23). This is depicted in Figure 4.
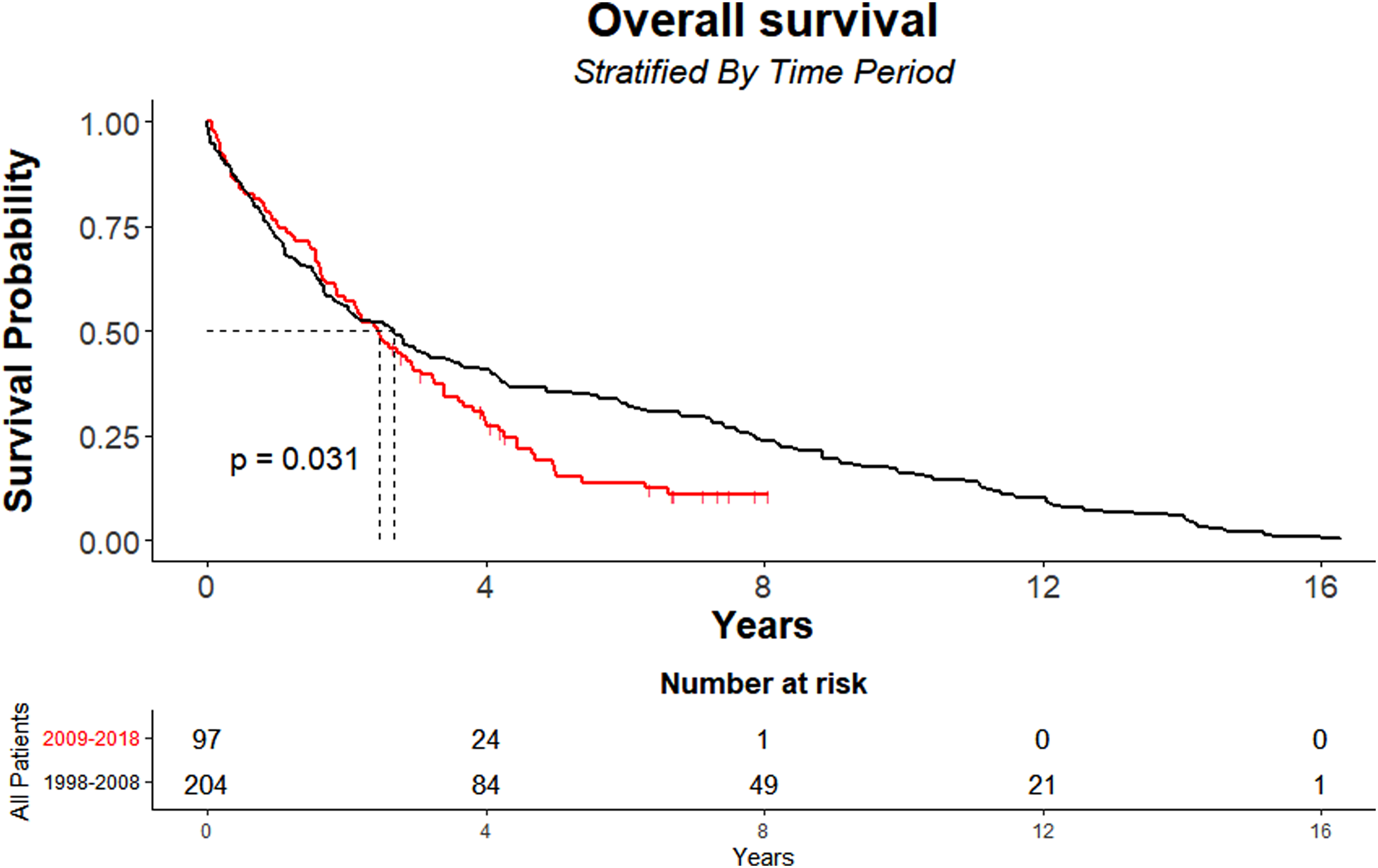
KM curves showing 10-year survival stratified by time period.
In addition, overall survival was significantly higher for those who had SCC (p = 0.00012) with a median survival of 3.23 years (IQR: 1.45–7.79; 95% CI: 2.68–4.16) compared to 1.54 years with AD (IQR: 0.66–4.05; 95% CI: 1.09–2.17). This was the case with 1-, 2-, 3-, 5-, 10- and 15-year survival (Figure 5). There was no hospital mortality observed amongst this group.
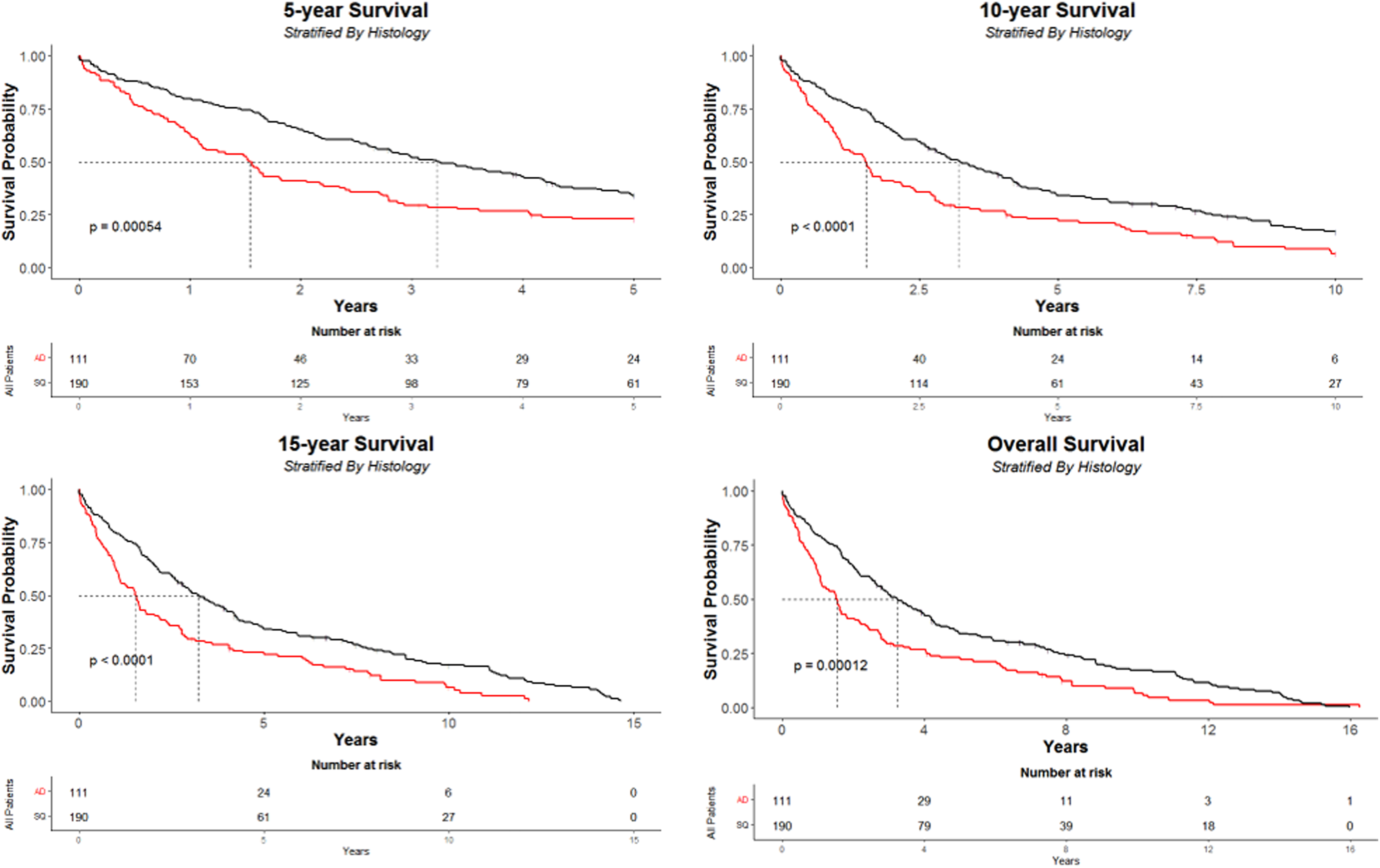
KM curves showing (1) 5-year overall survival, (2) 10-year survival, (3) 15-year survival, (4) overall survival, stratified by histology.
Age, laterality, histology and time period were placed in a multivariate Cox regression model which is presented in Figure 6. Mortality was significant for both patients above the age of 70 (1.4; 95% CI [1.08–1.83]; p = 0.012) and right-sided pneumonectomies (hazard ratio [HR] 1.44; 95% CI [1.13–1.83]; p = 0.004). The opposite was true for SCC (HR 0.6; 95% CI: 0.47–0.75; p < 0.001).
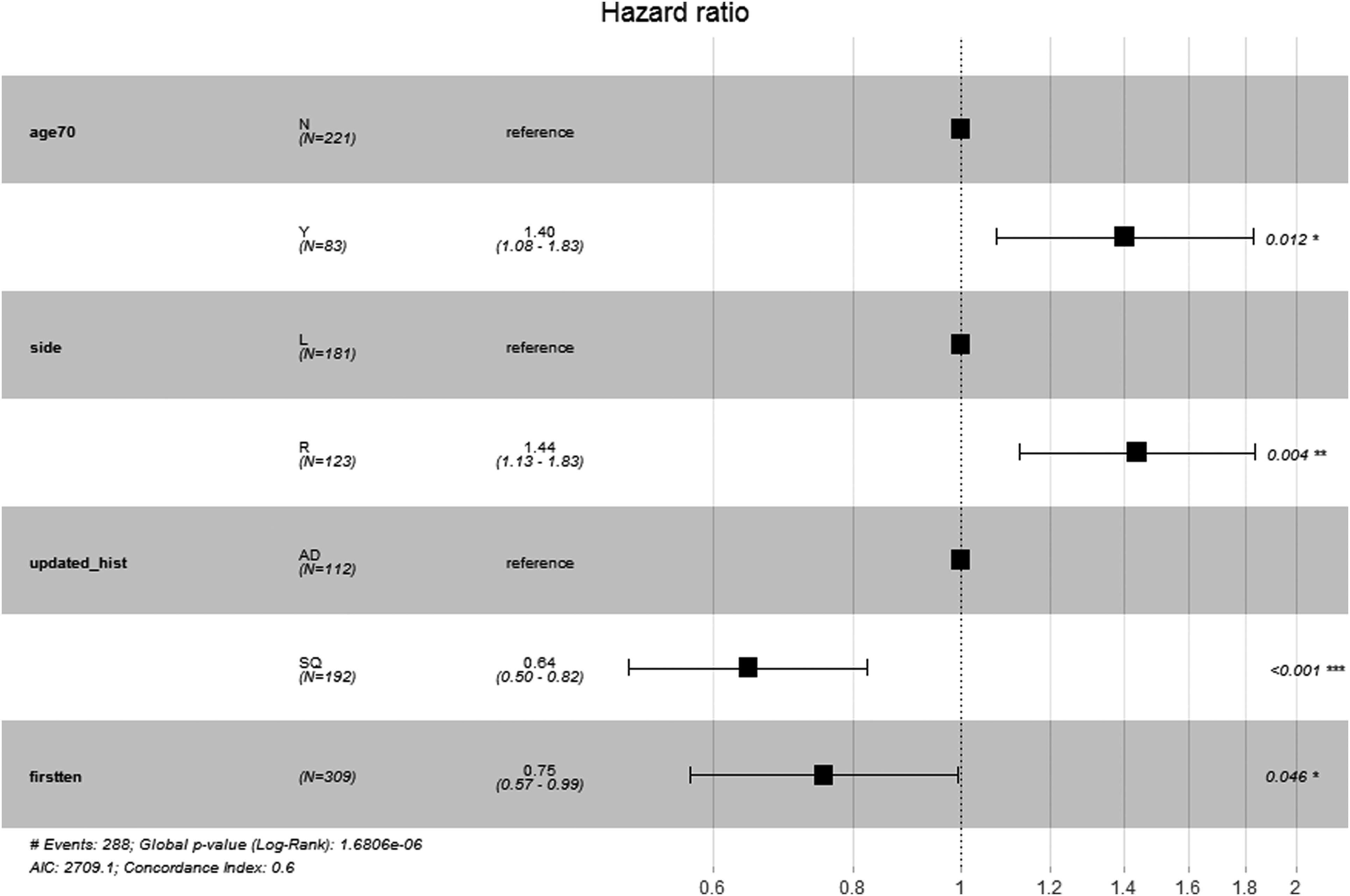
Cox-Hazard model.
Discussion
Despite the decreasing incidence of pneumonectomy as identified by the NCLA, 6 there is evidently still a need for the procedure in recent times. This is often the case with large and centrally located tumours which a lobectomy cannot excise. Our overall survival after one year was 73.4% which was lower compared to the average stated in the NCLA 2021 audit (76.8%). However, the 2021 audit was run from 2018 onwards; our unit undertook 11 pneumonectomies in 2017 and 2018 from when the study concluded. There were only two deaths totalling a survival of 81.8%, higher than the national average. Similarly, compared to our previous paper, our overall one-year survival containing patients from 1998 was 70%, thus showing an improvement. In addition, when comparing 1998–2008 group and the 2009–2018 group, one-year survival was 72.3% and 75.5%, respectively (p = 0.57).
Furthermore, there has been no 30-day mortality in the last five years in our cohort, which is a greater survival than that of the 2021 NCLA audit (94.2%). 6 Our 30-day mortality was also superior in our previous paper. 10
Similarly, survival following a left-sided pneumonectomy was significantly better (p = 0.0015). Differences in survival with regards to laterality have been documented in several studies and our results follow current literature. Yang and colleagues ran a similar and recent retrospective study comparing long-term outcomes with regards to induction therapy and laterality. 11 Akin to our study, results showed a greater mortality for right-sided pneumonectomy's and long-term survival, defined as five years, showed no difference between both sides. 11 This is mirrored by Fernandez et al., where right pneumonectomies had significantly superior survival at both one and three months as well as one and three years. 12 Likewise, Dhanasopon et al. investigated pneumonectomy survival through a National Lung Cancer Database of 79, 853 patients with NSCLC. 13 Their study concluded with significantly worse results for right-sided pneumonectomies. Furthermore, our dataset showed superior survival in left-sided pneumonectomy at 10- and 15-years.
However, as identified by Wu et al., 14 several studies have shown different results. Wang and colleagues, with a total population of 100, found there to be no difference in five-year survival between both sides. There has been speculation as to why this is the case. As identified by Darling et al. in their study retrospective study, right pneumonectomies carried an increased risk of bronchopleural fistula (BPF) formation. 15 This is thought to be attributed to a more extensive resection on the right side compared to the left. Other factors thought to contribute to BPF alongside pneumonectomy are thought to be barotrauma through ventilation to the bronchial stump,16,17 patients with COPD 18 and neoadjuvant therapy. 19 In addition, a study run by Deslauriers et al. assessing long-term cardiorespiratory function in 523 patients who underwent pneumonectomy, showed a greater decrease in FEV1 following right-sided pneumonectomy (44% vs. 34%). 20
Also following the trend of the current literature, overall survival following SCC was significantly higher compared to AD (p = 0.0001). Due to the likely central location of SCC, the majority of our patients, 192 (56.9%), had SCC compared to 112 patients (33.2%) who had AD. The difference in mortality with regards to histology has been thought to be due to the lymphatic spread of SCC compared to the systemic spread of AD. 21 Furthermore, multiple other studies have shown this association and included this as a significant prognostic factor.
With regards to the time period of operation, our results show there to be a greater survival in those operated between 1998 and 2008. This can be attributed to a few factors. Firstly, our unit has seen a shift from dual practicing to specialising in cardiac or thoracic surgery alone. Presentations for resectable lung carcinoma could potentially have been more likely to be through a pneumonectomy as techniques such as sleeve resections may not have been feasible with a broader skillset in earlier years. In later years, our unit had dedicated thoracic surgeons to optimise operative resection thus reducing the incidence of pneumonectomies. The disease stage should also be considered. In earlier years, it could be likely that the presentation on opening could be less advanced than in later years which may have been resectable with a bilobectomy or sleeve resection, but the decision was made to undertake a pneumonectomy. With an earlier stage of presentation, this could have led to a reduced incidence of mortality during the first 10 years within this cohort. Similarly, in later years, these options would have been considered and a pneumonectomy undertaken due to the advanced presentation of disease leading to a greater mortality.
Our previous paper indicated no statistical significance in mortality when comparing individual surgeons within the unit, 10 and this result maintained to be the same with current data. Although there was no difference in age, side, gender or pathology with regards to the cohorts from both time periods, there is a possibility that patients from the latter cohort elicited a more complex presentation due the increased incidence of lobectomies.
Strengths and limitations
Our study has several strengths; it is one of the few studies that provide long-term follow-up over 20 years. Furthermore, our centre is high volume and provides and accurate representation of cases and mortality. There was also no significant difference in mortality between surgeons (p = 0.089), thus allowing an accurate representation. Post-operative management following surgery was also consistent with regular thoracic physiotherapy and fluid management, being careful to avoid overload.
However, our study did have some limitations. Although our centre is indeed high volume and good indication of case variety and surgical outcome, it may not be truly representative of the UK population. In addition, we have a small sample size of 304 patients which may also have skewed our results.
Conclusion
Our updated retrospective study has built on our previous results by reinforcing the success of pneumonectomies. Our overall survival was higher than the national UK audit in 2018 at 73.4%. Furthermore, there was no 30-day mortality in the past five years at our centre compared to the national survival of 95.5%. Our study has also identified right pneumonectomies, SCC and age above 70 years old to be significant prognostic factors with regards to short and long-term outcomes. Additionally, it can also be a useful modality in emergency situations where lobar and/or bilobar resections are not possible.
With the deployment of a national lung screening programme in the UK, the incidence of pneumonectomies is likely to decrease to earlier cancer detection. Although we agree with the recommendation that centres should be audited if >5% of cases are pneumonectomies and that lobar or bilobar excision should be undertaken where possible, we have shown that pneumonectomy is an effective and safe operative strategy for both lung cancer and non-cancerous pathologies.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable
Informed consent
Not applicable
