Abstract
Leiomyoma is the most common benign tumor of the esophagus. Open thoracotomy, the traditional approach adopted for the enucleation of the esophageal leiomyoma, over the years, has been gradually replaced by video-assisted thoracoscopic surgery. However, this minimally invasive approach has limitations, such as two-dimensional vision and reduced range of motion, which have recently been overcome by technical advantages of robot-assisted surgery. In the surgical management of circumferential esophageal leiomyoma, a combined use of robotic surgery and intraoperative endoscopy may be helpful to facilitate tumor enucleation and to prevent esophageal mucosal injury during the surgical procedure.
Keywords
Introduction
Esophageal leiomyoma is the most common benign submucosal tumor of the esophagus, typically reported in middle-aged patients with a male predominance and located in lower two-thirds of the esophagus. In most cases, patients are asymptomatic while others may present nonspecific symptoms such as epigastric discomfort, dyspepsia or dysphagia, and retrosternal pain usually found in tumors larger than 5 cm. For lesions in the proximal and middle third of the esophagus, the surgical treatment of choice is a transthoracic extramucosal enucleation. 1 In this case, we present the surgical management of a circumferential esophageal leiomyoma and a safe procedure to reduce the risk of esophageal mucosal injury that is the most common intraoperative complication associated with enucleation of esophageal leiomyoma. 2
Case report
A 35-year-old man, with history of nonproductive cough lingering for several months, was admitted to our hospital for surgical treatment of esophageal leiomyoma diagnosed by endoscopic ultrasonography-guided fine-needle aspiration. A preoperative upper gastrointestinal endoscopy revealed an esophageal submucosal protrusion in the middle thoracic esophagus and a normal overlying mucosa. Contrast-enhanced computed tomography of the thorax and abdomen showed a well-circumscribed mass in the posterior mediastinum narrowing the esophageal lumen (Figure 1(a) and (b)). A robot-assisted thoracic surgery enucleation of the tumor using the da Vinci Xi System was planned. Under general anesthesia, the patient was intubated with a double-lumen endotracheal tube in the supine position and subsequently turned to the left semi-prone position. The trocars were inserted into the 5th intercostal space, on the posterior axillary line; the 7th intercostal space, on the middle axillary line; and the 3rd intercostal space, on the anterior axillary line. After retraction of the right lung toward the anterior mediastinum, the posterior mediastinal pleura was incised longitudinally, and the azygos vein arch was severed using an endostapler to better expose the esophagus. A longitudinal esophageal myotomy was then performed with the robot cautery spatula in order to visualize the tumor developed circumferentially around the esophagus (Figure 2(a)). A meticulous dissection with cauterization of adhesions between the tumor and the esophageal muscular layer and esophageal mucosa allowed a complete and safe enucleation of the leiomyoma. In order to prevent esophageal mucosal injury during the enucleation of the leiomyoma, an esophagoscope introduced via the mouth was used throughout the duration of the surgical dissection allowing both endoscopists and, through transillumination, surgeons direct visualization of the esophageal mucosa (Figure 2(b) and (c)). Finally, the tumor was removed in an endobag, and the esophageal muscular layer was closed with a 3/0 barbed V-loc suture using the robot needle driver, while the intraoperative endoscopy ruled out any degree of esophageal narrowing. A 24F chest tube was inserted into the pleural cavity, and incisions were closed. The postoperative course was uneventful and barium swallow study on postoperative day 1 showed no esophageal leaks. Macroscopic examination of the resected specimen revealed a tumor measuring 10 × 4.5 × 3 cm. Histopathology showed neoplastic spindle cells arranged in fascicles (Figure 3(a) and (b)). Immunohistochemical markers were positive for smooth muscle actin, desmin, and caldesmon. The diagnosis of esophageal leiomyoma was confirmed.
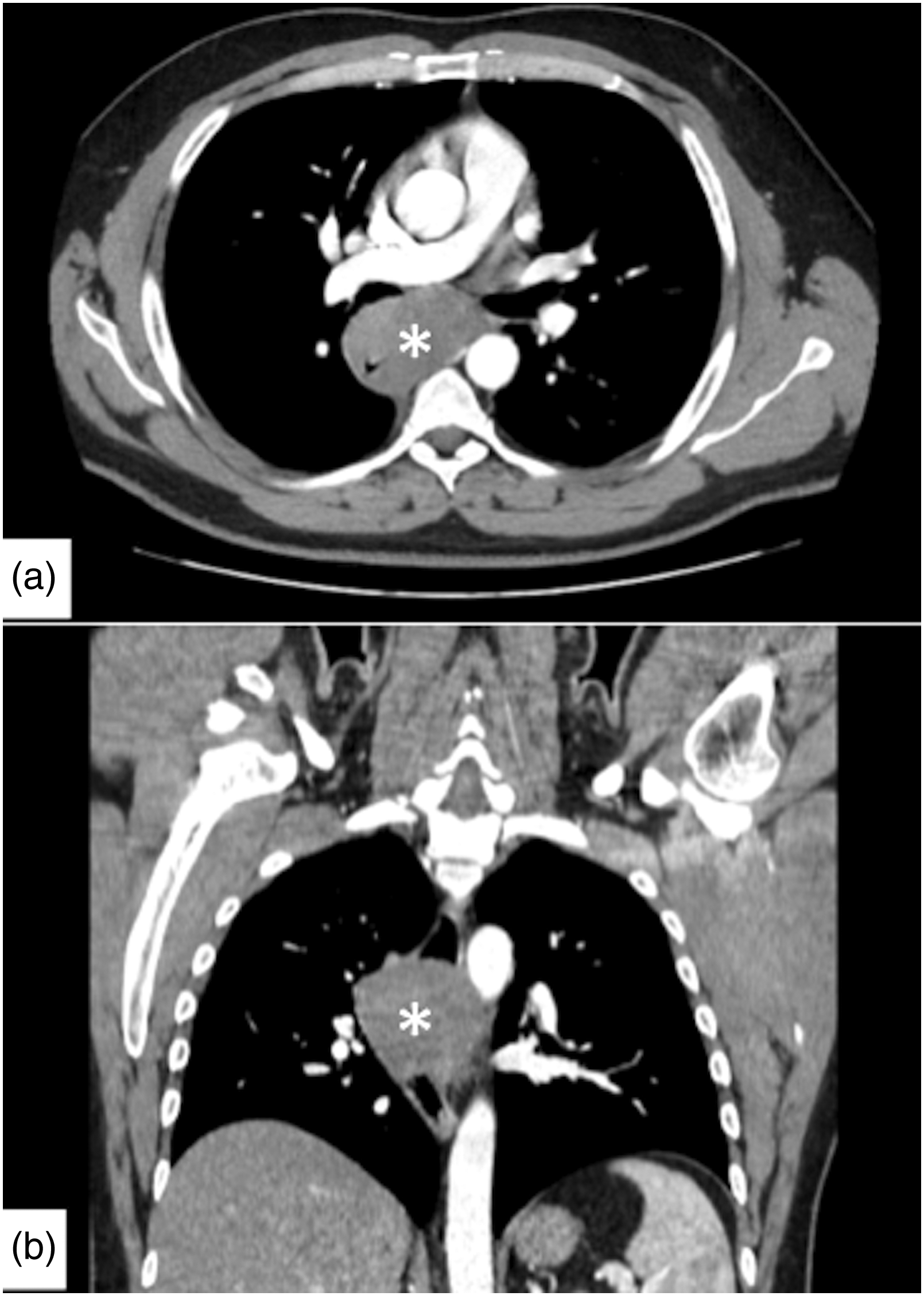
(a,b) Axial and coronal images from contrast-enhanced chest computed tomography show a large, well-circumscribed minimally enhancing mass (white asterisk) in the middle thoracic esophagus, with associated luminal narrowing.
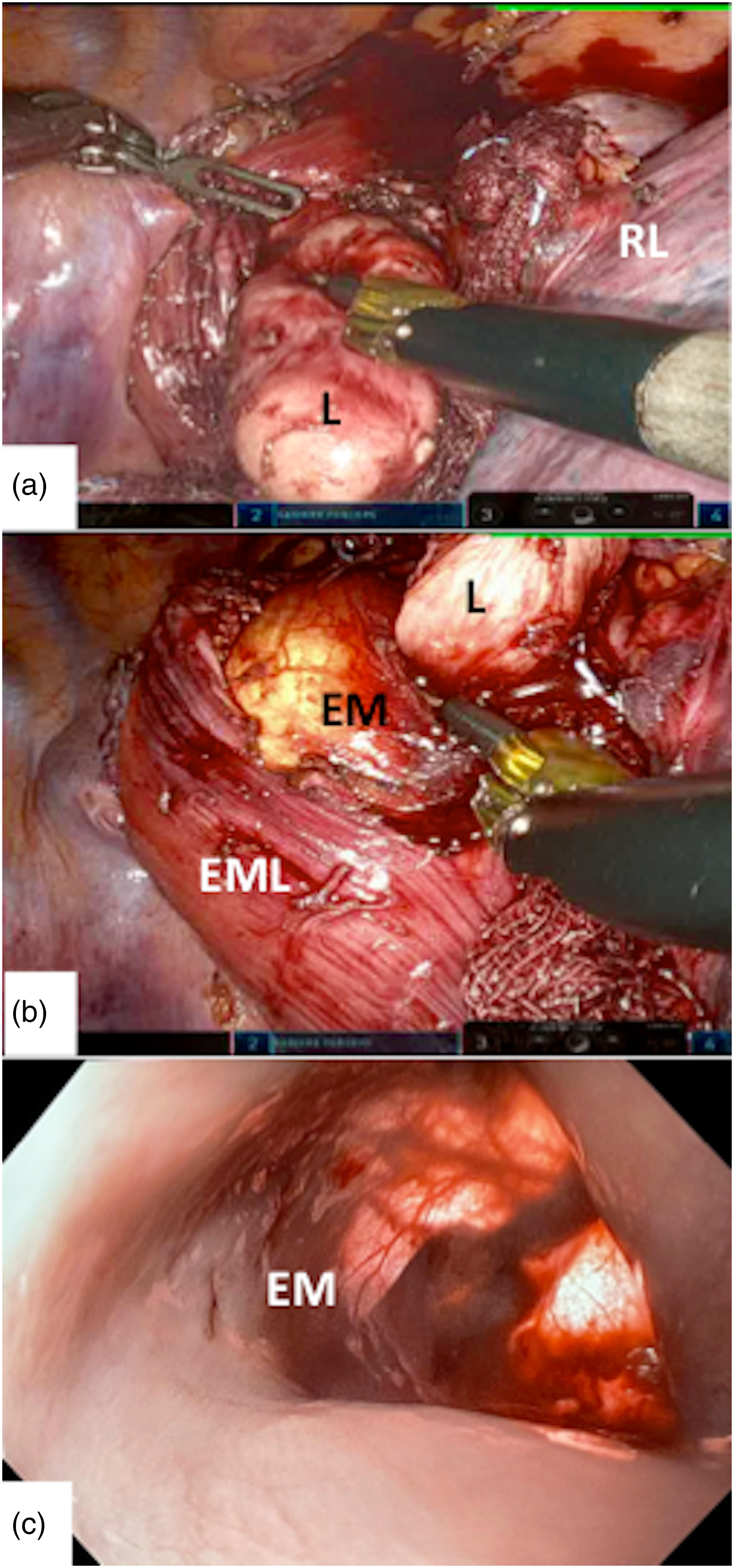
(a) Intraoperative identification of the leiomyoma after esophageal myotomy. (b) Thoracoscopic view of the esophageal mucosa, after extramucosal myotomy, improved by intraoperative esophagoscopy and transillumination. (c) Endoluminal view of the esophageal mucosa during intraoperative esophagoscopy. EM: esophageal mucosa; EML: esophageal muscular layer; L: leiomyoma; RL: right lung.
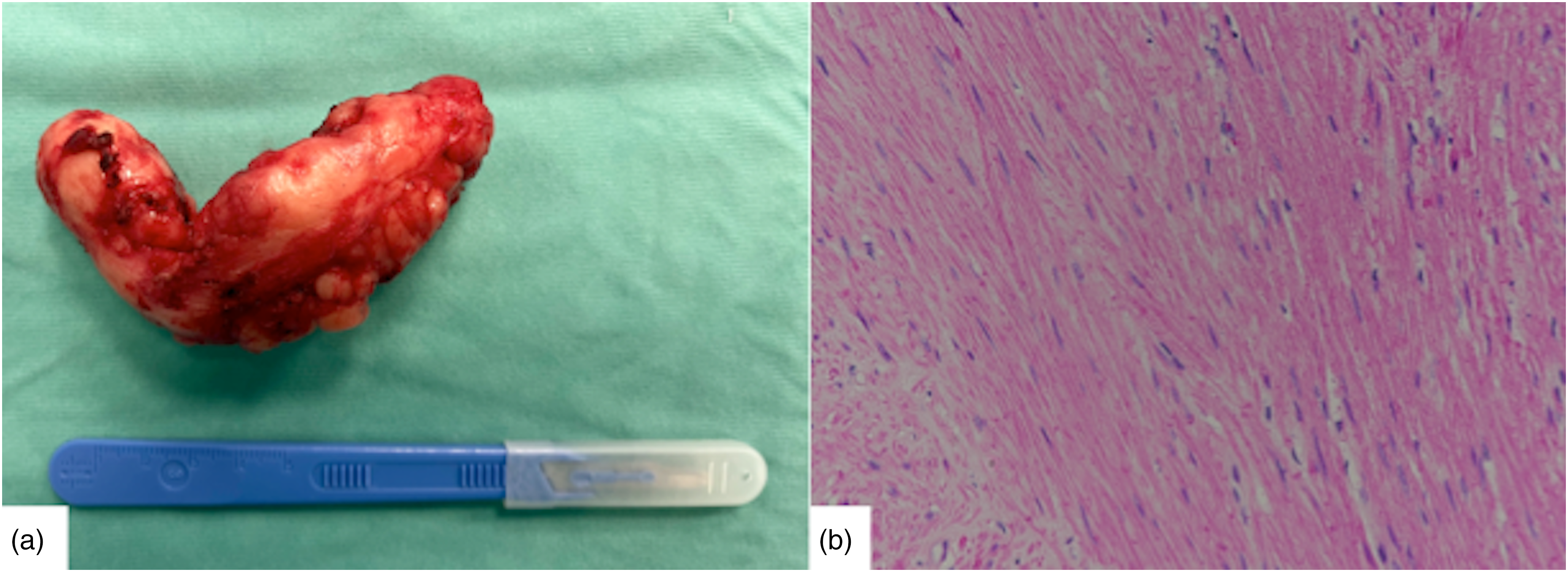
(a) Operative specimen. (b) Histopathological slide with hematoxylin and eosin demonstrating fascicles of elongated spindle cells with abundant cytoplasm and cigar-shaped nuclei.
Discussion
Surgical treatment of esophageal leiomyoma is usually recommended for tumors that are symptomatic, larger than 5 cm or growing. For many years, leiomyoma enucleation by thoracotomy has been the most traditional and common approach.3,4 With the advent of minimally invasive surgery, the thoracoscopy approach became the first choice for esophageal leiomyoma enucleation, providing several advantages over open thoracotomy as limited operative trauma, less postoperative pain, and shorter hospital stay. However, when the tumor is large, the use of video-assisted thoracoscopic surgery may result difficult due to only two-dimensional vision and straight rigid instruments. 5 In our case, the widely known advantages of robot-assisted thoracic surgery, as magnified three-dimensional vision and wrist-like mobility of the robot appliance,6,7 allowed a safe enucleation of the leiomyoma that encircled the esophagus and made closure of the esophageal muscular layer easier. Some authors have reported a significant reduction in the incidence of injuries to the esophageal mucosa during enucleation of esophageal leiomyoma by robot-assisted thoracic surgery.5,8 Our case shows that, for the surgical management of circumferential esophageal leiomyoma, combined robotic enucleationand intraoperative endoscopy may be particularly useful to preserve the integrity of esophageal mucosa during the surgical procedure.
Conclusions
In the treatment of a circumferential esophageal leiomyoma, the surgical approach with combined use of robot-assisted thoracic surgery and intraoperative endoscopy may be considered a safe and useful method.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Informed consent
Informed consent was obtained from the patient.
