Abstract
Objective
To evaluate expression of potential molecular imaging targets epidermal growth factor receptor (EGFR), glycoprotein nonmetastatic melanoma protein B (GPNMB), and vascular endothelial growth factor (VEGF) in lymph nodes (LNs) with or without head and neck squamous cell carcinoma (HNSCC) metastases after (chemo)radiation.
Study Design
Retrospective study comparing receptor expression in paired lymph nodes after initial treatment.
Setting
A tertiary referral hospital.
Subjects and Methods
Salvage neck dissection specimens of 40 patients treated with (chemo)radiation were selected. LNs that contained viable tumor, reactive changes after initial treatment, and normal LNs were analyzed using immunohistochemically determined H-scores and by calculating sensitivity and specificity rates and positive/negative predictive values (PPVs/NPVs).
Results
EGFR expression was found in 86% and GPNMB expression in 100% of the LNs with viable tumor. VEGF expression was present in all lymph node types. For EGFR, the sensitivity rate was 86%, and specificity rate was 81%. For GPNMB, these were 100% and 75%, respectively. PPV of EGFR was 61.8% and NPV was 98.2%. These were 56.4% and 100% for GPNMB, respectively.
Conclusion
In residual or recurrent HNSCC lymph node metastases, both EGFR and GPNMB show tumor-specific expression in immunohistochemistry, which may prove useful in future molecular imaging in salvage neck dissections. Immunohistochemically detected VEGF expression indicates that this target is not feasible for imaging purposes in salvage surgery. Therefore, GPNMB could be a new potential imaging target showing comparable results to EGFR in immunohistochemistry.
Keywords
Head and neck squamous cell carcinoma (HNSCC) presents with cervical lymph node metastasis in 60% of patients at the time of diagnosis. 1 After primary treatment, >50% of the patients with locally advanced HNSCC develop recurrence within 2 years. 1 When residual or recurrent metastatic disease after previous (chemo)radiation is resectable, salvage surgery may be performed to cure the patient. 1
Despite these therapeutic attempts, treatment of HNSCC remains a challenge with a median survival of 6 to 9 months in patients with recurrent or metastatic HNSCC who have run out of therapeutic options. 2
After preoperative imaging with computed tomography (CT), positron emission tomography (PET)–CT, or magnetic resonance imaging (MRI), intraoperative surgical decisions are mainly based on visual inspection and palpation. The effects of radiotherapy (eg, necrosis, fibrosis) complicate the surgical procedure. In initially treated patients, conventional imaging techniques are unsatisfactory for reliable detection and delineation of regional recurrences, since viable tumor is found in only 41% of salvage neck dissections. 3 Positive margins are found in up to 31% of salvage neck dissections compared to 9% of primary neck dissections and are associated with diminished regional control rates.4-6
Better visualization and detection of involved lymph nodes might reduce both positive margins, as the extent of surgery. This is especially important in salvage neck dissections, which are associated with high complication rates. 7 Intraoperative imaging using near-infrared (NIR) light is a new imaging modality that could be helpful in the surgical judgment during resections. 8
The fluorophore IRDye800, which can be conjugated with an antibody, is with excitation/emission wavelengths of 774/789 nm within the optimal range for (NIR) fluorescence imaging and has shown promising results in HNSCC.9,10 The IRDye800 may be conjugated with cetuximab, an antibody that binds to the epidermal growth factor receptor (EGFR) and is commonly overexpressed in HNSCC. In previously untreated cervical HNSCC lymph node metastases, Rosenthal et al 11 showed the possibility of NIR visualization with high sensitivity and specificity using cetuximab-IRDye800 during primary surgical treatment.
However, in patients with residual or recurrent lymph node metastases, reactive changes (fibrosis, necrosis, and calcifications) due to previous (chemo)radiation occur in both lymph nodes and surrounding tissues. No research has been conducted on how (chemo)radiation may affect receptor expression in these lymph nodes. Immunohistochemical receptor detection is the first step to identify new potential imaging targets. EGFR, glycoprotein nonmetastatic melanoma protein B (GPNMB), and vascular endothelial growth factor (VEGF) receptors are potential imaging targets, as previous studies indicated them to be (over)expressed in HNSCC.12-15 This study aims to evaluate receptor expression of these potential NIR fluorescence imaging targets in salvage neck dissections.
Methods
Fifty-five patients with mucosal HNSCC who had a salvage neck dissection for radiological suspicion of residual or recurrent disease after initial treatment with radiotherapy with or without concomitant systemic treatment between January 2005 and March 2015 at the University Medical Center Groningen were included. Retrospectively, demographics of all patients, relevant medical and treatment history, and human papillomavirus (HPV) status (for oropharyngeal carcinoma) were collected ( Table 1 ).
Patient and Tumor Characteristics.

Abbreviation: HPV, human papillomavirus.
Reactive lymph nodes were defined as lymph nodes with necrosis, fibrosis, or calcifications without viable tumor cells present on hematoxylin and eosin (H&E)–stained microscopic slide.
To avoid bias of preexisting variability in receptor expression between patients and tumors, we compared lymph nodes per patient for expression. Therefore, only patients with adjacent lymph nodes of at least 2 subtypes of lymph nodes (with viable tumor, reactive changes due to initial treatment or normal) were included. This led to exclusion of 15 patients because of unavailability of multiple lymph node types, resulting in a study group of 40 patients, which was used for further analyses ( Figure 1 ). Lymph nodes were preferably selected from the same neck level, but this was not possible in all patients. Formalin-fixed and paraffin-embedded blocks of the lymph nodes were gathered from the archives of the Department of Pathology, and 3-µm sections were cut and stained for standard H&E, EGFR (3C6, prediluted by supplier; Ventana Roche), GPNMB (clone AF2550, dilution 1:200; R&D Systems), and VEGF-A (clone A-20, dilution 1:50; Santa Cruz). Immunohistochemical staining of EGFR was performed on a BenchMark Ultra automated stainer (Ventana Roche), according to the manufacturer’s protocol. For GPNMB and VEGF-A, the following immunostaining protocol was performed: sections were deparaffinized and rehydrated in a series of decreasing concentrations of alcohol. Sections were then washed with demineralized water. For GPNMB, antigen retrieval was performed by microwaving the sections 15 minutes in 10 mM citrate buffer (pH 6.0). Endogenous peroxidase reaction was blocked by incubating the sections in 0.3% H2O2 in 50 mL phosphate-buffered saline (PBS) (0.15 M NaCl, 8.0 mM Na2HPO4· 2 H2O, 1.5 mM KH2PO4, pH 7.4) for 30 minutes. The primary antibody was diluted in PBS containing 1% bovine serum albumin (BSA) and incubated for 1 hour. The secondary antibody (polyclonal rabbit anti-goat [RAGPO], 1:100 diluted in PBS containing 1% BSA and 1% AB-serum; DAKO) was incubated for 30 minutes, after which the tertiary antibody (polyclonal goat anti-rabbit [GARPO], 1:100 diluted in PBS containing 1% BSA and 1% AB-serum; DAKO) was incubated for 30 minutes. For VEGF-A, avidin/biotin was blocked using a Blocking Kit (Vector Laboratories SP-2001). Primary antibody was diluted in PBS containing 1% BSA and incubated for 1 hour at room temperature. The secondary antibody (goat anti-rabbit biotin [GARbio], 1:300 diluted in PBS containing 1% BSA and 1% AB-serum; DAKO) was incubated for 30 minutes, after which the tertiary antibody (streptavidin polyclonal, 1:300 diluted in PBS containing 1% BSA and 1% AB-serum; DAKO) was incubated for 30 minutes. Visualization for both GPNMB and VEGF-A was performed using the diaminobenzidine peroxidase reaction. Sections were counterstained with hematoxylin and dehydrated in a series of increasing concentrations of alcohol.
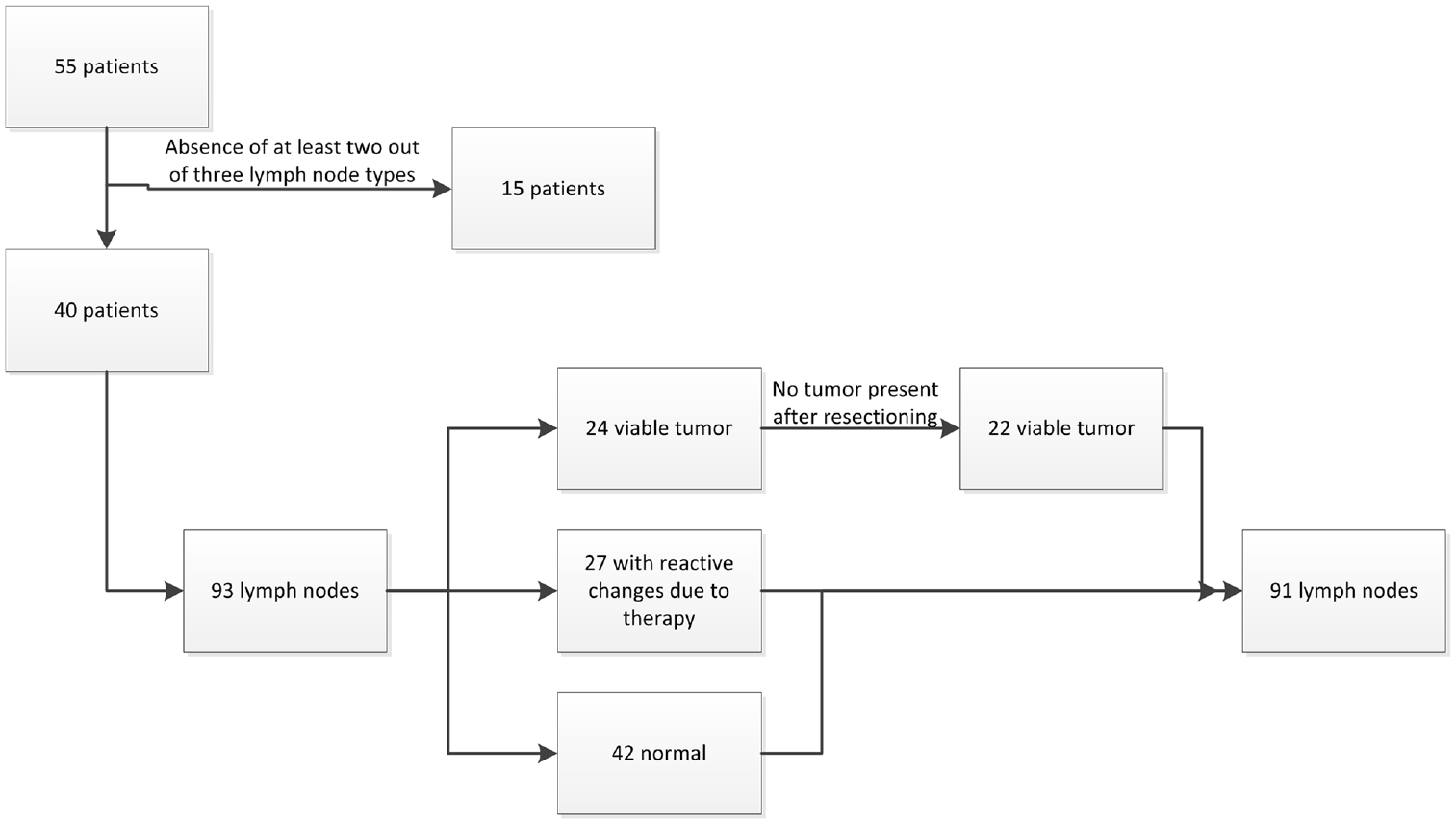
Inclusion process of patients and their lymph nodes.
After sectioning of the study slides, no viable tumor was present in both the study H&E and additional stainings of 2 lymph nodes that were tumor positive on the diagnostic H&E. These lymph nodes were excluded from further analysis. From these patients, a normal and a reactive lymph node were still available for analysis. Statistical analysis was therefore performed on 91 lymph nodes (of 40 patients) of which 22 contained viable tumor, 27 showed reactive changes, and 42 were normal ( Table 1 ). The division of matched lymph nodes for all patients is shown in Table 2 . Three patients who received a bilateral neck dissection have lymph nodes included from both sides.
Distribution of Matched Lymph Nodes per Type (Normal, With Reactive Changes, With Viable Tumor) From All Patients, Stratified for Initial Treatment.

Antibody staining was evaluated independently by 2 of the investigators (JvS and BvdV, who is a dedicated head and neck pathologist). Lymph node staining was scored on both percentage of positive tumor cells and intensity of the staining. Intensity of staining was scored as strong (2+), weak (1+), and negative (0). H-scores were calculated by multiplying the intensity of the staining with the percentage of positive tumor cells. The H-score ranges from 0 (no staining) to 200 (all tumor cells strongly positive).
Lymph nodes with viable tumor were classified as either positive (H-score ≥5) or negative (H-score <5).
Receptor expression was also evaluated in lymph nodes with viable tumor and lymph nodes with reactive changes in relation to the initial treatment (ie, radiotherapy or chemoradiation) and in normal lymph nodes.
Based on the Dutch Medical Research Law (Wet medisch-wetenschappelijk onderzoek met mensen [WMO]), no assessment was required by the hospital’s institutional review board.
Statistical Analysis
Groups were compared using the Mann-Whitney U test for nonparametric data, considering P < .05 to be statistically significant. Sensitivity, specificity, accuracy, positive predicting value (PPV), and negative predicting value (NPV) were calculated. Statistical analyses were performed using SPSS (version 23 for Windows; SPSS, Inc).
Results
Patients
Of the 40 patients available for analysis, 26 were male (65%) and 14 were female (35%) ( Table 1 ). The median age was 59 (range, 44-75) years. The mean follow-up time for patients with viable tumor was 27.6 (3.8-100.5) months. Thirteen patients had an HPV-positive primary tumor.
Thirteen patients were treated with radiotherapy and 27 received chemoradiation.
Receptor Expression
In lymph nodes with viable tumor, EGFR was expressed in >25% of viable tumor cells in 68% of lymph nodes with viable tumor, compared to 100% for GPNMB ( Table 3 ). Necrosis (partly) showed nonspecific staining in 95% and 100% of lymph nodes with viable tumor for EGFR and GPNMB, respectively.
EGFR, GPNMB, and VEGF Receptor Expression in the Different Types of Lymph Nodes and in Relation to Initial Treatment.
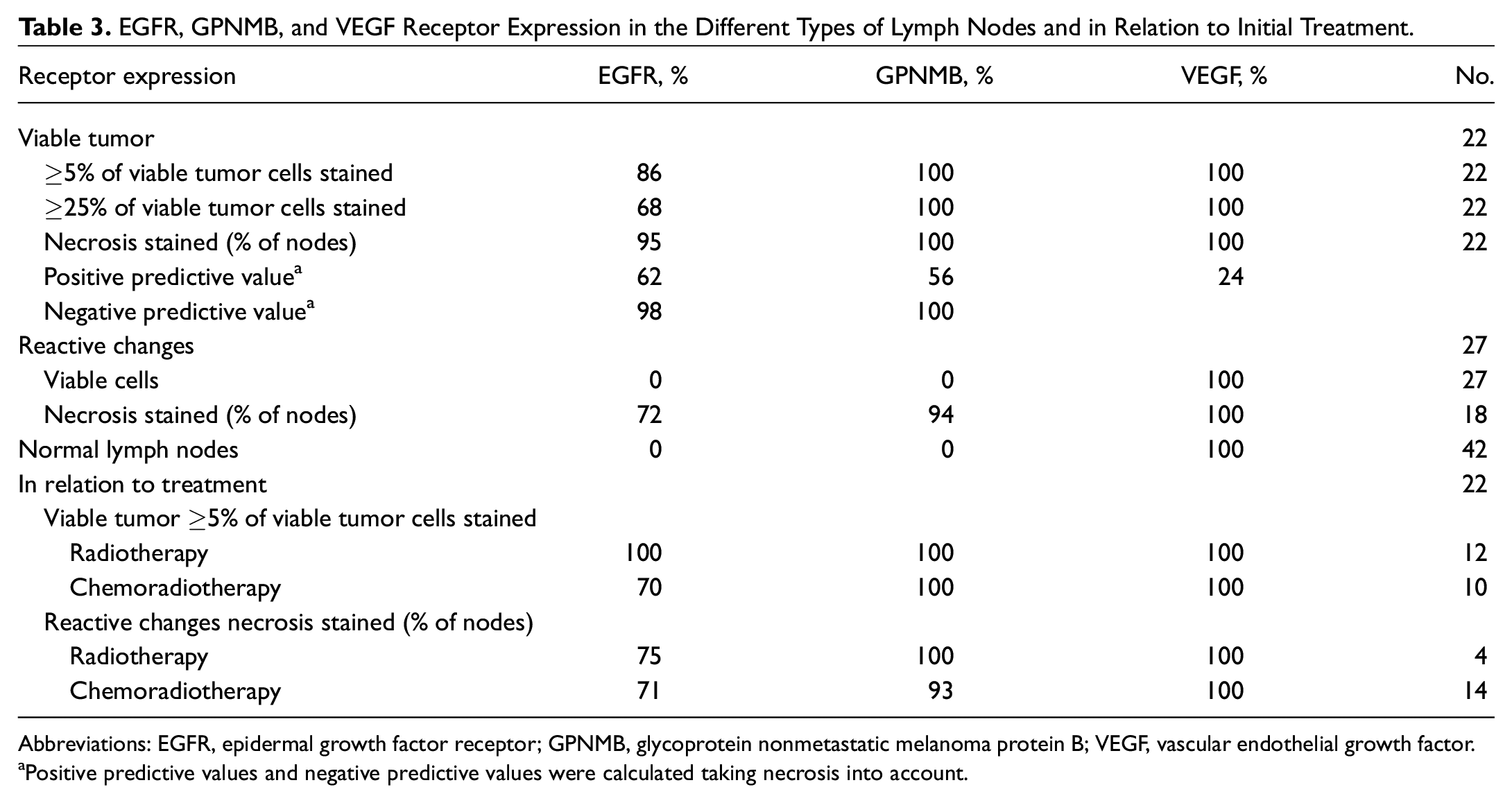
Abbreviations: EGFR, epidermal growth factor receptor; GPNMB, glycoprotein nonmetastatic melanoma protein B; VEGF, vascular endothelial growth factor.
Positive predictive values and negative predictive values were calculated taking necrosis into account.
In lymph nodes with reactive changes, GPNMB also showed higher nonspecific staining in necrosis. Fibrosis and cholesterol clefts only showed staining in 1 (EGFR) or 2 (GPNMB) lymph nodes, and calcifications did not stain in any of the lymph nodes.
Receptor expression of EGFR and GPNMB was absent in normal lymph nodes, except for a few histiocytes that showed GPNMB staining. VEGF showed weak to strong receptor expression in all lymph nodes with specific staining of both normal lymphoid cells as well as of viable tumor cells or necrosis ( Figure 2 ).
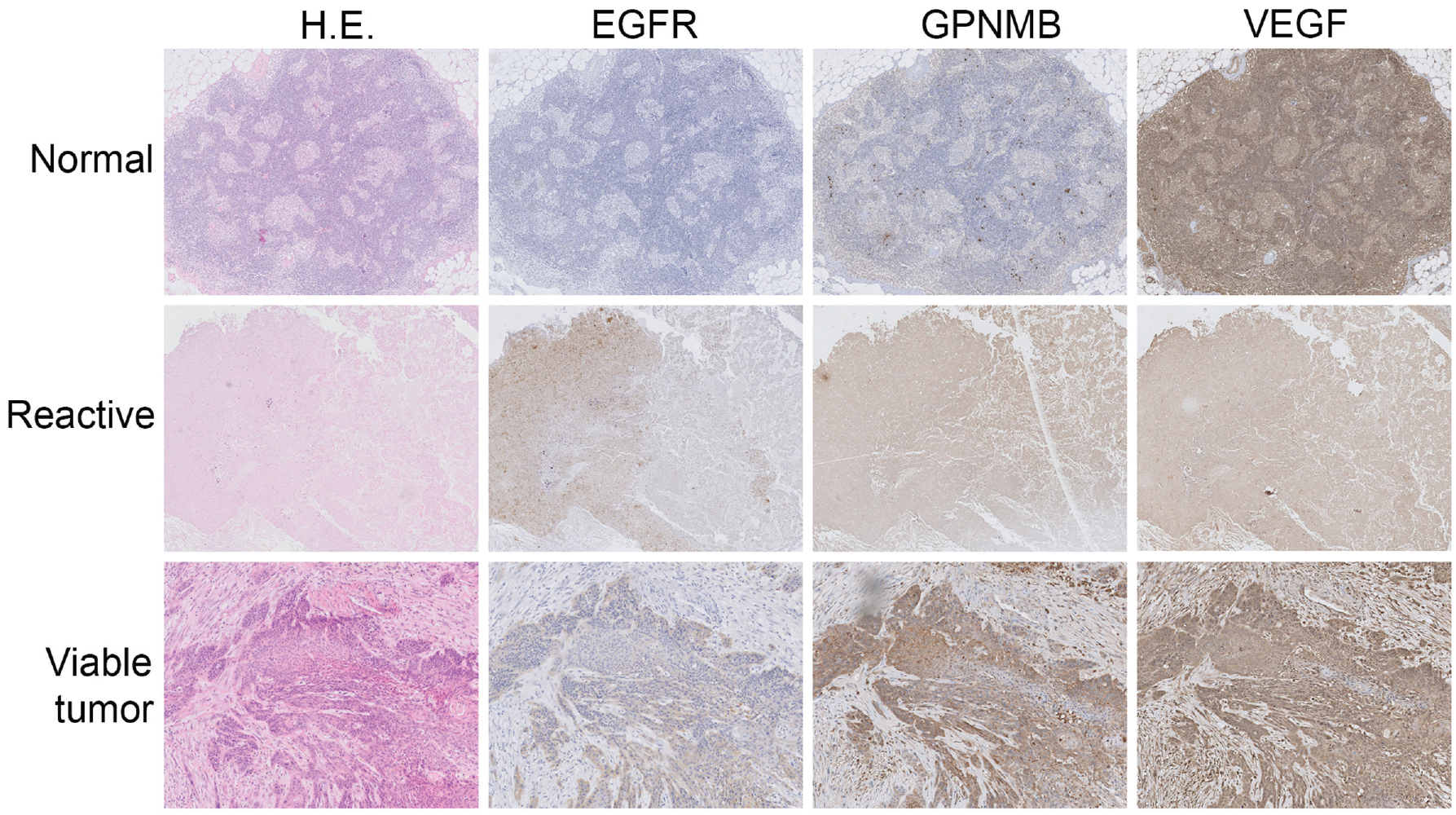
HE, EGFR, GPNMB, and VEGF staining of the 3 types of lymph nodes. EGFR, epidermal growth factor receptor; GPNMB, glycoprotein nonmetastatic melanoma protein B; HE, hematoxylin and eosin; VEGF, vascular endothelial growth factor.
Disregarding the staining of fibrosis and cholesterol clefts in 1 or 2 slides, nonspecific staining was only seen in regions of necrosis. Therefore, any further mentions of nonspecific staining will indicate necrosis.
The highest median H-score was seen in regional lymph node metastases from laryngeal carcinomas stained for EGFR, with a median (SD) of 160 (63.2; range, 10-200). For GPNMB, the medians of the different HNSCC sites lie at around 100. The median H-score was 95 for EGFR and 100 for GPNMB. The distribution of H-scores can be seen in Figure 3A .
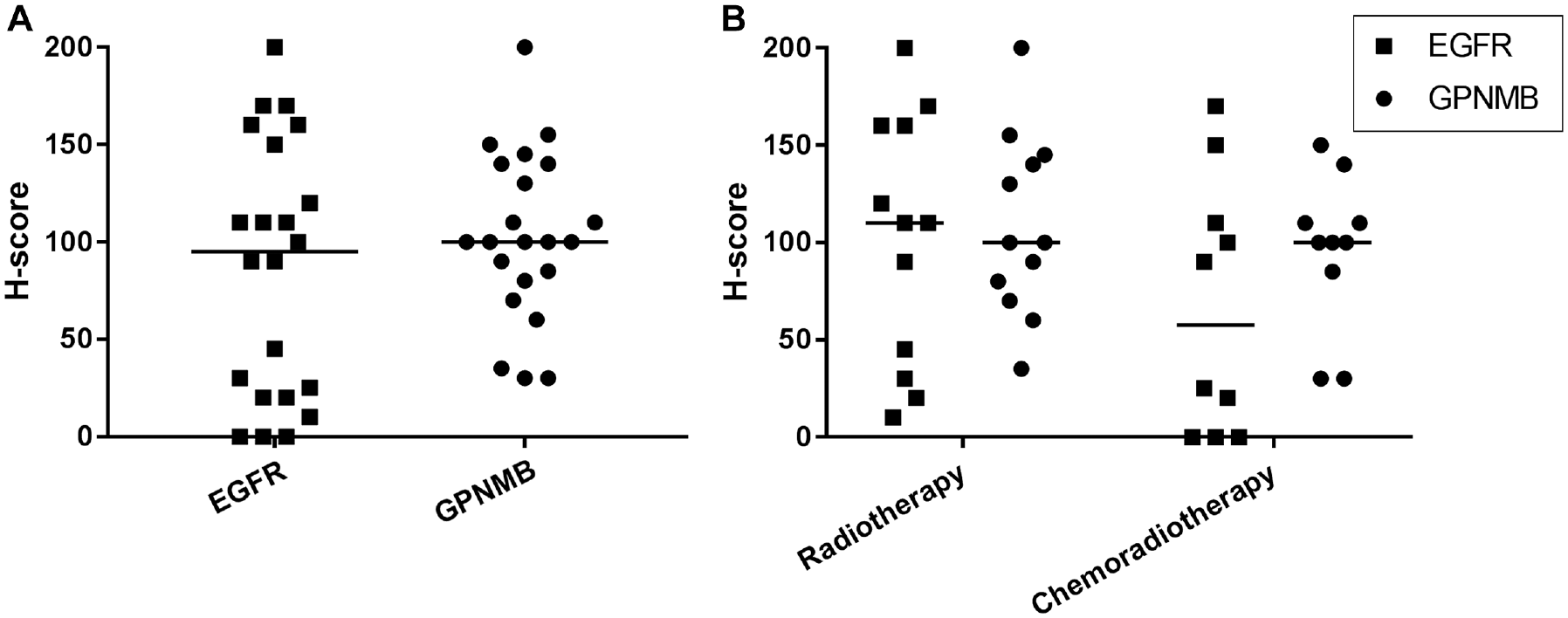
Distribution of H-scores in viable tumor metastases for EGFR and GPNMB (A), and in relation to initial treatment (B). EGFR, epidermal growth factor receptor; GPNMB, glycoprotein nonmetastatic melanoma protein B.
Receptor Expression in Relation to Initial Treatment
Out of 22 lymph nodes with viable tumor, 12 were treated with radiotherapy. All (100%) lymph nodes with viable tumor showed an H-score of ≥5 for EGFR and GPNMB. For the lymph nodes treated with chemoradiation, this was 7 (70%) for EGFR and 10 (100%) for GPNMB.
The H-score of EGFR was slightly but not significantly higher in lymph nodes after radiotherapy compared to chemoradiation (P = .137) ( Figure 3B ).
No differences were found within 1 HNSCC site and among sites between radiotherapy and chemoradiation for both EGFR and GPNMB.
Sensitivity, Specificity, and Positive and Negative Predictive Values
When any presence of staining (ie, both viable tumor cells as well as necrotic areas) was considered as “positive” and complete absence as “negative,” EGFR sensitivity was 86% and specificity was 81%. For GPNMB, sensitivity was 100% and specificity was 75%. The PPV and NPV of EGFR were 61.8% and 98.2%, respectively. For GPNMB, the PPV was 56.4%, and NPV was 100.0% ( Table 3 ).
When only viable cells were evaluated (ie, stained necrotic areas were ignored), sensitivity rates remained 86% for EGFR and 100% for GPNMB. The specificity rate was 100% for both EGFR and GPNMB, as none of the viable lymph nodes showed expression other than in viable tumor cells.
Discussion
To our knowledge, this is the first study to evaluate EGFR, GPNMB, and VEGF expression in lymph node metastases and adjacent lymph nodes without viable tumor in salvage neck dissection specimens. Using immunohistochemical detection, we revealed GPNMB as a potential imaging target after previous (chemo)radiation.
Although lymph node numbers of this study are relatively low, the results are based on a matched analysis of lymph node subtypes within the same patient as comparing lymph node subtypes from different patients might introduce bias due to interindividual differences. To minimize the influence of interindividual differences, we have decided to only include patients in whom we could match 2 or 3 types of lymph nodes ( Table 2 ) at the cost of including a greater number of patients in our analysis.
VEGF receptor expression was seen in all lymph nodes, regardless of its type. This could possibly be because patients in this cohort had recurrent or residual disease after either radiotherapy or chemoradiation and may therefore have had radioresistant tumors. Hypoxic tumors are known to be relatively radiotherapy resistant and have been shown to elevate systemic VEGF levels.16,17 However, we have not used hypoxia markers in the present study. Both internal and external positive and negative controls showed specific staining for VEGF, ruling out nonspecific staining as a cause for the extensive staining of VEGF.
In the evaluation of potential targets for molecular imaging in the detection of cervical lymph node metastasis in salvage surgery, a few factors should be considered. First of all, it should be sensitive for viable tumor cells (ie, ideally, all viable tumors should be stained). In this study, we assumed that all cells deemed viable tumor cells in immunohistochemical analysis have indeed been viable tumor cells in vivo. Second, it should be tumor specific, meaning that surrounding tissues and lymph nodes without viable tumor (either normal or with reactive changes) should not show staining. This would lead to the highest tumor-to-background ratios (TBRs; ie, tumor lighting up more brightly than surrounding tissues) and therefore assist in the intraoperative detection of HNSCC metastases.
In the case of salvage neck dissection patients, it is especially important that all lymph nodes containing viable tumor cells are detected. Leaving any positive lymph node behind has dramatic consequences for the prognosis of these patients, without any other treatment options remaining. Therefore, the surgeon must be absolutely sure that all nonresected lymph nodes do not contain any tumor (high NPV). Whether all positively stained lymph nodes do contain viable tumor cells (PPV) is less relevant for survival, as long as all viable tumor is detected and removed. However, unnecessary extended surgery may lead to loss of function and declined quality of life.
Necrosis was present in two-thirds of lymph nodes with reactive changes and showed EGFR and GPNMB staining in the majority of lymph nodes. As there were no viable tumor cells present in these lymph nodes, this staining would lead to false positives when trying to detect viable tumor metastases. However, this necrosis could possibly point to areas with tumor remnants since it resembled degraded tumor cells and was found in the same area as the lymph nodes containing viable metastatic tumor tissue.
No EGFR or GPNMB expression was found in normal lymph nodes, accentuating the high NPV of the markers. This could assist in the identification of lymph node levels that are not suspected to contain viable tumor metastases.
By immunohistochemical evaluation of viable cells, only tumor cells stained; all other cells were negative, leading to a 100% PPV and 96% NPV with EGFR staining and 100% PPV and 100% NPV with GPNMB staining. Macroscopically, however, as seen in intraoperative imaging, necrosis would confound these findings. Including necrosis, we found a PPV of 61.8% for EGFR, which is in line with the study by Rosenthal et al, 11 who reported a PPV of 51% for patients primarily treated with a neck dissection using molecular imaging in vivo.
As explained earlier, the NPVs are more relevant for imaging purposes in salvage neck dissections. The NPVs are very high, with 98.2% for EGFR and 100% for GPNMB, meaning that every GPNMB-negative node indeed does not contain any viable tumor cells. Therefore, the extent of neck dissection and thereby also morbidity could be reduced by leaving negative nodes behind. For instance, in this study, if the resection of lymph nodes in this study was based on GPNMB positivity, 52 of 69 tumor negative lymph nodes would not have been removed unnecessarily. Based on these immunohistochemically detected findings, GPNMB would be a more suitable target for molecular imaging than EGFR.
Another notable finding of our study is high EGFR expression in lymph node metastases of laryngeal carcinomas. Treatment type could be of influence, as laryngeal carcinomas are mostly treated with radiotherapy, while other HNSCCs were sites treated with chemoradiation in our study. However, no significant difference was found in EGFR expression in lymph nodes of patients treated with radiotherapy and those treated with chemoradiation. This finding may have importance in the future in the diagnostics of neck metastases of unknown primary tumors; however, it has to be further investigated.
A high TBR is essential in the identification of viable tumor cells in NIR fluorescence imaging. Laryngeal carcinoma seems to be the most suitable type of tumor, as it has the highest median H-score of 160. It will therefore definitely light up more brightly than surrounding tissues and necrosis expressing EGFR.
Another influence on EGFR expression is HPV-associated p16 expression. HPV-positive oropharyngeal carcinomas have been shown to have a low EGFR expression. 18 Although our series includes only 1 p16-positive oropharyngeal HNSCC case, an EGFR H-score of 0 is in accordance with this. The H-score for GPNMB, however, was 100.
GPNMB seems to have a steadier H-score, with a median of approximately 100 across all subsites. Furthermore, GPNMB has a higher NPV than EGFR. Therefore, GPNMB seems to be more reliable and preferable for NIR fluorescence imaging of HNSCC in general.
Future studies will have to prove which antibody is more useful for imaging purposes and whether immunohistochemically detected receptor expression correlates with in vivo imaging results. Depending on receptor expression patterns in the primary tumor, one of these antibodies may be more suitable for intraoperative detection of both the primary tumor and the lymph node metastases simultaneously. Future studies will therefore have to focus on the primary tumor too, as well as on the primary untreated neck, in which GPNMB has not yet been evaluated.
Conclusions
EGFR and GPNMB showed promising immunohistochemical results as potential molecular imaging targets for the specific detection of lymph node metastases of HNSCC in salvage surgery. With a high NPV of 98.2% and 100.0% for EGFR and GPNMB, respectively, they may facilitate the surgeon in the identification of tumor-positive lymph nodes. GPNMB is a promising new molecular imaging target for HNSCC, which detected all viable tumor cases and could have prevented 52 of 69 lymph nodes from unnecessary removal. In our study, it is at least as valuable as EGFR and should therefore be considered in future molecular imaging studies. VEGF showed strong staining in all lymph nodes and therefore seems unsuitable for identification of HNSCC lymph node metastases after previous (chemo)radiation.
