Abstract
The 31st edition of the slide seminar “Classic Examples in Toxicologic Pathology” was held at the Department of Pathology at the University of Veterinary Medicine in Hannover, Germany, in March 2024. The meeting series is jointly organized with the ESTP (European Society of Toxicologic Pathology) and aims at presenting and discussing classical and novel cases of toxicologic and experimental pathology. This article reflects on 6 out of 10 presentations given at the seminar and includes images of representative lesions. They comprise cases of toxicologic pathology, induced directly or indirectly by a test item. We herein summarize findings with a progesterone receptor antagonist and its impact on uterine tumor development in rats; an example of hybridization-dependent off-target hepatotoxicity in rats administered a small interfering RNA (siRNA) conjugated to N-acetylgalactosamine (GalNAc) and the case of an orexin-1/-2 receptor antagonist with liver enzyme induction. We further include a case on reactive metabolites with associated liver findings, a study on findings induced by a GPR40 agonist and a presentation on dysgeusia and its translatability.
Keywords
Introduction
The seminar concept of the “Classic Examples in Toxicologic Pathology” looks back at more than 30 years of tradition at the University of Veterinary Medicine in Hannover. Besides being among the major scientific meetings within the European Society of Toxicologic Pathology (ESTP), it is an integral part of the residency program and PhD training at the University of Veterinary Medicine.
It is different from any other conference in that participants are given the opportunity to investigate lesions on whole slide images (WSIs) which are being made available online before, during, and after the meeting. This interactive slide viewing fosters scientific discussions and enhances the learning experience for participating residents.
In 2024, a total of 10 presentations with 45 minutes duration each were given in 2 half days. These included cases from ongoing research or development projects. Thus, disclosing full information on chemical structures or compound names was not a requirement, but morphological changes and mechanistic investigations were in scope of these presentations.
6 out of 10 presenters were able to publish their cases within these proceedings.
The remaining four cases are described here in brief:
Sébastien Laurent (Sanofi, Montpellier, France) described effects of a selective estrogen receptor degrader with focus on hepatobiliary toxicity and cholestasis in dogs. This study has been published as an original article. 5
Mark Cesta, (NIEHS, Research Triangle Park, USA) gave a presentation on cholangiofibrosis and its extrahepatic manifestations.
Kuno Wuersch presented test-article-related effects and microscopic changes from a 5-day mouse study after intravenous administration of FR900359, a selective and potent Gq/11 alpha inhibitor. In addition, possible toxicity mechanisms have been proposed (publication pending).
Jan Hinrich Bräsen (Hannover Medical School, Hannover, Germany) presented a clinical perspective on toxic damage in cholemic nephropathy as a classic example of kidney injury in patients with liver disease.
Progesterone Receptor Antagonist
The first presentation of the 2024 ESTP Classical Examples series was given by Dr Krystyna Siudak (Bayer AG, Wuppertal, Germany) and showed a classic case of uterine tumor development in female rats after lifelong blockade of the progesterone receptor. The case study originated from a 104-week carcinogenicity study in RccHan:WIST rats with Vilaprisan, a selective progesterone receptor modulator intended for the treatment of uterine fibrinoids. The study was conducted in an AAALAC (Association for Assessment and Accreditation of Laboratory Animal Care International)–accredited laboratory in full accordance with animal welfare legislations and Bayer AG principles for animal use.
The presentation focused on organ weight changes, necropsy, and histopathological findings of test item-related changes in female reproductive organs as well as their mode of action and human relevance.
As a compound class member of selective progesterone receptor modulators (SPRM), Vilaprisan competitively binds to progesterone receptors and hereby modulates hormone-mediated gene expression. Pharmacodynamic studies revealed a potent antagonistic but no agonistic activity at the progesterone receptor in vitro as assessed by reporter gene assays. 64
The carcinogenicity study included 51 RccHan:WIST rats per group and sex supplied by Envigo, Bicester, UK. Animals were dosed with 6, 30, and 75 mg/kg/day (males) or 3, 15, and 75 mg/kg/day (females), respectively. The different dosing regime in males and females was based on toxicokinetic data showing sex-related differences of dose-normalized systemic exposure. Two control groups were included and received purified water or the vehicle 0.1% Myrj S-50-PA, 1.0% Klucel LF add 100% with 0.9% NaCl in purified water, respectively. In line with the use in humans, the chosen route of administration in animal studies was oral (via gavage). Since no Vilaprisan-related cancerogenic effects were observed in males, the focus of the presentation was on results in females. The overall number of female decedents was comparable between Vilaprisan treated rats and controls. However, a slightly increased incidence of decedents due to uterine tumors in females administered 3 or 75 mg/kg/day and a decreased incidence of decedents due to mammary gland tumors in females dosed at 3 mg/kg/day or higher were noticed. Treatment-related organ weight changes and macroscopic findings were recorded for the uterus and ovary. For the uterus, mean weights were increased at dose levels of 3 or 15 mg/kg/day and decreased at 75 mg/kg/day. Macroscopically, increased incidences of “distension,” “mass,” and “thin” were observed. Mean ovary weights were decreased at 3 mg/kg/day and increased at 15 or 75 mg/kg/day. Incidences of “cyst” and “small” were increased in females dosed at 3 mg/kg/day or higher.
Histopathologically, endometrial adenoma, adenocarcinoma (Figure 1E and 1F) and one anaplastic carcinoma were recorded in the uterus of Vilaprisan-treated groups without showing a clear dose-dependency (0/0/2/0/0; 0/0/4/2/6; 0/0/1/0/0, see Table 1). No such lesions were observed in the females of both control groups. In contrast, endometrial stromal polyp was seen in the purified water and vehicle control groups only, but not in Vilaprisan-treated females. Regarding hyperplastic lesions, an increased incidence and severity of diffuse endometrial hyperplasia, focal glandular hyperplasia, and squamous hyperplasia at the uterocervical junction were recorded starting at the low dose of 3 mg/kg/day. In addition, an increased incidence and severity of luminal dilatation and myometrial atrophy were noted in Vilaprisan-treated females which correlated with the macroscopic observations of “distension” and “thin.” The latter finding correlated also with the reduced uterus weights for females administered 75 mg/kg/day. When compared to controls, vaginal mucification was less prominent and less frequent in females administered 3, 15, or 75 mg/kg/day. In the ovary, a decreased incidence of mixed sex cord stromal hyperplasia was observed for Vilaprisan-treated females and the ovary showed fewer corpora lutea, a decreased incidence of corpus luteum hypertrophy and an increased incidence of follicular cysts. The reduced number of corpora lutea and the follicular cysts correlated to the necropsy observations of “small” and “cyst.” Additional histopathologic findings considered associated with the progesterone inhibitory effect of Vilaprisan were present in the mammary gland and pituitary gland and are mentioned for completeness: The incidence of mammary gland fibroadenomas was reduced at all dose levels and the incidence of diffuse lobuloalveolar hyperplasia at 75 mg/kg/day. Corpora amylacea, an increased incidence and severity of fibrosis, and a slight increase in incidence and severity of dilation/secretion occurred at all dose levels as well. In the pituitary gland, Vilaprisan increased the incidence of diffuse pars distalis hyperplasia at all dose levels. Of note in context of the new ICH S1B(R1) Weight of Evidence approach, in the 26-week study, a decrease in uterus size and mean weight, which correlated with myometrial atrophy, can be considered as a histopathologic and hormonal risk factor for carcinogenicity in rats. Considering the possible mechanism, Dr Siudak pointed out, that the lifelong daily treatment of rats with Vilaprisan led to a blockade of progesterone action starting at the low dose of 3 mg/kg/day. Under physiological conditions, the onset of reproductive senescence in rats leads to prolonged estrous periods with high estradiol levels or a pseudopregnancy state with moderately high estradiol levels resulting from age-related dysregulation of the hypothalamic-pituitary-gonadal axis. In the presented study, Vilaprisan administration resulted in the development of endometrial hyperplasia and potential progression into adenocarcinoma after life-long treatment. It is assumed that the histopathological findings were due to exaggerated and uninterrupted pharmacological action of Vilaprisan in the presence of high estradiol levels in senescent rats (Figure 1A-1D). Life-long Vilaprisan treatment seems to promote prolonged estrous periods with follicular cysts and reduced states of pseudopregnancy, thus, further enhancing estrogen dominance. All these effects led to a prolonged fully unopposed estradiol exposure resulting in a high estradiol (E2) to progesterone (P4) ratio which is known to induce endometrial adenocarcinomas. 20 The findings of unopposed estrogen action and estrogen dominance are also in line with the distribution of the findings across sexes, that is, tumors in female but not male animals. Estrogen dominance as mechanism for endometrial tumors has also been shown by the investigations on the endometrial tumor incidences in the Donryu and the Fisher F344 rat strains. 40
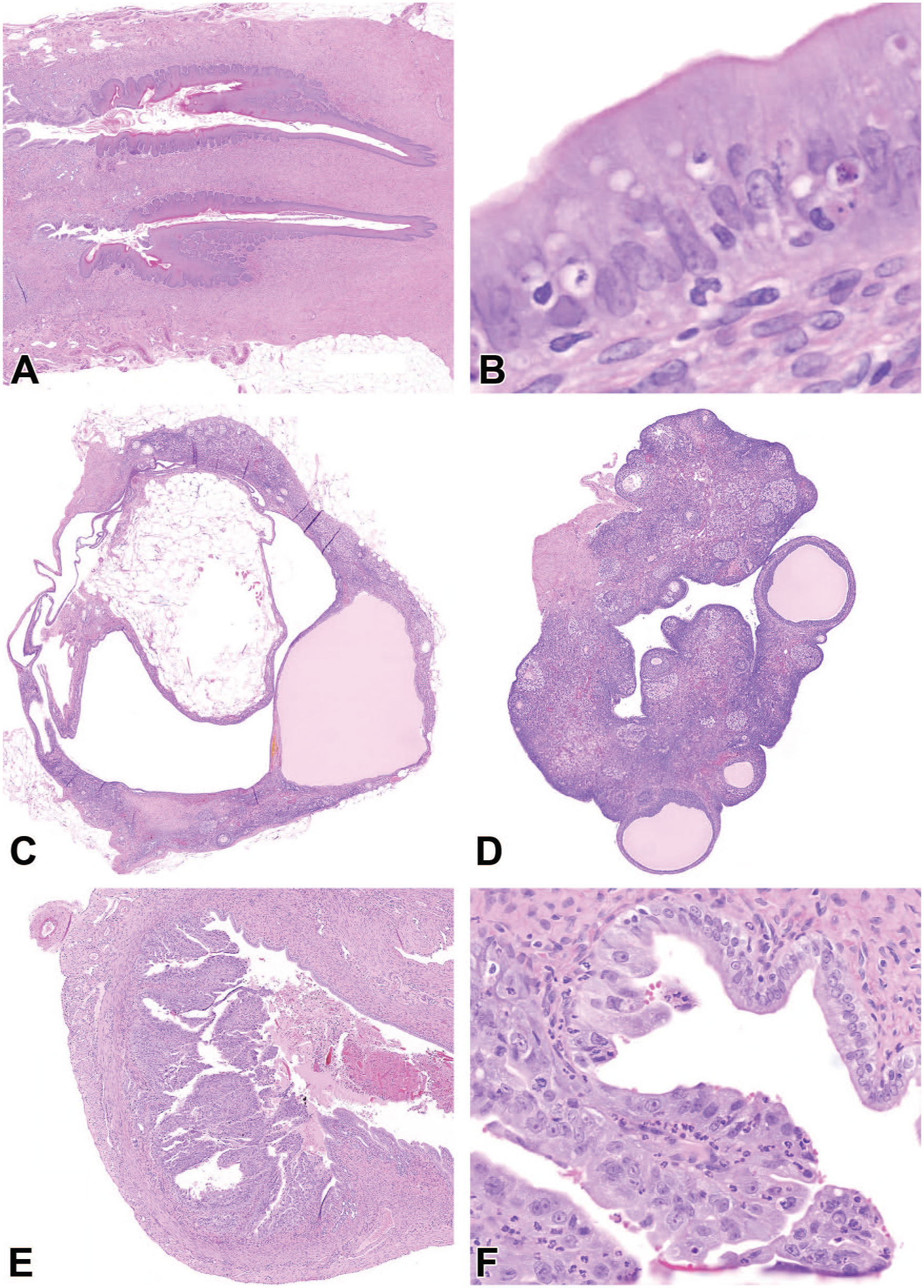
(A-D) Uterine cervix (A and B) and ovaries (C and D) of a female rat administered 75 mg/kg/day Vilaprisan for approximately 104 weeks, H&E stain. In the uterus, there is squamous hyperplasia of the uterocervical junction with associated marked keratinization, and hypertrophy of the endometrium (B). Corpora lutea are absent on both ovaries; the ovary (C) displays a cystic follicle; the other ovary (D) shows few antral follicles. Summed up, all these features point to a state of persistent estrous in this animal. (E and F) Uterus of a female rat administered 75 mg/kg/day Vilaprisan for approximately 103 weeks (moribund sacrifice), H&E stain. The endometrial epithelium turns from a single-layered epithelium to a hyperplastic multilayered epithelium (F) and eventually shows a solid and invasive growth pattern leading to the final diagnosis of an adenocarcinoma.
Incidence of selected findings in the uterus, vagina, and ovary.
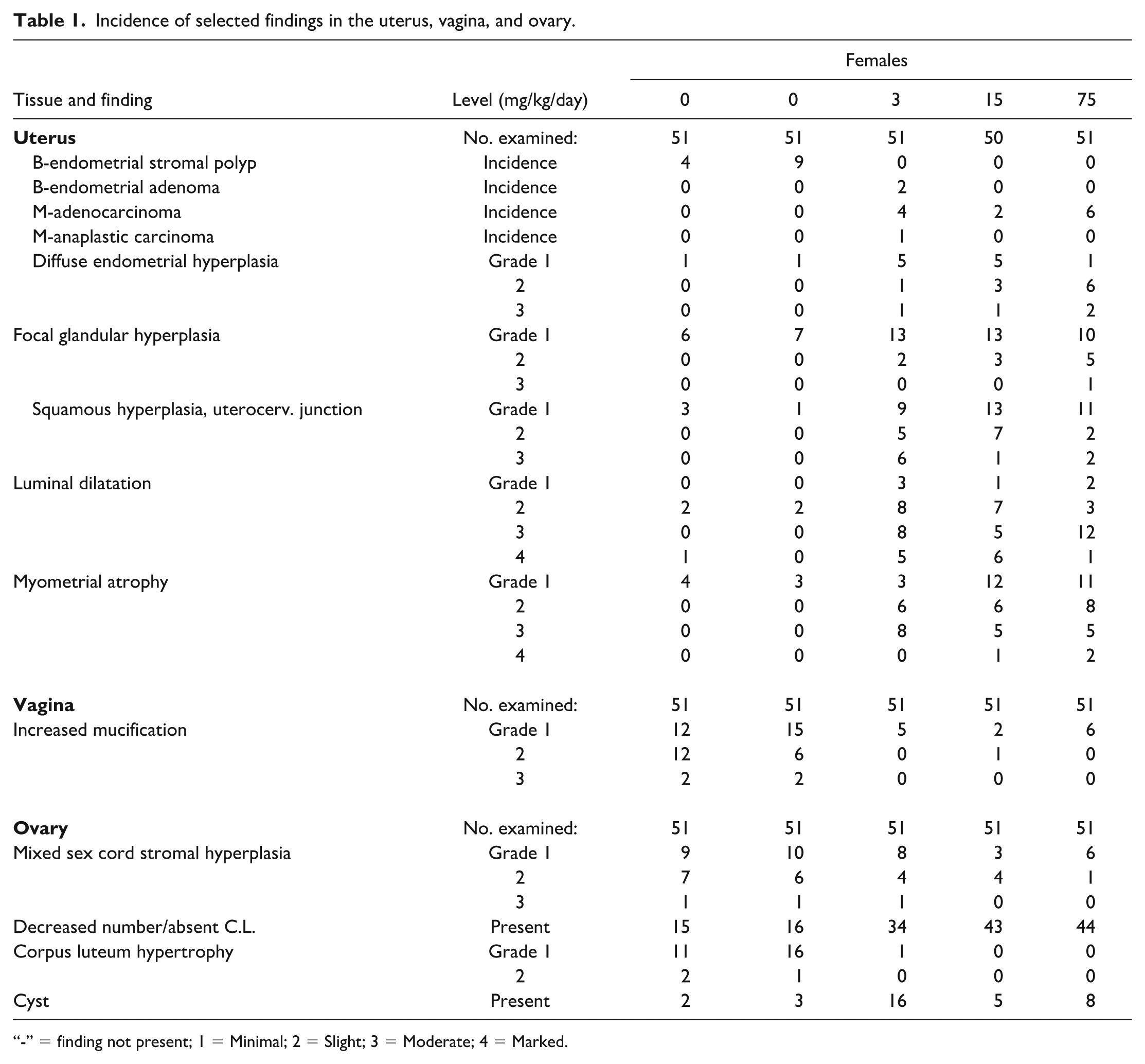
“-” = finding not present; 1 = Minimal; 2 = Slight; 3 = Moderate; 4 = Marked.
Regarding the human relevance, unopposed estrogenic states are an acknowledged risk factor for development of endometrial hyperplasia and neoplasms in women. The now contraindicated practice of giving unopposed estrogens (without a progestin) as hormone replacement therapy (HRT) to menopausal women historically created a comparable situation: In studies with estrogen-only HRT, increased incidences of endometrial hyperplasia were already seen after 6 months of continuous estrogen exposure in postmenopausal women.54,69 After 1 year of receiving systemic estrogen therapy without a progestin, endometrial hyperplasia (EH) rates in postmenopausal women were as high as 20% to 50%.14,48,69 The rate of progression from EH to endometrial carcinoma in women is largely dependent on continued exposure to unopposed estrogen. This may be caused by treatment with estrogenic compounds or by increased formation of endogenous estrogens (eg, obesity) or in women experiencing chronic anovulation. The risk of progression of atypical EH to endometrial carcinoma is 15% to 28% based on studies with up to 20 years of follow-up. Subjects were diagnosed with carcinoma in average of 6 years later. 31 Dr Siudak pointed out that although the general mechanism of prolonged unopposed estrogen effects leading to endometrial hyperplasia and subsequent development of carcinomas also applies to humans, however the situation in female rats (prolonged fully unopposed estradiol exposure and high estradiol [E2] to progesterone [P4] ratio) was considered not representative for women: All available clinical data indicated that no relevant unopposed estrogen effect occurred under Vilaprisan treatment in women. In contrast to the rodent’s reproductive senescence, human menopause is not associated with an increase in endogenous estrogen levels but characterized by low, sometimes undetectable, levels of 17β-estradiol and progesterone, and increased levels of the gonadotropins, FSH and LH. 29
Even more, women treated with Vilaprisan in first clinical trials had low estrogen levels under treatment, were treated intermittently (treatment cycles of 12 weeks with daily treatment, separated by 1 or 2 bleeding episodes, see SmPC of Esmya—Ulipristal acetate), and endometrial shedding occurred at regular intervals as well as ovulation and concomitant progesterone increases. 12 In conclusion, even though the mechanism Vilaprisan-induced uterine carcinogenesis in rats is relevant to humans, the different physiology of senescence and menopause in humans as well as different treatment regimens limit the translatability of the 2-year rat carcinogenicity study results to humans.
GalNAc-siRNA-Associated Liver Findings
Dr Johnson (Eli Lilly and Company) presented an example of hybridization dependent off-target hepatotoxicity associated with a GalNAc-siRNA. Slides were from an investigative study where the GalNAc-siRNA was administered as a single dose to male Sprague Dawley (SD) rats by subcutaneous injection to determine the time course of hepatic injury, characterize the dose response, and understand if hepatocellular injury markers preceded morphologic evidence of injury.
GalNAc-oligonucleotide conjugates are a therapeutic modality that is designed for targeted delivery of oligonucleotides to hepatocytes in order to modulate a variety of diseases for which hepatocytes are involved in the pathogenesis, particularly metabolic diseases. 55 The GalNAc moiety facilitates intracellular deliver of an oligonucleotide payload by binding the Asialoglycoprotein receptor (ASGPR) on the sinusoidal surface of hepatocytes. 55 siRNA is a type of oligonucleotide that is particularly attractive as a potential pharmaceutical. siRNA are double stranded RNA molecules that may be 19-23 nucleotides long and inhibit translation of targeted messenger ribonucleic acid (mRNA) sequences through the endogenous RNA interference pathway. 34 Approved GalNAc-siRNA conjugates include patisiran, 22 givosiran, 51 lumasiran, 52 inclisiran, 32 and nedosiran. 57
The siRNA conjugated to the GalNAc in this case was designed to target a human mRNA sequence and does not have activity against the target in rodents. The design of the investigative study was guided by the results of a 1-month repeat dose toxicity study where the GalNAc-siRNA was administered subcutaneously on day 1 and day 28 to SD rats for a total of two doses. On study day 15, following a single injection of the high dose, four rats were sacrificed in a moribund condition, which was attributed to hepatotoxicity characterized by marked increases in biomarkers of hepatobiliary injury and hepatocyte degeneration.
This investigative toxicity study in SD rats was subsequently designed to characterize the hepatotoxicity observed in the original 1-month repeat dose toxicity study. In the investigative study male rats were administered a single dose by subcutaneous injection and observed for up to 28 days or through the scheduled interim necropsies of the main study animals on study days 7, 14, 21, and 28. Rats were monitored for clinical signs daily and samples were collected for standard clinical pathology parameters and histopathology of the liver at the interim necropsy time points. There were four experimental groups in the study, including a vehicle control group and three dose levels of the GalNAc-siRNA. The high dose in the investigative study bridges to the high dose administered in the original 1-month repeat dose toxicity study and was, therefore, limited to a 14-day observation period because mortality had already been demonstrated at this dose level by 15 days after a single injection. The experimental procedures were approved by the Institutional Animal Care and Use Committee (IACUC) at the facility and conformed to the National Institutes of Health Guide for the Care and Use of Laboratory Animals (Eighth edition, 2011) and the U.S. Department of Agriculture’s (USDA) Animal Welfare Act (9 CFR Parts 1, 2, and 3).”
At all dose levels, test article related mortality occurred between study days 13-15 with no dose-dependency. One rat was found dead, and five rats were sacrificed because of moribund condition. Clinical observations noted on study days 13-15 were limited to rats in moribund condition and included hunched posture, decreased general activity, decreased body tone (whole body), thin appearance, whole body cool to touch, and/or piloerection. Unscheduled deaths were attributed to hepatocyte degeneration characterized by replacement of the normal hepatic cord/lobule structure by irregularly shaped aggregates of prominently enlarged hepatocytes with a hyperchromatic, densely staining cytoplasm and/or hepatocytes containing large or multiple small cytoplasmic vacuoles and nuclei with hyperchromatic nucleoli. Hepatocyte degeneration was typically associated with proliferation (hyperplasia) of oval cells and/or bile ducts, consistent with a regenerative response to the degenerative hepatocyte changes in these rats (Figure 2B). These liver findings correlated to moderate to marked increases in serum alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), and gamma-glutamyl transferase (GGT) activities and higher total bilirubin concentrations. Other test article related microscopic findings in the liver, which were also noted at the scheduled necropsy intervals (Kupffer cell pigment and increased hepatocyte karyomegaly, mitosis, and single cell necrosis), were also present in the unscheduled deaths.
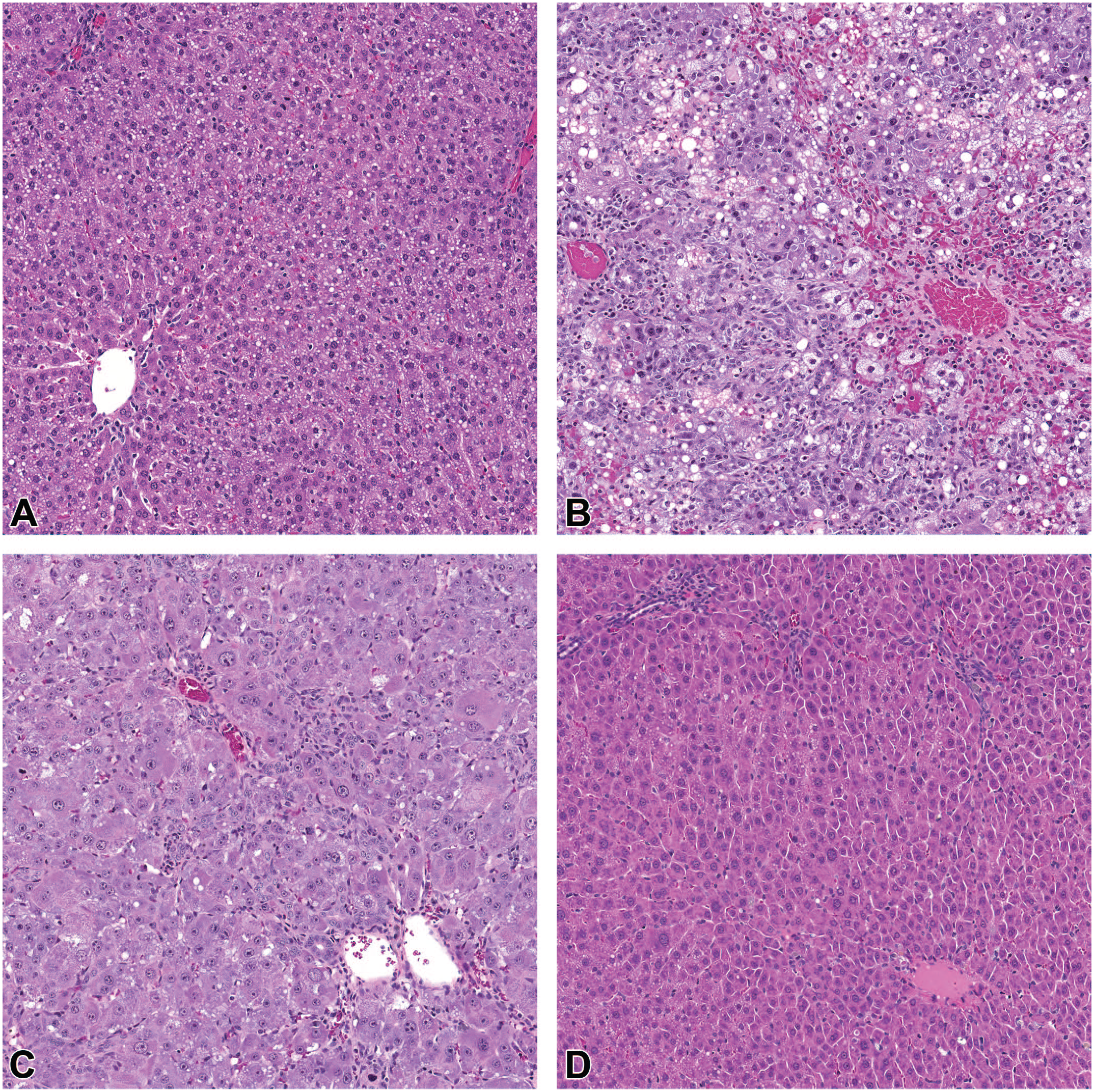
(A-D) Liver from rats administered a single subcutaneous dose of GalNAc-siRNA (original objective 20X), H&E. (A) Day 7 post-injection. There is diffuse minimal vacuolation of hepatocytes. (B) Rat necropsied in moribund condition on day 13 post-injection. There is diffuse hepatocellular degeneration accompanied by oval cell and biliary hyperplasia. (C) Day 14 post-injection. Diffuse hepatocellular and biliary regeneration. (D) Day 28 post-injection. Hepatic architecture is restored. GalNAc-siRNA-related increased pigment in Kupffer cells and minimal biliary hyperplasia remain.
In animals that survived to the study day 14 interim necropsy, all rats had minimal accumulation of pigment in the cytoplasm of Kupffer cells. Microscopic findings of increased single cell necrosis and vacuolation of hepatocytes were also of similar incidence and severity across all dosed groups. The pigment in cytoplasm of Kupffer cells scattered throughout sinusoidal spaces was yellow-gray to faintly-blue green staining and varied from a homogeneous to slightly granular or finely vacuolated in appearance. Other microscopic findings of hepatocyte karyomegaly and oval cell and/or bile duct hyperplasia present at this necropsy interval were consistent with an ongoing sublethal liver injury and regenerative response (Figure 2C).
At the study day 28 terminal necropsy, several test article-related microscopic findings observed at previous sacrifice intervals were absent or decreased in incidence and/or severity, consistent with a partial resolution and regenerative response to the earlier observed liver findings (Figure 2D). There was also an approximately 15% decrease in mean absolute liver weight and 10% decrease in liver weight relative to body weight at both dose levels relative to time-matched controls that were evaluated at study day 28 necropsy. None of the microscopic findings present in the liver at this time point were directly attributable to the lower liver weights.
At the earliest time point evaluated (study day 7), there was minimal accumulation of pigment in the cytoplasm of Kupffer cells, increased single cell necrosis and vacuolation of hepatocytes observed in all dose groups (Figure 2A). These findings were accompanied by minimal increases in serum ALT, AST, ALP, and GGT activities and higher total bilirubin concentrations relative to concurrent controls. Increases in these clinical chemistry parameters increased in magnitude correlating with liver microscopic findings to peak by study day 14 and then returned to concurrent control levels by study day 28.
The nonclinical safety profile of subcutaneous administration of GalNAc-siRNA conjugates is well characterized and has been reviewed by Janas et al. 24 The targeted delivery of potential therapies to hepatocytes and target selectivity provided by sequence specificity of the siRNA oligonucleotide result in a generally benign safety profile for this class of therapeutics. However, small interfering RNAs carry a low risk for silencing unintended RNA sequences through partial complementarity to the antisense strand of the siRNA. 23 As reviewed by Schlegel et al, 50 this is often due to binding of the siRNA to off-target transcripts due to base pairing in the seed region of the siRNA guide strand. Hybridization dependent off-target effects are not limited to siRNAs but have also been described for antisense oligonucleotides.
The available data suggest that hybridization dependent off-target effects are most likely responsible for the more severe hepatotoxicity phenotype observed in this case. As reported in the literature, this mechanism is usually associated with hepatocellular degeneration or coagulative necrosis and significant increases in hepatocellular injury biomarkers. 25 In additional investigative studies, the rat hepatoxicity was reproduced by conjugating the specific siRNA sequence to a distinct GalNAc moiety (unpublished results). These data further demonstrate that the observed toxicity was related to the specific sequence of the siRNA.
The prominence of oval cell and biliary hyperplasia accompanying the hepatocellular degeneration and necrosis in this case appeared to be greater than would be expected for other causes of diffuse hepatocellular necrosis. The authors speculate that the RNA that has been unintentionally disrupted by this GalNAc-siRNA may be involved in cellular pathways that influence proliferation or regeneration. Alternatively, the unique kinetics of this mechanism leading to massive hepatocellular degeneration between 7 and 14 days after administration may influence the regenerative phenotype captured on study days 13-15.
Hybridization-independent GalNAc-siRNA mediated effects observed in nonclinical species are typically observed at doses that significantly exceed clinically efficacious doses and are often given at greater frequencies in toxicity studies compared to clinical trials. These class effects have typically been interpreted as non-adverse in nonclinical species and are not thought to produce significant adverse events in humans.
Hybridization-dependent effects have most commonly been reported in rodents. Alnylam Pharmaceuticals describes their development paradigm in which potential GalNAc-siRNA sequences are screened for hepatoxicity in rodents. 25 Those that cause hepatoxicity in rodents are not advanced to testing in other species, so potential translation to higher species is largely unknown.
Alnylam also published the seminal investigation that implicates hybridization-dependent off-target effects as the likely cause of severe rodent hepatotoxicity associated with GalNAc-siRNAs. 25 In this publication, Alnylam reports that modification resulting in seed-pairing destabilization is an effective strategy to mitigate the observed hepatotoxicity. Interestingly, Alnylam recently applied this technique to a GalNAc-siRNA that advanced to clinical trials to mitigate treatment-related increases in ALT activity. 50 This report suggests that the mechanism of hybridization-dependent off-target effects is operative in humans. However, the nonclinical safety profile of that GalNAc-siRNA and potential concordance with the clinical observations was not disclosed.
Orexin-1/-2 Receptor Agonist
Enrico Vezzali (Idorsia Pharmaceuticals Ltd., Allschwil, Switzerland) presented data from the preclinical development of an orexin-1/-2 receptor antagonist with liver enzyme induction as a truly classical issue in drug development.
Orexin (OX, hypocretin) pathway modulation has been recognized as therapeutic target for decades, including investigations with single receptor (OX1 or OX2) or dual receptors (OX1 and OX2) agonism or antagonism, in the field of sleep, mood, addiction, alimentary, and other disorders. The orexin A and orexin B neuropeptides are involved in the regulation of sleep-wake cycles and promote wakefulness. Deficiency of orexins signals is one of the main causes of sleep disorders and insomnia. By blocking the actions of orexins and modulating sleep-wake cycles, orexin receptor antagonists reduce wakefulness and improve sleep mainly by blockade of the OX2 receptor in the lateral hypothalamus. Several drugs under investigation failed regulatory approvals or their development was discontinued, but not because of side effects specifically related to this class of compounds or to the related targets. Moreover, approved drugs from this compound class showed various toxicity profiles. However, the toxicologic profile that was common to these development compounds or approved drugs from the same dual orexin antagonist class, was limited to very common toxicologic pathology findings, generally considered to be not relevant for humans. The example described in this case aims to raise awareness on the lack of class-specific target-related effects, at the same time emphasizing a common mechanism of toxicity that is not relevant for humans, namely the liver adaptive changes related to drug metabolizing enzyme induction, and consequent secondary findings in the thyroid gland and in the pituitary gland, well described in the literature for decades. 10
The general toxicity profile of one orexin antagonist (a benzimidazole derivative small molecule, in the molecular weight range of 500 Da) was described in rats and dogs in repeat-dose oral toxicity studies with treatment durations of up to 26 and 39 weeks, respectively; among the activities enabling the registration, deserves to be mentioned for completeness of this presented case that genotoxicity potential was assessed in vitro and in vivo, carcinogenicity was assessed in 104-week rats and 26-week rasH2 transgenic mice, and cytotoxicity was assessed in vitro.
Rat and dog are well-established toxicology models and are pharmacologically relevant for orexin antagonists as the function of the orexin system in rats and dogs is similar to that in humans; in addition, in vitro metabolic profiles obtained in rat, dog, and human hepatocytes and microsomes supported the selection of these two species for toxicity studies. Animal experiments were conducted in accordance with institutional and governmental guiding principles in their use. In detail, all non-clinical specimens were obtained from studies approved by the local animal ethical committee and conducted in accredited facilities adhering to international guidelines. Orexin antagonist treatment resulted in histological changes in the liver (in rats and dogs), thyroid gland (in rats and marginally in dogs), pituitary gland (in rats only), and gallbladder (in dogs). In detail, increased incidence and/or severity of centrilobular hepatocyte hypertrophy in the liver, follicular cell hypertrophy of the thyroid gland, hypertrophy in the pars distalis of the pituitary gland (in rats only), and increased secretory material in the lumen of the gallbladder (in dogs only), were noted in the whole GLP toxicology program from the 4-week studies enabling Phase I clinical trials to the carcinogenicity studies; noteworthy, the gallbladder structure was normal (especially, no inflammation was seen), and no precursor signs to gallstone development (eg, presence of crystals) were noted at any time for any dose. These changes were considered non-adverse and were reversible. As case examples, the results of the 4-week rat and dog GLP studies were presented in detail.
In the 4-week rat study, orexin antagonist-related clinical signs included reduced cage activity, hypothermia, and bradycardia, in line with the expected pharmacological effect. A minor mean body weight loss was noted during the first treatment days, correlating with lower mean food intake; thereafter, these animals had similar or higher mean body weight gain and similar food consumption when compared to controls. Increased organ weight and enlargement were noted for the liver and thyroid gland. These findings showed a trend for reversibility (after the recovery period, few animals showed minimal changes in the range of background variability, considered to be due to the too short planning of the recovery period). Microscopic findings consisted of centrilobular hepatocellular hypertrophy in the liver at the high dose, characterized by increased size of centrilobular hepatocytes (compared with concurrent controls), with finely granular, pale eosinophilic cytoplasm (Figure 3A), as well as follicular cell hypertrophy in the thyroid gland at middle and high doses (Figure 3B), cell hypertrophy of the pars distalis in the pituitary gland of males at the high dose, coherent with thyrotropic cells (Figure 3C and 3D), and increased extramedullary hematopoiesis in the spleen at middle and high doses. All findings were mostly of minimal severity, considered non-adverse, and were reversible after the 4-week treatment-free period.
(A) Rat, liver, hepatocellular hypertrophy, moderate (grade 3 out of 4), centrilobular. Orexin antagonist, 4-week treatment. Hematoxylin-Eosin. (B) Rat, thyroid, hypertrophy, follicular cells, moderate (grade 3 out of 4), diffuse. Orexin antagonist, 4-week treatment. Hematoxylin-Eosin. (C) Rat, pituitary gland, pars distalis, hypertrophy, slight (grade 2 out of 4). Orexin antagonist, 4-week treatment. Hematoxylin-Eosin. (D) Rat, pituitary gland, pars distalis, thyrotropic cells. Orexin antagonist, 4-week treatment. Anti-TSH IHC. (E) Dog, gallbladder, increased amount of secretory material, moderate (grade 3 out of 4). Orexin antagonist, 4-week treatment. Hematoxylin-Eosin.
In the 4-week dog study, most animals given the high dose showed vomiting, reduced food consumption, body weight loss, and decreased activity, along with hypersalivation, intermittent tremors and/or head shaking in some animals. During the treatment-free period, such clinical signs were not observed anymore, and body weight gain and food consumption returned to normal, therefore they were considered in line with the pharmacological effects of the modulation of receptors for orexins. Dose-dependent increases in liver weight were observed at all dose levels. Microscopically, centrilobular hepatocellular hypertrophy in the liver and follicular cell hypertrophy in the thyroid gland were observed. In addition, an increased amount of luminal secretory material in the gallbladder was observed at middle and high dose levels (Figure 3E): the material trapped among the mucosal folds was non-cellular, from finely granular to homogeneous, with pale basophilic tinctorial affinity. These changes occurred at exposures hundreds of times above the human exposure at maximum recommended human dose, they were considered non-adverse and were shown to be reversible after the 4-week recovery period.
To summarize, livers of rats and dogs treated with orexin antagonist showed adaptive changes that were considered to be related to drug metabolizing enzyme induction, non-adverse, and reversible upon cessation of treatment. They were associated with secondary findings in the thyroid gland in rats and dogs, and in the pituitary gland in rats. Evidence of orexin antagonist-mediated hepatic drug metabolizing enzyme induction consisted of a dose-dependent upregulation of the gene expression of members of cytochrome P450 (cyp3a12, cy3a1, cyp2b1, cyp2b2) and phase 1 UGT-glucuronyltransferases (ugt1a1, ugt1a6, and ugt2b1) enzymes. Drug metabolizing enzyme induction after treatment with orexin antagonist was further supported by a decrease in exposure of the orexin antagonist in rats, after repeated dosing. In rats after treatment with orexin antagonist, increased liver metabolism lead to decreased levels of thyroid hormones (triiodothyronine [T3] and thyroxine [T4]); decreased thyroid hormones promote a classic endocrinology feed-back mechanism, that increases the levels of thyroid-stimulating hormone (TSH), synthesized by the pituitary gland (characterized by hypertrophy in the pars distalis), followed by new synthesis of thyroid hormones by the thyroid gland (characterized by hypertrophy of the follicular cells).
In dogs, follicular cell hypertrophy of the thyroid gland occurred, as well. Increased metabolic clearance of thyroid hormones, often associated with UDP-glucuronyltransferases (UGT) induction and decreased T4 levels, can lead to a compensatory elevation of circulating TSH produced by the pituitary gland, followed by TSH-mediated stimulation of the thyroid gland and follicular cell hypertrophy.5,6 In rats, increased TSH production by the pituitary gland was shown to correlate histologically with cellular hypertrophy of the pituitary pars distalis. 7 A similar feedback response leading to follicular cell hypertrophy of the thyroid gland was also described in dog studies with other compounds in the literature. 8
Thyroid and pituitary findings secondary to drug metabolizing enzyme induction are commonly observed in animals during preclinical development, particularly in the rat, and are considered of no clinical relevance due to species differences in thyroid hormones transport, metabolism, and elimination. In humans, circulating thyroid hormones are bound to plasma thyroxine-binding globulin with high affinity, resulting in relatively low concentration of free T4 and a relatively long T4 half-life (up to 9 days). In contrast, in rats, which lack thyroxine-binding globulin, circulating thyroid hormones are bound to albumin and transthyretin with low affinity, leading to increased susceptibility to metabolism and clearance, thereby resulting in short T4 plasma half-life (1-2 days), higher fluctuations in T4 plasma concentrations, and higher TSH secretion by the pituitary gland.5,9,10 Since T4 levels are more prone to change in rats than in humans, the feed-back signal to compensate with thyroid hormones synthesis comes earlier in rats than in humans, therefore rats are believed to be more sensitive than humans to thyroid changes, and consequently to alterations of the hypothalamus-pituitary-thyroid axis. Male rats, which have higher circulating TSH levels, usually are more sensitive than female rats. 5 In dogs, thyroxine-binding globulin is present but only at 15% of the concentration found in human and has less affinity for T4, thereby accounting for relatively high plasma concentrations of free T4 and also short T4 half-life (one day). Secondly, although the metabolic route of thyroid hormones is generally conserved across species, elimination of T4 in bile after conversion by UGT is high in rats, leading to faster clearance in instances of UGT induction (as observed in rats treated with orexin antagonist). In addition, in clinical studies, no time-dependent loss in orexin antagonist exposure was observed and orexin antagonist did not change the pharmacokinetic profile of the sensitive cytochrome P450 substrates in drug-drug interaction studies with benzodiazepines, indicating that drug metabolizing enzyme induction did not occur. Therefore, the thyroid and pituitary changes secondary to drug metabolizing enzyme induction observed in rats and dogs after treatments with orexin antagonists are clinically irrelevant at the intended therapeutic dose for insomnia. Noteworthy, the gall bladder abnormal content observed in the dog had unknown origin, but it occurred ad exposures and in modalities that did not require further investigations to clarify the mechanism of action. Finally, no orexin antagonist-related findings could be identified.
Reactive Metabolite—Liver Findings
In his presentation, Dr Nolte focused on major aspects of the liver pathology induced by a small molecule drug candidate (compound A). The compound is a new chemical entity that has been under development for treatment of a CNS disorder. The liver findings were considered unrelated to the drug target but caused by a reactive metabolite. Physicochemical properties of compound A (cationic amphophilic) have contributed to the liver pathology.
It is well known that reactive metabolites of drugs may cause toxicity by binding covalently to endogenous molecules. The typical drug toxicities, which are attributed to a covalent binding of reactive metabolites are adverse drug reactions Type B (ADR/B), which are characterized by their delayed onset, low incidence and unclear, if any, correlation between pharmacokinetics, pharmacodynamics and toxicological outcome. 2 The mechanism of this toxicity is discussed to be either mediated through impairing physiological functions of the modified proteins, resulting in direct tissue toxicity or through indirect immune-mediated mechanisms triggered by the modified proteins. Such typical ADR/B are:
- idiosyncratic drug reaction (hypersensitivity reaction)
- hepatotoxicity
- blood dyscrasia
However, there is neither a well-defined nor a consistent direct link between the formation of drug-protein adducts and organ toxicity 13 or idiosyncratic drug reactions 60 as many modifying factors are considered to contribute to this process. The most important modifying factors seem to be (I) the amount of reactive metabolite formed, (II) binding to critical biomolecules, and (III) efficacy of detoxification mechanisms.
Idiosyncratic drug reactions affecting the liver are the types that most commonly lead to drug withdrawal from market or black box warnings.
Results from a 13-week repeat dose toxicity study in Wistar rats and from mechanistic studies in CD-1 mice and Wistar rats were presented. In these studies, the test compound was suspended in 0.5% aqueous hydroxyethlycellulose (Natrosol 250 HX) and administered once daily by oral gavage. In the 13-week toxicity study in rats, groups of 10 male and 10 female animals were treated at dose levels of 0 (Control), 50, 100, 250, or 500 mg/kg/day. Additional 10 males and females of the control and high dose group were kept for a 10-week recovery period following cessation of treatment. Clinical pathology parameters were monitored on days 3, 22, 92, 134, and 162. At the end of the respective study phases, all main study and recovery animals were subjected to complete necropsy and histopathological investigation.
In a mechanistic study, groups of male CD-1 mice were either dosed 0 (vehicle) or 1250 mg/kg/day for up to 14 consecutive days. Clinical pathology investigations were performed on days 3, 7 and 14 on plasma obtained from terminal bleeding. For the measurement of hepatic glutathione content, liver samples were taken 1, 2, 4, 6, and 24 hours after dosing on days 3, 7 and day 14. Both the reduced (GSH) and the oxidized form (GSSG) of glutathione were measured and the ratio GSH/GSSG was calculated as marker for the cellular redox status. At the end of the respective study phases all animals were subjected to necropsy and histopathological examination of the liver.
In a mechanistic study in male Wistar rats, compound A was administered to groups of 40 male Crl:WI(Han) rats by daily oral gavage for up to 28 consecutive days at doses of 0 (Control), 120, 300 and 750 mg/kg. Plasma samples were taken on days 2, 4, 8, 15, and 29 for determination of levels of TSH, T3 and T4 in addition to standard clinical pathology.
Cohorts of eight males per dose group each were sacrificed on days 2, 4, 8, and 15. All animals underwent full necropsy, collection of selected tissue and subsequent histopathological examination. For toxicogenomics, additional liver samples from animals necropsied on days 2, 15, and 29 were preserved in RNAlater. All in vivo studies were conducted in accordance with German animal welfare legislation and Boehringer Ingelheim’s Global Animal Welfare Policy.
Daily dosing to rats for 13 weeks did not induce any clinical signs or mortalities but slightly increased ALT, AST, and GLDH starting on day 3. At the end of the treatment phase increases in plasma enzyme activities were more prominent in males (increase of group arithmetic mean vs Control: ALT 4 x, AST 2 x, GLDH 4 x) and females (ALT 2.0 x, GLDH 2 x). Morphological findings of the liver were minimal to moderate in grade and included increased organ weight, centrilobular hepatocellular hypertrophy indicative of induction of drug metabolizing enzymes (Figure 4A and 4B), single cell necrosis, and increased hepatocellular mitoses. The cytoplasm of hypertrophic hepatocytes was diffusely to clumpy eosinophilic, but without “ground glass” appearance, indicating that the hypertrophy was not due to exclusive enlargement of smooth endoplasmic reticulum Additional liver findings were lipofuscin storage in hepatocytes and Kupffer cells (males), multinucleated hepatocytes, activation/proliferation of Kupffer cells, and foam cells (Figure 4C-4E). Kupffer cell proliferation as evidenced by Kupffer cell mitoses was accompanied by degeneration of individual Kupffer cells and increased leukocytes in sinusoids.
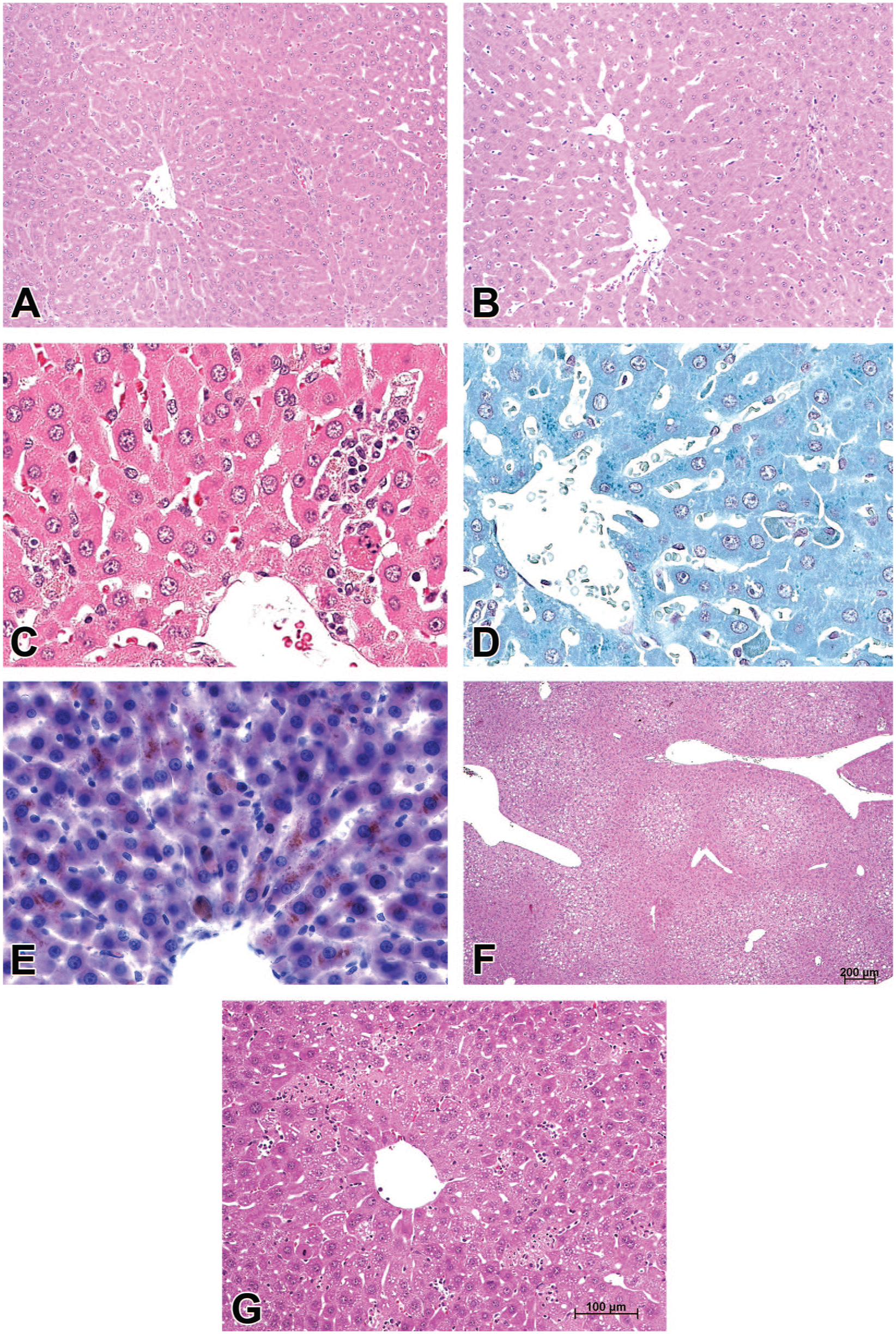
(A) Liver from a female rat after 13-week daily treatment with vehicle; for comparison to (B). H&E. (B) Liver from a female rat after 13-week daily treatment at high dose of compound A; slight centrilobular hepatocellular hypertrophy. H&E. (C) Liver from a female rat after 13-week daily treatment at high dose of compound A; centrilobular hepatocellular hypertrophy with granular eosinophilic cytoplasm; hepatocellular single cell necrosis and mononuclear inflammatory cell infiltrates. H&E. (D) Liver from a female rat after 13-week daily treatment at high dose of compound A; Schmorl’s stain for lipofuscin. Stippled positivity for lipofuscin within centrilobular hepatocytes. Hypertrophic Kupffer cells are also slightly positive for lipofuscin. (E) Liver from a female rat after 13-week daily treatment at high dose of compound A; frozen section stained with fat red 7B stain for neutral lipids. Lipofuscin within hepatocytes readily visible. (F): Liver from a male mouse after 7-day treatment with compound A; hepatocellular vacuolation with midzonal to centrilobular distribution. H&E. (G) Liver from a male mouse after 7-day treatment with compound A; necrosis of single to groups of hepatocytes with predominantly midzonal distribution pattern. H&E.
Most findings were ameliorated or reversed until the end of treatment free recovery periods of up to 10 weeks. Changes still present included increased activity of plasma enzymes in males (increase of group arithmetic mean vs Control: ALT 2 x, AST 2 x, and GLDH 7 x), centrilobular hepatocellular hypertrophy and pigment storage in centrilobular Kupffer cells in males.
Dosing at 1250 mg/kg/day to CD-1 mice resulted in five unscheduled deaths. At necropsy, a diffuse yellow discoloration of the carcass and soft consistency of the liver were noted in these animals. Terminal clinical chemistry investigations revealed time dependent increases in liver enzyme activities (ALT, AST, ALP, and GLDH) mainly and most prominent 6 h post dose on all days, but most prominent on day 7 (data not shown). Total bilirubin was markedly, and time dependently increased at all time points. Plasma cholesterol and triglycerides were slightly (≤ 2X) increased.
Liver tissue GSH, GSSG and total GSH levels were increased at 1250 mg/kg when compared to control at all days. A diurnal profile was evident on day 14 with the highest value 1 h after dosing, a nadir below Control values 6 h after dosing and recovery to control values after 24 h. In contrast, GSH/GSSG-ratio was found to be constant when compared to Control.
Test item-related histopathological effects on hepatocytes were noted from day 3 onwards, effects on Kupffer cells on day 14 only. Incidence and severity of most findings increased with time. Midzonal, and to a lesser extent, centrilobular microvesicular (Figure 4F) vacuolation was characterized as lipidosis with fat red 7B stain. The finding corresponded to the gross observations of discoloration and accentuated lobular pattern. Hepatocellular single cell necrosis and subsequent microgranuloma(s) were evident after 7 and 14 days treatment. Single cell necrosis was graded minimal to severe and occurred predominantly in the midzonal region (Figure 4G). A moderate to severe centrilobular necrosis present in isolated test item-treated animals on days 7 and 14 is considered an exacerbation of disseminated single cell necrosis. Hepatocellular (single cell) necrosis represented the morphological correlate to the clinical pathology findings of increases in ALT, AST, and GLDH activities. The hepatocellular mitotic activity was minimally to moderately increased on day 3 and up to severely increased on days 7 and 14, considered secondary to hepatocellular degeneration/necrosis. A minimal to moderate centrilobular hepatocellular hypertrophy was evident after 7 and 14 days, a moderate diffuse hepatocellular hypertrophy after 14 days only. The cytoplasm of hypertrophic hepatocytes was diffusely to clumpy eosinophilic, but without “ground glass appearance.” Centrilobular and diffuse hepatocellular hypertrophy correlated to an increase of absolute and relative liver weight (1.5X).
In the mechanistic study in Wistar rats, there were no deaths, and no clinical signs attributed to treatment with compound A. Starting at 300 mg/kg, plasma T4 levels were decreased on days 2, 8, 15, and 29, reaching a level of 0.02X Control mean at the high dose on day 29. Effects on T3 were less prominent with a reduction to ≥ 0.3X. The group mean liver to body weight ratios were increased at 750 mg/kg (≤ 1.4X), starting on day 8. The only gross finding of the liver was enlargement in two high dose animals terminated on day 29. Microscopic findings were generally minimal to mild. Kupffer cells of the centrilobular region were minimally to slightly hypertrophic on days 15 and 29. Occasionally, hypertrophic Kupffer cells formed small aggregates, similar to microgranulomas. Periportal hepatocytes were minimally to slightly vacuolated, starting on day 4. This finding occurred in only one animal sacrificed on day 29. An additional focal or diffuse vacuolation of hepatocytes occurred in individual animals on days 8, 15, and 29, graded minimal to moderate. Foam cell accumulation occurred on days 15 and 29, with a higher incidence and grade on day 15. Multinucleated giant hepatocytes were present in two animals on day 29. A minimal diffuse vacuolation of the epithelium of intrahepatic bile ducts was present on day 15 exclusively.
Microarray analysis of mRNA revealed differently expressed genes at the 750 mg/kg dose level, and to a lesser extent at 300 mg/kg. This affected Phase I enzymes involved in the glutathione metabolism, Examples of Phase I enzymes with increased expression include CYP 7A1 ≤ 10X and CYP 2C40 ≤ 7X. Phase II enzymes with increased expression included glutathione-S-transferases (≤ 14X), glutamate cysteine ligases, glutathione peroxidase, and glutathione reductase. Also, UDP glucuronosyltransferase and sulfotransferase mRNA was increased, both enzymes critical in T3 and T4 degradation. Expression of genes involved in proteasomal degradation was increased ≤ 2X.
The pattern of liver findings induced by compound A in repeat-dose toxicity studies exceeded what is frequently observed with molecules inducing hepatic microsomal enzymes. Findings of concern included multinucleated giant hepatocytes, activation/proliferation of Kupffer cells, a tendency of overall progression of findings over time, and a slow reversibility or even progression of findings (liver enzyme increase) after cessation of dosing. Early on, this pattern of liver findings raised the concern that the hepatic metabolism of compound A may have resulted in generation of a reactive metabolite. This was the reason for conducting mechanistic studies to investigate the mode of action that resulted in the liver pathology observed. They were complemented by studies in the department of drug metabolism and pharmacokinetics, encompassing investigation of enzyme induction and into the metabolism in rats with radioactively labeled compound A (data not shown).
The mechanistic studies strengthened evidence for the involvement of a reactive metabolite in the liver findings of compound A. The peak of the marker enzymes of hepatocellular injury, ALT, AST, and GLDH, 6 h after dosing to mice, was following the delayed maximum plasma levels that occurred 4 h after dosing. Marked increases of plasma total bilirubin coincided with those increases and almost fully reversed 24 h after dosing. Furthermore, degenerative hepatocellular findings in mice were most prominent in the midzonal region, a specific distribution pattern characteristic for metabolism-related hepatotoxicity. 59
The definitive cause for the marked hyperbilirubinemia in mice remained unclear, as investigations into transporter inhibition were not conducted. The prominent increases of plasma triglycerides corresponded to the lipidosis at microscopy and is considered to reflect alterations of hepatocellular lipid metabolism.
The increase of liver tissue GSH and GSSG content indicated an activation of the detoxifying glutathione pathways. The minimum of the total GSH levels was reached 6 h after dosing, d shortly after plasma Cmax of compound A. This was interpreted as evidence for detoxification of a reactive molecule by glutathione. The markedly increased mRNA expression of enzymes involved in the glutathione metabolism measured in the mechanistic study in rats were further support.
Centrilobular to diffuse hepatocellular hypertrophy are generally considered as the morphological correlate for the induction of drug metabolizing enzymes, specifically of the Phase I enzymes of the cytochrome family. 19 Indeed, mRNA and protein of hepatic microsomal enzymes were substantially increased after compound A treatment, in particular for CYP 3A1 as determined by qPCR (data not shown). This contrasted with markedly reduced CYP 3A1 enzymatic activity at the dose level affected (data not shown). It was explained by mechanism-based inhibition, that is, the generation of a reactive metabolite by CYP 3A1 that by covalent binding to the enzyme inactivated it. Further evidence for the generation of a reactive metabolite was generated in a 14C-ADME study in rats. In this study, a low proportion of 2% to 5% of radioactivity administered could not be recovered from feces, urine and exhaled air 96 h post-dose, indicating covalent binding to structural molecules. By extensive and sophisticated nuclear magnetic resonance investigations it was possible to identify a reactive quinonmethine intermediate that is stabilized by nucleophilic addition of nucleophilic protein substructures, for example, SH-groups, which is considered responsible for the covalent modification of proteins observed with compound A (data not shown).
The reduction of thyroid gland hormones secondary to the induction of hepatic microsomal enzymes is a well-known pathomechanism in rodents. The mechanism behind is an induction of uridine diphosphate glucuronosyl transferases (UGTs). UGT1A1 and UGT1A6 glucuronidase T4, while UGT2B2 glucuronidates T3. 61 The increase of UGT mRNAs in the 4-week mechanistic study in rats was clear evidence for this mechanism. The reduction of T4 in this study was extraordinarily prominent with T4 values down close to the detection limit.
The occurrence of lipofuscin is considered to represent a major hallmark for the alteration of structural molecules. Lipofuscin represents a poorly defined mixture of oxidized or cross-linked endogenous lipids and proteins. It is insoluble and not degradable by lysosomal enzymes or the proteasomal system.17,26 Lipofuscin deposits may, therefore, indirectly indicate an increased oxidative stress and/or the presence of reactive metabolite(s). Increased lipofuscin deposits are usually associated with an increased activity of the ubiquitin-proteasome system, evidenced for compound A by increased proteasomal mRNA expression in the mechanistic study in rats.
The vacuolation of Kupffer cells present after 14-day dosing in rats has been characterized by electron microscopy to represent phospholipidosis. Also, hepatocytes contained few lamellar bodies, which was considered to represent a minor contribution to their microvesicular vacuolation apparent histologically. It is likely that peroxidation of the excessively accumulated phospholipids has contributed to lipofuscinosis.
In conclusion, compound A induced a pattern of findings that together are considered causally related to its reactive quinonmethine intermediate. These included hepatocellular hypertrophy, hepatocellular midzonal single cell necrosis (mice), increased CYP mRNA production but reduced CYP activity (“mechanism based CYP inhibition”), activation of glutathione detoxification, as well as accumulation of lipofuscin and increased mRNA encoding for enzymes involved in proteasomal degradation.
GPR40 Agonist
Dr Emmanuelle Balme and Dr Arno Kalkuhl (both Boehringer Ingelheim Pharma GmbH&Co KG, Biberach an der Riss, Germany) presented the toxicity profile of a G protein-coupled receptor 40 (GPR40) agonist in rat and dog. Molecular techniques were added as non-routine endpoints for mechanistic investigation of drug-induced toxicity.
GPR40 agonists are indicated for the treatment of type 2 diabetes mellitus. They act by potentiating glucose-stimulated insulin secretion (GSIS) by two pathways45,46: one is the activation of GPR40 on pancreatic β-cells and subsequent stimulation of insulin secretion, the other is the activation of GPR40 on enteroendocrine L cells and subsequent release of gut incretins, including glucagon-like peptide 1 (GLP1), glucagon-dependent insulinotropic peptide (GIP) and peptide tyrosine tyrosine (PYY). Clinical proof of concept (reduction of glycosylated hemoglobin HbA1c) for this mechanism was shown for the first-in-class GPR40 agonist Fasiglifam (TAK-875) in a phase II clinical trial.
The study cases came from two oral (gavage) repeat-dose toxicity studies in which a GPR40 agonist was administered at graduated dose levels to Wistar rats and Beagle dogs. The experimental facilities where the studies were performed complied with the standard of animal husbandry and welfare.
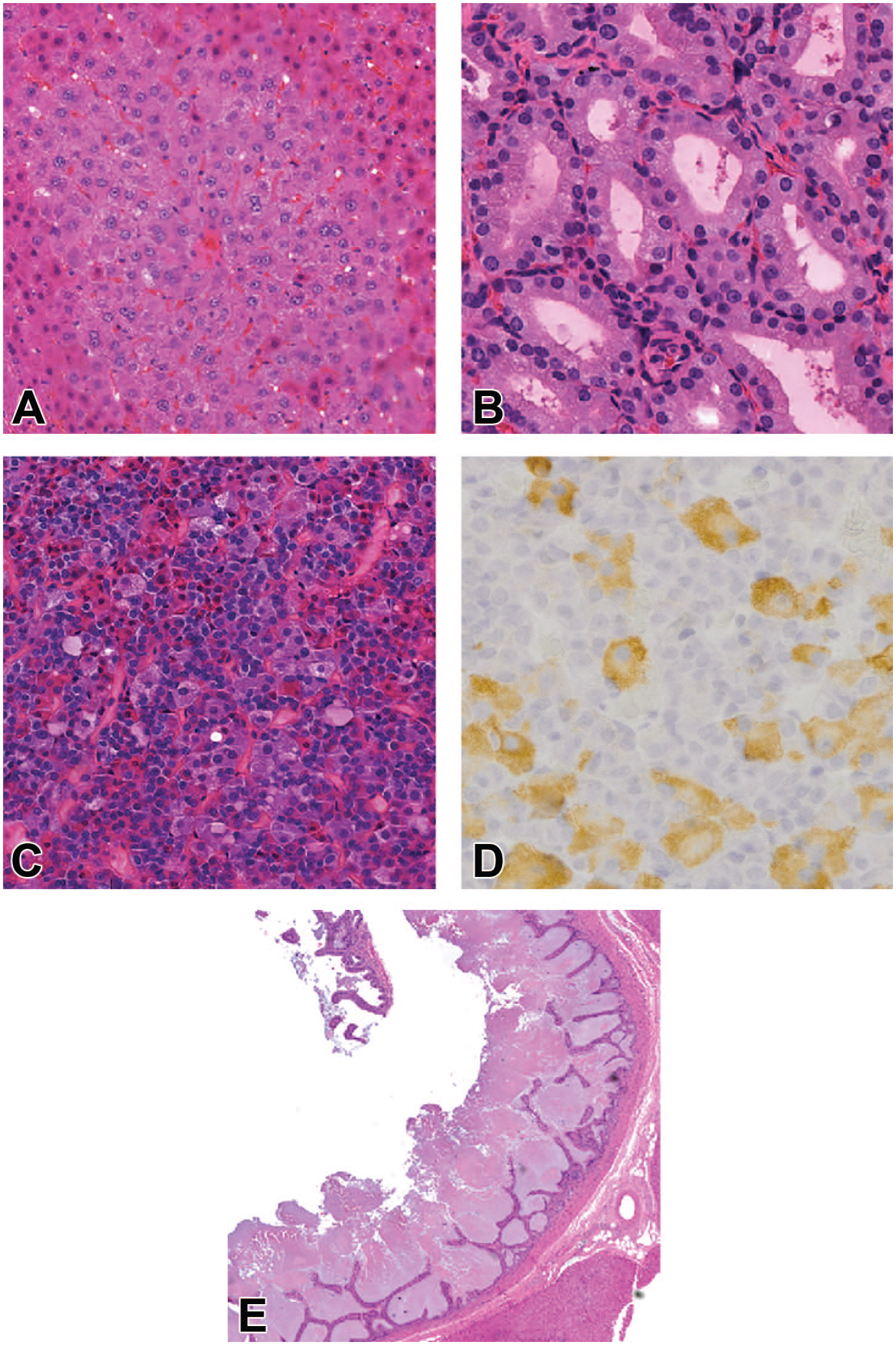
In Crl:WI(Han) rats, the compound was administered daily for 28 days at doses up to 1000 mg/kg. In the high-dose group, treatment led to the premature death/sacrifice of two animals with severe hepatic findings. Changes in clinical chemistry included increased plasma hepatic enzyme activities AST (1.5X), ALT (2.9X), and GLDH (3.0X), and increased plasma total bilirubin (4.9X). Histopathological findings were present in the liver and the pancreas. Most findings were reversible until the end of the 4-week recovery period. In the liver, hepatocytes, biliary system, and Kupffer cells were affected. Apoptosis of midzonal hepatocytes and diffusely increased hepatocellular mitoses were noted. In addition, diffuse or centrilobular (adaptive) hypertrophy of hepatocytes correlated with increased liver weight. Inflammation of bile ducts was characterized by mononuclear to mixed cell infiltrates and accompanied by degeneration/necrosis of the epithelium (Figure 5A). Basophilia and hyperplasia of the bile duct epithelium were regenerative in character and secondary to the degenerative process (Figure 5B). Kupffer cells were vacuolated, hypertrophic, and with foamy appearance. In the pancreas, the islet cells were vacuolated or apoptotic (Figure 5C). The vacuolated cells were positive for insulin by immunohistochemistry (IHC). Double staining for caspase-3 and insulin indicated that caspase-3 positive cells were frequently also positive for insulin.
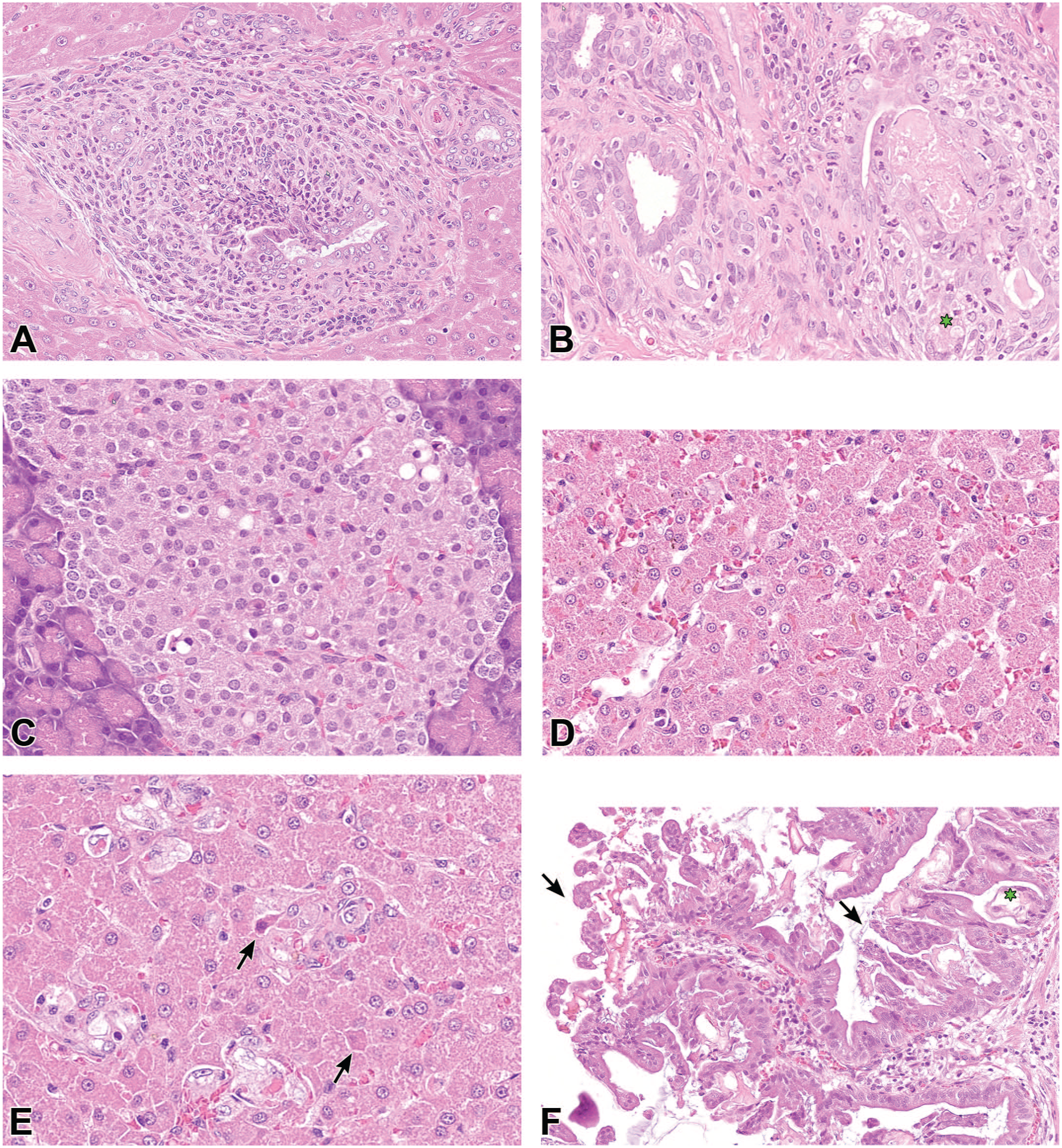
(A) Liver (H&E) from GPR40 agonist-treated rat. Inflammation, mostly composed of lymphocytes, macrophages, and neutrophils, was centered on bile ducts in the portal tract. Bile duct epithelium was infiltrated by inflammatory cells and focally replaced by necrotic cell debris. There was an increased number of small bile duct profiles reflecting hyperplasia. (B) Liver (H&E) from GPR40 agonist-treated rat. Granulomatous inflammation, characterized by infiltration of macrophages and multinucleated giant cells (asterisk), with fewer lymphocytes and neutrophils, surrounded bile ducts in portal tracts. Bile duct epithelium was infiltrated by inflammatory cells and appeared disorganized. Epithelial cells were either degenerated, hypertrophic (activation), or small and basophilic (hyperplasia). The lumen of bile ducts was sometimes obstructed by an eosinophilic acellular material (bile). (C) Pancreas (H&E) from GPR40 agonist-treated rat. A few islet cells were either vacuolated or appeared as rounded, shrunken, and hypereosinophilic apoptotic bodies surrounded by a clear halo. (D) Liver (H&E) from GPR40 agonist-treated dog. Brown pigment in hepatocytes and bile canaliculi was identified as bile with the Hall staining method (not shown here). (E) Liver (H&E) from GPR40 agonist-treated dog. Kupffer cells were hypertrophic, with large nucleus and nucleolus, marginated chromatin, and enlarged, foamy cytoplasm, sometimes engulfing cell debris, brown pigment, or red blood cells (phagocytosis). Few hepatocytes had shrunken and hypereosinophilic cytoplasm, and condensed nucleus (apoptosis, arrows). (F) Gall bladder (H&E) from GPR40 agonist-treated dog, showing diffuse mucosal chronic inflammation, and degeneration (asterisk), erosion, or reactive hyperplasia (cell stratification, arrows) of the mucosa-lining epithelium.
In Marshall Beagle dogs, the compound was administered daily for 14 days at doses up to 400/800 mg/kg. Clinically, dogs showed increased incidence of emesis, reduced body weight, and reduced food consumption. In clinical chemistry, increases in plasma were noted for AST (5.8X), ALT (17.0X), ALP (9.5X), GLDH (21.9X), GGT (5.8X), and total bilirubin (12.7X). These changes correlated with macroscopic yellow discoloration of mucous membranes indicative of jaundice, and histopathological findings. In urinalysis, orange colored urine and increased bilirubin and urobilinogen were noted. In the liver, multifocal chronic inflammation of bile ducts and increased bile duct profiles (hyperplasia) were sometimes associated with bile duct erosion. Brown pigment in hepatocytes and bile canaliculi was identified as bile with the Hall staining method (Figure 5D). In addition, parenchymal mononuclear cell infiltrates, hepatocellular apoptosis, and hypertrophic Kupffer cells were present (Figure 5E). The gall bladder showed mucosal chronic inflammation, reactive hyperplasia, degeneration, and erosion of the epithelium (Figure 5F).
Genomic analysis of the liver by quantification of mRNA via fluorescent tagging and hybridization to specific cDNA/oligonucleotide probes (Affymetrix) provided mechanistic information by molecular characterization of altered signaling pathways leading to hepatotoxicity. Gene expression changes were indicative of impaired fatty acid β oxidation, mitochondrial dysfunction, and perturbation of bile acid transport, as reported for other GPR40 agonists of the same “fatty acid-like” structure (carboxylic acids). Indeed, as medium- and long chain fatty acids are endogenous ligands of GPR40, carboxylic acid headgroup is a key feature of most GPR40 agonists.
Mitochondrial Injury as a Consequence of Impairment of Mitochondrial ß-Oxidation
Hepatic cellular processes that were affected based on the gene expression profiles included fatty acid biosynthesis, transport, and activation; triglyceride and cholesterol biosynthesis; α- and β-oxidation; PPAR target genes; oxidative stress; proteasomal degradation; inflammation; complement system; and cell cycle/regeneration. Genomic analysis indicative of interference with endogenous lipid metabolism was supported by drug metabolism investigation. Formation of GPR40 agonist-acyl-CoA thioester and GPR40 agonist-glycerol adduct was shown by in vivo radioactivity 14C-ADME studies. Accumulation of fatty acids and lipid intermediates in non-adipose tissue, along with diminished fat oxidative capacity, could have resulted in cytotoxicity. Indeed, the fatty acids that are not oxidized become trapped as anions inside the mitochondrial matrix and induce reactive oxygen species (ROS) production/oxidative damage and proinflammatory pathways with concomitant mitochondrial stress. 62 In addition, it is postulated that GPR40 agonists can inhibit fatty acid β-oxidation by blocking carnitine palmitoyltransferase-1 (CPT-1), which is responsible for importing fatty acids into the mitochondria for oxidation. GPR40 agonists do not directly inhibit CPT-1. However, the activation of GPR40 leads to enhanced GSIS, which is associated with the inhibition of CPT-1. 8 This inhibition prevents the import of fatty acids into the mitochondria, thereby promoting glucose metabolism over fatty acid oxidation. Inhibition of mitochondrial β-oxidation can also disrupt cell respiration by perturbing the energy-coupling properties and migration of electrons along the respiratory chain. Disruption of cell respiration leads to decline of ATP synthesis and impairs processes with high energy demand like apoptosis and protein synthesis, in favor of necrosis and inflammation. Further, depletion of GSH and ATP (a critical defense system within mitochondria which lack catalase) can trigger cell death and inflammation through the release of inflammatory cytokines, which in turn can activate nonparenchymal cells and contribute to the development of liver injury. Overall, impairment of mitochondrial ß-oxidation and mitochondrial toxicity were considered to be important mechanisms of hepatotoxicity. Pharmaceutical agents that affect mitochondrial function possess the propensity to cause mitochondrial injury, and the lesions are diverse, including steatosis, inflammation, apoptosis, necrosis, and fibrosis. 62
Alteration of Bile Acid Transport
Gene expression changes indicated perturbation of bile acid (BA) biosynthesis, and BA and drug conjugate transport in the liver, as reflected by the morphologic and clinical pathologic changes affecting the biliary system. GPR40 agonists have been shown to be in vitro inhibitors of multiple influx and efflux hepatobiliary BA transporters in human and animal models. Inhibition of hepatic transporters, specifically the bile salt export pump (BSEP) and multidrug resistant associated protein (MRP 2/3/4) have been identified as a potential cause for cholestatic drug induced liver injury (DILI).44,68 BAs are toxic to hepatocytes, and notably to mitochondria, and adaptive processes such as efflux via sinusoidal and canaliculi transporters are important for cellular protection. Also, glucuronides, including drug glucuronides and endogenous (bilirubin) glucuronides, are substrates for MRPs. Therefore, inhibition of BSEP and/or MRPs by drugs can be a hazard for cholestatic DILI because of the potential for accumulation of toxic BAs or reactive/toxic drugs in hepatocytes.
Pathogenesis of pancreatic lesions remained unclear. In the literature, GPR40 has been controversially discussed to be involved in lipotoxicity.42,63 Lipotoxicity is a metabolic syndrome that results from the accumulation of lipid intermediates in non-adipose tissues, leading to cellular dysfunction and death. Whereas short-term exposure to free FAs led to insulin secretion, it was shown that prolonged exposure of isolated islets to elevated free FA levels was associated with β-cell dysfunction and apoptosis.
Finally, relevance of findings to humans was discussed. Fasiglifam was withdrawn from Phase III clinical trials due to DILI. 33 Mechanistic studies revealed that formation of a reactive acyl glucuronide, and possibly an acyl CoA thioester intermediate in hepatocytes, coupled with inhibition of hepatic transporters and mitochondrial dysfunction, may have been key contributors of Fasiglifam-mediated DILI.18,44 Our clinical, pathologic and mechanistic data suggest that similar mechanisms (inhibition of ß-oxidation and mitochondrial toxicity, perturbation of endogenous lipid metabolism and effects on hepatobiliary transport) have led to hepatotoxicity in our preclinical studies and were therefore considered of human relevance.
Translatability of Dysgeusia
Dr Smitha Pillai (Pfizer Inc, San Diego, California) presented on the pathophysiology of dysgeusia (altered taste perception). Her presentation is the result of efforts to elucidate mechanisms of clinical dysgeusia from an oncology target, compound X, where 60% of patients reported grade 1-2 dysgeusia, with at least 50% experiencing bitter or metallic taste.
The physiology of gustation is complex and involves multiple receptors, pathways, and brain centers. The functional unit of taste is the taste bud, located on tongue papillae, palate, pharynx, and epiglottis. 67 In most animal species, the tongue generally contains 4 types of papillae: fungiform, circumvallate, foliate and filiform papillae. 6 Fungiform papillae occur on the anterior 1/3 of the dorsal surface, and each papilla usually contains one taste bud. The average number of fungiform papillae in rat tongue ranges from 100 to 200 with density of 1.3 papillae/mm2 of dorsal surface. 37 There is a single circumvallate papilla on the posterior aspect of rat tongue, which contains an average of 300 to 500 taste buds. 39 About 10 to 15 foliate papillae are located bilaterally along the posterolateral margins of the tongue and contain around 100 taste buds per papilla. Filiform papillae are distributed along the entire dorsal surface of the tongue, are mechanical and nongustatory (no taste buds), and account for the tongue’s rough and uneven surface. 9 Taste buds are clusters of up to 100 polarized neuroepithelial cells that form compact, columnar pseudostratified “islands” embedded in the stratified epithelium of the oral cavity. 67 The taste cells are divided into the following 4 types: type I (glia-like), type II (receptor cells), type III (presynaptic cells), and type IV (basal cells).16,28 The apices of the taste cells extend into the taste pore, an opening in the epithelium, and directly contact the external environment of the oral cavity. When one eats or drinks, taste or flavor molecules dissolve in saliva, interact with taste receptors on taste cells and activate signaling cascades. 70 Taste bud cells, similar to olfactory neurons, comprise a continuously renewing population.43,56 In the adult, each taste bud is innervated by 3 to 14 sensory ganglion neurons, depending on the species and oral region.30,66 Gustatory nerve fibers comingle with a rich plexus of other nerve fibers under the taste epithelium. The transmission of taste signals to the brain stem occurs through facial, glossopharyngeal, and vagus nerves, whose cell bodies are located in the geniculate, petrosal, and nodose cranial ganglia.7,67 There is a blend of olfaction, gustation, and texture in taste perception. The orbitofrontal cortex, the basolateral amygdala, and the insular cortex have been reported to be associated with interactions among odor, taste, and flavor.3,38
Dr Pillai further elaborated on taste disorders that can range from dysgeusia (abnormal taste sensation) to ageusia (complete loss of taste sensation), depending on the degree of severity of the taste abnormality. Any factor affecting components of chemosensation and peripheral sense organs (health of oral mucous membranes, tongue, papillae, salivary glands, olfactory epithelium), and/or neuronal unit (peripheral and central nervous system) can contribute to dysgeusia.11,21,58 Several genes and their receptors (ENaC, T1R, T2R, TAS2R, and TRPV1) are known to code for specific taste sensations and any sequence variation in these genes and receptors can cause taste disorders.1,4,28 More than 50% of the drugs prescribed routinely in the United States have been associated with taste disorders. The most notable of these are antiproliferative drugs used in cancer treatment that affect the normal turnover of taste and olfactory receptor cells. 49 Aging is also a factor for reduction in taste sensation.11,53
Routine Hematoxylin and Eosin (H&E) evaluation of tongue specimens collected in the standard toxicological assessment in preclinical species is not optimized for evaluation of taste bud numbers and morphology. In routine toxicological evaluation in rodents, typically a section of tongue is collected from the anterior aspect of the tongue. 47 Owing to the low density of taste buds in this region, taste buds are rarely included for evaluation in the section. Largest concentration and density of taste buds are present along the posterior aspect of tongue, within circumvallate papilla. Focused sampling of circumvallate papilla was done to ensure that adequate numbers of taste buds were collected for evaluation, as described in literature. 65 To optimize the technique, the entire tongue was collected from naïve rats, and a series of 8 transverse sections (with the last through the single grossly visible circumvallate papilla) were made. The sectioned circumvallate papilla was then placed face down for embedding. Ten serial microtome sections at 5 µm through the processed blocks of the tongue and circumvallate papilla provided adequate numbers of taste buds for histopathological evaluation. Once optimized in naïve rats, the same technique was used to examine taste buds from rats treated with compound X.
All procedures performed on animals were in accordance with regulations and established guidelines and were reviewed and approved by an Institutional Animal Care and Use Committee or through an ethical review process.
Dr Pillai reviewed the histology of the circumvallate papilla and taste buds. A single circumvallate papilla is present in posterior part of the rat tongue (unlike 8-12 circumvallate papillae along the sulcus terminalis in human) and is a dome shaped projection of connective tissue core lined by keratinized stratified squamous epithelium (Figure 6A). The connective tissue core is well innervated and vascularized. Embedded along the edges of the papilla are variable numbers of taste buds, which are polarized clusters of neuroepithelial cells. Serous salivary glands (von Ebner glands) are located in the submucous connective tissue of the tongue below the circumvallate papillae and their excretory ducts empty into the deep sulcus of the papilla (Figure 6A). There are adjacent mucous glands (Weber’s glands), whose ducts empty into the surface epithelium.
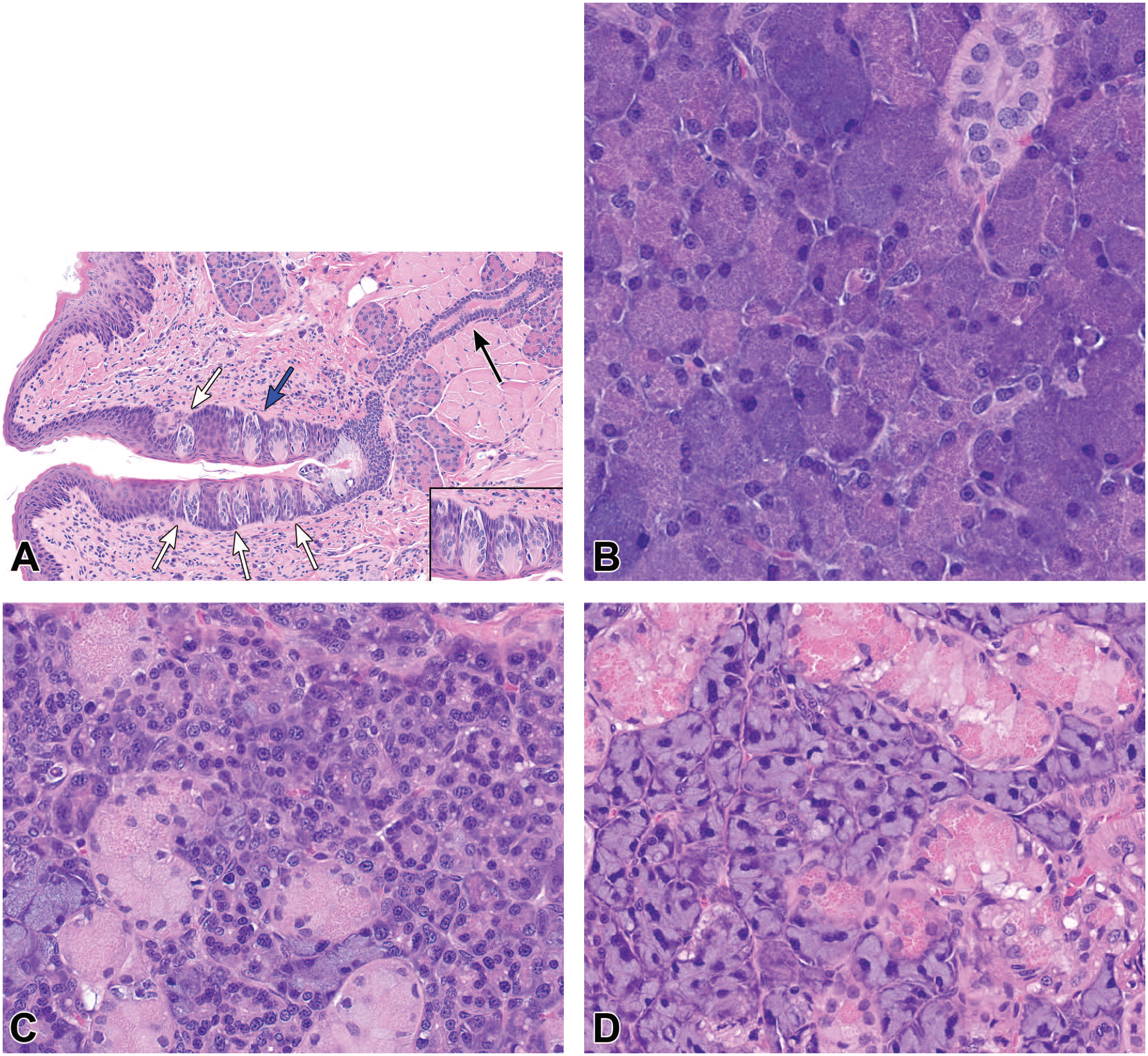
(A) Section from posterior aspect of rat tongue (H&E) showing circumvallate papilla with taste buds embedded in the epithelium (white arrows) along the lateral edges. von Ebner salivary glands are present in the muscularis with ducts draining into sulcus of circumvallate papilla (black arrow). Inset (from area of highlighted by arrow) showing taste buds at higher magnification. (B) Parotid salivary gland (H&E) from compound X-treated rat showing “foci of cellular alteration, basophilic” characterized by small to large clusters of acinar cells with increased cytoplasmic basophilia, with occasional enlarged or pyknotic nuclei. (C) and (D): Submandibular salivary gland (H&E) from compound X-treated male rat showing secretory depletion characterized by acinar and ductular cells containing fewer secretory granules (C) compared to control male rat (D).
Enumeration of the taste buds in serial sections from 3-month repeat dose oral gavage toxicity study in rats, as well as assessment of their morphology did not reveal a compound X-related effect, which could account for dysgeusia. There was minimally increased orthokeratotic hyperkeratosis of the squamous epithelium of circumvallate papilla, considered secondary to decreased mechanical exfoliation due to compound X- related inanition, resulting in poor body condition.
Compound X- related effects were present in the parotid and submandibular salivary glands. In the parotid salivary glands, there were small to large clusters of acinar cells with increased cytoplasmic basophilia, with occasional enlarged or pyknotic nuclei, diagnosed as “foci of cellular alteration, basophilic” (Figure 6B). Because of the lack of apparent hypertrophy of most of the affected acinar cells, the INHAND (International Harmonization of Nomenclature and Diagnostic Criteria for Lesions in Rats and Mice) term “hypertrophic basophilic focus” was not used. 41 In the submandibular salivary glands, there was minimal diffuse secretory depletion characterized by smaller acinar cells containing decreased secretory granules. Additionally, there was often decreased eosinophilic secretory granules within the lining cells of the convoluted (granular) ducts (Figure 6C).
Dr Pillai discussed the similarities between human and rodent salivary glands in terms of their anatomy, histology, and physiology. The major salivary glands in both species secrete fluids of similar composition.27,35 Saliva is the principal fluid component of the external environment of the taste receptor cells and plays a role in taste sensitivity. Saliva acts as a solvent and transporter of taste substances to the taste receptor sites. During this process, some salivary constituents chemically interact with substances and affect taste perception. For example, salivary buffers like bicarbonate ions decrease the concentration of free hydrogen ions (sour taste), and some salivary proteins bind with bitter taste substances. 36 Additionally, some salivary constituents can continuously stimulate the taste receptor, resulting in an alteration of taste sensitivity. Also, saliva protects the taste receptor from damage brought about by dryness and bacterial infection, and from disuse atrophy via a decrease in transport of taste stimuli to the receptor sites. Altered salivary protein composition (carbonic anhydrase VI/gustin, proline-rich proteins/PRPs, cystatins, α-amylases, histatins, salivary albumin, mucin, etc), flow rate, buffer capacity, and ionic composition are known to modulate taste sensation and have been associated with altered taste sensation.15,36 It was hypothesized in this case that the salivary gland changes could result in changes in saliva composition and thus contribute to dysgeusia. However, this needs to be investigated further as potential cause of clinical dysgeusia.
Dr Pillai acknowledged that dysgeusia is a common clinical side effect of many drugs, though mechanisms are poorly understood. Two of the highest prevalent symptoms reported by cancer patients are taste dysfunction (17.6%-93%) and dry mouth/xerostomia (40.4%-93%). The pathophysiology of taste alterations in cancer patients is complex. It may result from one or more of the following: damage to the gustatory system, loss or distortions of olfactory functions, systemic disease, local oropharyngeal conditions, hyposalivation, changes in the salivary composition, and the usage of specific drug types. 15 Changes in quantity/quality of saliva and/or salivary gland alterations might be considered, to investigate compound X-related dysgeusia in the clinic.
Footnotes
Acknowledgements
The authors acknowledge the key contribution to the success of the seminar by the local organization by Dr Theresa Störk and colleagues from the Institute of Pathology of the University of Veterinary Medicine, Hannover, Germany. Dr Siudak thanks Ute Bach and Claudia Stark of Bayer AG for scientific support during manuscript preparation. Dr Johnson thanks John Vahle and Armando Irizzary of Eli Lilly and Company for their review of the manuscript. Dr Pillai thanks the colleagues in Drug Safety Research and Development unit at Pfizer Inc. for their collaboration on this project.
Author Contributions
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Enrico Vezzali declared that the information presented is without guarantees of completeness or accuracy, and it represents the opinion of the author, not necessarily the position of Idorsia Pharmaceuticals Ltd., which commissioned the GLP and non-GLP toxicity studies performed to comply with the international guidelines.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
