Abstract
The cerebellum has intricate connections either directly or indirectly with most levels of the brain. While many of these are functionally relevant, prominent neuropathologic changes that reflect the underlying anatomy arise from injury to a few major pathways. Identifying these pathologic sequelae can help to identify the cells involved in the underlying disease. The cerebellum has two distinct components: the aesthetically pleasing cortical dendritic folia and the deep nuclei. The uniform and monotonous cerebellar cortex contains molecular, Purkinje, and granular layers. Several deep nuclei are the main efferent projections from the cerebellum. Key neuron types relevant to cerebellar pathology include the cortical excitatory granular input and inhibitory Purkinje output neurons, the deep nuclear excitatory projection neurons, as well as key afferent nuclei (eg, inferior olivary, pontine, and sensory neurons) and efferent targets (eg, red nucleus, thalamus). Pathologic processes that affect one or more of these neuron groups lead to stereotypic anatomical changes, which in turn signal the existence of those processes. This article first reviews key aspects of human cerebellar anatomy and histology, then discusses some key connections, and finally presents a selection of cases from humans that illustrate these interconnections.
Keywords
Cerebellar Anatomy
The cerebellum derives its name from the Latin for “little brain,” because it a distinct, smaller structure beneath the larger cerebrum. It is situated dorsal to the brainstem and beneath the cerebrum (see Figure 1A), in a bowl-like posterior fossa that is lined by dura below and the tentorium above. The cerebellar vermis (Latin for “worm”) is a midline division that indeed looks a bit like a worm (see Figure 1C) and is best viewed in a sagittal section (Figure 1B). This view demonstrates the leaflike or folia structure of the cerebellar cortex. It also shows the relationship between the cerebellum and the three levels of the brainstem: the medulla below, the pons, and the midbrain above (Figure 1B). Two related small folia are the midline nodulus, which is at the caudal end of the vermis (Figure 1B), and the flocculus, which are small, paired excrescences at the junction of the inferior and middle peduncles (Figure 1C).
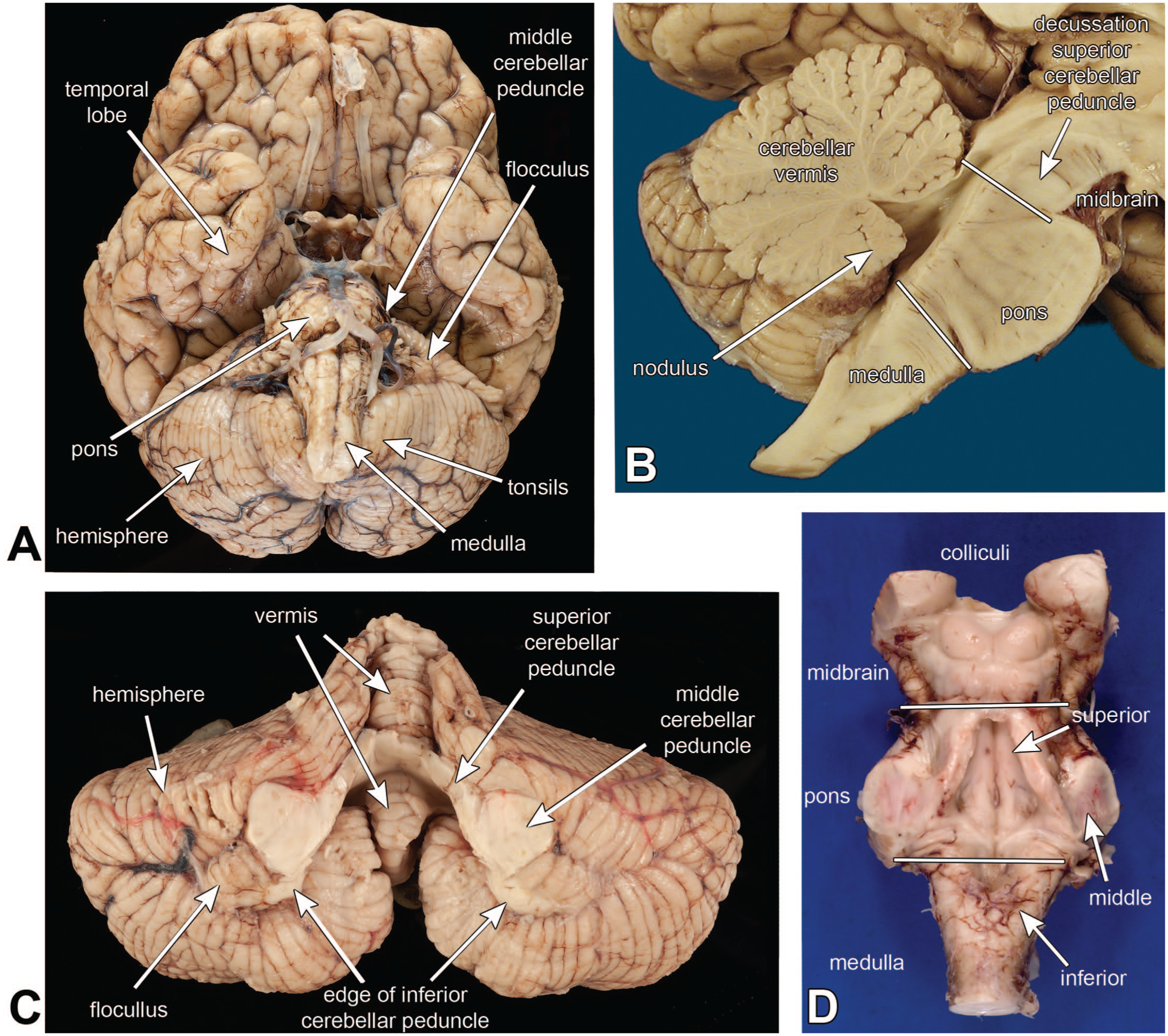
Cerebellar macroscopic anatomy. The basal view of the brain in panel A shows the cerebellum’s relationship with the surrounding brain. The cerebellum lies dorsal to the pons and medulla and ventral to the temporal lobes. In this view, only the middle cerebellar peduncle is visible, as it connects the pons base to the cerebellum. The cerebellar tonsils are immediately adjacent to the medulla. Panel B is from a sagittal section of the brain. This view shows the multiple leaflike folia of the vermis. It also shows the three levels of the brainstem (medulla, pons, midbrain). The superior cerebellar peduncle, which connects the cerebellum to the midbrain, decussates at the most caudal aspect of the midbrain and is visible as a great midline white matter structure. Dissection of the cerebellum (panel C) from the brainstem (panel D) reveals all three of the peduncles. In the central part of the cerebellum in panel C, the superior and middle peduncles blend together, and only the edge of the inferior peduncle is visible. The vermis in this view looks like a curled-up worm (compare with panel B). The cerebellar hemispheres are lateral (1A and 1C) and the region between these is the paravermal area. The flocculus is a small, paired collection of folia just lateral to the middle peduncle (1A and 1C). This structure is functionally connected to the nodulus and together they are sometimes referred to as the flocculonodular lobe. Panel D is a view of the brainstem, as if it had been removed from panel C and flipped over. It better illustrates the three levels of the brainstem and shows the superior peduncle in the rostral pontine tegmentum as it heads toward the midbrain, the middle peduncle connecting the pontine base to the cerebellum, and the inferior cerebellar peduncle, which connects the medulla and spinal cord afferents to the cerebellum.
The cerebellum is attached to the three levels of the brainstem by three great peduncles: the inferior cerebellar peduncle (icp) attaches to the dorsolateral medulla, the middle cerebellar peduncle (mcp) attaches to the lateral portion of the pontine base, and the superior cerebellar peduncle (scp) travels through the dorsal pons and attaches to the midbrain. The scp is the major output from the dentate nucleus (DN) and decussates to form a large white matter structure at the caudal midbrain (see Figure 1B). These three peduncles form a continuous region of white matter in the deep cerebellum (Figure 1C), and only in the context of the brainstem can they be distinguished grossly (Figure 1D).
Unlike the many different regions of the cerebral cortex, with their complex layering and multiple different cell types, the cerebellar folia are notably regular and monotonous. Only in the flocculus and nodulus show slight histologic variations. Although the cerebellum has functionally different areas that map very precisely to other regions of the brain and onto the body like a homunculus, these regions are all histologically identical. It does not have an equivalent of 40-some different-looking cerebrocortical Brodmann areas, since one small region of cerebellar cortex looks like any other small region (excluding the flocculus and nodulus). This is one reason why it can be easier to identify pathological changes in the cerebellum, compared to the cerebral cortex.
In histological sections (Figure 2A) using a combination of Luxol fast blue and hematoxylin-eosin (HE/LFB), the folia form dendritic structures that have a handful of branches and sub-branches, as well as lighter blue core of white matter in this myelin stain. This combination stain exquisitely highlights brain anatomy, since myelin, neuropil, and nuclei all stain different colors. The entire cerebellar cortical surface has three layers: an outer, pauci-cellular molecular layer, a Purkinje layer that forms a thin sheet of cells, and a granular layer containing innumerable tiny granular neurons. An external granular layer is present during early development but regresses away by the end of the first year of infancy. Although the external layer is absent in adults, the deeper layer is typically referred to as the internal granular layer or IGN (Figure 2B). The molecular layer is composed largely of fine neuropil, where granular neuron axons synapse on Purkinje neuron dendrites, and also has a sparse collection of other cells (Figure 2C). Notably, the molecular layer lacks any myelinated axons in this H&E/LFB (hematoxylin-eosin/Luxol fast blue myelin) stain. Purkinje neurons are large cells that have abundant cytoplasmic Nissl substance and a large nucleus having a single, large nucleolus (Figure 2D). They form a monolayer of cells. This layer also contains the Bergmann astrocytes that elaborate the radial glial fibers important for development. The Nissl substance represent abundant rough endoplasmic reticulum and indicates these cells produce ample proteins. The granular neurons in internal granular layer are arranged around large eosinophilic synaptic glomeruli (Figure 2E), which represent the large axon terminals of the afferent mossy fibers (mf) inputs. Although the molecular layer lacks myelinated axons, some of these axons are present in both the Purkinje and internal granular layers (small, light-blue staining linear structures in the LFB stains).
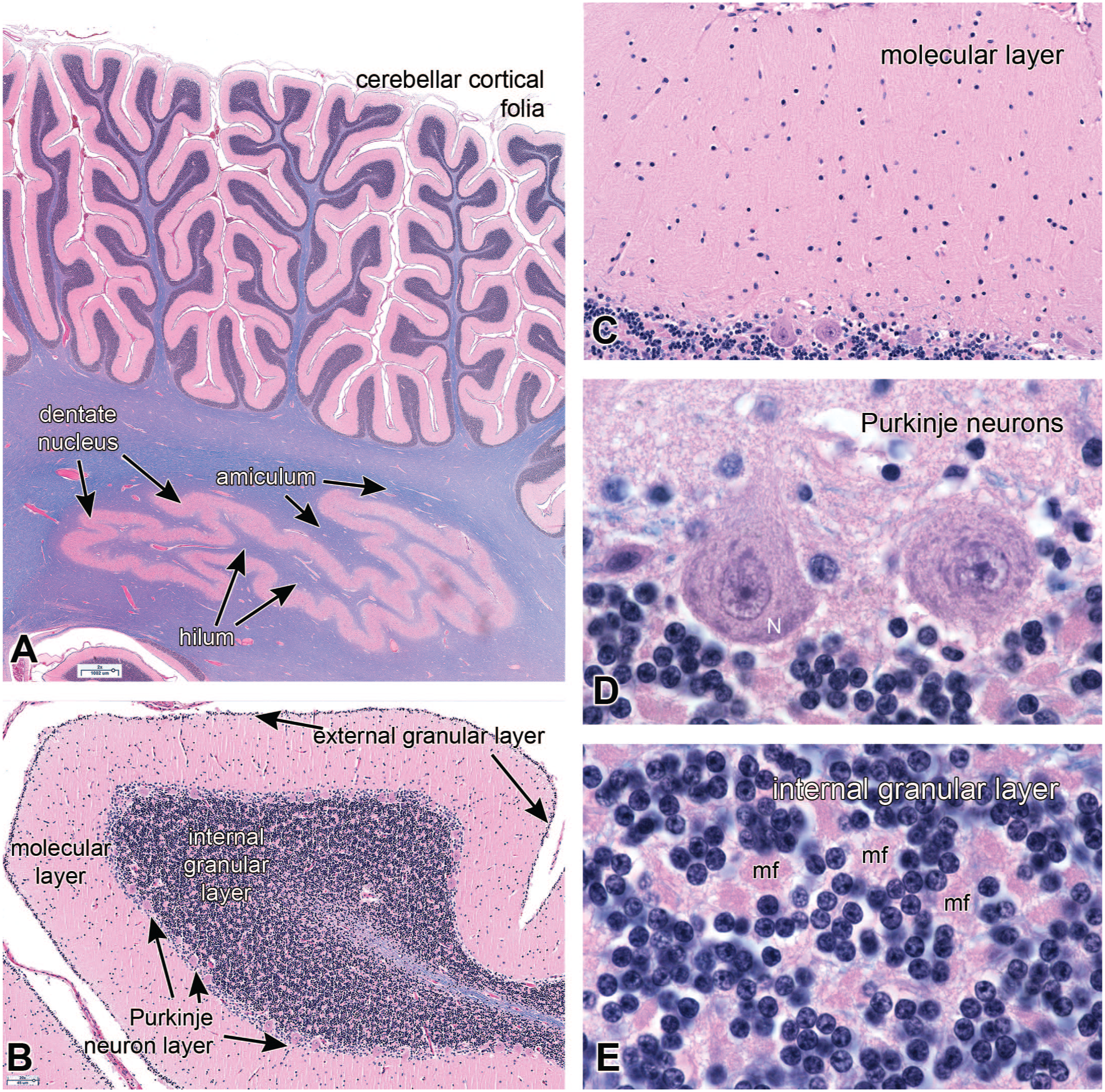
Cerebellar cortical histology; hematoxylin-eosin plus luxol fast blue (HE/LFB) stain. Panel A is a low-magnification view of several cerebellar folia and the deep dentate nucleus. In this view, the cortex has two prominent layers: the pink molecular layer and the darker blue internal granular neuron layer. White matter, which stains light blue with LFB, reaches up within the center of each folium. The dentate nucleus receives afferents through the white matter of its exterior amiculum and sends its efferents into the interior hilum and on into the superior cerebellar peduncle. Panel B shows a single folium from a several-month-old infant. At this age, the cerebellum still has a few remaining external granular neurons. The three additional mature layers are the pink molecular layer, the single-cell wide Purkinje layer, and the thick internal granular neuron layer. The molecular layer in panel C has a uniform, fine, pink-staining neuropil, and has a few embedded small stellate and basket neurons. The Purkinje layer (panel D) is defined by its single layer of large Purkinje neurons. In optimal conditions, these neurons have a large nucleus with a distinct nuclear envelope and a prominent nucleolus, as well as cytoplasmic Nissl substance (N), which represents accumulations of ribosomes. Internal granular neurons in panel E are small, non-descript neurons that have a small, dark nucleus and lack distinct cytoplasm. Embedded among the granular neurons are the mossy fiber (mf) synaptic glomeruli, which are the axon termini of cerebellar afferents from the spinal cord, medulla, and pons.
The cerebellar output nuclei lie in the deeper white matter closer to the ventricle. Within the cerebellum these are arranged from medial to lateral as the fastigial, globus, emboliform, and dentate nuclei. The globus and emboliform nuclei often blend together and in non-primate mammals are typically grouped together as the interposed nucleus. All of these nuclei can be identified in well-placed transverse cuts through the cerebellum and brainstem (Figure 3A and B), although this view only transects a small part of the dentate nucleus (compare with Figure 7A or Figure 2A). Each of these nuclei, from medial to lateral, receive projections from specific parts of the cerebellar cortex, also roughly from medial to lateral (vermis, paravermal region, hemisphere). The dentate nucleus has a continuous, undulating structure similar to a collapsed balloon (see Figure 2A). It is also composed of neuropil (np; see 3C and D) that has fine myelinated axons crossing through it (Figure 3D). Axons projecting to the dentate nucleus travel through the outer amiculum of white matter, while axons projecting from the nucleus to more distant regions travel through the inner hilum white matter (Figure 3C). See Kebschull 11 for a more detailed review of the cerebellar nuclei.
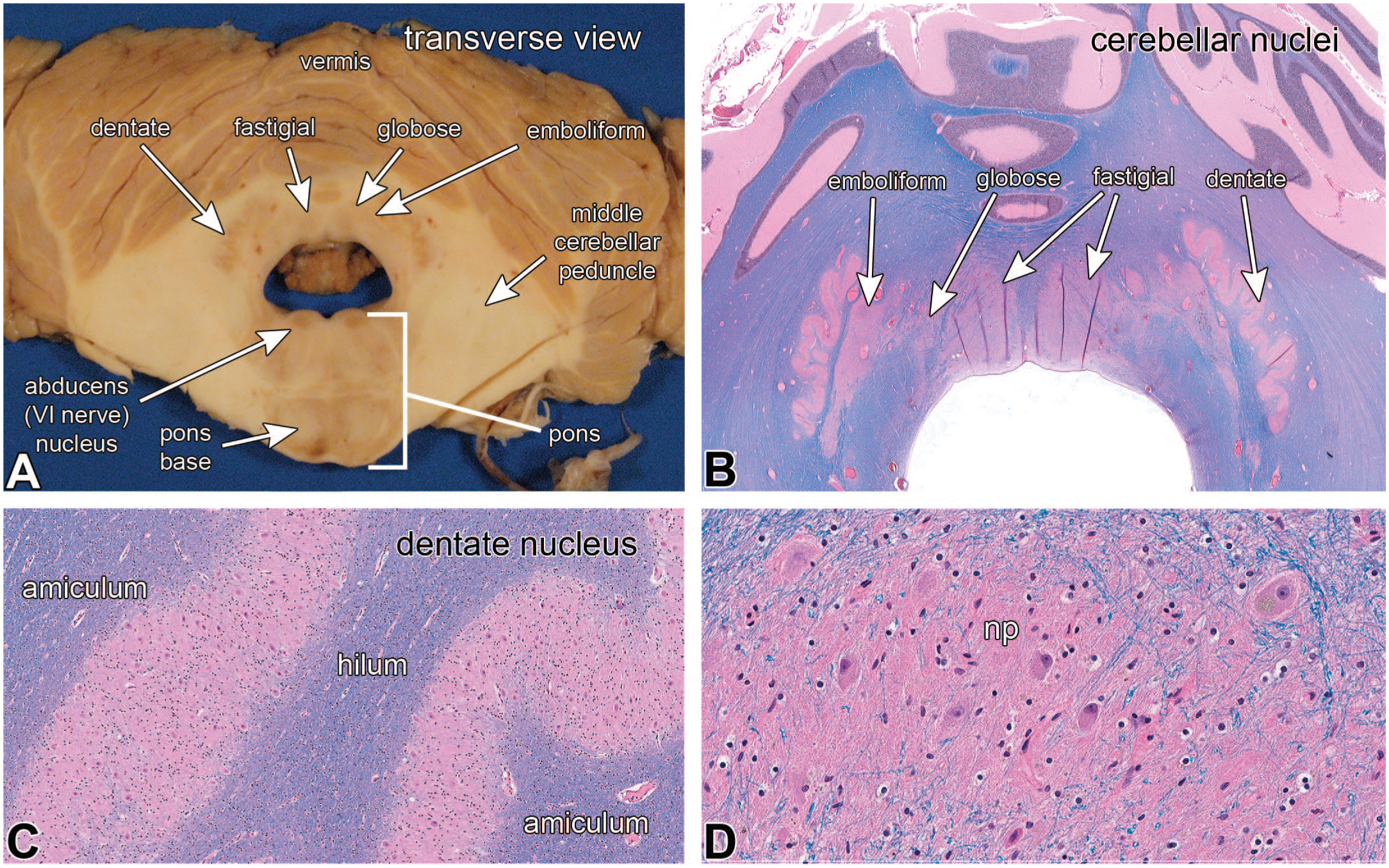
Cerebellar nuclei (hematoxylin-eosin-Luxol fast blue stains in panels B, C, and D.) In the transverse macroscopic view in panel A, the four paired cerebellar nuclei are from medial to lateral, the fastigial, globose, emboliform, and dentate nuclei. The globose and emboliform nuclei often seem to merge. This section also shows the caudal pons at the level of the abducens nucleus. This view is misleading because in most sections, only the much larger dentate nucleus is visible. Panel B is a histological view that is nearly identical as in panel A and again shows the four cerebellar nuclei. At intermediate magnification in panel C, the amiculum white matter inputs to the dentate nucleus extend to the border of its neuropil. Higher magnification of the dentate nucleus in panel D demonstrates its fine neuropil (np), some crossing myelinated axons, and the large neurons.
Cerebellum Development
Several aspects of cerebellar development provide a deeper understanding of its anatomy and pathological changes. The cerebellar nuclei and Purkinje neurons arise from the ventricular zone early in gestation, at around weeks 5 to 6 gestational age. 19 Granular neuron precursors develop near the end of the embryonic period, a few weeks after the Purkinje neurons. The Purkinje neurons migrate outward to eventually form a monolayer in the cortex. In contrast, the glutaminergic granular neurons arising from the rhombic lip migrate across the surface of the cerebellum within the external granular layer (see Figure 2C), where they continue to proliferate before migrating down Bergmann radial glial fibers within the molecular layer, past the Purkinje neurons and into the internal granular layer. Some granular neuron precursors remain in the external layer until late in the first year after birth. As the cerebellum matures, the cerebellar folia continue to develop until they reach their full complexity late in gestation. The cerebellum develops over a long time period, from the early embryo until late in the first year. Injuries to the cerebellum during this interval can alter its stereotypic developmental sequence, and hence deviations in its anatomy or histology can act like a timer for when those injuries had occurred.
Figure 4 is from an autopsy on an infant who died at the equivalent gestational age of 42 weeks. The infant had been diagnosed prenatally with pontocerebellar hypoplasia and was later found to have a mutation in the mitochondrial arginyl transfer RNA synthetase gene RARS2. 9 Compared to normal full structure (see Figure 1), the cerebellar hemispheres were thin wings of tissue, and the pontine base, composed of neurons projecting to the cerebellum, was greatly reduced (Figure 4A and B). In the sagittal section, the normally nearly circular vermis (see Figure 1B) was greatly reduced in size and complexity (Figure 4C). While the nodulus in the normal vermis is relatively small, in the infant it appeared relatively large, compared to the remaining vermis. Microscopically, the cerebellar vermis was greatly simplified. It retained features from early development, including the primary fissure, the anterior and posterior lobes, and the nodulus. However, the folia were fewer in number and lacked the typical sub-branches (compare 4C and D with Figure 1B). Three distinct cortical layers were only present in the nodulus, while the molecular layer was rudimentary in the anterior lobe, and parts of the posterior lobe lacked any real layers. The cerebellum had developed early but later stopped. The timing of the injuries could roughly be assessed by comparing the infant’s cerebellar vermis with those from other fetuses at different gestational ages (Figure 5). Comparing Figures 4D and 5, the infant’s cerebellum had developed to between 14 and 18 weeks, after which development ceased and then began to degenerate. The injury slowed and then stopped normal development before and up to about an estimated 16 gestational weeks, 9 based on published diagrams of cerebellar vermal development. 17
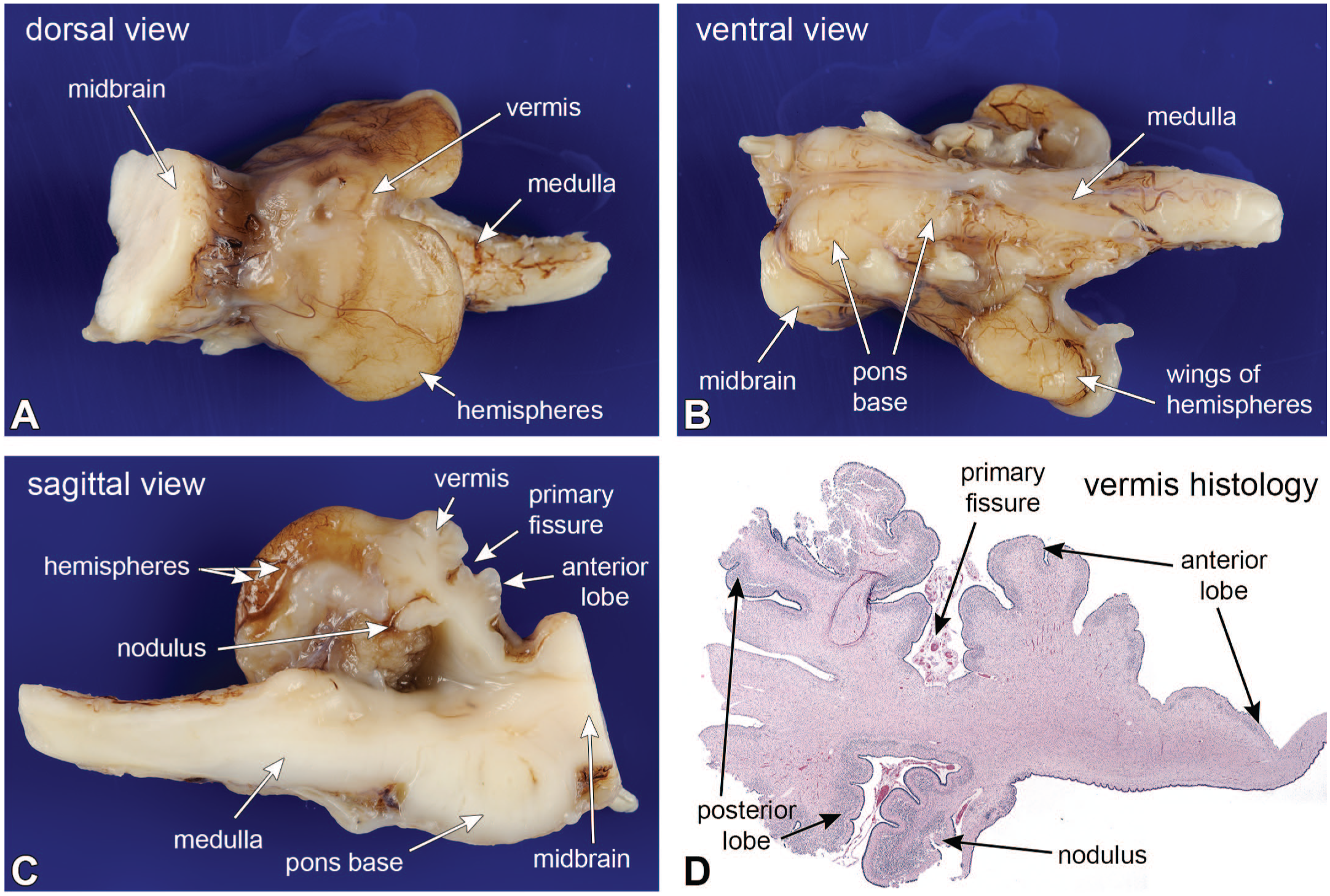
Pontocerebellar hypoplasia; cerebellum as developmental chronometer. At autopsy, the cerebellar hemispheres were flattened (panels A, B, and C), while the vermis was shrunken and had reduced folia (compare panel C with Figure 1B). The more phylogenetically ancient nodulus was the best preserved (compare the nodulus in pane C with Figure 1B), while the remaining folia were markedly reduced. The gross changes were recapitulated in the sagittal histology in panel D. The nodulus was better preserved and had a distinct external granular, molecular, and external granular layers. In the anterior lobes, the molecular and internal granular layers merged, while parts of the posterior lobe showed no demonstrable cortical layering. Contrast the infant’s vermis in panel D with normal cerebellar vermis development at different ages with Figure 5. The infant’s cerebellar vermis had developed to between 14 and less than 18 gestational weeks and then began to regress.
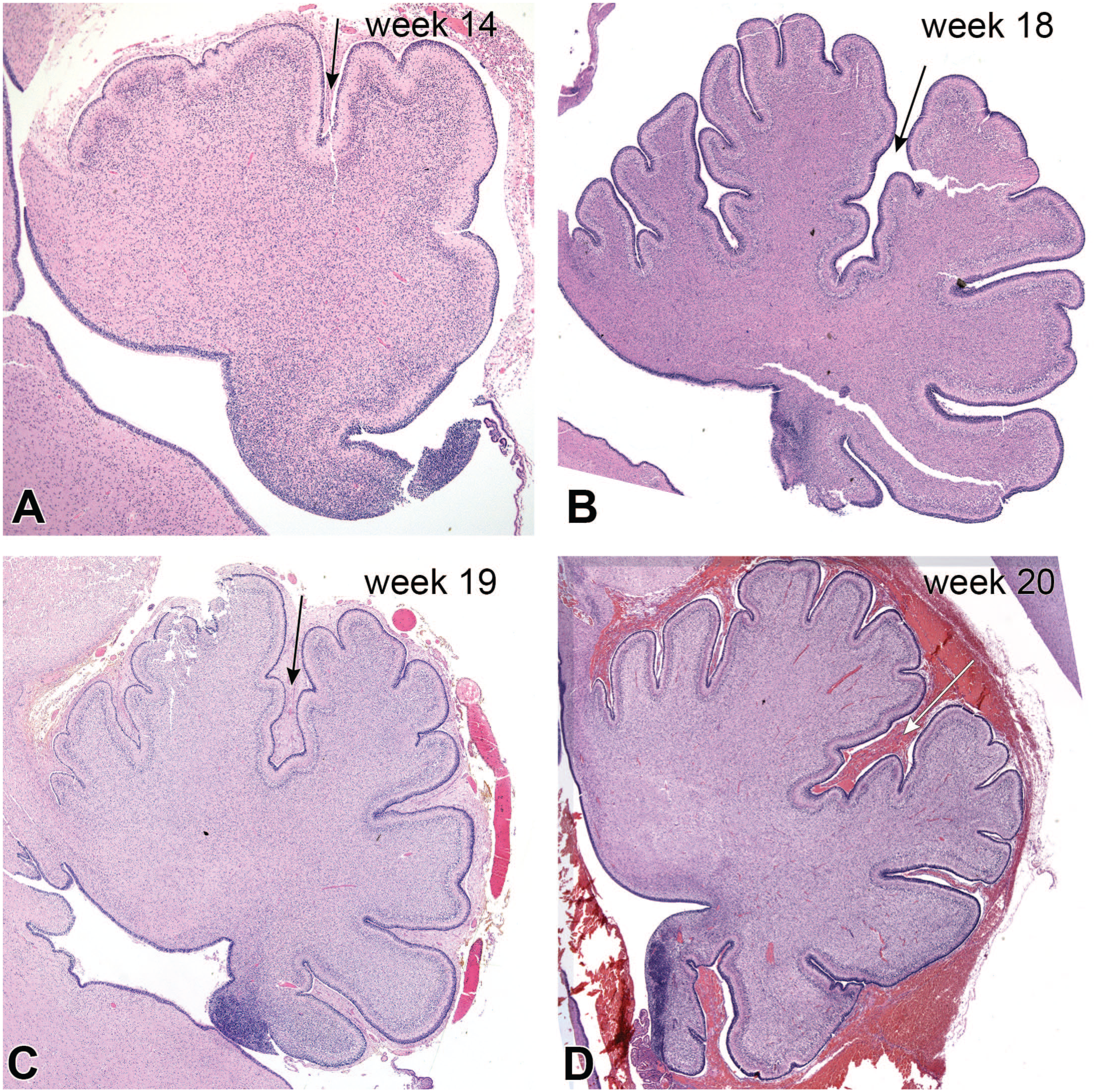
Cerebellar vermis development: 14 (A), 18 (B), 19 (C), and 20 (D) weeks gestational ages. Hematoxylin-eosin stains. The arrows in each figure denote the primary fissure of the cerebellum. Notice in all panels, the cerebellar folia have distinct, multilayered external granular layers (blue-staining layer on exterior of folia). This layer becomes depleted by about a year old. During development, the cerebellar folia start as a few knobs separated by a pair of fissures and later become increasingly complex. Their neurons and the cortical layers also become increasingly differentiated. The blue areas at the lower right sides of each figure are part of the cerebellar primordia that associates with the nodulus.
Cerebellar Connections: Afferents, Intrinsic Connections, Efferents
The cerebellum receives afferent sensory information from most regions of the body, which connect through spinal and select cranial nerves to relay nuclei in the spinal cord and brainstem (Figure 6A). For example, spinal motor afferent fibers from muscle spindles and Golgi tendon organs in the legs enter the spinal cord and synapse on Clarke’s nucleus. Clarke’s neurons then project in the laterally placed spinocerebellar tract to the ipsilateral cerebellum as mossy fibers, with collaterals to the deep nuclei. Similar information from the arms projects to the ipsilateral accessory cuneate nucleus in the caudal medulla. These sensory data enter the cerebellum through the inferior cerebellar peduncle (icp) in the lateral medulla.
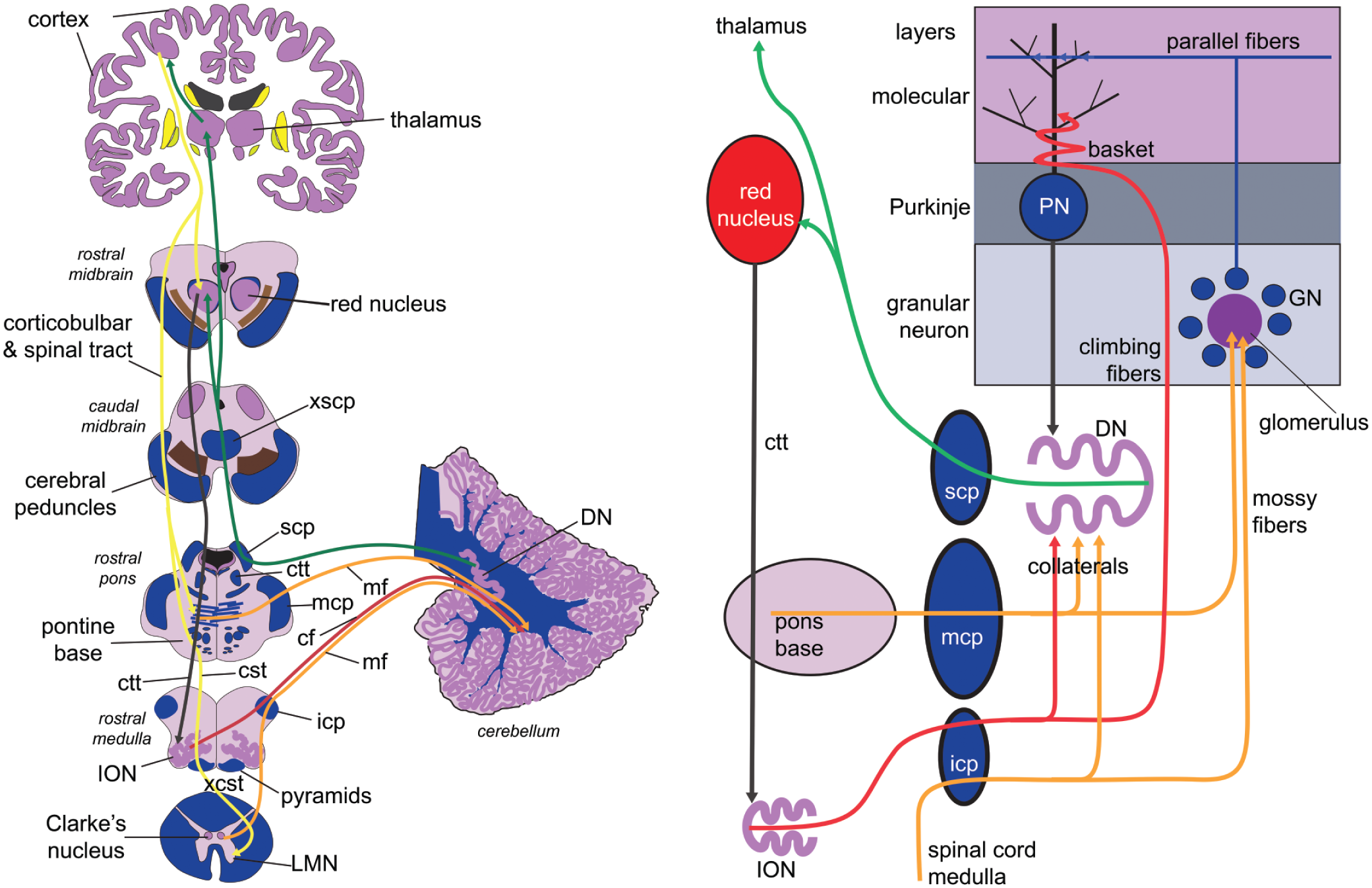
Cerebellar connections. Part A is a diagram of a section of major cerebellum connections with other brain regions, while part B is a schematic of intrinsic cerebellar connections, including some extrinsic nuclei. Tract colors: yellow tracts—projections from cortex; red tracts—climbing fiber cerebellar inputs; orange tracts—mossy fiber cerebellar inputs; black tract—Purkinje-dentate projection; green tracts—dentate nucleus efferents projections. cf, climbing fibers; cst, corticospinal tract; ctt, central tegmental tract; DN, dentate nucleus; GN, granular neuron; icp, inferior cerebellar peduncle; ION, inferior olivary nucleus; LMN, lower motor neuron; mcp, middle cerebellar peduncle; mf, mossy fiber afferent fibers or termini; PN, Purkinje neuron; scp, superior cerebellar peduncle; xcst, decussation of corticospinal tracts; xscp, decussation of superior cerebellar peduncles.
Input from the cerebral cortex (Figure 6A) arrives via two indirect pathways, one mossy fiber and one climbing fiber: (1) Corticobulbar fibers project ipsilaterally to the pontine base, which then sends mossy fibers through the massive middle cerebellar peduncle into the contralateral cerebellum. (2) Cortical fibers also project onto the ipsilateral red nucleus, which in turn projects in the central tegmental tract (ctt) through the pontine tegmentum to the ipsilateral inferior olivary nucleus (ION), which then projects as climbing fibers through the contralateral icp to the Purkinje neurons.
The outputs from the cerebellum arise from the cerebellar nuclei in the deep white matter. For the dentate nucleus, this information travels through the superior cerebellar peduncle (scp) in the pons, crosses in the decussation of the superior cerebellar peduncle (xscp) in the caudal midbrain, then projects to the contralateral red nucleus (RN) or on to the ventral tier thalamic nuclei, which then project on to cortex. Outputs from other cerebellar nuclei transmit to the pons and medulla.
Two distinct afferent fibers project to the cerebellar cortex: the mossy fibers and the climbing fibers (Figure 6B). The climbing fibers arise exclusively from the inferior olivary nucleus (ION) or one of its two accessory nuclei, project into the ION hilum, then cross the midline medulla and project out through the medial aspect of the contralateral inferior cerebellar peduncle (icp) to the contralateral cerebellum. These fibers terminate as a series of branches around the Purkinje neuron and synapse on its proximal dendrites. This projection is very narrow and precise: each Purkinje neuron is innervated by only a single climbing fiber, and each inferior olivary neuron projects only onto about ten or so Purkinje neurons. Important collaterals also project to corresponding cerebellar nuclei. The mossy fiber input basically comes from everywhere else, including the spinocerebellar, cuneocerebellar, and pontocerebellar afferents. As shown in Figure 2E, these mossy fiber terminal axons are huge and project onto multiple granular neuron dendrites in the granular layer. The small granular neurons project into the molecular layer, then split into two long axons that run parallel to the axis of the folium. These cross the field of Purkinje neuron dendrites, which lie in a plane that is perpendicular to the parallel fiber axons and the long axis of the folium. Unlike the precision of the climbing fibers, a single granular neuron axon can synapse on thousands of Purkinje neuron distal dendrites. The granule cell input to Purkinje neurons is modified by intrinsic neurons in the granular and molecular layers. The GABAergic Purkinje neurons provide the exclusive inhibitory output from the cerebellar cortex to their corresponding nuclei. The inhibitory Purkinje input is counterbalanced by the collateral excitatory inputs from the afferent fibers.
Given all of the different connections and decussations, it is easy to lose track of a general principle: many cerebellar lesions result in ipsilateral clinical deficits. To see this, imagine an infarct in the right cerebellar hemisphere (see Figure 6A). The missing Purkinje neuron output would normally project to the right dentate nucleus. The projection from this nucleus would decussate in the superior cerebellar peduncle decussation in the caudal midbrain before synapsing on the left thalamic ventral lateral nucleus. From there, the missing signal would have projected to the left cerebral motor cortex. The large cortical Betz neurons would project as corticospinal fibers through the left internal capsule, left cerebral peduncle, left pontine base, and left medullary pyramids. These fibers would then decussate at the base of the pyramids in the pyramidal decussation and project to right spinal lower motor neurons (LMN). Information from the right cerebellum decussates twice before reaching its target spinal motor neuron. This “double-cross” system explains why many cerebellar lesions cause ipsilateral motor deficits.
An interesting feedback loop involves the dentate, red, and the inferior olivary nuclei, known as the Guillain-Mollaret triangle (see Figure 6B and also Figure 15B). Beginning at the dentate nucleus, the first leg of the triangle is the superior cerebellar peduncle, which crosses in the caudal midbrain and projects to the contralateral red nucleus. The red nucleus then projects via the central tegmental tract (ctt) in the pontine tegmentum (dorsal pons) down to the ipsilateral inferior olivary nucleus (ION). From there, the pathway crosses again in the midline of the medulla, exits through the contralateral inferior cerebellar peduncle (icp), and collaterals return to the contralateral dentate nucleus. Interruption of this pathway can cause palatal myoclonus (a repetitive clicking of the palate) and olivary pseudohypertrophy (see Figure 15 for details).
Pathological Cases Illustrating Aspects of Cerebellar Anatomy or Diseases
The following examples of cerebellar pathologic changes illustrate aspects of cerebellar anatomy. Several pathological principles are important. (1) The loss of a neuron results in the loss of its axon and its dendritic arborization, which is often accompanied by a reactive astrocytosis or gliosis. (2) Larger neurons are often more sensitive to insults than smaller neurons, while glia tend to be the most resistant to various injuries. (3) Sometimes, loss of a neuron can lead to transsynaptic degeneration of either afferent (retrograde) or efferent (anterograde) neurons.
A common finding at autopsy of patients who die from respiratory failure is illustrated in Figure 7. Granular neurons are very sensitive to acidemia and can autolyze quickly after death.1,2 The autolysis makes the granular layer pale on dissection (Figure 7B), which has been informally termed an icy state or état glacés. Microscopically, the internal granular neurons undergo rapid autolysis, including dissolution of their nuclei, which renders the granular layer pale, instead of basophilic (compare with Figure 2B and E). This change occurs close to or after death, since it leaves no other anatomic sequelae. The phenomenon is also present in Figure 13C.
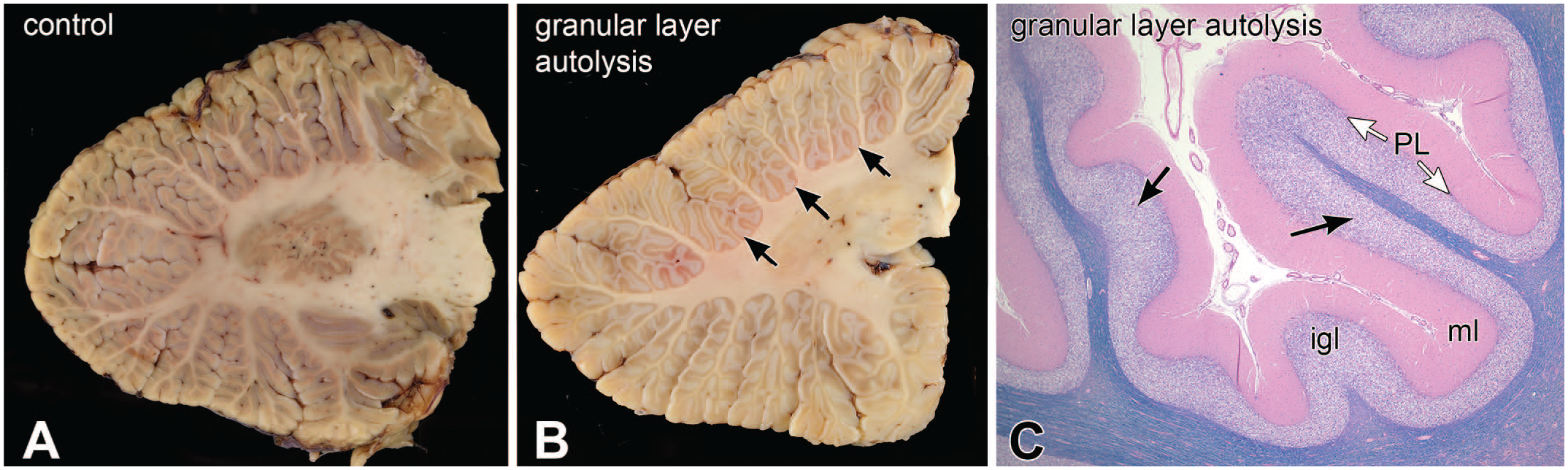
Acidosis; autolysis of granular neurons. Panel A is a parasagittal view of a normal cerebellum. Panels B and C illustrate the gross and histologic features, respectively, of granular neuron autolysis or “état glacé.” Compared to the control, the affected granular cellular layer is white (black arrows). Microscopically, the internal granular neuron nuclei have lost their integrity and broken down. Nuclei are smudged, which causes staining pallor in this hematoxylin-eosin-Luxol fast blue stain. igl, internal granular layer; ml, molecular layer; PL, Purkinje layer.
Cell death following ischemia depends on multiple factors, including the duration and completeness of the injury and the vascular territory involved. Figure 8 illustrates the cerebellum from a patient who developed a temporary clot in his cerebellar vasculature, which caused a partial infarct in the lateral cerebellum (Figure 8A and B). A complete infarct would have killed all cell types and left a cavitated lesion, while this incomplete infarct left some remaining but shrunken cortex (Figure 8B and C). As is typical of partial and borderline infarcts, the changes more affected the depths of the folia while relatively sparing the outer surface of folium (Figure 8C). Microscopically, the Purkinje and granular neurons were lost, the internal granular layer (IGL) neuropil became cavitated, while Bergmann astrocytes were relatively retained in the Purkinje layer (PL; white arrows in Figure 8D and black arrows in Figure 8E). Since both types of neurons have processes in molecular layer, this layer was no longer supported and had involuted or collapsed (Figure 8E). However, the surviving Bergmann glia retained their primordial radial glia fibers, which left fine radial processes that span the involuted molecular layer and is called “Bergmann gliosis.” (Figure 8E).
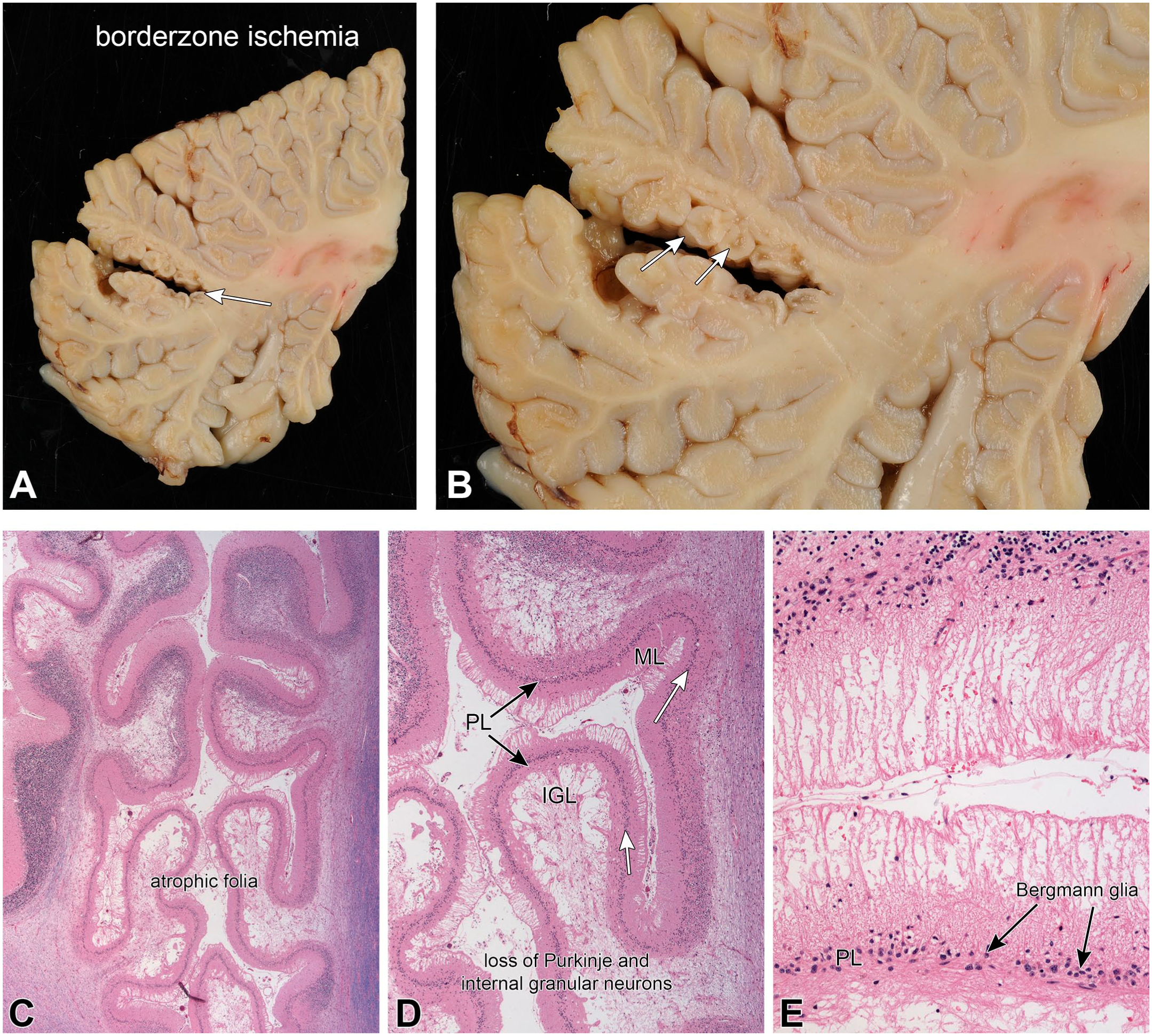
Partial ischemia; molecular layer involution. Panels A and B are macroscopic images of the lesion, showing its location (white arrow in A) and the shrunken but retained small folia (white arrows in B). This caused a loss of both Purkinje and internal granular neurons. The H&E/LFB stains in panels C–E demonstrated the extensively atrophic folia (C and D) that had a marked loss of neurons. In the most involved regions, the internal granular layer (IGL) was devoid of neurons (D) and the molecular layer (ML) had collapsed. The nuclei in the remaining Purkinje layer (PL) were residual Bergmann glia. During early cerebellar development, these astrocytes elaborate the original radial glia network down which the external granular layer precursor neurons migrate past the Purkinje neurons, and into the internal granular layer (also see external granular neurons in Figure 2 panel B and Figure 4). Bergmann astrocytes are more resistant to ischemia than the neurons. In some regions from this brain, the molecular layer had retained only these residual radial glia and had completely lost its normally fine neuropil (E). This pattern of pathology is known as “Bergmann gliosis.”
The chemotherapeutic agent ara-C (cytosine arabinoside) is used to treat certain types of leukemia and has a known toxic side effect on the cerebellum. 13 Figure 9 illustrates the cerebellum from a middle-aged individual who developed ara-C cerebellar toxicity during treatment for acute myeloid leukemia. The macroscopic image of the cerebellar vermis at autopsy (Figure 9A) demonstrated widespread folia atrophy, that was greatest in the anterior lobe but extended past the primary fissure into the posterior lobe (panel A). Microscopy from an affected region (Figure 9B) revealed an extensive loss of Purkinje neurons and a partial involution of the molecular layer secondary to degeneration of Purkinje dendrites. A brown discoloration was prominent around the dentate nucleus, especially its more lateral parts (Figure 9C). This brown discoloration represents the macroscopic correlate of an underlying reactive gliosis. Dentate nucleus neurons were preserved but shrunken in the H&E/LFB microscopy (Figure 9D and F), while the white matter of the surrounding amiculum had a significant loss of myelinated axons (pale blue staining) and an accompanying reactive gliosis (staining a slightly more eosinophilic or reddish color in H&E stains of the involved tissues in Figure 9F). The diagram in Figure 9E emphasizes that the loss of Purkinje neurons dendrites and axons leads to both an involution of the molecular layer and axonal loss with and accompanying gliosis in areas projecting to the dentate nucleus.
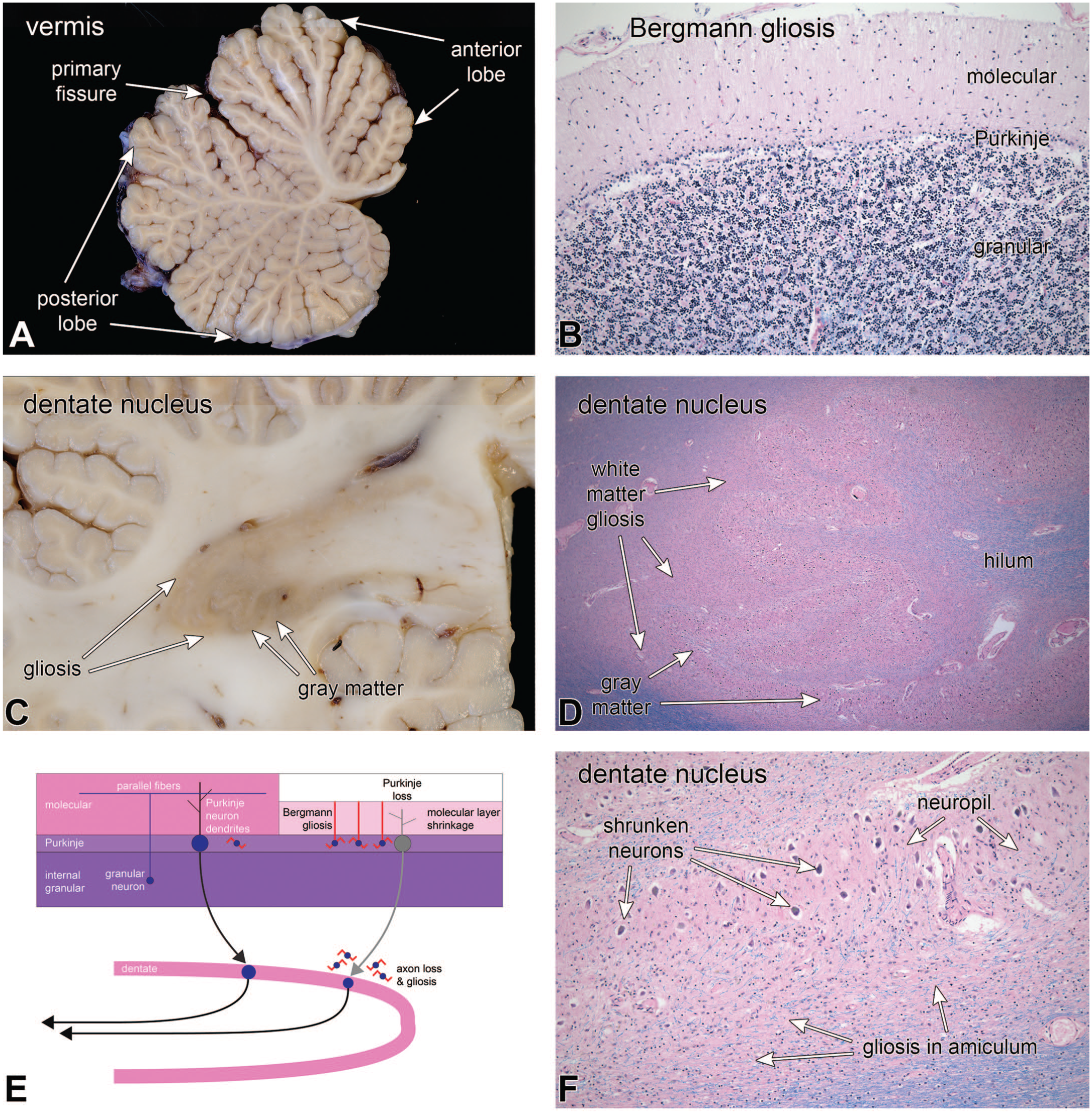
Ara-C toxicity; selective loss of Purkinje neurons. At autopsy, folia in the vermis, especially in the anterior lobe, were somewhat atrophic (A). Histology of an affected region in (B) demonstrated a loss of Purkinje neurons, more prominent Bergmann astrocytes in the Purkinje layer, as well as shrinkage of the molecular layer. The molecular layer had lost its uniform neuropil and instead had fine ascending processes from the Bergmann glia. Purkinje neurons project their inhibitory axons down to the cerebellar nuclei, including the dentate nucleus. Loss of Purkinje axons led to myelinated axon loss and myelin pallor (panel D, HE/LFB), as well as reactive gliosis (light brown color in macroscopic sections in C or eosinophilia in D) in the white matter (amiculum) immediately surrounding the nucleus (panels D and F). Dentate neurons also appeared shrunken (F). The diagram in E summarizes changes from chronic Purkinje neuron loss (molecular layer thinning, Bergmann gliosis, loss of axons, and gliosis around their target nuclei).
In patients who develop Purkinje neuron loss but have not lost corresponding inferior olivary neurons, the climbing fibers can remain intact around the empty Purkinje neuron space. The patient illustrated in Figure 10 was a woman who presented in her late 60s with frequent falls and progressive dysarthria. Before her death, neuroimaging had demonstrated severe, generalized cerebellar volume loss. At autopsy, she had widespread phosphorylated tau deposition, including in her dentate nucleus (for discussions of similar phenotypes, see Houlden et al 8 , Kano and Watanabe 10 , and Koga, Josephs and Ogaki. 12 In Figure 10A, her cerebellum displayed generalized atrophy of the folia that was slightly greater in the anterior lobe. Microscopically, the involved folia showed extensive Purkinje neuron loss and concomitant molecular layer thinning (Figure 10B; H&E/LFB stains). Moderate magnification demonstrated the expected Bergmann gliosis with distinct radial glial fibers in the shrunken molecular layer (Figure 10C; H&E/LFB). Purkinje neurons were nearly totally lost; however, prominent pink tufts remained in the Purkinje layer (long white arrows). In the Bielschowsky silver stain (Figure 10D), the pink tufts were silver-impregnated processes of empty baskets, each of which represented the original climbing fiber termini around their now-absent Purkinje neuron cell body and proximal dendrites. The inferior olivary nucleus had retained many neurons (black arrows, Figure 10F) but showed an overall moderate neuron loss and an intense gliotic reaction (white arrowheads, Figure 10F). The diagram in E schematizes the loss of Purkinje neurons and Bergmann gliosis, while retaining olivary climbing fibers as empty baskets. In this patient, it was unclear how the Purkinje neuron loss was related to losses elsewhere in the brain (eg, dentate nucleus), since no abnormal tau had aggregated in the rare remaining Purkinje neurons nor in inferior olivary neurons.
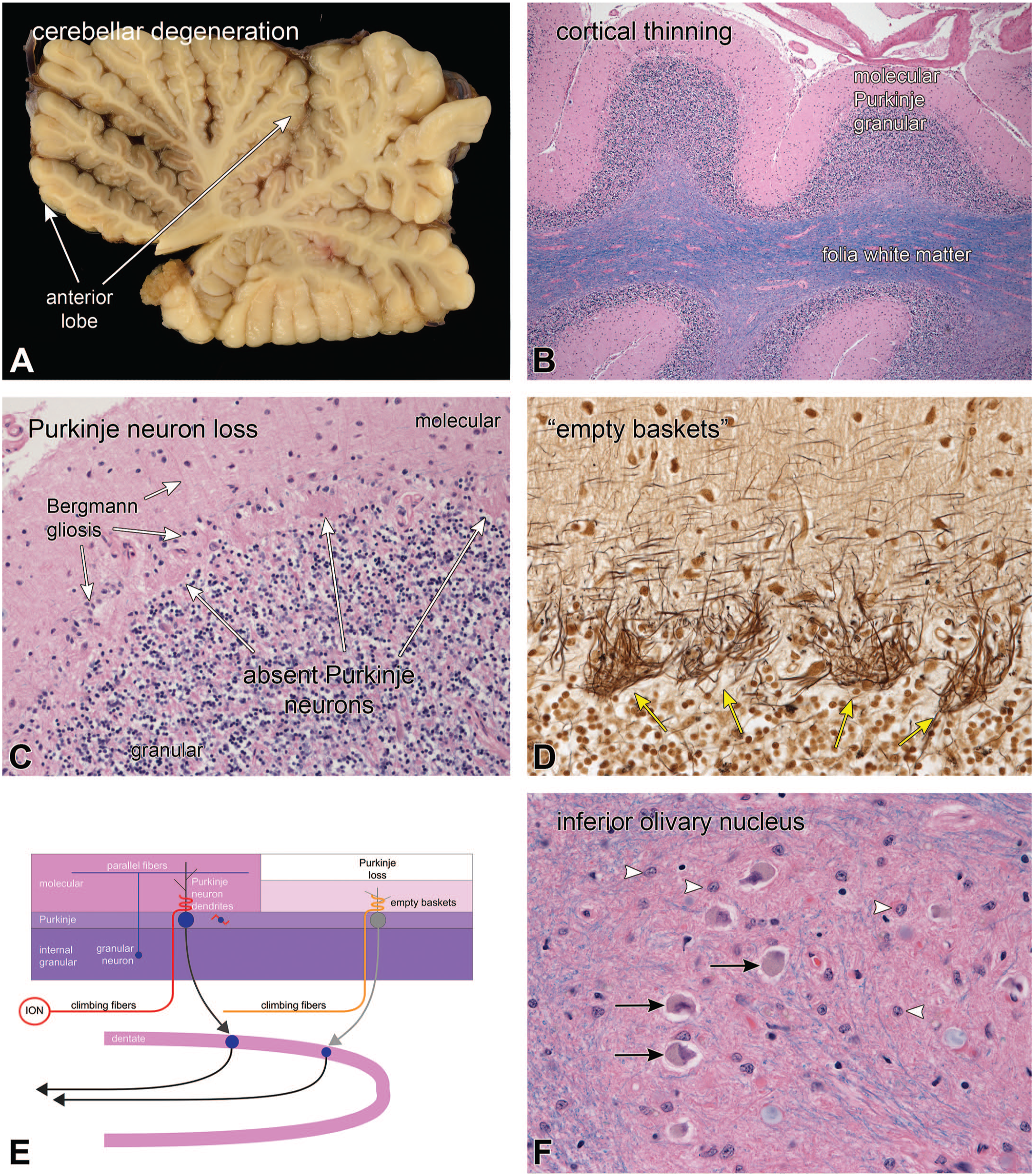
Tauopathy-related cerebellar degeneration; empty baskets. Panel A demonstrated the vermal foliar atrophy, which was greatest in the anterior lobe but was also present more posteriorly. The low magnification, H&E/LFB-stained view in B demonstrated the cortical thinning, which was most notable in the molecular layer. A higher magnification view of the same stain in C showed marked Purkinje neuron loss and a reactive Bergmann gliosis (shorter white arrows). Purkinje neurons had been replaced by tufts of pink staining material (long white arrows). The Bielschowsky silver-stained view of the Purkinje layer in D demonstrated the absence of Purkinje neurons but still-present climbing fiber baskets. These baskets retained silver and were black in this stain (yellow arrows). This pattern of retained climbing fibers axons and absent Purkinje neurons is termed “empty baskets.” The inferior olivary nucleus (ION) in F had some remaining neurons (black arrows) that were surrounded by a reactive gliosis (white arrowheads). The diagram in E illustrates the normal climbing fibers, which project to a few proximal Purkinje neuron dendrites. These axon termini become prominent in silver stains after the Purkinje neuron loss.
While many pathological processes affect Purkinje neurons, only rarely are granular neurons selectively affected by disease. Progressive multifocal leukoencephalopathy (PML) is a demyelinating disease of the immunosuppressed that is caused by an infection of cells with the JC polyoma virus. Figure 11 illustrates an HIV-positive patient who died from widespread PML. His cerebrospinal fluid viral PCR was positive for JC virus. In addition to other symptoms, he had developed dysarthria and balance difficulties. At autopsy, cerebellar tissues that were sampled as control tissues lacking plaques had high titers of JC virus. In Figure 11A, the low-magnification H&E/LFB section demonstrated patches and confluent loss of internal granular neurons (white arrows). At moderate magnification, immunoperoxidase staining for polyoma virus (Figure 11B) showed frequent granular neuron positivity. Further work and double labeling demonstrated JC-positive infection of granular neurons.4,21 Comparing uninvolved (Figure 11C) and involved (Figure 11D) folia, the H&E/LFB stains demonstrated patchy and confluent regions of granular neuron loss. Purkinje neurons were relatively preserved (black arrows). The retention of some myelinated axons in the granular layer (light blue staining lines) indicated that the granular neuron loss was not related to a demyelinated PML plaque. Like the examples above of Purkinje neuron loss, the loss of granular neurons led to shrinkage of the molecular layer (compare Figure 11D and C), with a concomitant Bergmann gliosis in the Purkinje layer and Bergmann radial glia in the molecular layer (Figure 11F). In the diagram in Figure 11E, granular neurons project their axons into the molecular layer and synapse on Purkinje neuron dendrites. Similar to the loss of Purkinje neurons, granular neuron axonal loss results in molecular layer involution.
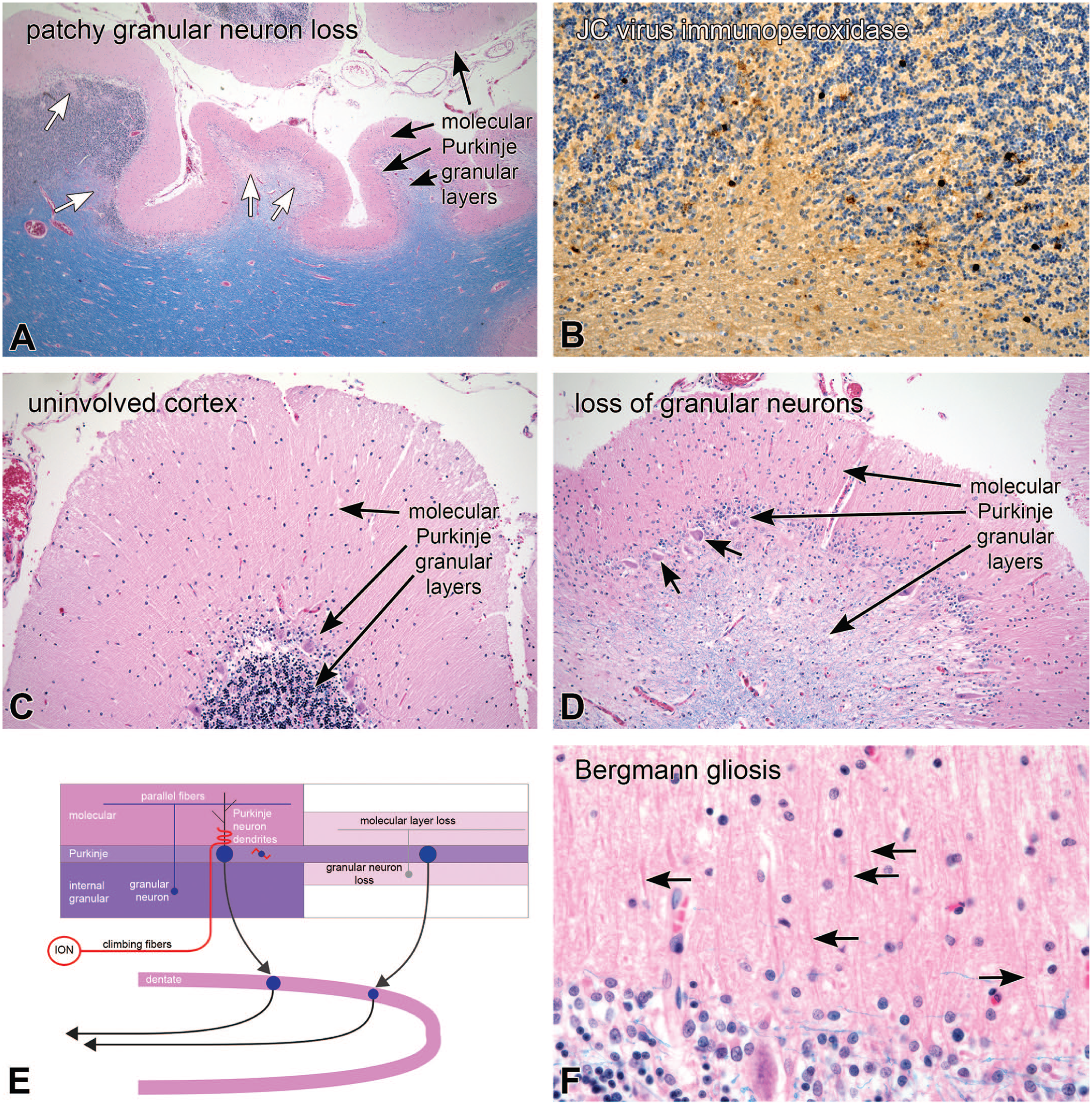
JC virus infection; selective granular neuron loss. At autopsy, the patient had patches and confluent loss of granular neurons (white arrows in A; low magnification H&E/LFB stain). Immunoperoxidase staining for JC virus was positive in granular neurons around areas of loss (brown nuclei in the internal granular layer in B). Panels C and D contrast the patient’s uninvolved (C) and affected (D) folia. The uninvolved cortex (C) had all three normal cerebellar cortical layers, while the end-stage affected areas (D) had a nearly total loss of internal granular neurons and a thinning of the molecular layer. Even in the most severely affected regions, Purkinje neurons were relatively preserved (short arrows in panel D). Panel F is a high-magnification view of the molecular and Purkinje layers that demonstrated the more prominent Bergmann nuclei in the Purkinje layer and their radiating radial glial fibers in the molecular layer (black arrows in F). The diagram in E emphasizes that selective loss of internal granular neurons can also lead to loss of the molecular layer, similar to Purkinje neuron loss. Abbreviation: ION = inferior olivary nucleus.
While pathologic changes can affect specific cell types, they can also affect regions of the cerebellum. An example of phenytoin (Dilantin) cerebellar toxicity is shown in Figure 12. This patient had developed seizures following a cortical infarct and had been treated with phenytoin. She had trisomy 21 and had developed dementia, which may have complicated identifying any toxicity. In contrast to some of the prior examples of greater anterior lobe disease (Figures 9 and 10), panel 12A demonstrated marked inferior cerebellar foliar atrophy (white arrows). This was corroborated in the H&E/LFB stain field view of the from the same section (B); the inferior lobe had undergone extensive involution. In addition, lateral superior cerebellum was partially involved, and only the most medial aspects were spared. The intermediate magnification H&E/LFB view, the extensively involved folium (Figure 12D), in contrast to an uninvolved (Figure 12C) folium, demonstrated a near-total loss of Purkinje neurons and an extensive loss of internal granular neurons, as well as shrinkage of the molecular layer. The higher magnification view in Figure 12F again showed a Bergmann gliosis with residual radial glial fibers, as well as an empty basket. This process also had increased cellularity at the base of the molecular layer that was due to an overall increased gliosis. Although not as apparent as the ara C toxicity example (Figure 9), the region around the dentate nucleus also showed some myelin pallor (Figure 12B, just to the right of the dentate). The summary diagram indicates the loss of granular and Purkinje neurons, the involution of the molecular layer from axon and dendrite loss, a Bergmann gliosis, and peri-dentate axon loss that relate to the underlying cerebellar anatomical connections. Cerebellar toxicity is a well know side effect of phenytoin use.3,5
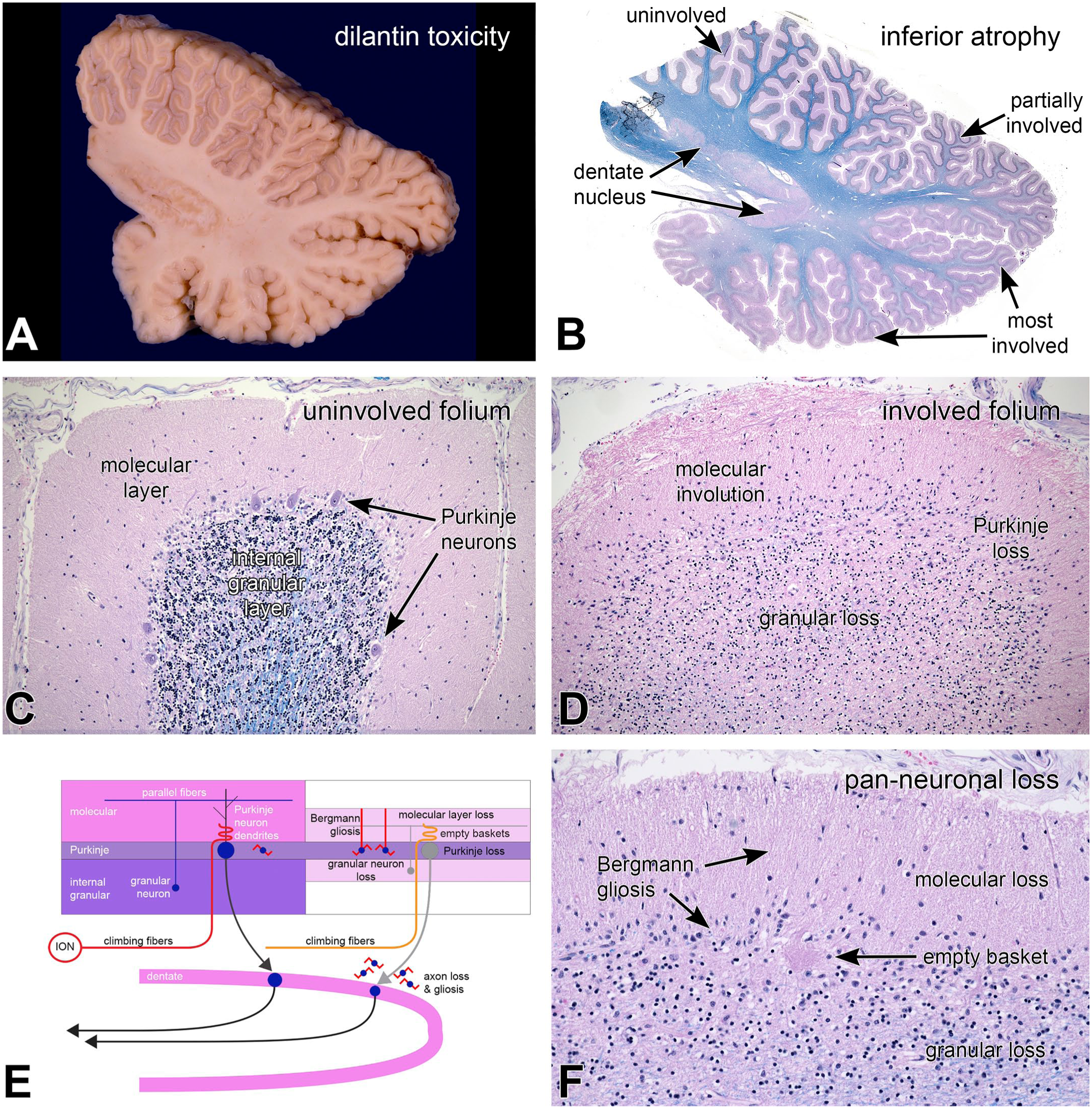
Phenytoin toxicity; regional neuronal loss. At autopsy, the patient’s inferior cerebellum was firm, and the folia were atrophic (white arrows in panel A). In the corresponding field view in panel B demonstrated the extensive atrophy in the inferior cerebellum. Dorsolateral folia were partially involved between the superior uninvolved and inferior extensively involved folia. The superior folium (C) had preserved cerebellar molecular, Purkinje, and granular layers (B). The most involved folium had an extensive loss of Purkinje and a moderate loss of internal granular neurons, as well as thinning of the molecular layer. The high-magnification view in panel F demonstrated Bergmann gliosis and radial Bergmann processes in the thinned molecular layer, an empty basket in the Purkinje layer (see Figure 10), and loss of granular neurons. The diagram in panel E emphasizes the anatomic distribution of the changes related to loss of both granular and Purkinje neurons. Panels B—D and F, H&E/LFB stains. H&E, hematoxylin-eosin stain; ION, inferior olivary nucleus; LFB, Luxol fast blue myelin stain.
Diseases that damage the dentate nucleus neuron will manifest both in the loss of cells as well as the loss of their projecting axons in the superior cerebellar peduncle(scp). These changes are most easily identified where the superior cerebellar peduncle decussates in the caudal midbrain (see Figure 1B).
Progressive supranuclear palsy (PSP) is a 4-repeat tauopathy that typically affects the dentate nucleus. The patients illustrated in Figure 13 presented with balance and gait issues and had difficulty coordinating his right limbs. A similar patient is presented in Figure 14. At autopsy, the patient had a 4R tauopathy that involved multiple structures, including his dentate nucleus. His sagittal MRI demonstrated atrophy and thinning of the rostral midbrain. This thinning has been called the “hummingbird sign” of PSP, with the midbrain as its head and the pons base as its body (Figure 14A). The midbrain had a much less prominent decussation of the superior cerebellar peduncle in this patient (Figure 14B) compared to the normal decussation in Figure 1B, which contributed to the midbrain atrophy. On a parasagittal section through the cerebellum, the dentate nucleus was indistinct, especially in its lateral aspects (white arrows; compare with the normal dentate nucleus Figure 7A). Similar to Figure 7, this patient had prominent autolysis of the internal granular layer (white arrowheads), which indicated that he likely had pre-mortem acidosis. The low magnification H&E/LFB view of the dentate demonstrated some loss of myelinated axons in the hilum, which is the output region of the dentate gyrus. Figure 13F at the same magnification demonstrates abundant abnormally phosphorylated tau staining of dentate neurons (AT8 monoclonal antibody), which is greater in the more lateral aspects, similar to the gross image in Figure 13C. The output from the dentate nucleus through the hilum travels in the large superior cerebellar peduncle through the dorsolateral pontine tegmentum (Figure 14D) and decussates in the caudal midbrain (Figure 14B). In this patient, the superior cerebellar peduncle was greatly reduced in the dorsolateral pons (Figure 14C) and its decussation in the caudal midbrain was nearly imperceptible (Figure 14A). Figure 14C also showed loss of myelinated axons in the central tegmental tract (ctt). This tract carries axons projecting from the red nucleus to the ipsilateral inferior olivary nucleus and is part of the Guillain-Mollaret triangle. PSP affects both the red nucleus and the substantia nigra and their loss contribute to the hummingbird sign (Figure 13A and B). Degeneration of the superior cerebellar peduncle following dentate nucleus degeneration is diagramed in Figure 13E.
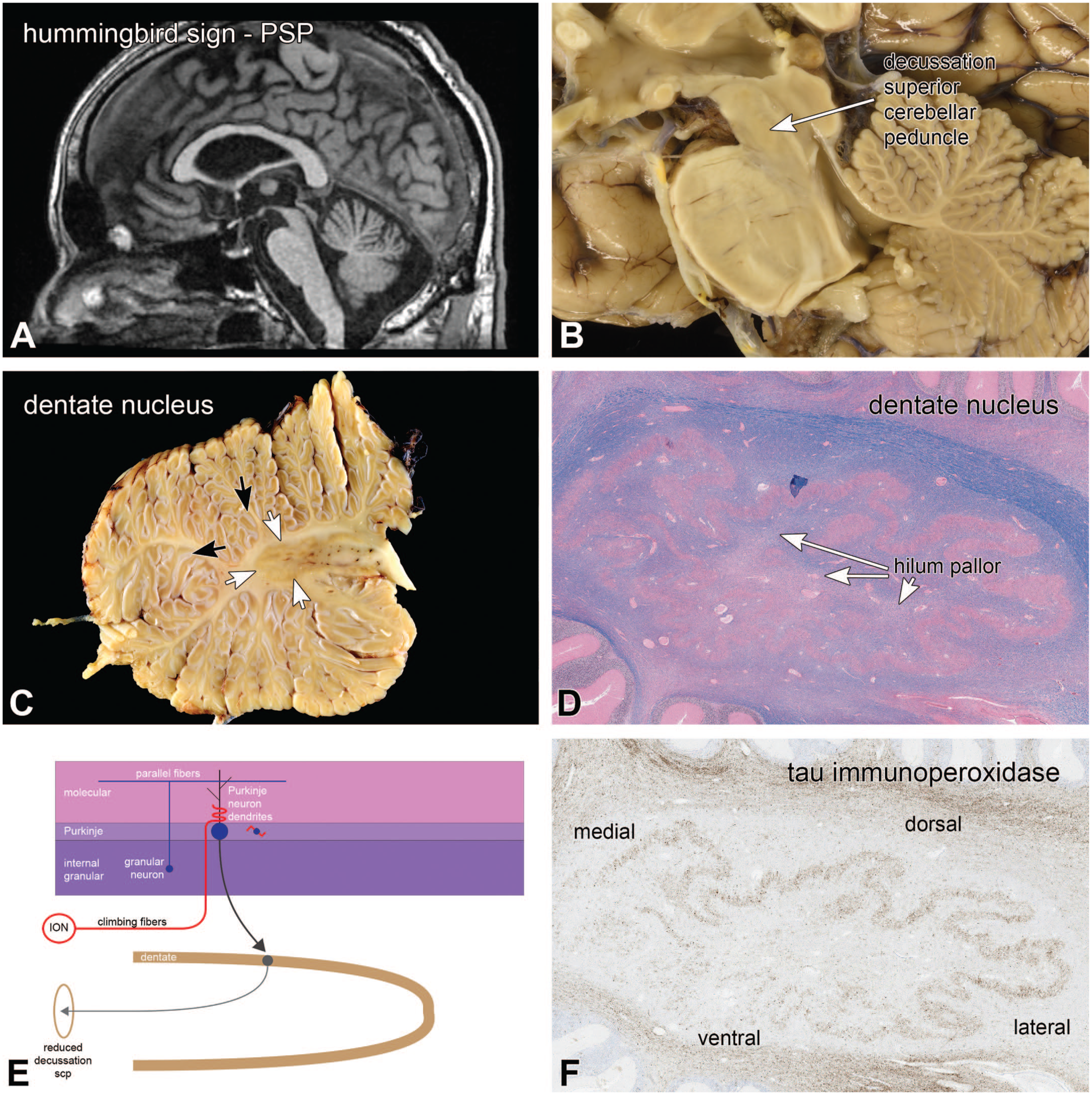
Progressive supranuclear palsy; dentate nucleus degeneration. The patient’s sagittal T2 MRI scan in panel A identified the “hummingbird sign,” which involved the midbrain and pons. The midbrain was flattened, compared to normal individuals. The corresponding gross image from the autopsy had a barely visible decussation of the superior cerebellar peduncle (compare with Figure 1B). The gross image of the dentate nucleus in panel C demonstrated a loss of a clearly delineated gray-white matter border in its lateral aspects (white arrows). Notice the prominent état glacis in the folia (white arrowheads) (also see Figure 7). Panels D and F are the corresponding but flipped images of the dentate nucleus in H&E/LFB stains (D) and tau immunoperoxidase (F). While degeneration is difficult to discern in D, the pathologic hyperphosphorylated tau staining clearly outlines this nucleus, and has greater staining laterally and dorsally. Myelinated axon loss was apparent in the hilum of the dentate nucleus (panel D). Panel E diagrams the changes, from a loss of dentate nucleus to a reduced decussation of the scp. H&E, hematoxylin-eosin stain; ION, inferior olivary nucleus; LFB, Luxol fast blue myelin stain; scp, superior cerebellar peduncles.
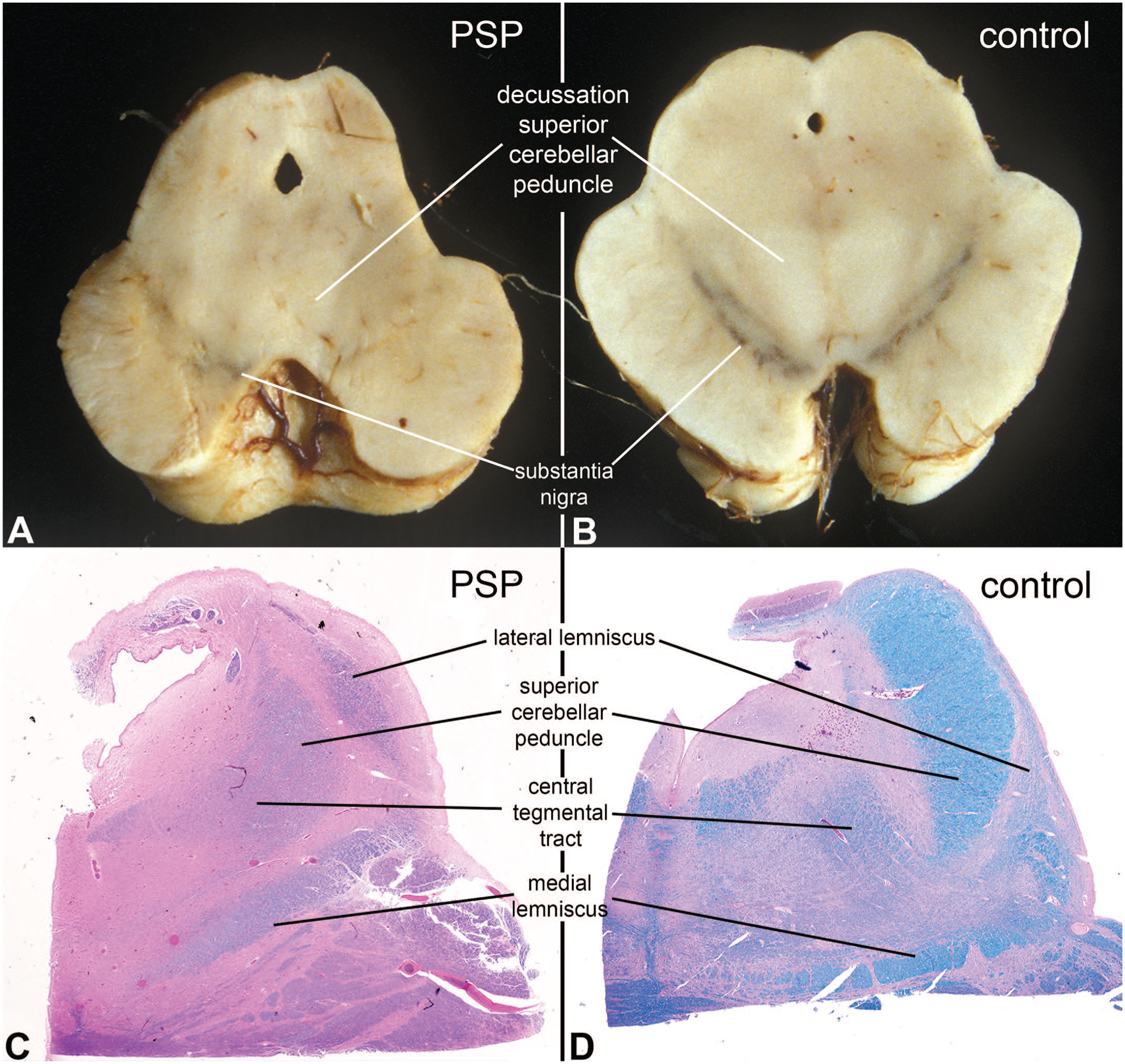
Progressive supranuclear palsy; degeneration of superior cerebellar peduncle. Panels A and B illustrate gross images at the level of the caudal midbrain from another patient who died with PSP (A) and an age-matched control (B). In the control, the substantia nigra had two dark bands and the decussation of the superior cerebellar peduncle was large and white (B), while in the affected patient, the decussation was barely discernible and the nigra was pale. Similarly, in the histologic H&E/LFB sections at the level of the rostral pons, the control superior cerebellar peduncle (panel D) is large and well myelinated, while it was shrunken and only weakly myelinated in the patient (panel C). The central tegmental tract descending from the red nucleus (part of Guillain-Mollaret triangle), was also distinct in the normal individual and pale in the patient with the 4R-tauopathy, since his red nucleus was also affected by the disease. The lateral lemniscus and medial lemniscus were not involved and serve as internal controls. H&E, hematoxylin-eosin stain; LFB, Luxol fast blue myelin stain.
Inhibitory, GABAergic Purkinje neurons are the only output from the cerebellar cortex to the cerebellar projection nuclei. Excitatory inputs to these nuclei come from cerebellar afferent collaterals, including mossy fiber inputs from the spinal cord, medulla, and pontine base, and the olivary climbing fibers (see Figure 6B). One circular loop from these collateral projections involves the dentate, red, and inferior olivary nuclei, which together with their connections form the Guillain-Mollaret triangle.
The patient with pathology illustrated in Figure 15 was an elderly man who had known vascular disease and died two years after developing “gait issues.” His brain had several old infarcts, including one that involved the medial aspect of his left dentate nucleus (white arrows in Figure 15A). H&E/LFB microscopy from the infarct in panel B demonstrated a non-cavitated area of old, incomplete ischemic injury, including extensive neuron loss (white arrows) and surrounding gliosis, which imparted a reddish blush around involved region. The dentate nucleus projects through the superior cerebellar peduncle (scp) to the contralateral red nucleus (RN) in the rostral midbrain, which then projects via the central tegmental tract (ctt) to the inferior olivary nucleus (ION) on the same side. In this patient, his right inferior olive would have projected collaterals to his left dentate nucleus through the inferior cerebellar peduncle (icp). His right inferior olivary nucleus in Figure 15C appeared grossly enlarged (white arrows), compared to his left olive (black arrows), which was corroborated by the H&E/LFB histology in Figure 15D (white arrows). The hilum of his right inferior olive had minimal LFB staining, which indicated a loss of myelinated axons projecting from this nucleus to the contralateral cerebellum, including the dentate nucleus. The inferior olive also had scattered neuron cell bodies that were filled with neurofilament protein (black arrows in Figure 15F).
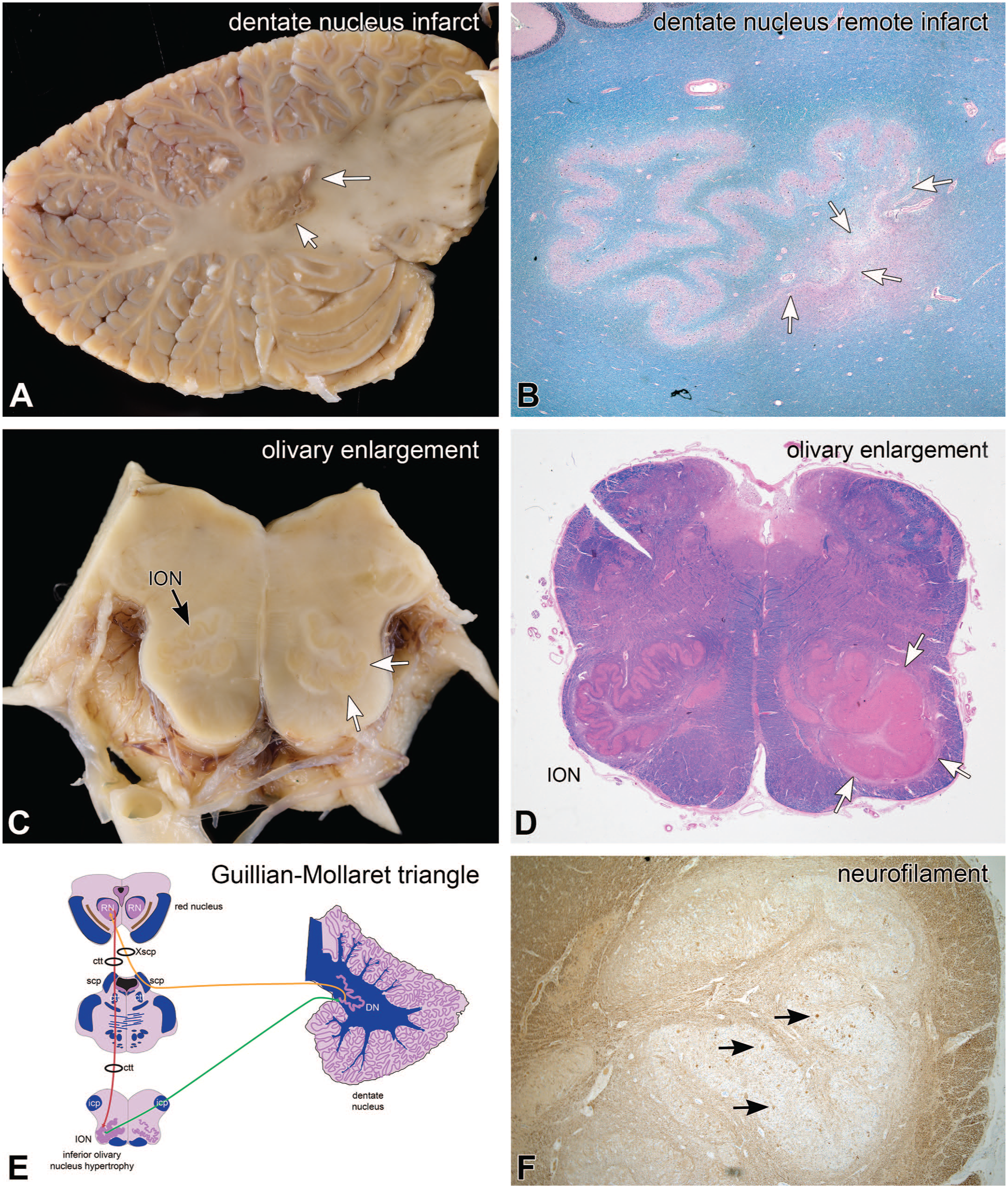
Remote infarct; pseudohypertrophy of inferior olivary nucleus. At autopsy, the patient had an old infarct in the left dentate nucleus (white arrows, panel A). In the H&E/LFB stain in panel B, this infarct caused loss of dentate neurons and neuropil (white arrows), as well as surrounding loss of myelinated axons and gliosis, including in the hilum and amiculum (pink staining around the infarct). The contralateral right inferior olivary nucleus (ION) (white arrows, panel C) was enlarged, compared to the left side (black arrow). This enlargement (white arrows) is move obvious in the H&E/LFB stain in panel D. Panel F is a neurofilament immunoperoxidase stain at a higher magnification view of the inferior olivary nucleus, which shows scattered positive staining neurons. H&E, hematoxylin-eosin stain; ION, inferior olivary nucleus; LFB, Luxol fast blue myelin stain.
The apparent enlargement of the ION after damage to elements in Guillain-Mollaret’s triangle is called “pseudohypertrophy of the inferior olivary nucleus.” This pattern of loss, with degeneration in axons that project to the damaged dentate nucleus, and the accumulation of neurofilaments in the corresponding ION, is suggestive of transsynaptic retrograde degeneration of the olivo-dentate projection. Olivary pseudohypertrophy has been described from lesions involving different aspects of the Guillain-Mollaret triangle (see Figure 15E), including in its major nuclei (DN, RN) as well as in its connecting white matter tracts (icp, scp, ctt).15,16
Discussion
This paper has reviewed the macroscopic and histologic features of cerebellar anatomy and the connections of some of its components and provided examples of how these connections help explain phenomena related to cerebellar pathology The summary of the connections in Figure 6 diagrams only a few of the many complex connections in networks that involve the cerebellum. However, these major components are useful to consider when examining cerebellar pathology, since they can indicate both the involved anatomic structures as well as the timing of the injuries. The anatomic changes discussed here typically require a prolonged period to develop and become obvious. Over a shorter time frame, the loss of components or connections will demonstrate more active features of degeneration, including macrophage infiltration and microgliosis of axonal or neuronal loss. Molecular layer involution requires weeks to develop, and olivary pseudohypertrophy evolves over months to years. As illustrated in Figures 4 and 5, the cerebellum can also be a useful chronometer for injuries that affect the developing fetus.
This review has not discussed several important topics about cerebellar anatomy. Since the emphasis of the review has been related to cerebellar disease pathology, important functional aspects of the cerebellum have largely been ignored. Related to the clinical functions, the modular organization of the cerebellum from medial to lateral has not been reviewed: the main cerebellar “lobes” (floccular-nodular, vermis, paravermal area, cerebellar hemispheres) have corresponding deep nuclei, and the divisions map onto several distinct cerebellar functions (eg, eye movements, axial musculature, precision of movements). Another more refined organizational principle that was not discussed is the longitudinally oriented functional cerebellar stripes that are delineated by developmentally expressed proteins and map in a precise stereotypic pattern with associated structures, such as the inferior olivary nucleus.6,7 In addition, this review has only discussed a few cell types. The cerebellum has several other types of neurons (eg, stellate, granular, Golgi) and glia. See Voogd and Ruigrok 20 and Kano and Watanabe 10 for additional information on these various cells.
Many references discuss cerebellar anatomy and its connections in much greater detail. These include the Cerebellum and Precerebellar Nuclei chapter by Voogd and Ruigrok from The Human Nervous System by Mai and Paxinos 20 and the cerebellum chapter from “The Human Nervous System: a Synopsis.” by Nieuwenhuys et al. 14 Development of the brain, including a chapter on the cerebellum, can be found in the excellent text by ten Donkelaar et al. 18
Footnotes
Acknowledgements
The author thanks Dr Kristopher Langdon for discussion and references about the Guillain-Mollaret triangle.
Author Contributions
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
