Abstract
The virtual control group (VCG) concept was originally developed in the IMI2 project eTRANSAFE, using data of control animals which pharmaceutical companies have accrued over decades from animal toxicity studies. This control data could be repurposed to create virtual control animals to reduce or replace concurrent controls in animal studies. Initial work demonstrated the general feasibility of the VCG concept, but implementation requires significant further collaborative efforts. The new Innovative Health Initiative (IHI) project VICT3R aims to address these challenges and to obtain regulatory acceptance for the VCG concept. To achieve these goals, VICT3R will build a database comprising high-quality, standardized, and duly annotated control animal data from past and forthcoming toxicity studies. The VICT3R project will create workflows and computational tools to generate adequate VCGs based on statistical and artificial intelligence (AI) approaches. The validity, reproducibility, and robustness of the resulting VCGs will be assessed by comparing the performance of their use with that of real control groups.
Introduction
The concept of virtual control groups (VCGs) is based on the availability of large data sets of high-quality control data recorded and stored in standardized terminologies originating from highly regulated animal safety studies. The collected historical control data are proposed to be used to construct VCGs which could partly or entirely replace the concurrent controls in future animal toxicity studies. 1 The impact of the 3R principles (Replacement, Reduction, and Refinement) on animal studies and the associated cost savings particularly for the larger species (dog, nonhuman primates [NHPs]) has generated gained broad interest in the VCG concept. However, regulatory acceptance and implementation of the VCG concept require a thorough assessment of the procedures to avoid any impairment of study outcomes of new safety studies, which might result in a safety risk for patients, workers, or consumers. 2
To address the aforementioned requirements, we developed the VICT3R project (“Developing and Implementing Virtual Control Groups to Reduce Animal Use in Toxicology Research”) submitted to the Innovative Health Initiative (IHI) in January 2024 under call 5, topic 1 (“Accelerating the implementation of New Approach Methodologies and other innovative non-animal approaches for the development, testing and production of health technologies”). IHI is a public-private partnership (PPP) between the European Union and the European life science industries. The partners of IHI are the European Union, represented by the European Commission, and the European life science industry, represented by the industry associations COCIR, EFPIA (including Vaccines Europe), EuropaBio, and MedTech Europe. The VICT3R proposal was positively evaluated, and the project started in September 2024 with a duration of 42 months. 3
The VICT3R consortium (https://www.vict3r.eu) consists of 6 academic institutions, 7 small- and medium-sized enterprises, and 20 industry partners from the pharmaceutical and the agrochemical sector, which collectively provide the necessary expertise in data provision and handling, software development, statistics, bioinformatics, artificial intelligence (AI), and regulatory science. The primary aim of VICT3R is to significantly reduce the number of animals required for experimental studies in nonclinical evaluations by implementing VCGs, which aligns with the European Union’s 3Rs requirements. 4
The VICT3R project heavily builds on preliminary work conducted under the Innovative Medicines Initiative (IMI) eTRANSAFE project. The initial work performed under eTRANSAFE demonstrated the feasibility of the VCG concept 5 but also identified significant challenges which need to be overcome before its regulatory implementation can be envisaged. While eTRANSAFE ended in February 2023, some eTRANSAFE partners continued to further develop the VCG concept. This involved creating a prototype of a VCG database and exploring approaches for qualification of the VCG procedure. These activities culminated in a workshop with industry and regulatory participation under the auspices of the Center for Alternatives to Animal Testing (CAAT, Johns Hopkins Bloomberg School of Public Health, Baltimore, MD, US), which took place in May 2023 in Washington. The results of this workshop were summarized by Golden et al. 6 The momentum of the workshop and the increasing interest in the VCG topic led to the formation of the European consortium which submitted the VICT3R proposal.
The development of a database designed to host the high-quality historical control data from different animal test facilities using controlled terminologies (e.g., the Standard for Exchange of Nonclinical Data [SEND]) will be at the core of the VICT3R project. The database will also include links to digital histopathological images from control animals. The database’s functionality will be assured by combining it with tools for data retrieval and mining, data visualization, and statistical analysis. The control data workflow of the VICT3R project is depicted in Figure 1.
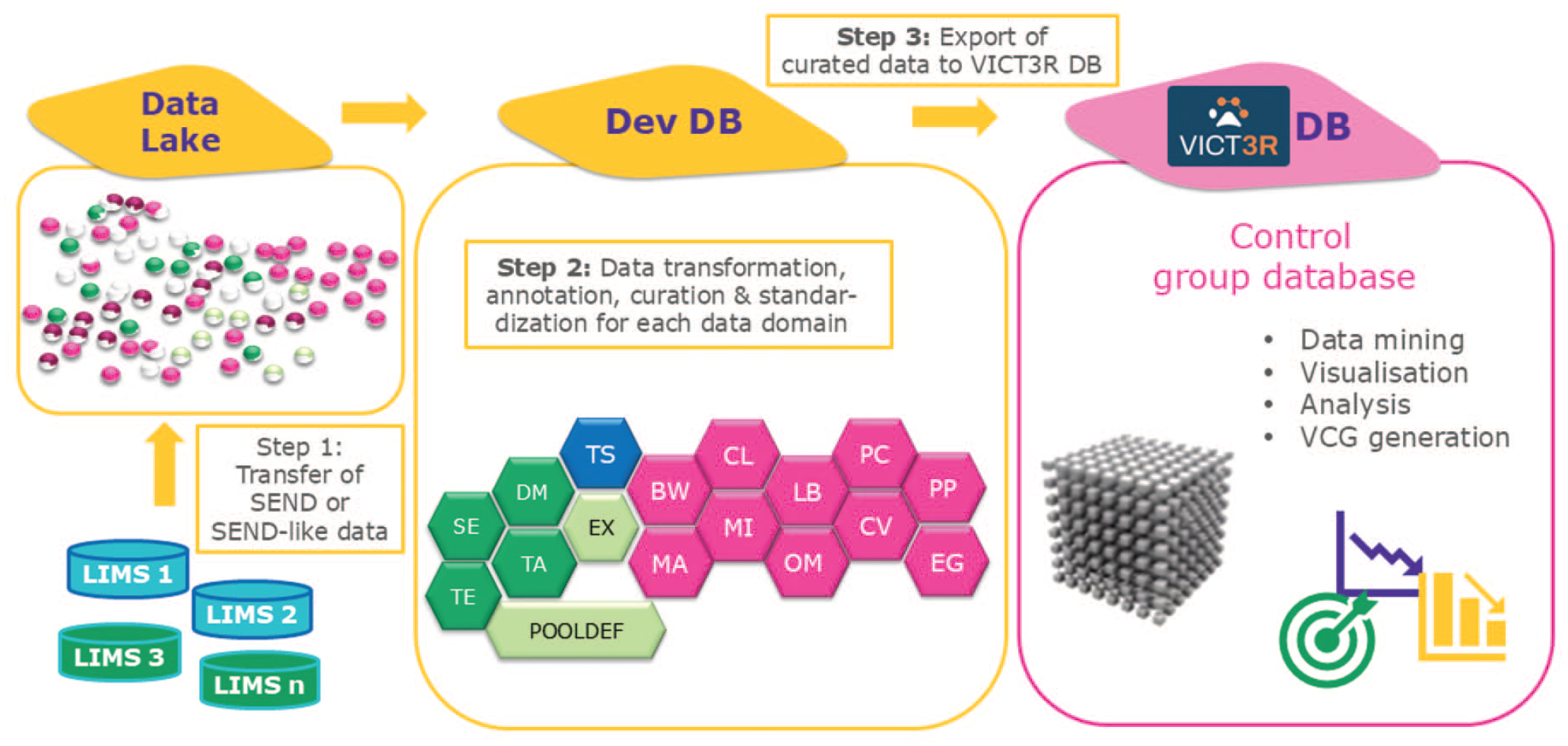
Data workflow in the VICT3R project from the collection of control study data (individual animal data) to the final database for VCG generation. LIMS, laboratory integrated management system; the abbreviations in step 2 stand for the different SEND domains.
In parallel, operational procedures for data curation and quality control will be established. Statistical procedures (trend analysis and distribution characteristics) will be developed to characterize the collected data and detect hidden confounders (e.g. changes in biochemical analysis of clinical pathology parameters). This will allow establishing procedures for the generation and optimal matching of the VCG animals with the animals in the treatment groups. Machine learning and AI workflows will be applied to bridge data gaps and to generate synthetic VCGs.
The performance of the VCG procedure will be assessed by comparing the outcome of existing studies before and after replacing the concurrent controls with VCGs by applying procedures as recently described by Gurjanov et al. 7 Potential challenges will be addressed through rigorous data standardization, stakeholder engagement, and transparent communication with regulatory bodies. The results of this performance testing will be presented to regulatory authorities across the different industry sectors (EMA & FDA, EFSA, and ECHA) spanning regions including Europe, North America, and beyond, to obtain feedback and eventually global regulatory acceptance of the VCG concept. The initial focus of VICT3R will be on systemic toxicity studies, reflecting the abundance of available data for this study type, but it is also intended to explore the extension of the concept to other types of animal studies, such as embryo-fetal and developmental studies, carcinogenicity studies or certain types of in vivo pharmacology studies. Beyond toxicology, the VCG concept could be extended to other areas of biomedical research, such as pharmacokinetics and disease modeling, further reducing animal use across various scientific disciplines.
Since most of the in vivo studies for safety assessment are performed under Good Laboratory Practice (GLP), the database and the developed IT tools are subject to Computer System Validation (CSV) practices. To ensure that the database and other developed assets remain applicable and maintained after the end of the VICT3R project, a sustainability plan will be designed from the start. Figure 2 provides an overview of the activities described.
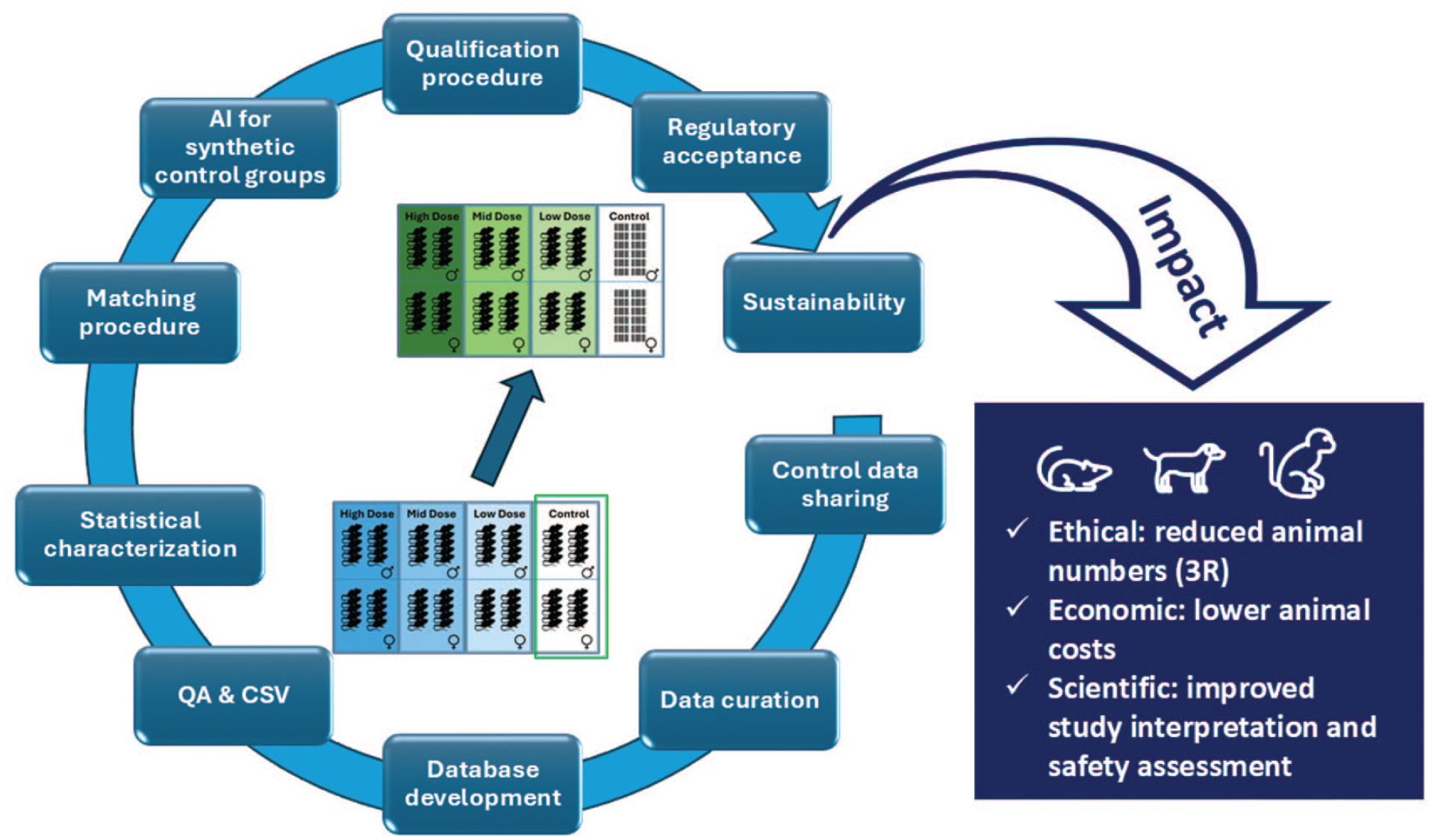
The iterative cycle of activities in the VICT3R project to achieve regulatory acceptance for replacing concurrent controls with VCGs resulting in positive impact on 3R, economics, and science. CSV indicates computer system validation; QA, quality assurance.
The VICT3R project is anticipated to have significant impact benefits in various societal fields (see Figure 2). First, by reducing animal usage, VICT3R aligns with ethical standards and public expectations regarding animal welfare. Since control groups usually account for around 25% of the animals in a toxicity study used in drug and chemical safety assessment, a significant reduction of animals can be reached through the application of VCGs. Second, enhanced methodologies for safety assessments will improve the quality, reliability, and predictability of study outcomes, ultimately benefiting patient or consumer safety. Third, the reduction in animal testing is expected to yield substantial cost savings for pharmaceutical and chemical industries, particularly by reducing the number of required NHPs for studies. 2 Finally, the project’s goal is to influence future Organisation for Economic Cooperation and Development (OECD) and International Council for Harmonisation (ICH) guidelines, promoting a shift toward more humane and efficient testing practices.
Conclusion
VICT3R represents a transformative step toward modernizing toxicological research by integrating innovative methodologies into regulatory frameworks. By harnessing historical data and advanced data science, the project aims to significantly reduce animal use while improving the quality and efficiency of safety assessments in the pharmaceutical and chemical industries. The anticipated outcomes of this project have the potential to reshape the landscape of nonclinical research while aligning it with progressing ethical standards and scientific advancements. We invite researchers, regulatory bodies, and industry stakeholders to join us in this transformative effort, as the successful implementation of VCGs will not only advance scientific research but also set new ethical standards in toxicology.
Footnotes
Author Contributions
All authors equally contributed equally to the concept of this work, the drafting and revision of the article and the approval of the final version.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TS-H is an employee of Bayer AG, Pharmaceuticals and holds stocks from Bayer AG. FB is an employee of Merck Healthcare KGaA and holds stocks from Merck KGaA.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project receives funding from the Innovative Health Initiative Joint Undertaking (IHI JU) under grant agreement no. 101172693. The JU receives support from the European Union’s Horizon Europe research and innovation program and COCIR, EFPIA, Europa Bío, MedTech Europe, and Vaccines Europe and Instem Scientific Limited. This project is funded by the European Union, the private members, and those contributing partners of the IHI JU. Views and opinions expressed are however those of the author(s) only and do not necessarily reflect those of the aforementioned parties. Neither of the aforementioned parties can be held responsible for them.
