Abstract
Ginsenoside Rh2 (GRh2) exhibits significant potential as an anticancer agent; however, progress in developing chemotherapeutic drugs is impeded by their toxicity toward off-target tissues. Specifically, anemia caused by chemotherapy is a debilitating side effect and can be caused by red blood cell (RBC) hemolysis and eryptosis. Cells were exposed to GRh2 in the antitumor range and hemolytic and eryptotic markers were examined under different experimental conditions using photometric and cytofluorimetric methods. GRh2 caused Ca2+-independent, concentration-responsive hemolysis in addition to disrupted ion trafficking with K+ and Cl– leakage. Significant increases in cells positive for annexin-V-fluorescein isothiocyanate, Fluo4, and 2,7-dichlorofluorescein were noted upon GRh2 treatment coupled with a decrease in forward scatter and acetylcholinesterase activity. Importantly, the cytotoxic effects of GRh2 were mitigated by ascorbic acid and by blocking casein kinase 1α (CK1α) and mixed lineage kinase domain-like (MLKL) signaling. In contrast, Ca2+ omission, inhibition of KCl efflux, and isosmotic sucrose aggravated GRh2-induced RBC death. In whole blood, GRh2 selectively targeted reticulocytes and lymphocytes. Altogether, this study identified novel mechanisms underlying GRh2-induced RBC death involving Ca2+ buildup, loss of membrane phospholipid asymmetry and cellular volume, anticholinesterase activity, and oxidative stress. These findings shed light on the hematologic toxicity of GRh2 which is crucial for optimizing its utilization in cancer treatment.
Introduction
Ginseng (Panax ginseng) is a significant medicinal plant in the Araliaceae family.15,62 Ginsenosides, the plant’s primary active constituents, are steroid-like saponins that possess a wide range of pharmacological effects related to cardiovascular, immunological, metabolic, and cognitive functions.9,20,34 Approximately 100 different ginsenosides have been isolated from roots, stems, and leaves of Panax species, of which, ten have been characterized and are known for their pharmacological properties including Rb1, Rg1, Rd, Re, CK, and Rh2.15,49,60 Globally, ginseng-based products, such as functional foods, dietary supplements, and traditional herbal remedies, are widely available and consumed.8,39 It has also been shown in numerous clinical trials that ginsenosides and root extracts improve cognitive function and immune response and reduce diabetes and cardiovascular disease risk.47,53,55
Ginsenoside Rh2 (GRh2), a protopanaxadiol glycoside, is a bioactive ginsenoside in P. ginseng as it exhibits anti-proliferative activity in multiple tumor cells by inhibiting invasion and metastasis and inducing cell cycle arrest. 34 GRh2 modulates several cell death signaling pathways and stimulates apoptosis through caspase-dependent and independent mechanisms involving TRAIL and NFκB.30,54 Previous efforts have also demonstrated that reactive oxygen species (ROS) produced by GRh2 can also trigger mitochondrial dysfunction and set off endoplasmic reticulum stress, which in turn activates the caspase cascade to trigger apoptosis. 57
Like nucleated cell apoptosis, erythrocytes may undergo a form of cell death called eryptosis. Senescent and damaged red blood cells (RBCs) undergo eryptosis followed by quick clearance from the circulation to prevent the prolonged presence of these dysfunctional cells in the systemic circulation and avoid intravascular hemolysis. This elimination must be simultaneously counterbalanced by efficient erythropoiesis in the bone marrow to maintain physiological RBC count in the circulation. 12 However, when the rate of eryptosis exceeds that of erythropoiesis and reticulocyte maturation, as is the case in chemotherapy-related anemia, a fall in RBC count ensues. 22 Eryptotic cells are characterized by loss of membrane asymmetry, leading to the externalization of phosphatidylserine (PS) on the cell surface. In addition, there is a rise in intracellular calcium levels, excessive production of ceramide, ROS buildup, and the stimulation of signaling enzymes including p38 MAPK, protein kinase C (PKC), casein kinase 1α (CK1α), mixed lineage kinase domain-like (MLKL) pseudokinase, and caspases. 6
Although paclitaxel and cisplatin are known to cause anemia in patients in part through the induction of eryptosis,25,36 no reports have thus far examined the potential side effects or hematologic toxicity of GRh2 administration. In mouse xenograft models of lung, prostate, and colon cancer, GRh2 at 1-20 mg/kg at varying intervals was effective in reducing tumor growth but hematotoxicity was not assessed.13,59,63 Therefore, the aim of this study is to examine the hemolytic and eryptotic potential of GRh2 as a safety assessment which informs further therapeutic applications of this gensinoside.
Materials and Methods
Chemicals and Reagents
All chemicals and reagents, unless otherwise specified, were provided by Solarbio Life Science (Beijing, China). A 10 mM stock solution of GRh2 (CAS #78214-33-2) was prepared in DMSO (6.23 mg/ml). The resulting solution was thereafter divided into aliquots and stored at −80°C. Ringer buffers were prepared essentially as previously detailed. 1
Experimental Design
Ethical approval was received from the Ethics Committee of King Saud University Medical City (E-23-7764). Informed consent was obtained from every participant in accordance with the principles outlined in the Declaration of Helsinki. Blood samples were collected in lithium heparin or ethylenediaminetetraacetic acid (EDTA) vacutainer tubes from 27 nonsmoking, healthy participants, 19 males and 8 females aged 24-38 years, with normal complete blood count (CBC) results and body mass index values. RBCs were separated and washed in PBS and resuspended in either PBS or Ca2+-free Ringer solution (1:3 v/v) at 4°C for a maximum of 48 hr. Cells were suspended in Ringer solutions at a hematocrit of 5% (950 μl Ringer buffer + 50 μl RBCs) and were subjected to antitumor concentrations of GRh2 (10-100 µM) for 24 hr at 37°C. This cell density is widely accepted and ensures consistency allowing for accurate comparisons of results among studies. It also rejuvenates RBCs by maintaining energy metabolism and deformability during incubation.3,11,27,35 For experiments on purified RBCs, a negative control was prepared by exposing cells to 1% DMSO, whereas cells suspended in distilled water were used as a positive control. In addition, EDTA blood was diluted 1:2 in PBS and incubated with 80 µM of GRh2 for 24 hr at 37°C. 4
Hemolysis and Hemolytic Markers
Supernatants of control and experimental cells were assayed for hemoglobin (Hb) at 405 nm using an LMPR-A14 microplate reader (Labtron Equipment Ltd., Surrey, UK). LDH, AST, and CK activities were measured using a BS-240Pro clinical chemistry analyzer (Mindray Medical International Limited, Shenzhen, China). 44
Measurement of Electrolytes
Supernatant K+, Cl–, Na+, and pH were detected using an EXIAS e|1 electrolyte analyzer (EXIAS Medical GmbH, Graz, Austria). 2
Acetylcholine Esterase (AChE) Activity
The photometric measurement of AChE activity was conducted in pooled hemolysates using Solarbio’s AChE Activity Assay Kit which is based on Ellman’s method. 41 Galantamine was used as a positive control for AChE inhibition.
Osmotic Fragility
Cells treated with 1 or 5 μM of GRh2 were incubated for 1 hr at 37oC in saline solutions of varying tonicity ranging from 0.9% to 0.1%. 7
Eryptotic Markers
Eryptosis was evaluated by the Northern Lights™ flow cytometer (Cytek Biosciences, Fremont, CA, USA). PS-exposing cells were labeled with 1% annexin-V-FITC for 10 min at RT, away from light, intracellular Ca2+ was detected by 5 μM of Fluo4/AM for 30 min at 37°C, cell size was inferred from forward scatter (FSC), and ROS were determined by 10 μM of 2′,7′-dichlorodihydrofuorescein diacetate (H2DCFDA) for 30 min at 37°C in the dark. All three dyes were excited by the blue laser at 488 nm, and the emitted green light was detected at 520 nm. In all cases, a total of 10,000 events were recorded and analyzed. 17 Flow cytometric fluorescence signals, measured in arbitrary units (a.u.) or percentages, were analyzed using FlowJo™ v10.7.2 software (Becton, Dickinson and Company, Ashland, OR, USA).
Erythrocyte Sedimentation Rate (ESR)
Westergren tubes were used to observe the sedimentation rate (mm/hr) of RBCs in whole blood as described elsewhere. 65
Ultrastructural Analysis
Cells were prepared for examination by JSM-7610F ultra-high-resolution Schottky field emission scanning electron microscope (JEOL Co., Ltd., Akishima, Tokyo, Japan) as previously detailed. 42
Systemic Toxic Effects
Control and treated (80 µM) whole blood was tested for a CBC using a BC-6200 hematology analyzer (Mindray Medical International Limited, Shenzhen, China). 5
Statistical Analysis
Statistical tests were performed using the Prism v9.5.1 software (GraphPad Software, Inc., San Diego, CA, USA). Results are shown as means ± standard deviation of three independent experiments each run in triplicates on cells from three donors. Two groups were compared using Student’s t-test whereas three or more groups were analyzed by one-way ANOVA followed by Dunnett’s test.
Results
There was a concentration-dependent hemolysis with statistical significance in cells exposed to 10-100 μM of GRh2 in PBS. In comparison to control values of 2.36% ± 0.70%, the hemolytic rate of GRh2 significantly increased to 12.36% ± 2.51% (p < 0.0001) at 10 µM, 17.19% ± 1.40% (p < .0001) at 20 µM, 22.79% ± 1.88% (p < .0001) at 40 µM, 28.42% ± 4.09% (p < .0001) at 80 µM, and 28.59% ± 4.57% (p < .0001) at 100 µM, as depicted in Figure 1B. Likewise, GRh2 showed increased hemolytic activity at all concentrations when the suspension medium was changed to standard Ringer solution (Figure 1C) from 0.59% ± 0.34% in the control group to 4.57% ± 1.13% (p = .0009), 7.91% ± 1.26% (p < .0001), 16.88% ± 2.667% (p < .0001), 32.54% ± 2.49% (p < .0001), and 36.96% ± 3.15% (p < .0001) after treatment with 10, 20, 40, 80, and 100 µM of GRh2, respectively.
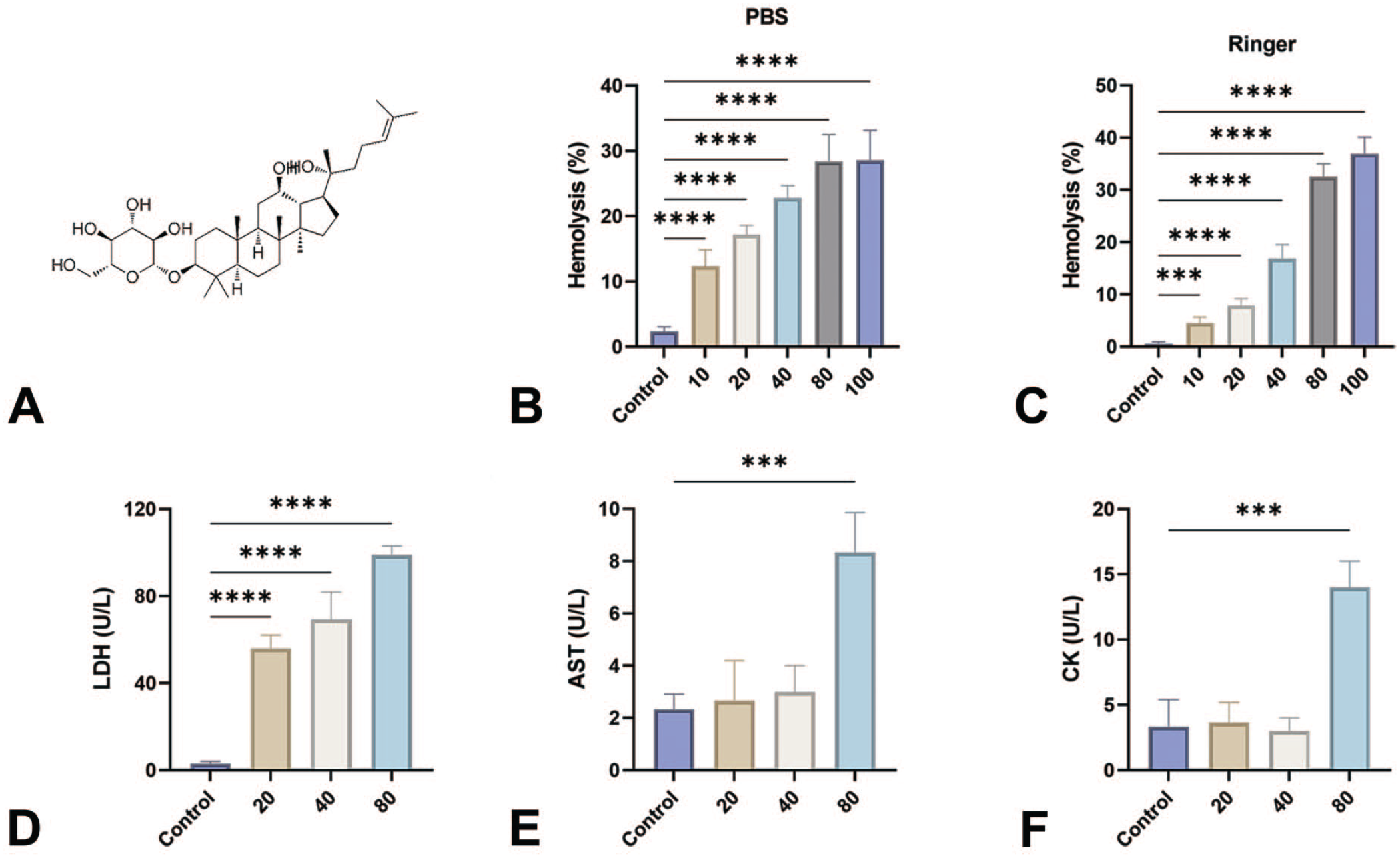
The leakage of LDH (Figure 1D) significantly increased from 3.00 ± 1.00 to 56.00 ± 6.08 U/L (20 µM, p < .0001), 69.33 ± 12.50 U/L (40 µM, p < .0001), and 99.00 ± 4.00 U/L (80 µM, p < .0001). Significant increases in AST (2.33 ± 0.58 U/L to 8.33 ± 1.53 U/L, p = .0008) and CK (3.33 ± 2.08 U/L to 14.00 ± 2.00 U/L, p = .0002) activities, however, were only observed at 80 μM as illustrated in Figure 1E and 1F, respectively.
As illustrated in Figure 2A, hemolysis caused by GRh2 resulted in a significant K+ efflux at 40 μM (6.04 ± 0.17 mmol/L, p < .0001), 80 μM (6.74 ± 0.26 mmol/L, p < .0001), and 100 μM (7.15 ± 0.27 mmol/L, p < .0001) compared with untreated cells (4.95 ± 0.18 mmol/L). This was similarly accompanied by Na+ loss at 80 μM (137.7 ± 4.35 to 147.1 ± 0.59 U/L, p < .01) and at 100 μM (151.7 ± 6.183 U/L, p < .0001) as seen in Figure 2B. Cl– efflux (Figure 2C) followed a similar pattern as significant increases were noted at 80 μM (113.7 ± 5.22 to 122.4 ± 3.28 mmol/L, p = .0054) and at 100 μM (127.9 ± 5.71 mmol/L, p < .0001). GRh2 did not cause a significant change in extracellular acidity and did not provide any protection against the hypotonic lysis of RBCs, as seen in Figure 2D and 2E, respectively.
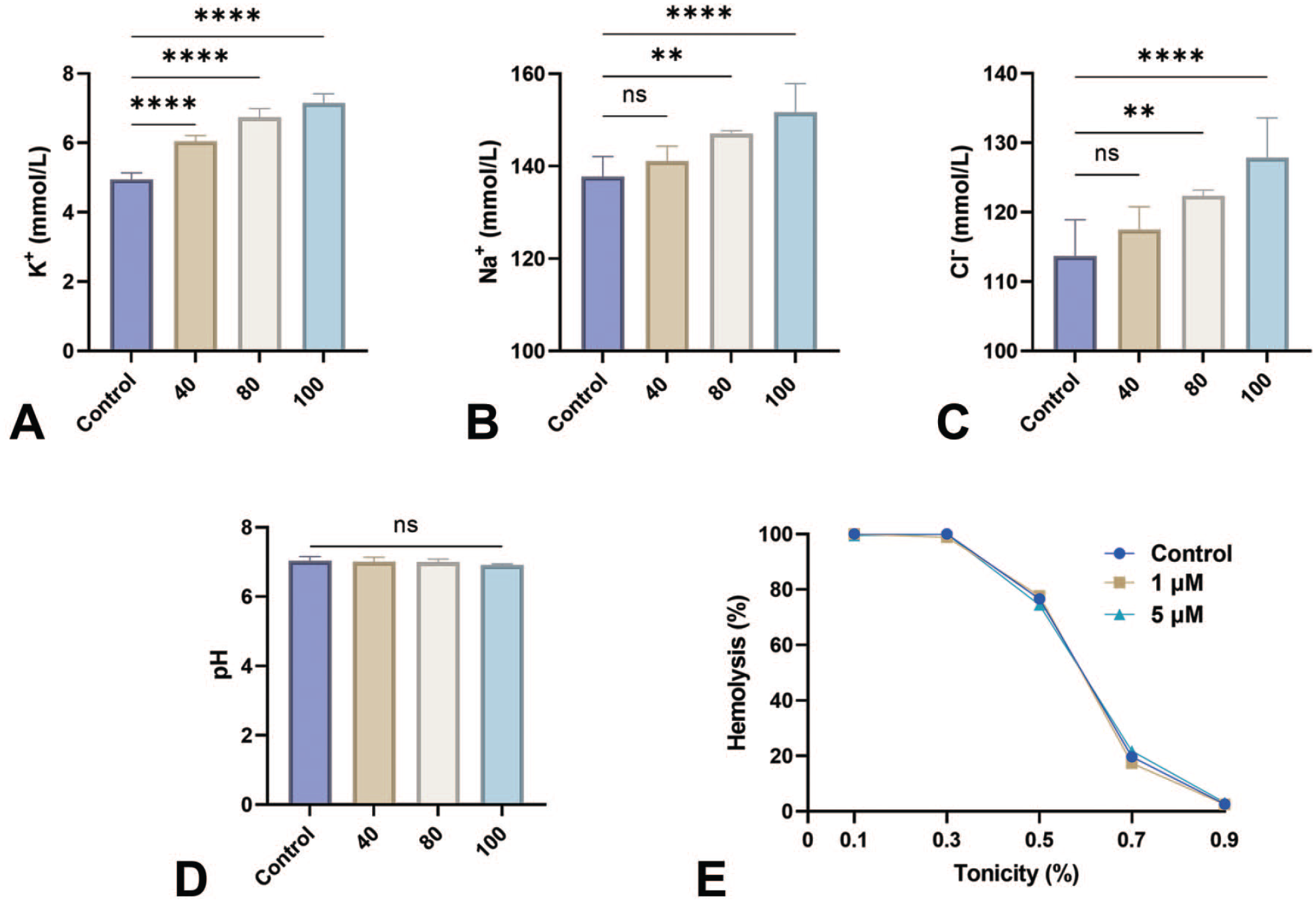
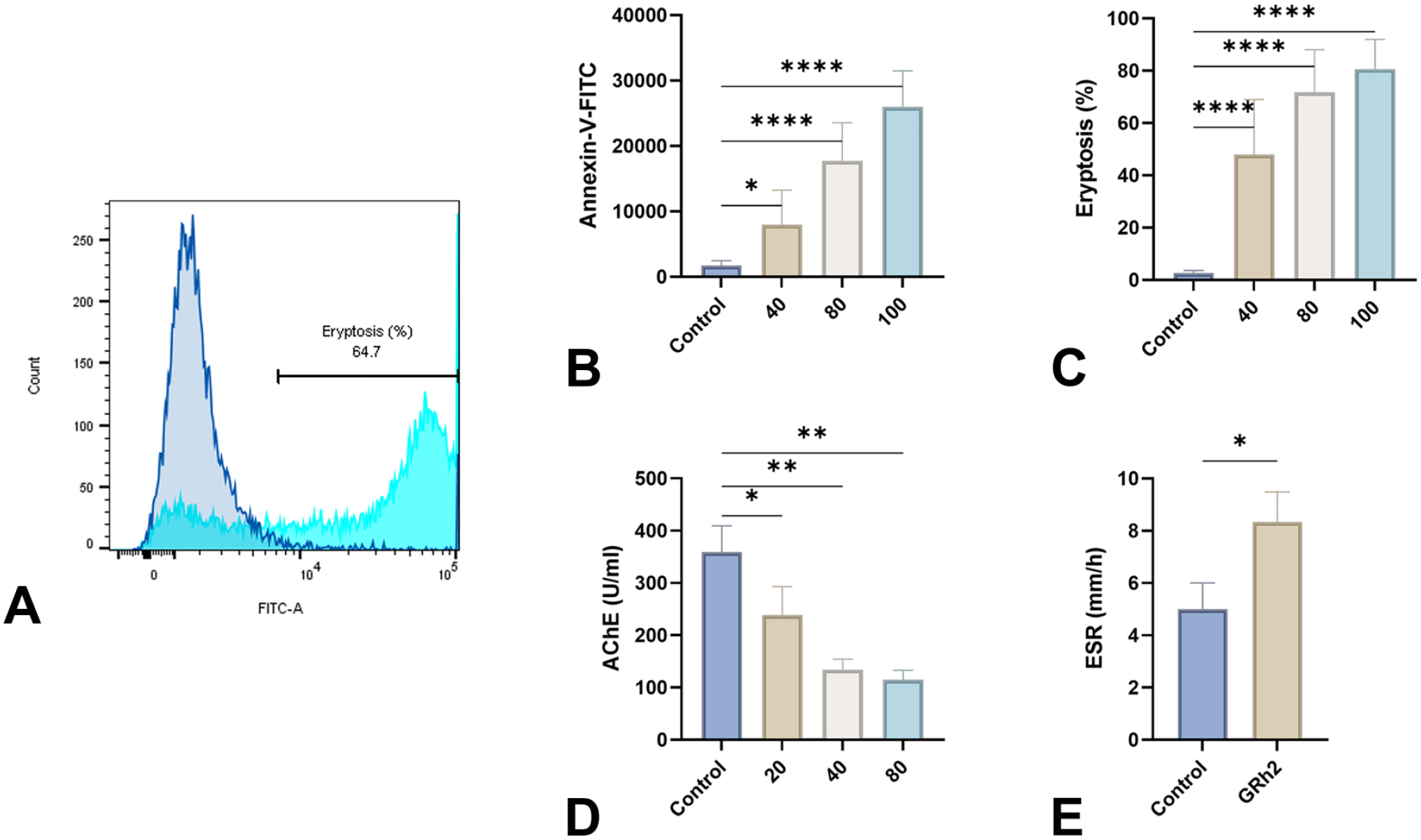
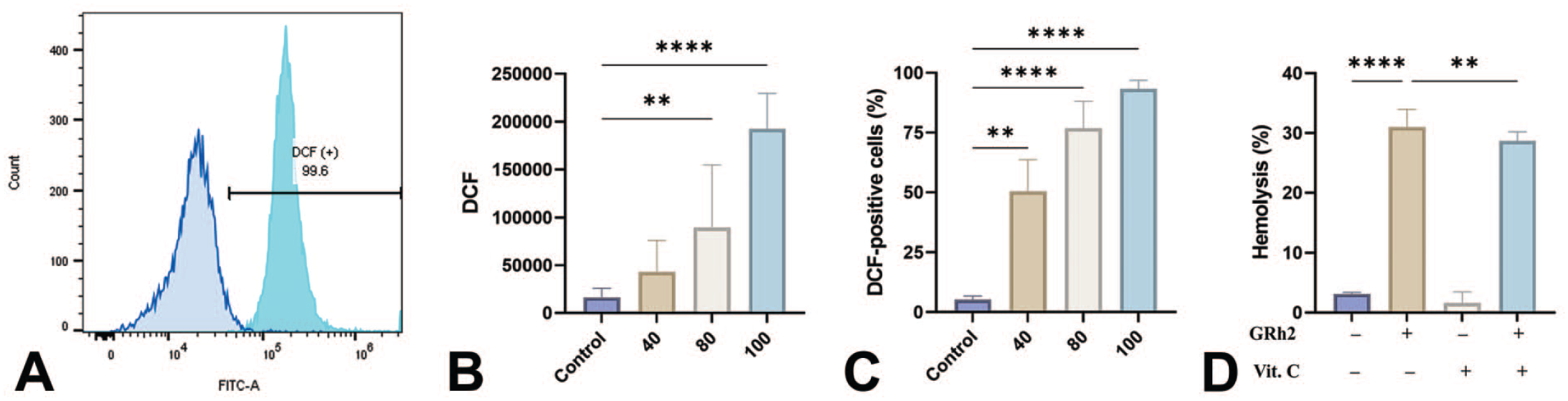
Figure 3A demonstrates that GRh2 enhances the fluorescence of annexin-V-FITC, which indicates the exposure of PS. The geomean of annexin-V-FITC fluorescence showed a significant rise from 1722 ± 725.8 a.u. in control cells to 7950 ± 5311 a.u. (40 μM, p < .05), 17,747 ± 5807 a.u. (80 μM, p < .0001), and 25,982 ± 5491 a.u. (100 μM, p < .0001) as depicted in Figure 3B. In a similar vein, the percentage of eryptotic cells was found to be 47.96% ± 21.01% (40 μM, p < .0001), 71.74% ± 16.32% (80 μM, p < .0001), and 80.63% ± 11.36% (100 μM, p < .0001), compared with control values of 2.64% ± 0.97% (Figure 3C). Figure 3D demonstrates a significant reduction in AChE activity after treatment with GRh2 from 358.9 ± 50.46 to 239.0 ± 54.21 U/L (20 μM, p = .0489), 134.5 ± 19.70 U/L (40 μM, p = .0026), and 114.6 ± 18.58 U/L (80 μM, p = .0026). In contrast, the ESR was significantly increased in cells exposed to 80 μM of GRh2 (5.0 ± 1.0 to 8.33 ± 1.16 mm/hr, p = .0194, Figure 3E).
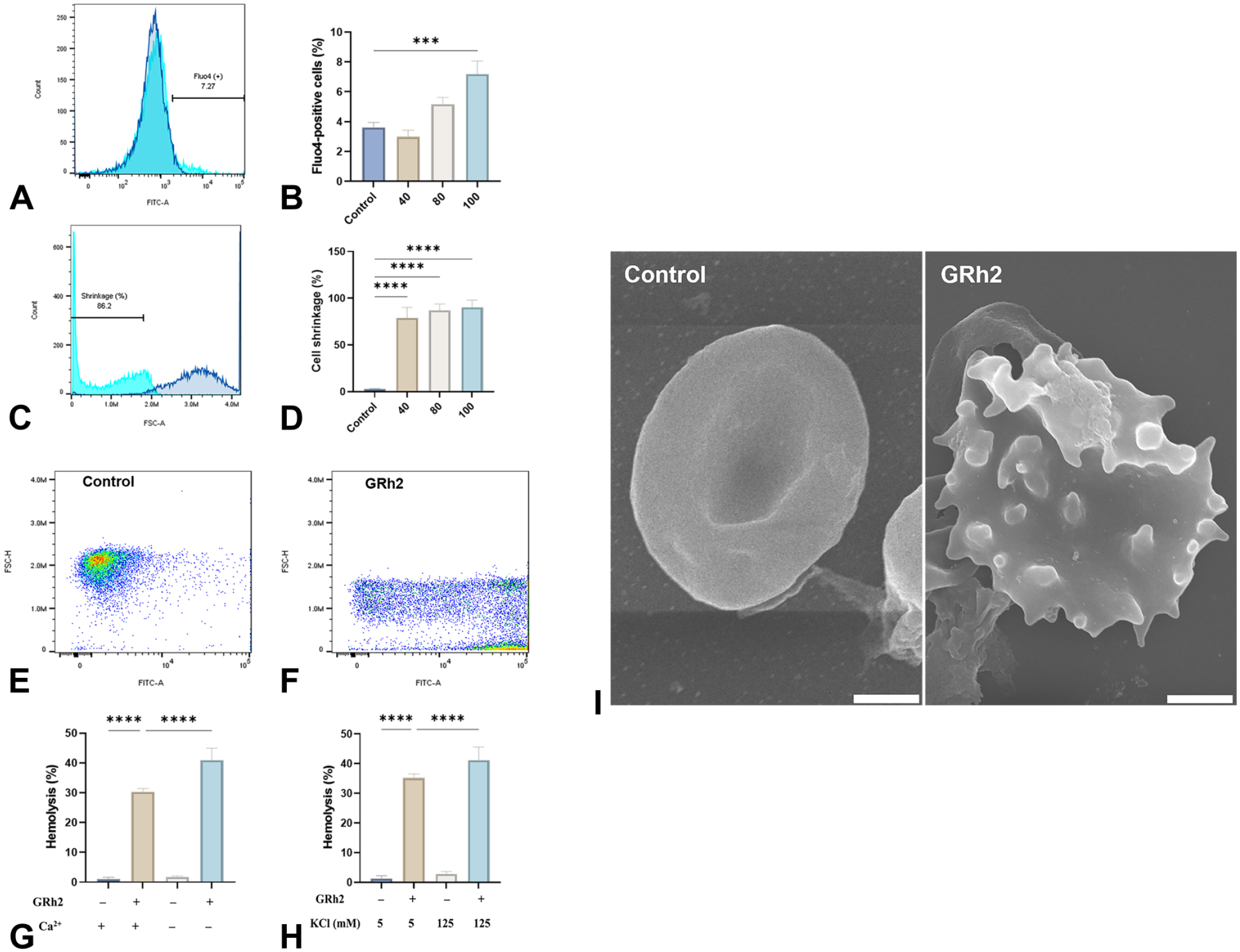
As shown in Figure 4B, the percentage of RBCs with increased levels of Ca2+ was significantly higher in cells exposed to 100 µM of GRh2 (3.62% ± 0.96% to 7.19 ± 2.59, p = .0002). As a result, notable cell shrinkage was detected in treated cells which significantly increased from 2.88% ± 0.43% to 79.04% ± 11.09% (p < .0001), 87.04% ± 6.95% (p < .0001), and 90.21% ± 7.70% (p < .0001) following treatment with 40, 80, and 100 μM, respectively (Figure 4D). Extracellular Ca2+ elimination did not protect the cells from the hemolytic activity of GRh2 but rather exacerbated it as shown in Figure 4G. A similar effect was also observed following the addition of 125 mM of KCl to the incubation medium (p < .0001, Figure 4H). Analysis of the ultrastructural changes induced by 40 μM of GRh2 revealed the formation of membrane blebbing; a characteristic eryptotic feature (Figure 4I).
A significant increase in DCF fluorescence from 16,280 ± 9487 to 88,812 ± 66,046 a.u. (80 μM, p = .036) and to 137,986 ± 91,538 a.u. (100 μM, p = .0004) was noted (Figure 5B). The percentage of cells with elevated ROS (Figure 5C) was thus significantly greater at 40 μM (50.43% ± 39.87%, p = .0032), 80 μM (76.78% ± 34.03%, p < .0001), and 100 μM (93.34% ± 10.54%, p < .0001) in comparison with control cells (5.29% ± 4.58%). Furthermore, cotreatment with 1 mM of vitamin C (Figure 5D) significantly reduced GRh2-induced hemolysis from 31.01% ± 2.94% to 25.96% ± 4.94% (p = .0039).
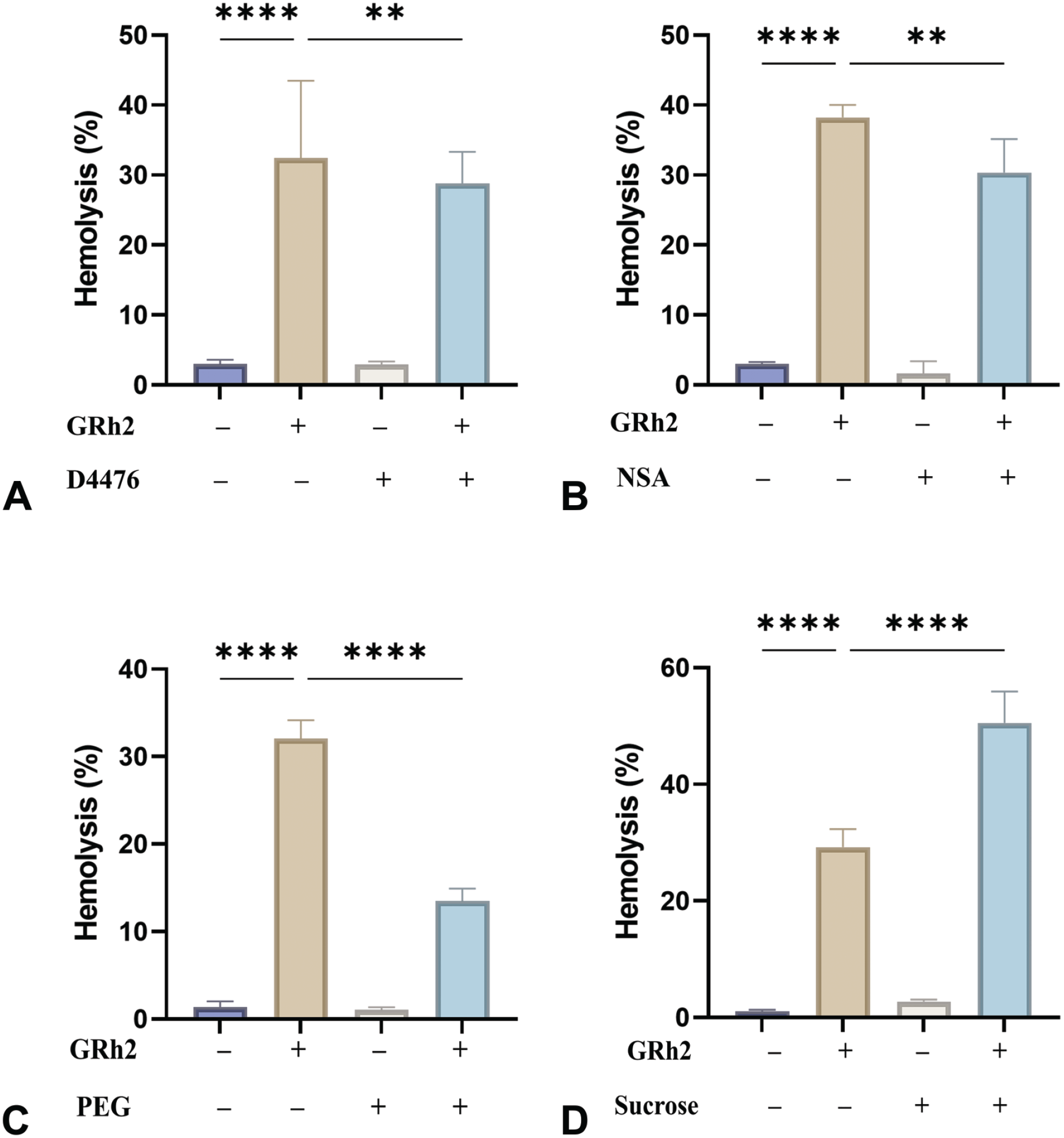
The hemolytic effect of GRh2 (80 µM) was significantly ameliorated by the administration of D4476 (36.73% ± 7.28% to 28.81% ± 4.50%, p = .0016, Figure 6A), NSA (36.63% ± 5.28% to 30.37% ± 4.80%, p = .0029, Figure 6B), and PEG 8000 (32.04% ± 2.11% to 12.71 ± 2.70%, Figure 6C). The addition of isosmotic sucrose exacerbated GRh2-induced haemolysis (29.96% ± 3.35% to 50.54% ± 5.37%, p < .0001, Figure 6D).
Although the number of RBCs in treated whole blood remained unchanged (Figure S1), there was a statistically significant elevation in Hb (70.56 ± 1.33 to 86.78 ± 19.98 g/L, p = .0272), hematocrit (23.61% ± 0.54% to 28.07% ± 6.11%, p = .0447), MCH (25.62 ± 5.13 to 31.10 ± 1.34 pg, p = .0069), MCHC (29.82 ± 0.35 to 30.82 ± 0.53 g/L, p = .0003), and MCV (85.87 ± 017.26 to 100.8 ± 3.08 fL, p = .0209). In addition, significant decreases in RDW-CV (0.16% ± 0.005% to 0.15% ± 0.009%, p = .0006), fragmented red cells (FRC) counts (0.021 ± 0.008×103 to 0.008 ± 0.0004×103 cells/μL, p = .0002), and FRC percentage (0.70% ± 0.17% to 0.29% ± 0.05%, p < .0001) were also observed. GRh2 also significantly raised the percentage of macrocytes from 6.29% ± 7.83% to 15.56% ± 5.62% (p = .0108) while reducing that of microcytes (7.84% ± 6.95% to 0.089% ± 0.08%, p = .0041).
In Figure S2, a significant decrease in reticulocyte count (0.035 ± 0.019×106 to 0.019 ± 0.008×106 cells/μL, p < .05) and percentage (1.13% ± 0.48% to 0.77 ± 0.07, p < .05) was observed upon treatment with 80 μM. Moreover, immature reticulocyte fraction (IRF) significantly decreased (1.78% ± 0.68% to 0.91% ± 0.45%, p = .0058), while low-fluorescence reticulocytes (LFR) increased (98.22% ± 0.68% to 99.09% ± 0.45%, p = .0058) upon GRh2 exposure. Of note, GRh2 caused significant elevations in reticulocyte Hb from (23.49 ± 4.4 to 27.42 ± 1.19 pg, p < .05).
Figure 3S shows that there was also a significant reduction in WBCs (3.13 ± 0.11×103 to 2.35 ± 0.49×103 cells/μL, p = .0003) that was specifically caused by the cytotoxic effect of GRh2 on lymphocytes (1.26 ± 0.11×103 to 0.50 ± 0.42×103 cells/μL, p < .0001). Accordingly, there was a significant decrease in total nuclear cell count (TNC) obtained through both the white cell nucleated (WNR) channel (3.13 ± 0.11×103 to 2.35 ± 0.49×103 cells/μL, p = .0003) and the white blood cell differential (WDF) channel (2.87 ± 0.11×103 to 2.33 ± 0.57×103cells/μL, p = .0125).
Discussion
The anticancer activity of GRh2 is well-established in the literature, but its effects on off-target tissue are understudied. 16 This work presents novel evidence of the in vitro cytotoxicity of GRh2 in human erythrocytes, highlighting the need for careful evaluation of the compound for use in anticancer therapy. The results of our study indicate that anticancer concentrations (10-100 μM) of GRh248,56 induce premature RBC death via hemolysis and eryptosis. This suggests that GRh2 disrupts the integrity of the RBC membrane, an effect expected to be more severe in susceptible individuals such as those afflicted with cancer, diabetes mellitus, or hemoglobinopathies. 22 As a result, various cellular components including Hb, LDH, AST, and CK are released from hemolyzed cells (Figure 1D-1F). The leakage of LDH and AST are well-established markers of erythrocyte membrane injury and hemolysis. 43 While CK activity is less commonly used as a marker of erythrocyte damage, as these cells are devoid of CK, the release of metabolic intermediates like adenylate kinase, ADP, and G6P from hemolyzed cells contribute to ATP formation in the assay reactions and lead to a falsely elevated CK activity. 21 Therefore, the observed increases in CK activity (Figure 1F) indicate GRh2-induced disruption of the erythrocyte membrane and leakage of intracellular components.
Furthermore, GRh2-induced hemolysis was accompanied by the release of K+, Na+, and Cl- ions (Figure 2A-2C), suggesting alterations in ion homeostasis and membrane permeability. 31
On the contrary, both acidity and osmotic fragility were unaffected by GRh2, as shown in Figure 2D and 2E, respectively. This unequivocally excludes the possibility of any effect on membrane elasticity, hence dismissing the hypothesis that GRh2 induces cell membrane expansion without causing significant rupture. A serious consequence of the presence of cell-free Hb in the circulation is the depletion of haptoglobin stores leading to Hb oxidation into methemoglobin. This leads to deleterious effects on the vasculature and other tissues including consumption of nitric oxide and inflammatory and oxidative damage particularly to renal tubules 45 .
In this report, we disclose for the first time that GRh2 triggers PS externalization (Figure 3A-3C); a pathognomonic marker of eryptosis that agrees with its apoptotic activity in nucleated cells.32,33,46 Eryptotic cells increase the likelihood of thrombotic events due to enhanced blood stasis and the attachment of dead RBCs to the endothelium. In congruence, we found that treated cells exhibit elevated ESR (Figure 3E) pointing at an exaggerated aggregation of RBCs in the form of Rouleaux formation which further compromises blood rheology. AChE activity is a significant indicator of cellular aging and membrane integrity. It also plays a crucial role in preserving the shape and size of RBCs albeit through ill-defined mechanisms. We found that GRh2 has anticholinesterase activity, thereby increasing the acetylcholine pool available to cells which could potentially impair oxygen transport. 14
Ca2+ signaling is the most important mechanism responsible for the expression of PS on the erythrocyte surface. 24 It was observed that GRh2 caused a significant rise in cytosolic Ca2+ activity (Figure 4A and 4B), which appeared to be unrelated to the entry of Ca2+ from outside the cell, as the removal of extracellular Ca2+ did not lead to a notable reduction in cell death (Figure 4G). Therefore, the activity of Ca2+ channels does not seem to be essential for GRh2 toxicity in RBCs. Of note, K+ channels facilitate the efflux of KCl, resulting in membrane hyperpolarization and water loss in response to elevated intracellular Ca2+. 10 The outcome is a significant reduction in cell size due to a decrease in cellular fluid volume (Figure 4C-4F). However, preventing the efflux of KCl does not protect the cells from hemolysis induced by GRh2 (Figure 4H) which suggests the existence of other mechanisms.
Eryptosis can be triggered by oxidative stress 23 in various pathological conditions and in response to xenobiotics.38,50 According to our findings, GRh2 induced a significant increase in ROS levels (Figure 5A-5C). When erythrocytes undergo oxidative stress, it allows Ca2+ entry into the cells which opens Ca2+-sensitive K+ channels leading to cell shrinkage 26 as well as the expression of PS on the outer membrane leaflet. 6 Consistent with this finding, the addition of vitamin C significantly mitigated the cytotoxicity of GRh2 (Figure 5D), indicating that oxidative damage is required for the full hemolytic activity of GRh2.
GRh2 toxicity was also greatly reduced by D4476; a specific inhibitor of CK1α which regulates cell death and survival in nucleated cells. 52 It has been reported that CK1α is activated in eryptotic cells undergoing hyperosmotic stress and energy depletion with elevated Ca2+, 61 which lends support to the involvement of a CK1α–Ca2+ signaling axis in GRh2 toxicity. Likewise, MLKL seems to participate in GRh2 cytotoxicity in erythrocytes. Necroptosis is known to occur in human RBCs following exposure to bacterial toxins and cryopreservation.28,37 In this process, a cascade of events eventually leads to the perforation of the cell membrane by MLKL. 18 The protective effect of NSA against GRh2-induced hemolysis suggests that GRh2 targets the canonical RIPK1/RIPK3/MLKL necroptotic pathway to exert its cytotoxic effects in RBCs. 64
The interaction between GRh2 and sucrose appears to be primarily characterized by ionic forces, 40 and the observed exacerbation of hemolysis by sucrose (Figure 6D) may be a direct result of this interaction. In contrast, PEG 8000 rescued the cells from hemolysis (Figure 6C), suggesting that PEG acts as a physical barrier against GRh2 and RBC interaction. 19
Previous studies have found that in the early stages of maturation, actin microfilaments shift to a disorganized structure and interact with RBC membrane structures to increase transverse connections between actin subunits and tropomyosin, protein 4.1R, and dematin. These proteins are linked to band3 by adducin. 51 Our study revealed that administration of GRh2 had no impact on the RBC count in whole blood (Figure S1); however, it had a notable influence on several RBC indices, suggesting that GRh2 may alter actin, which in turn affects the structure of the cell membrane and the cytoskeleton.
Our study on whole blood has revealed that reticulocytes are vulnerable to GRh2 toxicity (Figure S2). This suggests that GRh2 may compromise the ability of the bone marrow to respond to a decrease in RBC count by targeting nascent RBCs, further exacerbating anemic states. Moreover, GRh2 was also cytotoxic to lymphocytes (Figure S3) which aligns with the decreased count of WBCs reported for GRh2 in the spleen of mice. 58 Notably, the majority of anticancer medications cause significant lymphocytopenia with substantial immune sequelae. 29 Future studies must thus examine the immunomodulatory effects of GRh2 in relevant human model systems.
In conclusion, this study provides novel evidence of the toxicity of GRh2 in erythrocytes (Figure 7), which is characterized by disruption of membrane asymmetry, externalization of PS, elevated intracellular Ca2+ levels, cell shrinkage, membrane blebbing, and oxidative stress. GRh2 toxicity involves the activation of CK1α and MLKL and can be mitigated by vitamin C and PEG. Altogether, the anticancer potential of GRh2 may be augmented by the use of eryptosis inhibitors, ion channel modulators, and antioxidants to ameliorate the toxic effects on erythrocytes. Future studies must therefore focus on interrogating the hematological toxicity of GRh2 in relevant animal models.
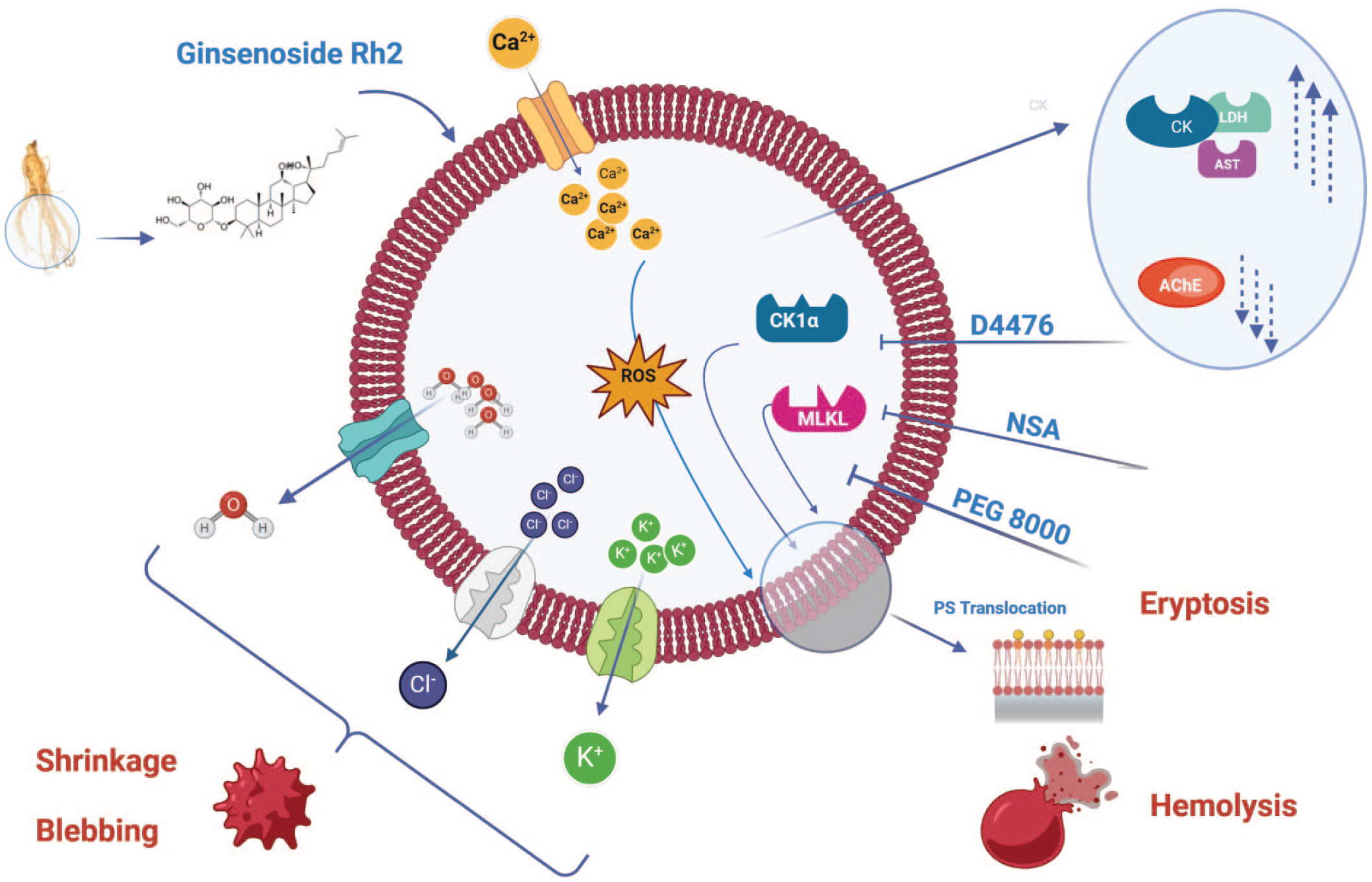
A working model of GRh2 cytotoxicity in RBCs. Created with BioRender.com
Supplemental Material
sj-docx-1-tpx-10.1177_01926233241268846 – Supplemental material for Ginsenoside Rh2 Regulates the Calcium/ROS/CK1α/MLKL Pathway to Promote Premature Eryptosis and Hemolysis in Red Blood Cells
Supplemental material, sj-docx-1-tpx-10.1177_01926233241268846 for Ginsenoside Rh2 Regulates the Calcium/ROS/CK1α/MLKL Pathway to Promote Premature Eryptosis and Hemolysis in Red Blood Cells by Sumiah A Alghareeb, Jawaher Alsughayyir and Mohammad A Alfhili in Toxicologic Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors extend their appreciation to the Researchers Supporting Project, King Saud University, Riyadh, Saudi Arabia, for funding this work (grant no. RSPD2024R554).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
