Abstract
Black cohosh is a readily available dietary supplement currently marketed as a remedy for dysmenorrhea and menopausal symptoms and is one of the top-selling herbal supplements in the United States. Black cohosh extract (BCE) was nominated to the National Toxicology Program (NTP) by the National Cancer Institute and the National Institute of Environmental Health Sciences due to its widespread use and lack of animal toxicity studies. Results of the NTP BCE subchronic mouse toxicity study revealed a dose-dependent, non-regenerative decrease in the erythron with an increase in the mean corpuscular volume (macrocytosis). Howell-Jolly bodies, or micronuclei, were significantly increased. These particular changes indicated an ineffective erythropoiesis consistent with a condition known as megaloblastic anemia. Megaloblastic anemia is due to disruptions in DNA synthesis during hematopoiesis and can be a result of an inherited or drug-induced disorder or a consequence of folate or cobalamin deficiency. Subsequent mouse studies revealed hematological and biochemical changes that were consistent with a functional cobalamin deficiency. This article will review basic mechanisms and laboratory features of megaloblastic anemia. The results of our studies including morphological abnormalities of the erythron and biomarkers of folate and cobalamin deficiencies, as well as hepatic microarray gene changes, are also discussed.
Black cohosh (Actaea racemose; previously Cimicifuga racemosa) is a botanical dietary supplement marketed as a remedy for menopausal symptoms including night sweats, hot flashes, and sleep disturbances. It is widely available as a stand-alone supplement or in combination with other botanical supplements such as gingko biloba or green tea. In 2020, black cohosh was ranked as the 17th top-selling supplement with over $22.9 million in sales. 1
Despite the widespread use of botanical supplements, there is very little safety data regarding many of them. To help fill this gap, the National Toxicology Program (NTP) has an ongoing botanical dietary supplement testing program (https://ntp.niehs.nih.gov/whatwestudy/topics/botanical/index.html). Within this program, black cohosh was nominated to the NTP for toxicity testing by both the National Cancer Institute and the National Institute of Environmental Health Sciences due its widespread use and lack of available data to assess safety. To date, several short-term rodent studies using a black cohosh extract (BCE) have been completed, and a 2-year bioassay is nearing completion.2,3 Test article procurement and characterization and the dose formulation development studies of BCE are described elsewhere. 2 The following article will review the results of the short-term studies as they relate to the hematological changes observed in the initial NTP 90-day or subchronic study.
Results of the Initial NTP Subchronic BCE Study
In the initial BCE study, 10 female rats and 10 female mice were gavaged daily for 90 days. 2 The dose groups were as follows: 0 mg/kg (vehicle control), 15 mg/kg, 125 mg/kg, 250 mg/kg, 500 mg/kg, and 1000 mg/kg. In the female mice, BCE caused a dose-dependent decrease in the erythrocyte count, hemoglobin concentration, and hematocrit with no change in the reticulocyte count and an increase in erythrocyte size (macrocytosis); erythrocyte count in the 1000 mg/kg mice was decreased by 20.3% compared to concurrent control. There were similar changes in the female rats but to a much lesser extent. In addition, occasional poikilocytes and basophilic stippling were observed on the mouse peripheral blood smears (Figure 1). BCE also caused a highly significant dose-dependent increase in micronucleated (also known as Howell-Jolly [HJ] bodies) reticulocytes and erythrocytes (Figure 2). In total, these hematological changes indicated an ineffective erythropoiesis consistent with a condition known as megaloblastic anemia (MA).
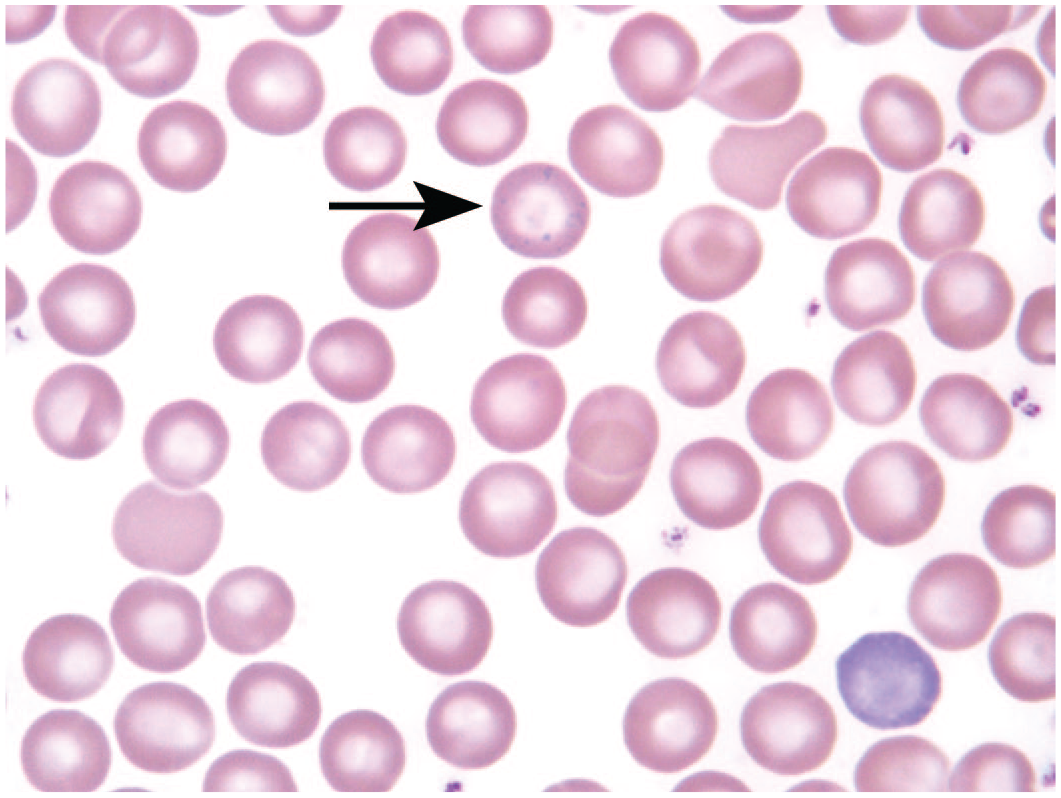
Basophilic stippling (arrow) was occasionally observed on the blood smears of mice treated daily with 1000 mg/kg black cohosh extract for 92 days; Wright-Giemsa stain. 3
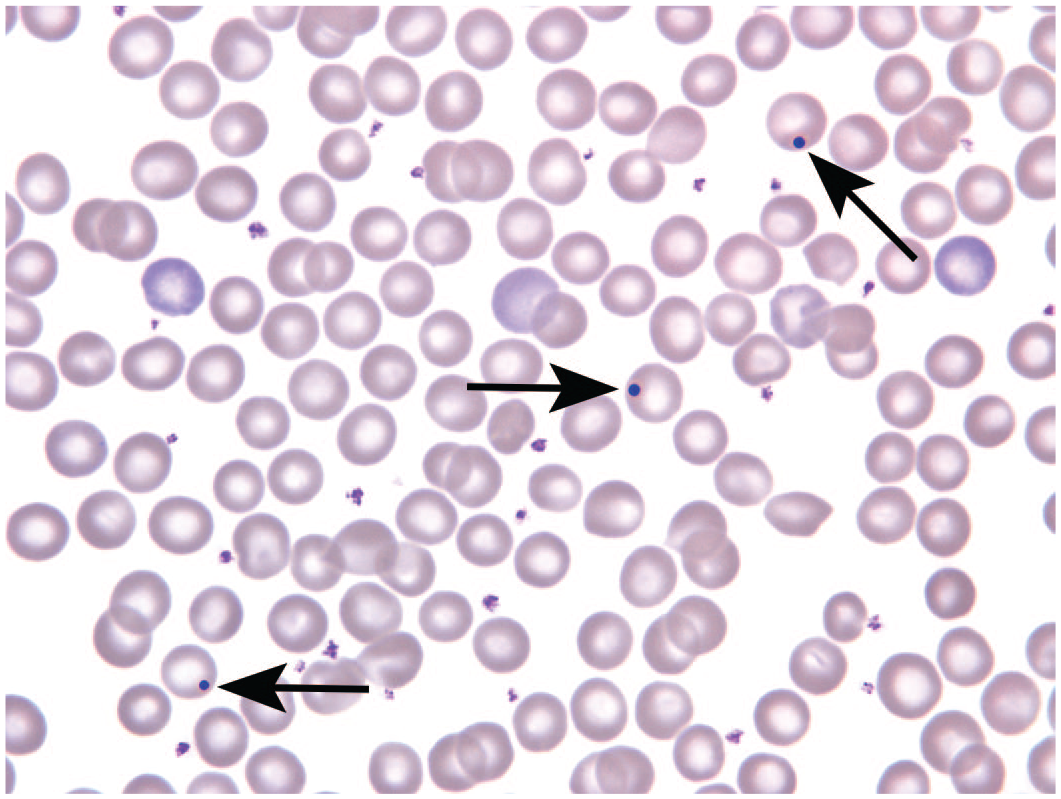
Howell-Jolly bodies were significantly increased in mice treated daily with 1000 mg/kg black cohosh extract for 92 days. Howell-Jolly bodies are present within 3 individual erythrocytes (arrows). 3
Key Points Regarding MA
Despite its given name, MA is a dyshematopoietic condition that affects all bone marrow cell lineages and is due to disruptions in DNA synthesis during hematopoiesis; however, abnormalities of the erythroid series are usually the predominant feature. Causes of MA include inherited disorders, drug-induced disorders (e.g., pyrimidine antagonists, antivirals), and dietary deficiencies or perturbations of folate and cobalamin (vitamin B12) metabolism.4,5 Perturbations of folate and cobalamin metabolism or what can be called functional deficiencies cause MA because they both are essential for several reactions involved in DNA synthesis including those required for the synthesis of thymidylate and purines. In addition, the remethylation of homocysteine to methionine also requires folate and cobalamin. Causes of functional folate and cobalamin deficiencies include changes in bacterial gut flora, decreased gastric intrinsic factor (pernicious anemia), interference with their uptake (e.g., colchicine, proton pump inhibitors), and inhibition of enzymes involved in their normal metabolism (e.g., methotrexate).6-9
Clinically, the two metabolites of homocysteine and methylmalonic acid (MMA) are measured for the diagnosis, confirmation, and differentiation of folate and cobalamin deficiencies.10,11 In mammals, only 2 cobalamin-dependent enzyme reactions are known to exist. 12 Cobalamin (as methylcobalamin) is a cofactor for the remethylation of homocysteine to methionine, in which 5-methyl-THF serves as a methyl donor; therefore, deficiencies in either folate or cobalamin result in an accumulation of serum homocysteine. The other cobalamin-dependent enzyme reaction involves the conversion of methylmalonyl-coenzyme A (CoA) to succinyl-CoA, where cobalamin (as adenosylcobalamin) serves as a cofactor for the enzyme methylmalonyl-CoA mutase. A cobalamin deficiency will lead to the accumulation of methylmalonyl-CoA and its hydrolysis product of MMA. Thus, increases in homocysteine may indicate a folate or cobalamin deficiency, while increases in MMA are specific only for the diagnosis of a cobalamin deficiency.
Results of the Mechanistic Subchronic BCE Study
To further explore folate or cobalamin deficiencies as a mechanism behind the BCE-induced hematological changes, a second subchronic mouse study was performed. 3 This study had two dose groups—control and 1000 mg/kg—with BCE administered via gavage for 92 days. Complete blood count (CBC) and blood smear findings (basophilic stippling, HJ bodies, and poikilocytes) of the initial NTP subchronic study were confirmed in this study (Table 1). In addition, the blood biomarkers discussed above—homocysteine and MMA—were elevated in the BCE-treated mice (Table 2). Serum folate and cobalamin levels and selected serum hepatic and kidney chemistries were unchanged in the treated mice (Table 2). As part of the ongoing BCE 2-year bioassay, interim bone marrow cytology was evaluated at 90 days. The M:E ratio was unchanged, but there were dose-dependent dysplastic changes in the metarubricytes. Dysplastic changes included multi-lobulated nuclei, nuclear to cytoplasmic asynchrony, and nuclear fragmentation. Taken together, the results of the mechanistic subchronic study and the 90-day interim bone marrow cytology were consistent with a MA caused by a functional cobalamin deficiency.
Hematology parameters of B6C3F1/N female mice after gavage treatment with vehicle or 1000 mg/kg black cohosh extract for 92 days.
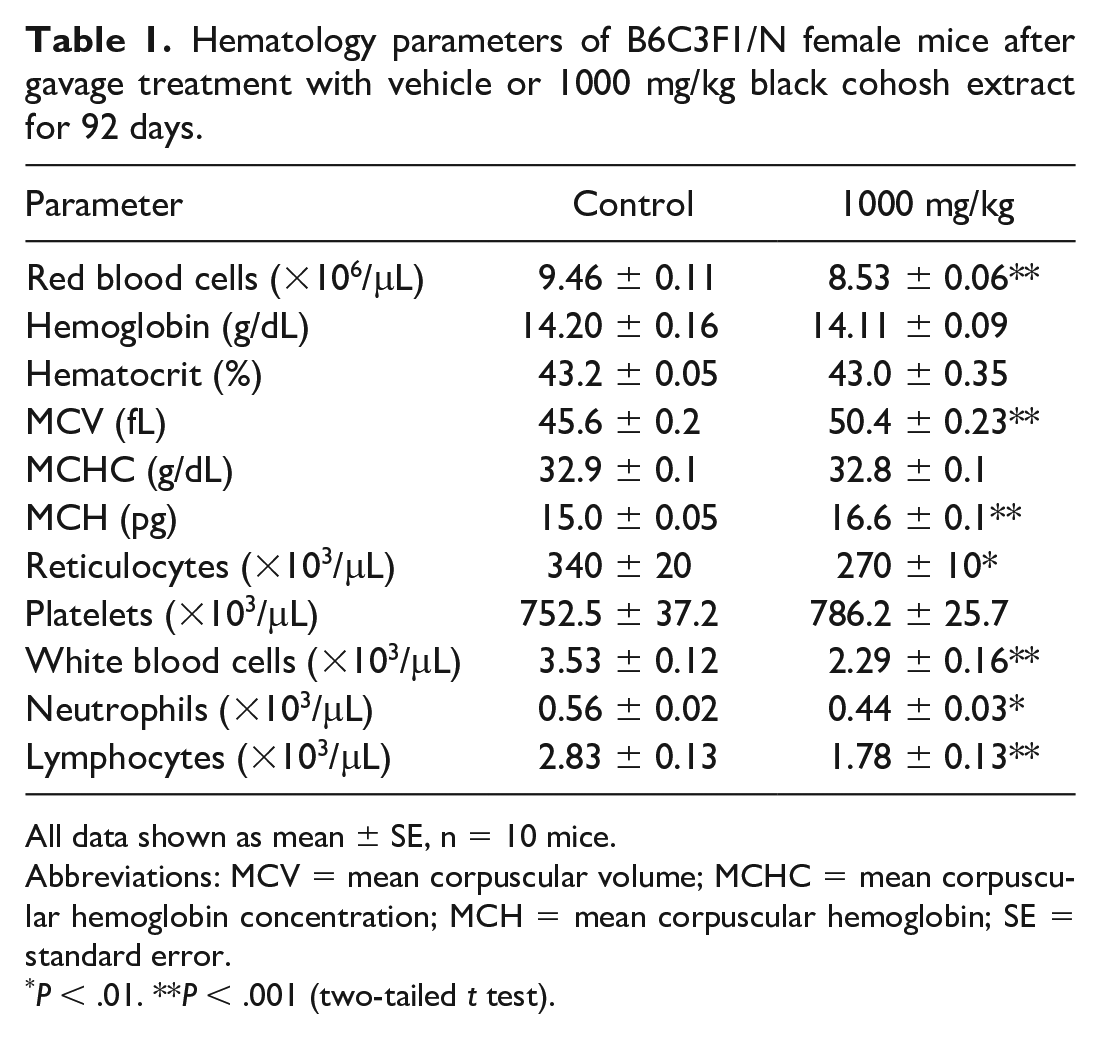
All data shown as mean ± SE, n = 10 mice.
Abbreviations: MCV = mean corpuscular volume; MCHC = mean corpuscular hemoglobin concentration; MCH = mean corpuscular hemoglobin; SE = standard error.
P < .01. **P < .001 (two-tailed t test).
Clinical chemistry parameters of B6C3F1/N female mice after gavage treatment with vehicle or 1000 mg/kg BCE for 92 days.
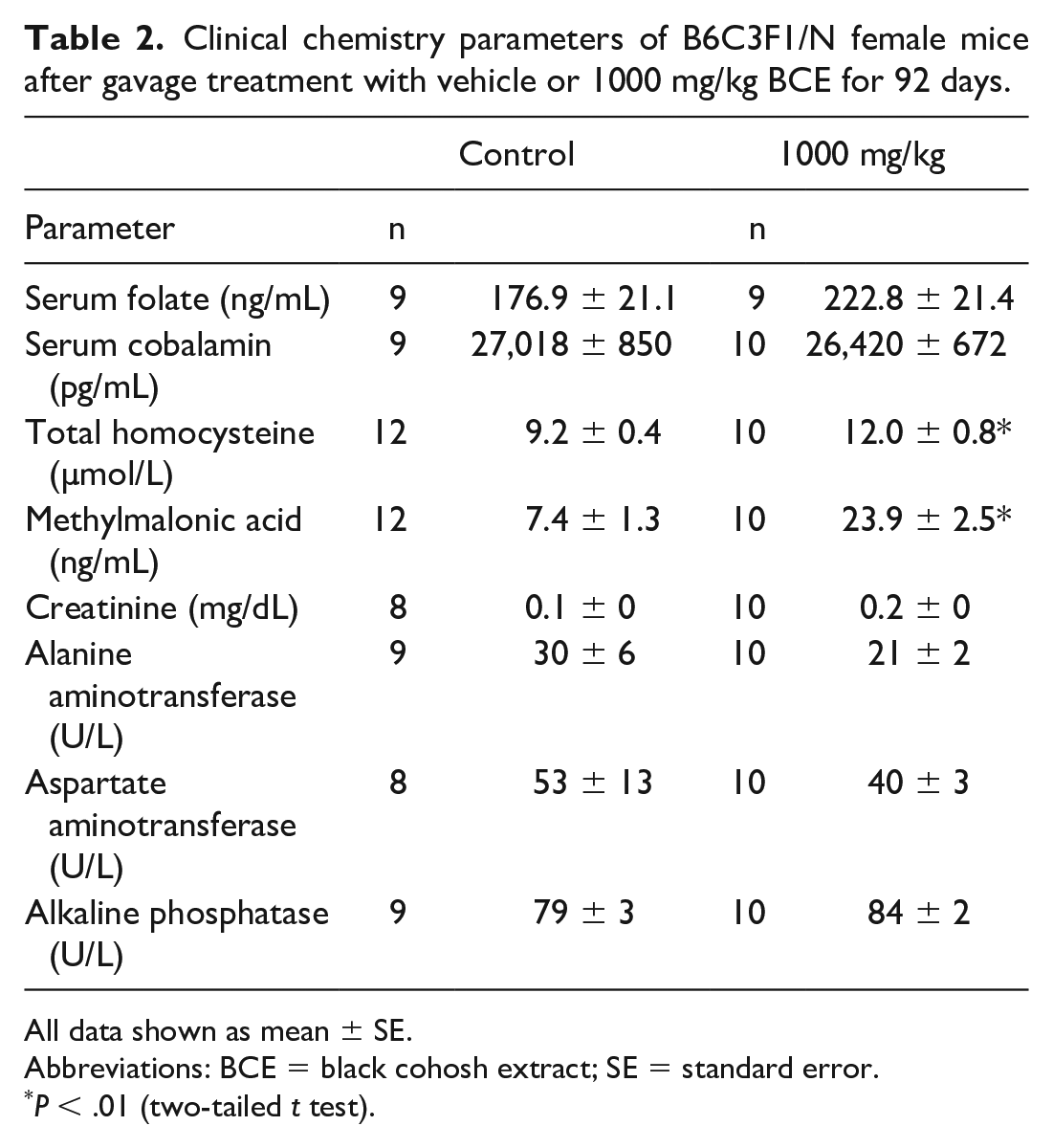
All data shown as mean ± SE.
Abbreviations: BCE = black cohosh extract; SE = standard error.
P < .01 (two-tailed t test).
Results of the Liver Microarray From the Subchronic BCE Study
Liver tissue from six control and six 1000 mg/kg mice from the mechanistic subchronic study was collected at necropsy and flash frozen. Liver microarray was performed using an Affymetrix GeneChip mouse genome array. Differential expression of transcripts between the two groups were identified using a P value of <.05. Significant differential transcripts were uploaded into QIAGEN Ingenuity Pathway Analysis (IPA; Redwood City, CA) to facilitate biological interpretation of the transcriptional changes.
When the differentially expressed transcripts were analyzed in IPA, 626 genes were found to be altered; 413 were downregulated and 213 were upregulated. Many of the top upregulated genes were drug-metabolizing enzyme associated and included CYP2C18, CYP7A1, and Akr1b7. The 3 most significantly enriched canonical pathways as determined by IPA were as follows: hypoxia signaling in the cardiovascular system, Nrf2-mediated oxidative stress response, and protein ubiquitination pathway. At the center of the hypoxia signaling pathway is HIF-1α. With normoxia, HIF-1α is constitutively expressed and continually degraded via proteolysis by the ubiquitin-proteasome pathway. 13 With hypoxia, the continual degradation of HIF-1α is stalled. Because the treated mice had decreases in their erythron, it was not surprising that the hypoxia pathway was activated, as HIF-1α is one of the main drivers for inducing transcription erythropoietin. Similarly, as noted above, the protein ubiquitination pathway is involved in regulation of HIF-1α. Thus, this pathway’s enrichment may also be directly related to the MA in the treated mice. Finally, Nrf2 is not only involved in the activation of antioxidant response elements such as glutathione S-transferase and cytochrome P450, but it also has recently been shown in renal tubular cells that with hypoxia, HIF-1α stabilization is Nrf2 dependent. 14 During hypoxia, with stabilization of HIF-1α, Nrf2 is downregulated.
Four genes related to cobalamin metabolism were enriched in the BCE-treated mice. They are as follows: metabolism of cobalamin associated D (MMADHC; downregulated), transcobalamin 2 (TCN2; upregulated), methionine adenosyltransferase 2A (MAT2A; upregulated), and butadiene-homocysteine S-methyltransferase 2 (BHMT2; upregulated). The MAT2A and BHMT2 are involved in an alternative metabolic pathway for the remethylation of homocysteine to methionine. 15 The TCN2 transports transcobalamin-cobalamin II into cells. The MMADHC, also known as CblD, is involved in the intracellular transport and processing of cobalamin. 16 More specifically, it appears to play a role in determining whether cobalamin remains in the cytoplasm or enters the mitochondria during its metabolism. 16 The increase of MMADHC suggests perturbation of steps upstream in the metabolism of cobalamin. In humans, there are several cobalamin-related remethylation genetic disorders that lead to impaired remethylation of homocysteine to methionine. 15 In particular, defects of MMADHC cause a MA with increases in both homocysteine and MMA, among other clinical signs and symptoms. These remethylation disorders in humans offer insight to the mechanism behind the functional cobalamin deficiency observed in mice after administration of BCE.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
